Introduction
Prostate cancer is the most common type of cancer
diagnosed in men, and the second cause of cancer-associated
mortalities in the USA and Western Europe (1,2). By
contrast, prostate adenocarcinoma rarely occurs in Japanese and
Chinese men (1,2). According to the statistical data
established from the demographic records in the USA, 1 out of 6 men
is diagnosed with prostate carcinoma during his lifetime (3), and ~1/2 of these patients experience the
final stage of malignancy despite certain local treatment
modalities, including radiotherapy and radical prostatectomy
(4,5).
Currently, the suppression of androgenic hormones is the only
effective systemic treatment approach during the advanced phase of
prostate cancer (6,7). However, the general outcome of this
approach is not essentially curative, although ~80% of the cases
demonstrate objective or subjective responses to anti-androgenic
therapy (6,7). As a result, the disease becomes
insensitive to the suppression approach during its course, and is
then defined as hormone-refractory prostate carcinoma (HRPC)
(6,7).
In the majority of cases that receive androgenic suppression
treatment, HRPC develops in ~1–4 years (6,7), depending
on various prognostic factors, and the mean survival rate has been
reported to be 12–18 months (8). By
contrast, a minimal favorable effect on the mean survival rate of
these patients has been reported with docetaxel, a derivative agent
resembling taxol, which may suggest the beginning of a novel era
for the treatment of HRPC (9,10). In regards to the possibility of total
cure, chemotherapy is far from clinical expectations (9,10). Thus,
no current standardized treatment exists for HRPC, and ongoing
research studies on this field are currently being conducted
(9,10). The ubiquitin proteasome pathway may
aid the investigation or discovery of novel targets for cancer
treatment (11,12). Bortezomib is a highly specific and
reversible inhibitor of the 26S proteasome that has been approved
for the treatment of multiple myeloma (11,12).
Although, bortezomib has demonstrated strong antitumor activity in
clinical settings against hematological malignancies (11), the drug has not been approved for the
treatment of solid tumors to date. Bortezomib was previously
indicated to cause apoptosis and growth inhibition through tumor
protein p53 (p53)-dependent and p53-independent mechanisms in a
number of cells in vitro (13,14). In
the present study, the effect of bortezomib on the
androgen-independent and p53-deficient cell line PC-3, alone and in
combination with chemotherapeutic agents such as irinotecan (an
inhibitor of topoisomerase I, a nuclear enzyme maintaining the DNA
structure) and etoposide (a topoisomerase II inhibitor) were
investigated. The combination of bortezomib with etoposide produced
synergistic effects at several doses, and may be tested in clinical
settings against prostate cancer as an alternative treatment in the
future.
Materials and methods
Materials
Dulbecco's modified Eagle's medium (DMEM) cell
culture media, fetal bovine serum (FBS), trypsin, penicillin and
streptomycin were obtained from Sigma-Aldrich (St. Louis, MO USA).
Bortezomib was provided by Dr Engin Ulukaya (Uludağ University,
Bursa, Turkey), and PC-3 cells were provided by Dr Serap Kuruca
Erdem (İstanbul University, İstanbul, Turkey). Water-soluble
tetrazolium (WST)-1 cell proliferation reagent was purchased from
Roche Diagnostics GmbH (Mannheim, Germany). All other reagents were
purchased from Sigma-Aldrich, unless otherwise stated.
Cell culture and maintenance
The human prostate cancer cell line used in the
present study, PC-3, was cultured in DMEM containing 4.5 g/l
glucose, 0.375% sodium bicarbonate, 100 µg/ml streptomycin and 100
U/ml penicillin. The medium was supplemented with 10% FBS. Stock
cultures were maintained in 25-cm2 flasks (Corning
Incorporated, Corning, NY, USA). Cells were grown in 96-well cell
culture plates (Corning Incorporated), and subcultured or seeded at
~70% confluence for subsequent experiments (13).
WST-1 cell proliferation assay
PC-3 prostate cancer cells were seeded at 1,000
cells/well in 96-well plates, and allowed to attach to the wells
for 24 h. Cells were then treated with various concentrations of
bortezomib (1 nM, 10 nM, 100 nM, 500 nM, 1 µM, 10 µM, 50 µM and 100
µM), etoposide (100 nM, 500 nM, 1 µM, 10 µM, 50 µM and 100 µM) and
irinotecan (100 nM, 500 nM, 1 µM, 10 µM, 50 µM and 100 µM) for 24
h. Following treatment, the medium was replaced with DMEM
containing 0.5% FBS and 10% WST-1. Cells were incubated with WST-1
for 1 h at 5% CO2 in an incubator, and the absorbance of
each sample was next recorded with an enzyme-linked immunosorbent
assay reader (RT-21000; Rayto Life and Analytical Sciences Co.,
Ltd., Shenzhen, China) using 450 and 630-nm filters as the
reference filters.
