Introduction
Focal adhesion kinase (FAK), a 125 kDa cytoplasmic
tyrosine (Y) kinase that is localized in focal contacts, is
hypothesized to perform a crucial role in the control of
integrin-mediated cellular functions, such as cell invasion,
migration, cycle progression and survival (1–3).
Autophosphorylation of FAK on Y397 (pY397), the most important
autophosphorylated site of FAK (4),
leads to the activation of FAK and the formation of a multiprotein
signaling complex containing FAK at the core (5). Previous reports have revealed the
overexpression of FAK in numerous solid cancers (6–12) and
metastatic tumors (13–15). Subsequent studies have also revealed
that FAK and phosphorylated FAK (p-FAK) are involved in the
carcinogenesis of cancers of the digestive organs (14,16). In
particular, FAK and p-FAK (pY397 and pY407) expression levels have
been found to be closely associated with the differentiation of
human colon cancers (17). In
addition, Owen et al (18)
demonstrated the importance of pY397 FAK signaling in the promotion
of cell spreading and migration, suggesting that the interaction
between FAK and effector molecules, which bind to this site
presents a key step in these processes (18–20). As a
result, it was hypothesized that pY397 FAK exhibits an extremely
important role during cancer progression.
To the best of our knowledge, only a small number of
studies investigating the expression of pY397 FAK in cancer have
been performed (17,21–23) and no
studies investigating the expression of pY397 FAK in non-small cell
lung cancer (NSCLC) have been published in the literature.
Therefore, the aim of the present study was to analyze the
expression of pY397 FAK in NSCLC to investigate the association
between pY397 FAK and the development and prognosis of NSCLC.
Materials and methods
Ethics statement
The present study was approved by the Institutional
Research Board of Harbin Medical University (Harbin, Heilongjiang,
China). Samples were obtained with informed consent and written
informed consent was obtained from each patient.
Tissue specimens
Paraffin-embedded surgical tissue specimens were
obtained from 145 patients with NSCLC who underwent curative
surgery between January 2001 and October 2005 at The First
Affiliated Hospital of Harbin Medical University (Harbin,
Heilongjiang, China). Samples were obtained from 145 primary tumor
tissues, 87 corresponding non-tumor tissues and 37 lymph node
metastases. None of the patients had received radiotherapy or
