Introduction
Esophageal cancer is the eighth most common type of
cancer and the sixth most common cause of cancer-related mortality
worldwide (1). To date, surgical
resection remains the main method of treatment for esophageal
cancer patients, although the long-term survival rate remains low,
even for localized diseases (2).
Currently, the tubular stomach and whole stomach approaches are
used for esophagectomy in patients with esophageal cancer (3,4). It has
been reported that the tubular stomach method is more effective
than the whole stomach method for esophageal reconstruction in
terms of dealing with postoperative anastomotic leakage,
intrathoracic syndrome and reflux esophagitis (4). However, the extent to which this
surgical method is correlated with postoperative recurrence and
metastasis remains unknown. Therefore, in the present study we
aimed to investigate the effect of the tubular and whole stomach
methods on postoperative recurrence and metastasis in esophageal
cancer patients who underwent esophagectomy.
Patients and methods
Patients
A total of 121 patients that were diagnosed with
middle (70 cases) and lower (51 cases) esophageal cancer by
gastroscopic biopsy between March 2010 and March 2011 at Taian
Central Hospital (Taian, China), were recruited into this study.
The patients were assigned into the gastric tube group (67 cases;
male, 43; female, 24; age, 48–78 years; average age, 62 years) and
whole stomach group (54 cases; male, 36; female, 18; age, 51–76
years; average age, 63 years) according to the order of admission.
None of the patients received chemotherapy or radiotherapy prior to
surgery. Further details of the patient characteristics are shown
in Table I. Written informed consent
was obtained from all patients.
 | Table I.Characteristics of esophageal cancer
patients enrolled in the study. |
Table I.
Characteristics of esophageal cancer
patients enrolled in the study.
| Characteristic | Gastric tube
(cases) | Whole stomach
(cases) | Total (cases) | P-value |
|---|
| Age (years) |
|
| ≤50 | 2 | 0 | 2 | 0.200 |
|
51–60 | 23 | 24 | 47 | 0.256 |
|
61–70 | 33 | 20 | 53 | 0.178 |
| ≥71 | 9 | 10 | 19 | 0.445 |
| Gender |
|
| Male | 43 | 36 | 79 | 0.775 |
|
Female | 24 | 18 | 42 | |
| Tumor location |
|
| Middle
esophagus | 38 | 32 | 70 | 0.778 |
| Lower
esophagus | 29 | 22 | 51 | |
| Stage |
|
| 0 | 0 | 1 | 1 | 0.263 |
| I | 3 | 2 | 5 | 0.832 |
| II | 41 | 35 | 76 | 0.682 |
| III | 23 | 16 | 39 | 0.582 |
Surgical methods
All patients underwent esophagectomy through the
left thorax, above the aortic arches. All surgeries followed the
principles of radical resection of the esophagus: stumping from the
esophageal tumor edge was >5 cm, and wild lymph node dissection
was performed at the same time. The tubular stomach approach was
used for the patients in the gastric tube group. The tubular
stomach was formed from the distal aspect of the lesser curvature
of the stomach with the application of linear staplers. It was
created by resection of the lesser curvature of the stomach. The
formation of the gastric conduit (4–6 cm in diameter) was based on
the preservation of the gastroepiploic vessels of the greater
curvature of the stomach. Then, the tubular stomach was pulled
upwards to the left cervical part through the posterior mediastinal
route and hand-sewn two-layer anastomosis was performed. The
patients in the whole stomach group underwent the full stomach
approach, whereby the stomach was pulled to the left cervical part
through the posterior mediastinal route, and hand-sewn two-layer
anastomosis was performed.
Assessment of recurrence and
metastasis
Following surgery, the patients were followed up
every 6 months for 2 years. Tests included chest and upper
abdominal enhancement computed tomography, emission computed
tomography and gastroscopy. Lymph nodes were determined to be
enlarged if their diameter was >1 cm.
Statistical analysis
The SPSS 13.0 statistical package (SPSS Inc.,
Chicago, IL, USA) was used for the statistical analysis.
Measurement data were expressed as the means ± standard deviation
and analyzed by Student's t-test. Enumeration data were calculated
by the Chi-square test. The survival analysis was performed using
the Kaplan-Meier method and the difference was assessed by the
log-rank test. P<0.05 was considered to indicate a statistically
significant difference.