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide
(MTT)-based cytotoxicity assay
A total of 50,000 PC-3 cells were seeded in each
35×10 mm plate. When cells were in the logarithmic phase of the
growth curve (following 24 h of seeding), the cells were treated
with bortezomib alone (10 nM or 40 nM), irinotecan alone (100 nM,
500 nM or 2 µM), etoposide alone (5 µM or 20 µM) or with a
combination of the above drugs for 48 h. Following exposure to the
inhibitor, cells were treated for 2 h with DMEM containing 0.5% FBS
and 0.5 mg/ml MTT at 37°C with 5% CO2. Following
incubation, cells were lysed with 3% sodium dodecyl sulfate (SDS;
200 µl) plus 1 ml 40 mM HCl/isopropanol for 15 min. The homogenate
was diluted 1:10 with the same solution used to lyse the cell (200
µl 3% SDS plus 1 ml 40 mM HCl/isopropanol), and the absorbance of
each sample was recorded at 570 nm with a SmartSpec Plus
spectrophotometer (Bio-Rad Laboratories, Inc., Hercules, CA, USA)
(13,15).
iCELLigence system
Following resistor plate verification, 150 µl cell
culture medium (DMEM with 10% FBS) was added to each E-Plate L8
well (ACEA Biosciences, San Diego, CA, USA) and incubated at room
temperature for 30 min. The E-Plates were then inserted into the
RTCA iCELLigence instrument (ACEA Biosciences) for background
measurement. Subsequently, 12,500 PC-3 cells were seeded in each
E-Plate L8 well in a final volume of 500 µl. Following 24 h of
seeding, cells were treated with bortezomib alone, irinotecan alone
or in combination for approximately 96 h. The cell index, which is
a measure of the relative change in electrical impedance to account
for the cell status, was monitored every hour.
DNA fragmentation
A total of 200,000 PC-3 cells were seeded in
60×15-mm sterile petri dishes. Subsequently, cells in the
logarithmic phase of the growth curve were treated with 10 nM
bortezomib, 40 nM bortezomib, 5 µM etoposide, 20 µM etoposide or in
combination (10 nM bortezomib+5 µM etoposide; 10 nM bortezomib+20
µM etoposide; 40 nM bortezomib+5 µM etoposide; and 40 nM
bortezomib+20 µM etoposide) for 48 h. Control cells were treated
with isotonic solution. Following treatment, cells were washed with
1 ml phosphate-buffered saline (PBS; 8 g NaCI, 0.2 g KCI, 1.44 g
Na2HPO4 and 0.24 g
KH2PO4), and then resuspended in 200 µl PBS.
DNA was then isolated using the Apoptotic DNA-Ladder kit (Roche
Diagnostics GmbH), according to the manufacturer's protocol. Equal
amounts of DNA (2 µg) from each sample were separated by 1.5%
agarose (Vivantis Technologies Sdn. Bhd., Selangor Darul Ehsan,
Malaysia) gel electrophoresis at 80 V for 2 h. DNA was visualized
by ethidium bromide staining under ultraviolet light, and the image
was recorded using a Canon PowerShot G2 digital camera (Canon,
Inc., Tokyo, Japan) and RemoteCapture 2.2 software. GelQuant.NET software (biochemlabsolutions.com/GelQuant-NET.html) was
used for quantification of images.
Statistical analyses and combination
index (CI) determination
Data were analyzed and plotted with GraphPad Prism
3.03 software (GraphPad Software, Inc., La Jolla, CA, USA). To
determine the IC50 values of each inhibitor, a sigmoid-dose
response curve was fitted to the data using nonlinear regression in
GraphPad Prism 3.03 software. The statistical differences between
the samples were evaluated by one-way analysis of variance, and the
Bonferroni test was used for multiple comparisons. P<0.05 was
considered to indicate a statistically significant difference. The
sample number (n) for each drug concentration varied between 2 and
6, and the results are presented as the mean ± standard error or
deviation of the mean as stated in each figure legend. To analyze
the effect of non-constant ratios of combinations of bortezomib and
etoposide, CalcuSyn software version 1.0 (Biosoft, Ferguson, MO,
USA) was used. The results of MTT assay were expressed as the
fraction of cells treated with bortezomib or etoposide alone or in
combination. The CI plot was then obtained, according to the
Chou-Talalay method (16).
Results
The growth of early prostate cancer cells requires
5α-dihydrotestosterone; therefore, these cells are described as
androgen-dependent (1). Since
prostate cancer cells require androgen for growth, patients with
prostate cancer are usually treated with hormonal intervention
(1). However, following a period of
remission, the prostate cancer recurs, and these cancer cells then
become androgen-independent (1). In
order to develop novel treatment strategies for prostate cancer,
the present study tested the effect of the highly specific 26S
proteasome inhibitor bortezomib, either alone or in combination
with irinotecan or etoposide, on PC-3 prostate cancer cells. As
represented in Fig. 1A, PC-3 prostate
cancer cells were observed to be highly sensitive to bortezomib
following 24 h of treatment with this drug, whose IC50
value was determined to be 53.9 nM. The effects of irinotecan and
etoposide on PC-3 cells were also examined following a period of
24-h incubation. The IC50 values of irinotecan and
etoposide were calculated to be 2.1 and 26.5 µM, respectively
(Fig. 1B).