chemotherapy prior to surgical treatment. Representative tissue
sections were used for immunohistochemical analysis, and the NSCLC
diagnosis for each lesion was pathologically confirmed. The
clinicopathological data of the patients are reported in Table I.
 | Table I.Associations between pY397 FAK
expression and the clinical and pathological characteristics of 145
non-small cell lung cancer patients. |
Table I.
Associations between pY397 FAK
expression and the clinical and pathological characteristics of 145
non-small cell lung cancer patients.
|
|
| pY397 FAK
expression |
|
|---|
|
|
|
|
|
|---|
| Parameter | n | Weak, n | Strong, n | P-value |
|---|
| Age |
|
|
| 0.405 |
| ≤60
years | 80 | 35 | 45 |
|
| >60
years | 65 | 24 | 41 |
|
| Gender |
|
|
| 0.133 |
| Male | 94 | 34 | 60 |
|
|
Female | 51 | 25 | 26 |
|
| Size |
|
|
| 0.704 |
| <3
cm | 49 | 21 | 28 |
|
| ≥3
cm | 96 | 38 | 58 |
|
| Differentiation |
|
|
|
|
|
Well-moderate | 91 | 41 | 50 | 0.165 |
| Low | 54 | 18 | 36 |
|
| Histotype |
|
|
| 0.754 |
|
Adenocarcinoma | 69 | 29 | 40 |
|
|
Squamous cell carcinoma | 76 | 30 | 46 |
|
| Lymph node
metastasis |
|
|
| 0.764 |
|
Present | 71 | 28 | 43 |
|
|
Absent | 74 | 31 | 43 |
|
Immunohistochemical studies
Immunohistochemical analysis of the
paraffin-embedded sections was performed using the PV-9000 kit
(Beijing Zhongshan Golden Bridge Biotechnology Co., Ltd., Beijing,
China) according to the manufacturer's instructions. For antigen
retrieval, the sections were autoclaved for 10 min at 121°C. The
sections were then incubated with rabbit polyclonal anti-pY397 FAK
primary antibodies (cat no. 07-012; Upstate Biotechnology, Inc.,
Lake Placid, NY, USA) at a dilution of 1:200 at 4°C overnight. Two
board-certified pathologists scored each tissue section for pY397
FAK expression based on a scoring system (24,25) that
measured staining intensity as follows: 0, none; 1, borderline; 2,
weak; 3, moderate; and 4, strong. The percentage of positive cells
(1–100%), cellular localization (cytoplasm, nucleus, membrane or a
combination) and overall distribution (homogeneous or
heterogeneous) were also assessed. The expression of pY397 FAK was
considered to be strong if the tissue sections exhibited a staining
intensity of 3–4 and ≥90% of the cells exhibited positive
immunoreactivity, whereas tissue sections exhibiting a staining
score of 0–2 with <90% of cells exhibiting positive
immunoreactivity were considered to exhibit weak pY397 FAK
expression.
Statistical analysis