Results
There were no mortalities during surgery, and all of
the patients recovered and completed the follow-up period. There
was one case of anastomotic fistula and two cases of gastric
fistula in the gastric tube group, and three cases of anastomotic
fistula in whole stomach group (Table
II). All of the patients recovered within 2 months following
adequate drainage, irrigation and nutrition support postoperation.
With the exception of six patients in stage 0-I, all patients
underwent radiotherapy or chemotherapy according to the
pathological results. There was no significant difference between
the two groups in terms of the rates of recurrence or metastasis 6
months after surgery (Table II).
However, the rates of recurrence or metastasis 1 year and 2 years
after surgery in the gastric tube group were lower than those in
the whole stomach group (Table II).
There were three and seven cases of mortality 1 year after surgery
in the gastric tube and whole stomach group, respectively. In the
two-year follow-up period, there were 34 cases of mortality due to
local tumor recurrence or metastasis; 13 cases in the gastric tube
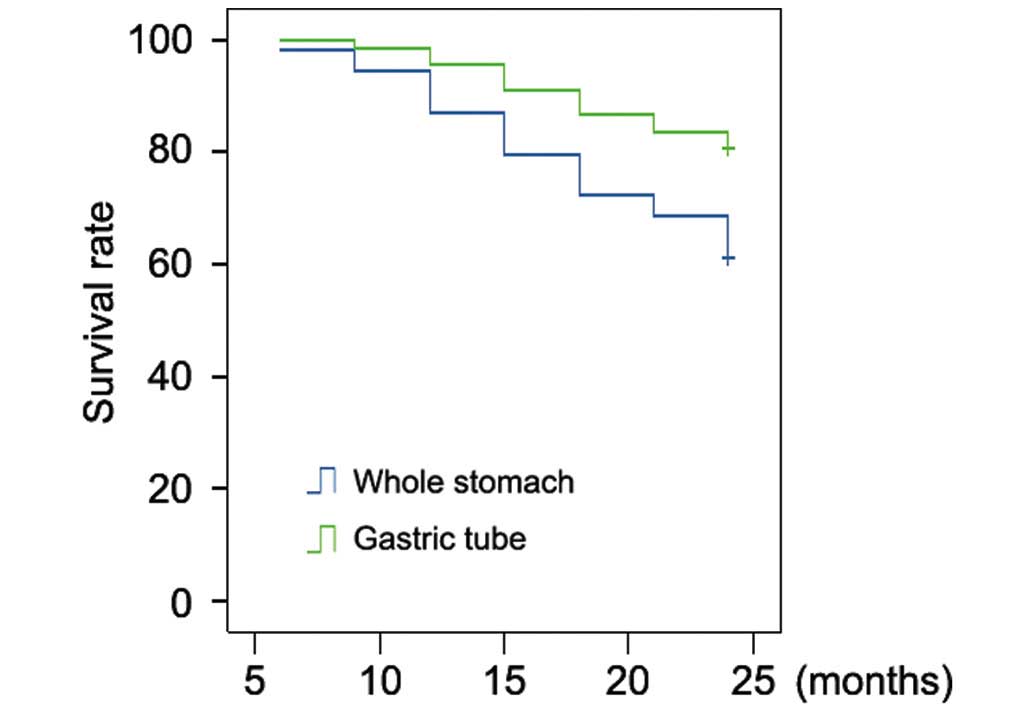
group and 21 cases in the whole stomach group. The survival rates
of the gastric tube and whole stomach groups were 80 and 61%,
respectively (Fig. 1). A significant
difference in the survival rate was identified between the two
groups (P=0.016; Fig. 1).
 | Table II.Fistula and postoperative recurrence
or metastasis in esophageal cancer patients. |
Table II.
Fistula and postoperative recurrence
or metastasis in esophageal cancer patients.
| Result | Gastric tube | Whole stomach | P-value |
|---|
| Gastric fistula | 2 | 0 | 0.200 |
| Anastomotic
fistula | 1 | 3 | 0.214 |
| Recurrence or
metastasis |
|
|
|
| Half a
year | 1 | 2 | 0.437 |
| 1
year | 4 | 10 | 0.032 |
| 2
year | 15 | 24 | 0.010 |
Discussion
In the present study, the data revealed that
patients in the gastric tube group had a lower rate of recurrence
or metastasis and a higher survival rate compared with the patients
in the whole stomach group. Our results suggest that patients with
esophageal cancer may obtain mid-term benefits from tubular stomach
reconstruction.