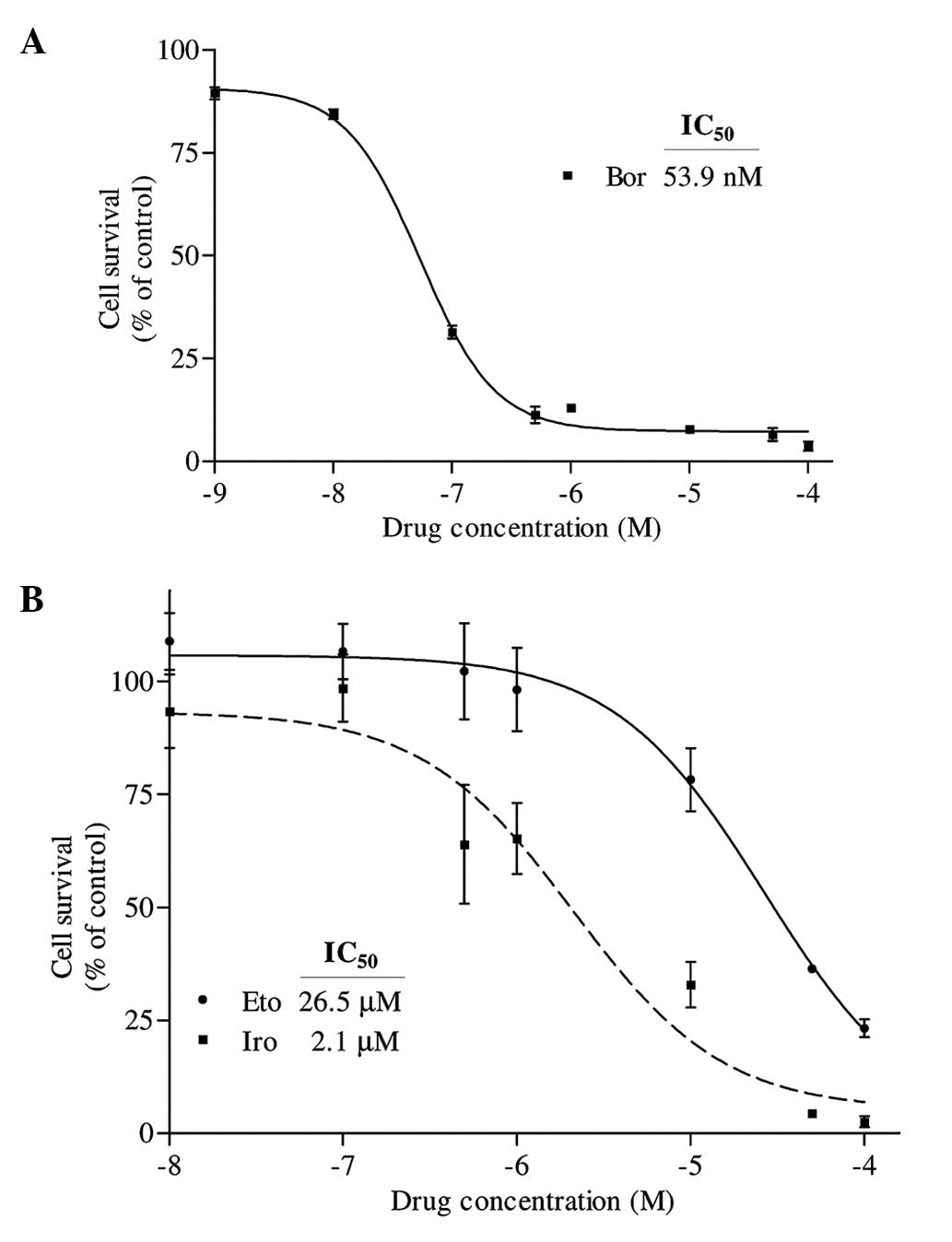 | Figure 1.(A) Determination of the
IC50 value of Bor on PC-3 cells. Cells were seeded in
96-well enzyme-linked immunosorbent assay plates and treated with 1
nM, 10 nM, 100 nM, 500 nM, 1 µM, 10 µM, 50 µM and 100 µM Bor for 24
h. Following treatment, the percentage of cell survival was
determined by water-soluble tetrazolium-1 assay. The
IC50 value of Bor was determined by fitting a sigmoidal
dose-response curve to the data, using the GraphPad Prism 3.03
program. (B) Determination of the IC50 values of Iro and
Eto on PC-3 cells. Cells were treated with various concentrations
of Eto and Iro (100 nM, 500 nM, 1 µM, 10 µM, 50 µM and 100 µM) for
24 h, and the IC50 values were similarly determined by
analyzing the data with GraphPad Prism 3.03. The X axes in each
graph is presented as log10 values, and the data are plotted as the
mean ± standard error. IC50, half maximal inhibitory
concentration; Bor, bortezomib; Iro, irinotecan; Eto,
etoposide. |
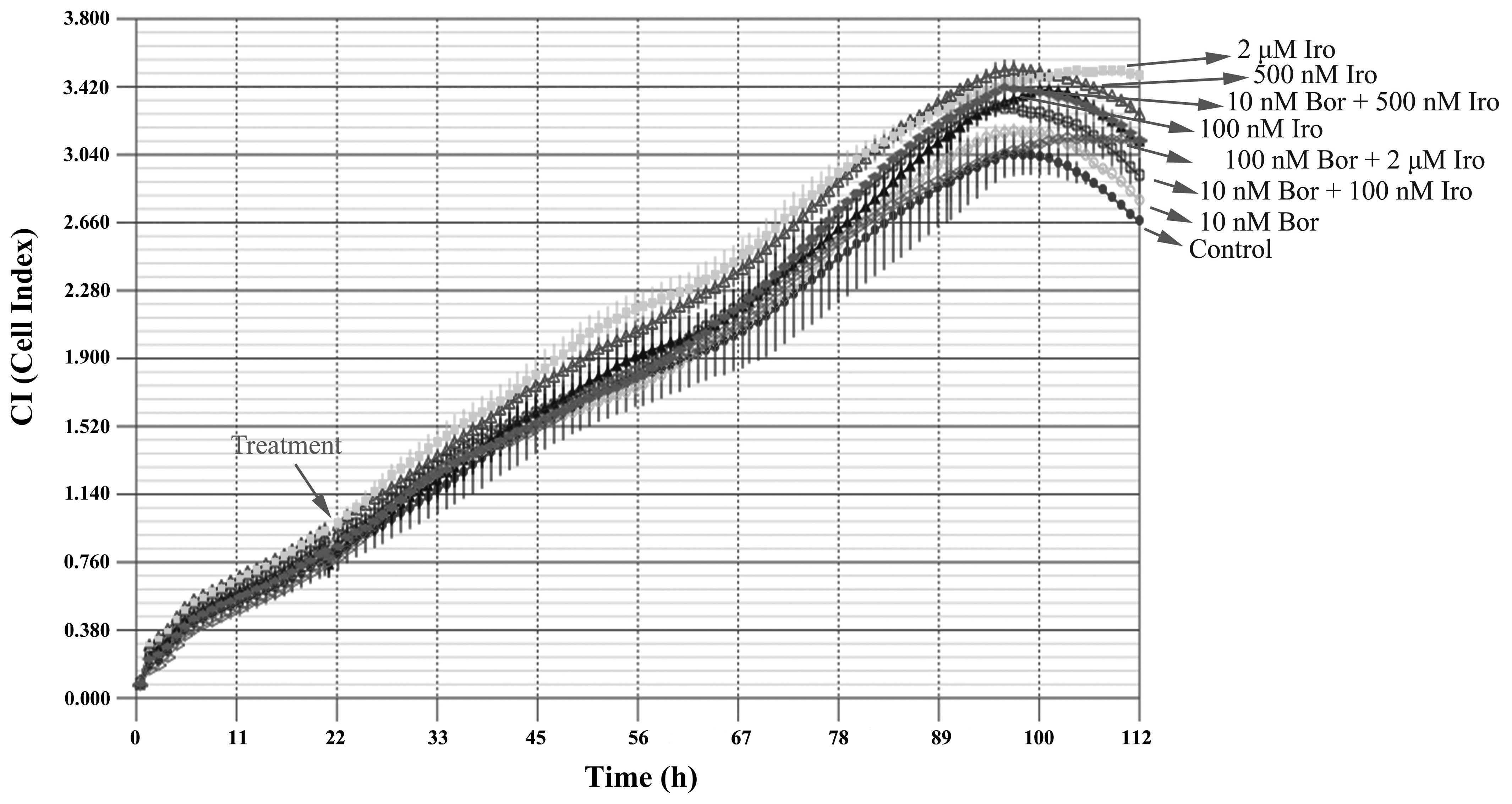
The combined effect of bortezomib and irinotecan on
the PC-3 cell line was then examined with the iCELLigence system,
an impedance-based system used for label-free and real-time
monitoring of cytotoxicity. Based on the IC50 values
obtained, the cells were treated with 10 nM bortezomib, 100 nM
irinotecan, 500 nM irinotecan or 2 µM irinotecan, alone or in