All statistical analyses were performed using SPSS
version 17.0 statistical software (SPSS, Inc., Chicago, IL, USA).
The χ2 test was used to compare the intensity of pY397
FAK immunostaining in the non-tumor lung tissues, primary tumors
and the corresponding metastatic tissues. In addition, the
association between clinicopathological factors and pY397 FAK
expression was analyzed using the χ2 test. A survival
curve was calculated using the Kaplan-Meier product limit estimate,
and differences between patient survival times were analyzed using
the log-rank method. P<0.05 was considered to indicate a
statistically significant difference.
Results
pY397 FAK overexpression in primary
NSCLC tumors and corresponding lymph node metastases
To confirm whether pY397 FAK was involved in the
tumorigenesis of NSCLC, immunohistochemical analyses were conducted
to analyze pY397 FAK expression in paraffin-embedded specimens
obtained from 145 primary carcinomas, 87 corresponding non-tumor
lung tissues and 37 lymph node metastases.
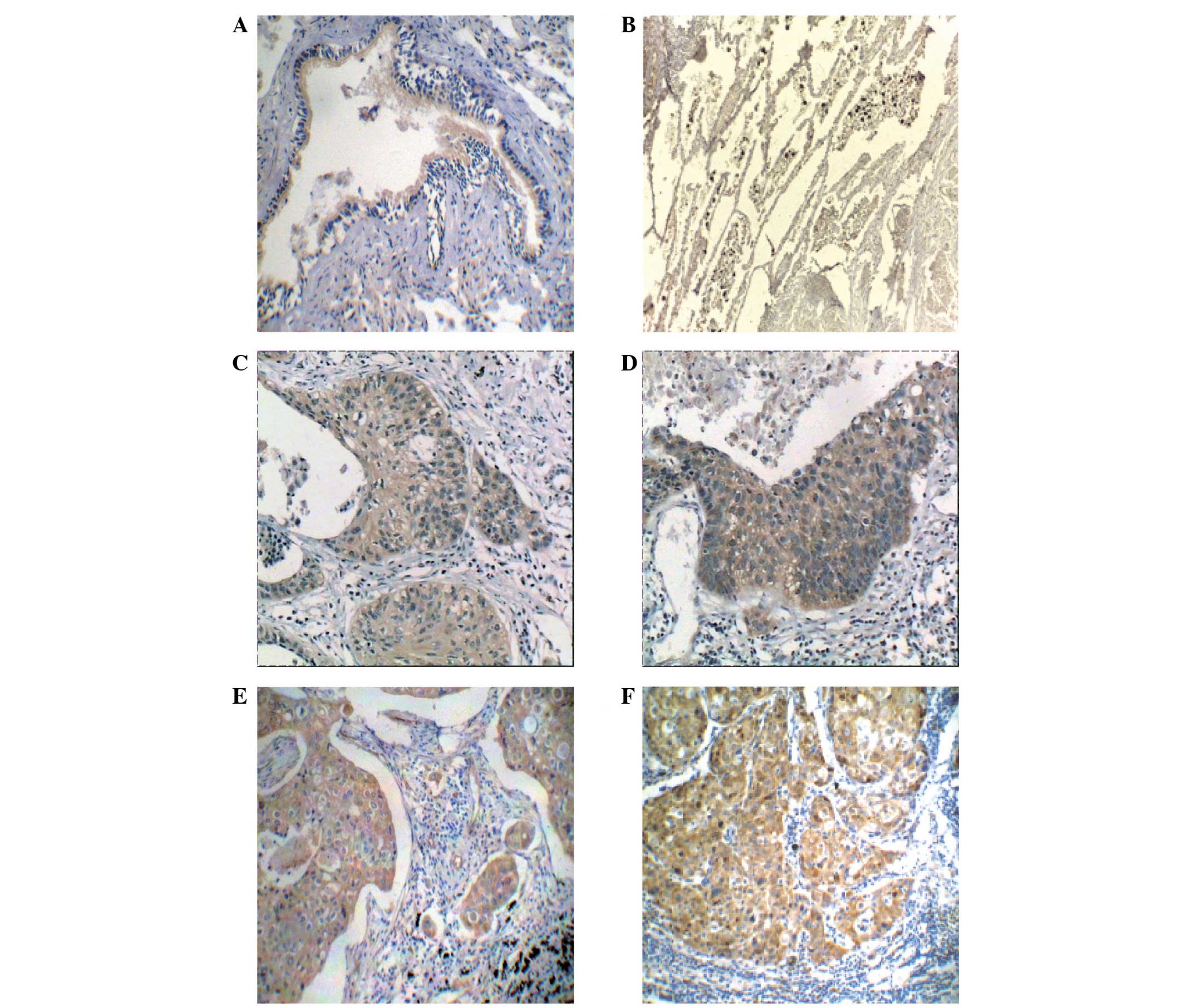
Cytoplasmic pY397 FAK expression was observed in
NSCLC and non-tumor lung cells. In non-tumor cells, pY397 FAK
protein expression was mainly identified in the normal ciliated
columnar epithelium of the bronchus, particularly the apical cilia
and the basal surface, as well as in the type 2 alveolar epithelial
cells (Fig. 1A). pY397 FAK expression
was also identified in non-tumor cells, indicating that FAK
autophosphorylation is required for the normal development of cells
(26).
In the matched samples, a total of 86 (59%) tumor
tissues and 21 (57%) lymph node metastases exhibited strong pY397
FAK expression, while 87 (100%) non-tumor tissues revealed weak
immunostaining (Table II; Fig. 1). From these results, a strong level
of pY397 FAK expression was identified in primary NSCLC tumors and
the associated lymph node metastases compared with non-tumor
tissues (P<0.001). No significant difference was identified
between the pY397 FAK expression levels of the primary tumors and
the associated lymph node metastases (P=0.778). Notably, the
expression of pY397 FAK was increased in precancerous lesions and
in situ primary carcinoma lesions compared with normal
epithelia (Fig. 1). These results
indicate that pY397 FAK is involved in tumorigenesis, particularly
the early development of NSCLC.
 | Table II.Expression of pY397 FAK in non-tumor
tissues, primary NSCLC tissue and lymph node metastases. |
Table II.