The presence of lymph node metastases affects the
prognosis of patients with esophageal cancer. It was demonstrated
that the lower the location, the lower the grade and the greater
the depth of invasion, the more likely a patient was to have
independent lymph node metastasis, while there was no significant
difference in lymphatic metastasis status among the four stages of
esophageal cancer (5). Van de Ven
et al (6) reported that lymph
node metastasis of thoracic esophageal cancer frequently occurred
on the cervicothoracic junction of the recurrent laryngeal nerve
chain lymph node, paraesophageal lymph nodes and the cardia→left
gastric artery→celiac lymph node chain. Currently, surgical
resection and subsequent lymph node dissection remain the most
significant and effective methods in the treatment of esophageal
cancer. Abdominal lymph node metastasis of thoracic esophageal
cancer normally involves the lesser curvature of the stomach.
Recurrent cancer in the thoracic stomach following surgical
treatment for esophageal cancer is mainly due to the incomplete
resection of lymph nodes in the lesser curvature region. The lesser
curvature of the stomach and the cardia are resected in the gastric
tube reconstruction surgery, which reduces the recurrence of the
gastric tumor. Complete abdominal lymph node dissection may also
reduce the risk of postsurgical chest and abdominal lymph node
recurrence or metastasis. In addition, the resection of the lesser
curvature may help to remove more fat and more of the lymphatic
network, and subsequently lead to thorough resection of the omentum
lymphatic network and microlymphatic metastasis. Therefore,
resection of the lesser curvature may increase the extent of lymph
node dissection and decrease the risk of local recurrence and
metastasis. The advantages of gastric tube reconstruction,
including the ease of surgery, reduction in thoracic stomach
syndrome and reflux, and improved quality of life of patients
following surgery, have been widely recognized in the clinical
treatment of esophageal cancer (7,8). In the
present study, our data revealed that the rates of recurrence or
metastasis one and two years after surgery in the gastric tube
group were lower than those in the whole stomach group. The
two-year survival rate of the gastric tube group was also higher
than that of the whole stomach group. These data indicate that
esophageal cancer patients may obtain greater benefit from the
tubular stomach approach compared with the whole stomach
approach.
In conclusion, this study demonstrated that the
gastric tube may decrease the rate of recurrence or metastasis and
improve the two-year survival rate following esophagectomy in
esophageal cancer patients. The current study suggests that
esophageal cancer patients may gain a mid-term benefit from gastric
tube reconstruction.
References
|
1
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Gaur P, Kim MP and Dunkin BJ: Esophageal
cancer: recent advances in screening, targeted therapy, and
management. J Carcinog. 13:112014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Shen C, Yang H, Zhang B, Chen H, Chen Z
and Chen J: Improved quality of life in patients with
adenocarcinoma of esophagogastric junction after gastric tube
reconstruction. Hepatogastroenterology. 60:1985–1989.
2013.PubMed/NCBI
|
|
4
|
Shu YS, Sun C, Shi WP, Shi HC, Lu SC and
Wang K: Tubular stomach or whole stomach for esophagectomy through
cervico-thoraco-abdominal approach: a comparative clinical study on
anastomotic leakage. Ir J Med Sci. 182:477–480. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Ge H, Liu C, Qiu R, Lu Y, Ye K, Yang C,
Liu X, Zheng X and Zhai C: The pattern of lymphatic metastasis and
influencing factors of thoracic esophageal squamous cell carcinoma.
Pract Radiat Oncol. 3(2 Suppl 1): S32–S33. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
van de Ven C, De Leyn P, Coosemans W, Van
Raemdonck D and Lerut T: Three-field lymphadenectomy and pattern of
lymph node spread in T3 adenocarcinoma of the distal esophagus and
the gastro-esophageal junction. Eur J Cardiothorac Surg.
15:769–773. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Okamoto N, Ozawa S, Kitagawa Y, Shimizu Y
and Kitajima M: Metachronous gastric carcinoma from a gastric tube
after radical surgery for esophageal carcinoma. Ann Thorac Surg.
77:1189–1192. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ninomiya I, Okamoto K, Oyama K, Hayashi H,
Miyashita T, Tajima H, Kitagawa H, Fushida S, Fujimura T and Ohta
T: Feasibility of esophageal reconstruction using a pedicled
jejunum with intrathoracic esophagojejunostomy in the upper
mediastinum for esophageal cancer. Gen Thorac Cardiovasc Surg.
62:627–634. 2014. View Article : Google Scholar : PubMed/NCBI
|