combination. Contrary to what it was expected, none of the above
treatments caused significant cytotoxicity, according to the
results of iCELLigence system analysis (Fig. 2). This phenomenon was hypothesized to
be partly due to the enlargement of cell morphology following
treatment with the aforementioned drugs as determined by
visualization of E-plates under an inverted microscope (AE21; Motic
Europe, Barcelona, Spain) (data not shown), which may consequently
cause an increase in impedance despite the low cell number.
To confirm whether the bortezomib+irinotecan
combination is more cytotoxic than the corresponding monotherapies,
PC-3 cells were treated with the same concentrations used for of
iCELLigence system, and tested by MTT assay. The iCELLigence data
revealed that the doubling time of PC-3 prostate cancer cells under
the experimental conditions used (cells were seeded at a density of
12,500 cells/E-Plate L8 well) was 44.8±3.8 h. Therefore, cells were
treated for 48 h with the aforementioned concentrations of drugs.
As indicated in Fig. 3, only 2 µM
irinotecan induced significant cytotoxicity as compared to the
control (P<0.05). However, 10 nM bortezomib was combined with
three different concentrations of irinotecan (100 nM, 500 nM and 2
µM), and resulted in no significant enhancement of cytotoxicity
compared with each treatment alone (Fig.
3), which is in agreement with the data obtained using the
iCELLigence system.