Expression of pY397 FAK in non-tumor
tissues, primary NSCLC tissue and lymph node metastases.
|
|
| pY397 FAK
expression |
|
|---|
|
|
|
|
|
|---|
| Tissue | n | Weak, n (%) | Strong, n (%) | P-value |
|---|
| Non-tumor | 87 | 87
(100) | 0 (0) | 0.000a |
| Primary NSCLC | 145 | 59 (41) | 86 (59) | 0.778b |
| Lymph node
metastases | 37 | 16 (43) | 21 (57) |
|
Association between pY397 FAK
expression and clinicopathological features of NSCLC patients
To investigate the clinical significance of pY397
FAK in NSCLC, the clinicopathological profiles of patients with and
without pY397 FAK overexpression were analyzed, as reported in
Table I. No significant differences
were identified between pY397 FAK overexpression and patient age,
gender, tumor size, differentiation, histotype or lymph node
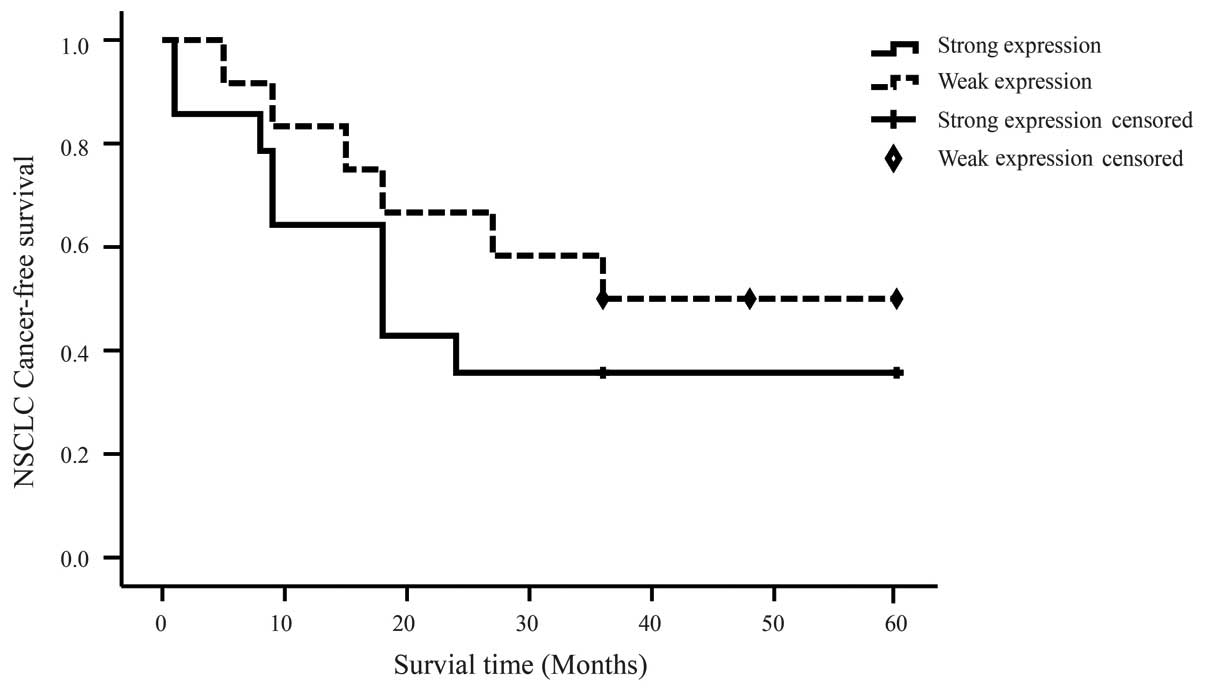
metastasis (P>0.05). In addition, although no significant
difference was identified between the five-year recurrence-free
survival rate of patients with and without pY397 FAK overexpression
(P>0.05), patients with strong pY397 FAK expression exhibited a
trend towards shorter survival time (Fig.
2). These results indicate that pY397 FAK overexpression is not
a prognostic marker in NSCLC.
Discussion
FAK, which is activated by phosphorylation of Y397,
regulates numerous cellular functions and is critical for cancer
progression. FAK overexpression has been observed in various
cancers (26). However, to the best
of our knowledge, only a small number of studies have demonstrated
an association between FAK phosphorylation on pY397 and cancer
using human tissue samples (17,21–23).