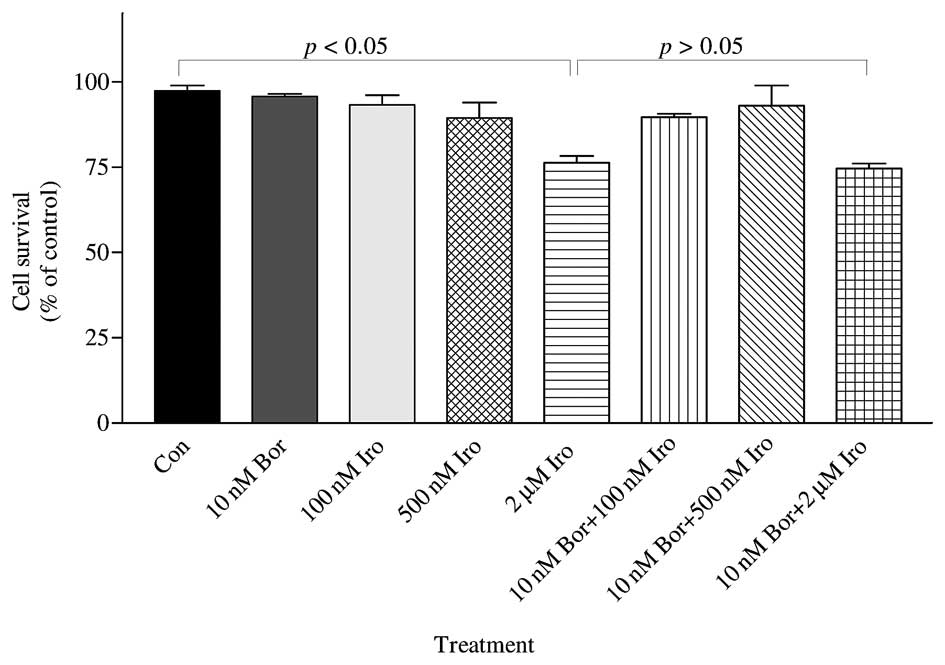 | Figure 3.The combined effect of Bor and Iro on
the survival of PC-3 cells was determined by MTT assay. PC-3 cells
(50,000) were seeded in 35×10-mm plates and treated with 10 nM Bor,
100 nM Iro, 500 nM Iro and 2 µM Iro, alone or in combination (10 nM
Bor+100 nM Iro; 10 nM Bor+500 nM Iro; and 10 nM Bor+2 µM Iro), for
48 h. The number of surviving cells was determined by MTT assay.
The results are presented as the mean ± standard error of the mean
(n=3). 2 µM irinotecan caused significant cytotoxicity as compared
to the control (P<0.05); however, 2 µM irinotecan did not cause
significant cytotoxicity compared with 100 nM irinotecan, 500 nM
irinotecan or 10 nM bortezomib treatment. By contrast, 10 nM
bortezomib + 2 µM irinotecan was significantly different compared
with the control, but this combination was not significantly
different than each drug treatment alone (100 nM irinotecan, 500 nM
irinotecan or 10 nM bortezomib)(P>0.05). Con, control; Bor,
bortezomib; Iro, irinotecan; MTT,
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide. |
The effects of two concentrations of bortezomib in
combination with two concentrations of etoposide were also
investigated by MTT assay. Cells were treated for 48 h with doses
selected based on the aforementioned IC50 data. As shown
in Fig. 4A, the effect of 40 nM
bortezomib in combination with 5 µM etoposide was significantly
different to that of 10 nM bortezomib alone (P<0.001) and 5 µM
etoposide alone (P<0.001). Similarly, the effects of combining
40 nM bortezomib with 20 µM etoposide differed significantly,
compared with 40 nM bortezomib-alone treated cells (P<0.001) and
20 µM etoposide-alone treated cells (P<0.001). In addition, the
effect of 40 nM bortezomib+20 µM etoposide combination was
significantly different, compared with that of 40 nM bortezomib+5
µM etoposide treatment (P<0.05). Analysis of DNA fragmentation
also corroborated the results of the MTT assay. As shown in
Fig. 4B and C, the 40 nM bortezomib+5
µM etoposide combination resulted in a ~2.8-fold increase in DNA
fragmentation (evaluated by the level of smearing), compared with
40 nM bortezomib-treated cells, and in a 1.8-fold increase,
compared with 5 µM etoposide-treated cells. Similarly, the 40 nM
bortezomib+20 µM etoposide combination increased DNA fragmentation
by ~2.9 fold, compared with 40 nM bortezomib-treated cells, and by
~1.4 fold, compared with 20 µM etoposide-treated cells.
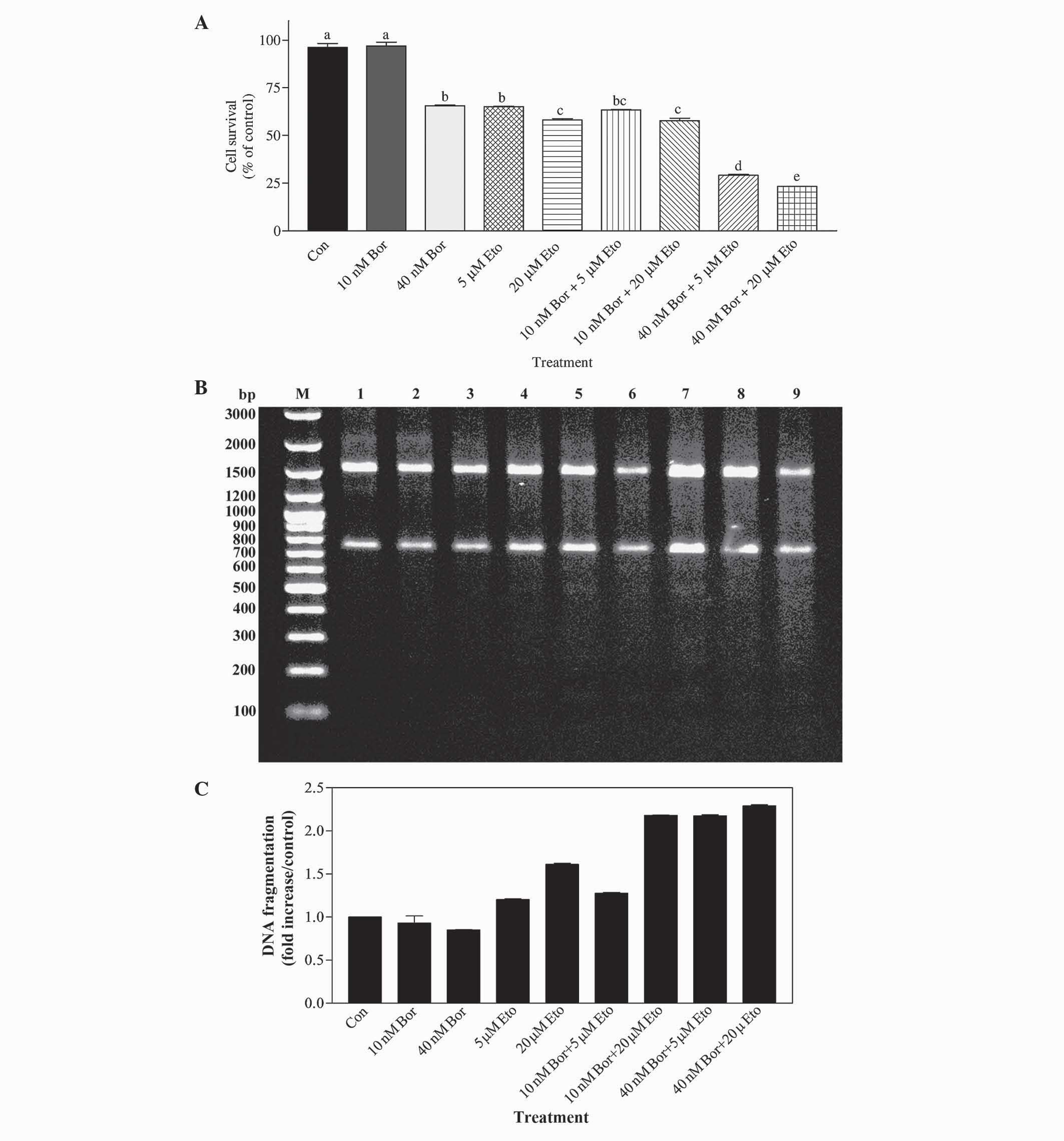 | Figure 4.(A) Determination of the effect of Bor
and Eto by MTT assay. PC-3 cells were treated with 10 nM Bor, 40 nM
Bor, 5 µM Eto and 20 µM Eto, alone or in combination (10 nM Bor+5
µM Eto; 10 nM Bor+20 µM Eto; 40 nM Bor+5 µM Eto; and 40 nM Bor+20
µM Eto). The number of surviving cells was determined by MTT assay.
The data are presented as the mean ± standard error of the mean
(n=3). Means with the same letter are not significantly different
from each other (P>0.05); means with different letters are
significant different (P<0.05). (B) Determination of the
combined effect of Bor and Eto treatment by DNA fragmentation
analysis. PC-3 cells were treated with 10 nM Bor, 40 nM Bor, 5 µM
Eto and 20 µM Eto, alone or in combination. DNA fragmentation was
determined with the Roche Apoptotic DNA-Ladder kit, according to
the manufacturer's protocol, using the 100 bp DNA ladder as a
standard. M, marker; lane 1, control; lane 2, 10 nM Bor; lane 3, 40
nM Bor; lane 4, 5 µM Eto; lane 5, 20 µM Eto; lane 6, 10 nM Bor+5 µM
Eto; lane 7, 10 nM Bor+20 µM Eto; lane 8, 40 nM Bor+5 µM Eto; and
lane 9, 40 nM Bor+20 µM Eto. (C) Quantification of the DNA smearing
observed in panel B. Quantification was performed using the
GelQuant.NET program. Con, control; Bor, bortezomib;
Eto, etoposide; MTT,
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; bp,
base pairs. |
In order to determine whether the effect of the
bortezomib+etoposide combination was synergistic or additive, the
CI values were calculated according to the Chou-Talalay method,
using the data presented in Fig. 4A.
The findings indicated that treatment with the 40 nM bortezomib+5
µM etoposide combination resulted in a CI value of 0.46, and 40 nM
bortezomib+20 µM etoposide combination resulted in a CI value of
0.39, indicating that these combinations cause synergistic effects
on PC-3 cells.