In the present study, pY397 FAK expression in NSCLC
tissues was analyzed. Strong pY397 FAK expression was identified in
the majority of primary cancer tissue samples, as well as
metastatic lymph nodes, whereas weak pY397 FAK expression was
identified in non-tumor lung tissues. In addition, no significant
difference in pY397 FAK expression was identified between primary
lesions and lymph node metastases. However, increased pY397 FAK
expression was observed in precancerous lesions and in situ
primary carcinoma lesions compared with the expression in normal
epithelia. These results are consistent with those of previous
studies. For example, Madan et al (22) observed no pY397 FAK immunostaining in
benign epithelium samples, while strong pY397 FAK staining was
observed in all breast carcinoma tissues analyzed. Furthermore,
Grisaru-Granovsky et al (27)
observed high levels of pY397 FAK expression in invasive ovarian
samples, but not in normal ovarian epithelium and Aronsohn et
al (28) found that pY397 FAK was
overexpressed in laryngeal squamous carcinoma tissues compared with
normal squamous epithelium. Madan et al (22) also demonstrated that pY397 FAK
overexpression was found to be correlated with malignant
transformation, but not the development of invasive tumor
properties. By contrast, Carelli et al (29) performed western blot analysis, which
demonstrated that pY397 FAK was equally phosphorylated in the
neoplastic and non-neoplastic regions of lung cancer tissue.
However, the present study hypothesizes that the tissue homogenates
analyzed in the study by Carelli et al (29) may have contained a mixture of
epithelium and surrounding stromal contaminants. In addition, the
sample size used was relatively small. Consequently, these two
factors may have affected the expression of pY397 FAK observed in
NSCLC tissues. In the present study, the data was obtained from the
analysis of 145 NSCLC samples by immunohistochemistry, which may be
used to stain the epithelium specifically. The results of the
present study indicate that pY397 FAK is involved in the process of
malignant transformation, particularly the early stages. However,
pY397 FAK was not found to correlate with the metastatic phenotype
in NSCLC.
The upregulation of pY397 FAK observed in primary
and precancerous lesions indicates that the overexpression of pY397
FAK may be a feature of early stage NSCLC cells. The inhibition of
pY397 FAK expression of breast cancer and oral squamous cell
carcinoma by TAE226, a FAK inhibitor, was found to significantly
suppress the proliferation and caspase-mediated apoptosis (30,31),
indicating that pY397 FAK may contribute to the malignant
transformation of NSCLC by promoting proliferation and suppressing
apoptosis. Compared with primary lesions, the lack of change in
pY397 FAK expression in the metastatic lymph nodes may predict a
completely anchorage-independent phenotype in additional tumor
progression, or the lack of change in expression may suggest that
certain signals are no longer required while other signals emerge.
For example, Slack et al (32)
demonstrated that increased FAK expression in combination with
pY861 FAK contributed to an increase in the cell motility of
prostate cancer cells. However, further studies are required to
investigate these findings.
Additionally, in the present study, no association
was identified between pY397 FAK expression and clinicopathological
factors, including the presence of lymph node metastases and the
survival rate, suggesting that pY397 FAK is not a prognostic marker
for NSCLC.
In conclusion, the results of the present study
indicate that the overexpression of pY397 FAK during NSCLC
progression may be involved with the development and maintenance of
NSCLC. pY397 FAK positivity does not predict patient outcome,
however, its expression may serve as an attractive therapeutic
target. In addition, the mechanism of overexpression of pY397 FAK
in NSCLC has yet to be clarified.
Acknowledgements
This study was supported by the International
Science and Technology Cooperation Program of China (grant no.
2013DFA31610) and the Natural Science Foundation in Heilongjiang
Province of China (grant no. QC08C90).