Discussion
Bortezomib, the first proteasome inhibitor approved
by the Food and Drug Administration for the treatment of multiple
myeloma, induces apoptotic cell death in androgen-dependent and
androgen-independent prostate cancer cell lines (17). In order to develop an alternative and
novel treatment strategy for androgen-independent prostate cancer,
the PC-3 cell line was treated with bortezomib, alone and in
combination with irinotecan or etoposide, in the present study. The
IC50 value of bortezomib in PC-3 cells was determined to
be 53.9 nM following 24 h of treatment, which indicates that
bortezomib is highly cytotoxic in this prostate cancer cell line.
This result is in agreement with a number of previous studies
performed on prostate cancer cells. For instance, Kiliccioglu et
al (18) determined the
IC50 value of bortezomib to be 30 nM, following 24-h
treatment in LNCaP cells (bearing wild-type p53), and 50 nM in PC-3
cells (which are p53-deficient). By contrast, Williams et al
(19) indicated that the
IC50 value of bortezomib was ~10 nM following 48 h of
treatment in PC-3 cells. In another study, Sato et al
(20) estimated the IC50
value of bortezomib in the PC-3 cell line to be 23 nM. Overall,
these results suggest that prostate cancer cells are highly
sensitive to bortezomib and that bortezomib is effective in
p53-wild-type or p53-deficient prostate cancer cells.
In the present study, the IC50 values of
etoposide (26.5 µM) and irinotecan (2.1 µM) in PC-3 cells were
determined, and the effects of these drugs in combination with
bortezomib were subsequently tested. The effect of irinotecan in
combination with bortezomib was tested in the iCELLigence system;
however, no significant cytotoxicity was observed in this real-time
cell analysis system for ≤112 h of incubation. To the best of our
knowledge, the combination of bortezomib+irinotecan has not been
previously tested on prostate cancer cells. In one of the rare
studies performed on other types of cancer, Ocean et al
(21) investigated the effect of
bortezomib+irinotecan combination in patients with adenocarcinoma
of gastroesophageal junction (GEJ) or stomach. However, bortezomib
alone or in combination with irinotecan was not effective for the
treatment of advanced adenocarcinoma of the GEJ or stomach
(21). Similarly, in advanced
colorectal carcinoma (CRC), bortezomib alone or in combination with
irinotecan was ineffective in patients with relapsed or refractory
CRC (22).
The present study also examined the effect of the
bortezomib+etoposide combination on PC-3 cells. Different doses of
bortezomib (10 nM and 40 nM) were tested, either alone or in
combination with various doses of etoposide (5 µM and 20 µM).
Bortezomib at a dose close to its IC50 value was
observed to be more cytotoxic on PC-3 cells when in combination
with 5 µM or 20 µM etoposide. To the best of our knowledge, this
bortezomib+etoposide combination has not been previously
investigated on prostate cancer cells. However, the combination of
bortezomib plus etoposide has been studied in children with
relapsed, refractory or secondary acute myeloid leukemia (AML)
(23). In those studies, bortezomib
was reported to be tolerable in combination with chemotherapy
regimens for relapsed pediatric AML, but the regimens (1.3
mg/m2 bortezomib and 100 mg/m2 etoposide) did
not exceed the preset minimum response criteria to allow continued
accrual (23). The dose of bortezomib
combined with other chemotherapeutic drugs is hypothesized to be
critical for obtaining effective treatment modalities (12), and the present results imply that
doses close to the IC50 values of each inhibitor
(bortezomib and etoposide) are crucial for exerting synergistic
effects. Although both irinotecan and etoposide interfere with DNA
replication through destabilizing DNA structure and causing DNA
strand breaks (24,25), only etoposide was observed to cause
synergistic effects when in combination with 40 nM bortezomib in
the present study. Since various doses of irinotecan in combination
with 40 nM bortezomib were not tested in the present study, it is
possible to hypothesize that irinotecan may also exert synergistic
effects when in combination with 40 nM bortezomib. The present
authors are currently designing experiments to confirm the present
results and to determine the combined effect of
irinotecan+bortezomib.
In conclusion, the results of the present study
suggest that targeting various intracellular pathways with two
structurally and functionally different inhibitors (bortezomib,
which inhibits the proteasome, and etoposide, which blocks the DNA
topoisomerase activity) generated a more potent effect, compared
with monotherapies. Therefore, the use of bortezomib, either alone
or in combination with etoposide, requires additional studies in a
clinical setting, and may be an alternative strategy for the
treatment of androgen-independent prostate cancer.
References
|
1
|
Russell PJ and Kingsley EA: Human prostate
cancer cell lines. Methods Mol Med. 81:21–39. 2003.PubMed/NCBI
|
|
2
|
Whang PG, Gamradt SC, Gates JJ and
Lieberman JR: Effects of the proteasome inhibitor bortezomib on
osteolytic human prostate cancer cell metastases. Prostate Cancer
Prostatic Dis. 8:327–334. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Potosky AL, Miller BA, Albertsen PC and
Kramer BS: The role of increasing detection in the rising incidence
of prostate cancer. JAMA. 273:548–552. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Hanks GE, Krall JM, Hanlon AL, Asbell SO,
Pilepich MV and Owen JB: Patterns of Care and RTOG studies in
prostate cancer: Long-term survival, hazard rate observations, and
possibilities of cure. Int J Radiat Oncol Biol Phys. 28:39–45.