Glossary
Abbreviations
Abbreviations:
|
FAK
|
focal adhesion kinase
|
|
NSCLC
|
non-small cell lung cancer
|
References
|
1
|
Schaller MD, Borgman CA, Cobb BS, Vines
RR, Reynolds AB and Parsons JT: pp125FAK a structurally distinctive
protein-tyrosine kinase associated with focal adhesions. Proc Natl
Acad Sci USA. 89:5192–5196. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
van Nimwegen: MJ and van deW ater B: Focal
adhesion kinase: A potential target in cancer therapy. Biochem
Pharmacol. 73:597–609. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Megison ML, Stewart JE, Nabers HC, Gillory
LA and Beierle EA: FAK inhibition decreases cell invasion,
migration and metastasis in MYCN amplified neuroblastoma. Clin Exp
Metastasis. 30:555–568. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Schlaepfer DD, Mitra SK and Ilic D:
Control of motile and invasive cell phenotypes by focal adhesion
kinase. Biochim Biophys Acta. 1692:77–102. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Schaller MD: Biochemical signals and
biological responses elicited by the focal adhesion kinase. Biochim
Biophys Acta. 1540:1–21. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Oktay MH, Oktay K, Hamele-Bena D, Buyuk A
and Koss LG: Focal adhesion kinase as a marker of malignant
phenotype in breast and cervical carcinomas. Hum Pathol.
34:240–245. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Watermann DO, Gabriel B, Jäger M,
Orlowska-Volk M, Hasenburg A, Hausen Zur A, Gitsch G and Stickeler
E: Specific induction of pp125 focal adhesion kinase in human
breast cancer. Br J Cancer. 93:694–698. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Rovin JD, Frierson HF Jr, Ledinh W,
Parsons JT and Adams RB: Expression of focal adhesion kinase in
normal and pathologic human prostate tissues. Prostate. 53:124–132.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Schneider GB, Kurago Z, Zaharias R, Gruman
LM, Schaller MD and Hendrix MJ: Elevated focal adhesion kinase
expression facilitates oral tumor cell invasion. Cancer.
95:2508–2515. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Judson PL, He X, Cance WG and Van Le L:
Overexpression of focal adhesion kinase, a protein tyrosine kinase,
in ovarian carcinoma. Cancer. 86:1551–1556. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kim SJ, Park JW, Yoon JS, et al: Increased
expression of focal adhesion kinase in thyroid cancer:
Immunohistochemical study. J Korean Med Sci. 19:710–715. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Fujii T, Koshikawa K, Nomoto S, Okochi O,
Kaneko T, Inoue S, Yatabe Y, Takeda S and Nakao A: Focal adhesion
kinase is overexpressed in hepatocellular carcinoma and can be
served as an independent prognostic factor. J Hepatol. 41:104–111.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Lark AL, Livasy CA, Calvo B, Caskey L,
Moore DT, Yang X and Cance WG: Overexpression of focal adhesion
kinase in primary colorectal carcinomas and colorectal liver
metastases, Immunohistochemistry and real-time PCR analyses. Clin
Cancer Res. 9:215–222. 2003.PubMed/NCBI
|
|
14
|
Cance WG, Harris JE, Iacocca MV, et al:
Immunohistochemical analyses of focal adhesion kinase expression in
benign and malignant human breast and colon tissues: Correlation
with preinvasive and invasive phenotypes. Clin Cancer Res.
6:2417–2423. 2000.PubMed/NCBI
|
|
15
|
Miyazaki T, Kato H, Nakajima M, Sohda M,
Fukai Y, Masuda N, Manda R, Fukuchi M, Tsukada K and Kuwano H: FAK
overexpression is correlated with tumour invasiveness and lymph
node metastasis in oesophageal squamous cell carcinoma. Br J
Cancer. 89:140–145. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Murata T, Naomoto Y, Yamatsuji T, et al:
Localization of FAK is related with colorectal carcinogenesis. Int
J Oncol. 32:791–796. 2008.PubMed/NCBI
|
|
17
|
Matkowskyj KA, Keller K, Glover S,
Kornberg L, Tran-Son-Tay R and Benya RV: Expression of GRP and its
receptor in well-differentiated colon cancer cells correlates with
the presence of focal adhesion kinase phosphorylated at tyrosines
397 and 407. J Histochem Cytochem. 51:1041–1048. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Owen JD, Ruest PJ, Fry DW and Hanks SK:
Induced focal adhesion kinase (FAK) expression in FAK-null cells
enhances cell spreading and migration requiring both auto- and
activation loop phosphorylation sites and inhibits
adhesion-dependent tyrosine phosphorylation of Pyk2. Mol Cell Biol.
19:4806–4818. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Cary LA, Chang JF and Guan JL: Stimulation
of cell migration by overexpression of focal adhesion kinase and
its association with Src and Fyn. J Cell Sci. 109:1787–1794.
1996.PubMed/NCBI
|
|
20
|
Richardson A, Malik RK, Hildebrand JD and
Parsons JT: Inhibition of cell spreading by expression of the
C-terminal domain of focal adhesion kinase (FAK) is rescued by
coexpression of Src or catalytically inactive FAK, A role for
paxillin tyrosine phosphorylation. Mol Cell Biol. 17:6906–6914.
1997. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Fan DM and Shi HR: Pilot study: Alteration
of deleted in liver cancer 1 and phosphorylated focal adhesion
kinase Y397 cytoplasmic expression and the prognostic value in
advanced epithelial ovarian carcinoma. Int J Mol Sci. 12:8489–8501.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Madan R, Smolkin MB, Cocker R, Fayyad R
and Oktay MH: Focal adhesion proteins as markers of malignant
transformation and prognostic indicators in breast carcinoma. Hum
Pathol. 37:9–15. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Lai IR, Chu PY, Lin HS, Liou JY, Jan YJ,
Lee JC and Shen TL: Phosphorylation of focal adhesion kinase at
Tyr397 in gastric carcinomas and its clinical significance. Am J
Pathol. 177:1629–1637. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Lark AL, Livasy CA, Dressler L, et al:
High focal adhesion kinase expression in invasive breast carcinomas
is associated with an aggressive phenotype. Mod Pathol.
18:1289–1294. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Lightfoot HM Jr, Lark A, Livasy CA, Moore
DT, Cowan D, Dressler L, Craven RJ and Cance WG: Upregulation of
focal adhesion kinase (FAK) expression in ductal carcinoma in situ
(DCIS) is an early event in breast tumorigenesis. Breast Cancer Res
Treat. 88:109–116. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Corsi JM, Houbron C, Billuart P, et al:
Autophosphorylation-independent and-dependent functions of focal
adhesion kinase during development. J Biol Chem. 284:34769–34776.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Grisaru-Granovsky S, Salah Z, Maoz M,
Pruss D, Beller U and Bar-Shavit R: Differential expression of
protease activated receptor 1 (Par1) and pY397FAK in benign and
malignant human ovarian tissue samples. Int J Cancer. 113:372–378.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Aronsohn MS, Brown HM, Hauptman G and
Kornberg LJ: Expression of focal adhesion kinase and phosphorylated
focal adhesion kinase in squamous cell carcinoma of the larynx.
Laryngoscope. 113:1944–1948. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Carelli S, Zadra G, Vaira V, et al:
Up-regulation of focal adhesion kinase in non-small cell lung
cancer. Lung Cancer. 53:263–271. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kurio N, Shimo T, Fukazawa T, Takaoka M,
Okui T, Hassan NM, Honami T, Hatakeyama S, Ikeda M, Naomoto Y, et
al: Anti-tumor effect in human breast cancer by TAE226, a dual
inhibitor for FAK and IGF-IR in vitro and in vivo. Exp Cell Res.
317:1134–1146. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Kurio N, Shimo T, Fukazawa T, et al:
Anti-tumor effect of a novel FAK inhibitor TAE226 against human
oral squamous cell carcinoma. Oral Oncol. 48:1159–1170. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Slack JK, Adams RB, Rovin JD, Bissonette
EA, Stoker CE and Parsons JT: Alterations in the focal adhesion
kinase/Src signal transduction pathway correlate with increased
migratory capacity of prostate carcinoma cells. Oncogene.
20:1152–1163. 2001. View Article : Google Scholar : PubMed/NCBI
|