1994. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zincke H, Oesterling JE, Blute ML,
Bergstralh EJ, Myers RP and Barrett DM: Long-term (15 years)
results after radical prostatectomy for clinically localized (stage
T2c or lower) prostate cancer. J Urol. 152:1850–1857.
1994.PubMed/NCBI
|
|
6
|
Crawford ED, Eisenberger MA, McLeod DG,
Spaulding JT, Benson R, Dorr FA, Blumenstein BA, Davis MA and
Goodman PJ: A controlled trial of leuprolide with and without
flutamide in prostatic carcinoma. N Engl J Med. 321:419–424. 1989.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Eisenberger MA, Crawford ED, Wolf M,
Blumenstein B, McLeod DG, Benson R, Dorr FA, Benson M and Spaulding
JT: Prognostic factors in stage D2 prostate cancer; important
implications for future trials: Results of a cooperative intergroup
study (INT.0036). The National Cancer Institute Intergroup Study
#0036. Semin Oncol. 21:613–619. 1994.PubMed/NCBI
|
|
8
|
Newling DW, Denis L and Vermeylen K:
Orchiectomy versus goserelin and flutamide in the treatment of
newly diagnosed metastatic prostate cancer. Analysis of the
criteria of evaluation used in the European Organization for
Research and Treatment of Cancer - Genitourinary Group Study 30853.
Cancer. 72(Suppl 12): 3793–3798. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Bracarda S, Logothetis C, Sternberg CN and
Oudard S: Current and emerging treatment modalities for metastatic
castration- resistant prostate cancer. BJU Int. 107(Suppl 2):
13–20. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Sartor O, Halstead M and Katz L: Improving
outcomes with recent advances in chemotherapy for
castrate-resistant prostate cancer. Clin Genitourin Cancer.
8:23–28. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Papandreou CN and Logothetis CJ:
Bortezomib as a potential treatment for prostate cancer. Cancer
Res. 64:5036–5043. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Yerlikaya A and Yöntem M: The significance
of ubiquitin proteasome pathway in cancer development. Recent
Patents Anticancer Drug Discov. 8:298–309. 2013. View Article : Google Scholar
|
|
13
|
Yerlikaya A and Erin N: Differential
sensitivity of breast cancer and melanoma cells to proteasome
inhibitor Velcade. Int J Mol Med. 22:817–823. 2008.PubMed/NCBI
|
|
14
|
Yerlikaya A, Okur E and Ulukaya E: The
p53-independent induction of apoptosis in breast cancer cells in
response to proteasome inhibitor bortezomib. Tumour Biol.
33:1385–1392. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Freshney RI: Cytotoxicity. Culture of
Animal Cells: A Manual of Basic Technique (5th). John Wiley and
Sons Inc. (Hoboken, NJ). 3592005.
|
|
16
|
Pham LV, Tamayo AT, Li C, Bornmann W,
Priebe W and Ford RJ: Degrasyn potentiates the antitumor effects of
bortezomib in mantle cell lymphoma cells in vitro and in vivo:
Therapeutic implications. Mol Cancer Ther. 9:2026–2036. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Adams J, Palombella VJ, Sausville EA,
Johnson J, Destree A, Lazarus DD, Maas J, Pien CS, Prakash S and
Elliott PJ: Proteasome inhibitors: A novel class of potent and
effective antitumor agents. Cancer Res. 59:2615–2622.
1999.PubMed/NCBI
|
|
18
|
Kiliccioglu I, Konac E, Varol N, Gurocak S
and Bilen Yucel C: Apoptotic effects of proteasome and histone
deacetylase inhibitors in prostate cancer cell lines. Genet Mol
Res. 13:3721–3731. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Williams S, Pettaway C, Song R, Papandreou
C, Logothetis C and McConkey DJ: Differential effects of the
proteasome inhibitor bortezomib on apoptosis and angiogenesis in
human prostate tumor xenografts. Mol Cancer Ther. 2:835–843.
2003.PubMed/NCBI
|
|
20
|
Sato A and Asano T, Ito K and Asano T:
Vorinostat and bortezomib synergistically cause ubiquitinated
protein accumulation in prostate cancer cells. J Urol.
188:2410–2418. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ocean AJ, Christos P, Sparano JA, Shah MA,
Yantiss RK, Cheng J, Lin J, Papetti M, Matulich D, Schnoll-Sussman
F, et al: Phase II trial of bortezomib alone or in combination with
irinotecan in patients with adenocarcinoma of the gastroesophageal
junction or stomach. Invest New Drugs. 32:542–548. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kozuch PS, Rocha-Lima CM, Dragovich T,
Hochster H, O'Neil BH, Atiq OT, Pipas JM, Ryan DP and Lenz HJ:
Bortezomib with or without irinotecan in relapsed or refractory
colorectal cancer: Results from a randomized phase II study. J Clin
Oncol. 26:2320–2326. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Horton TM, Perentesis JP, Gamis AS, Alonzo
TA, Gerbing RB, Ballard J, Adlard K, Howard DS, Smith FO, Jenkins
G, et al: A Phase 2 study of bortezomib combined with either
idarubicin/cytarabine or cytarabine/etoposidee in children with
relapsed, refractory or secondary acute myeloid leukemia: A report
from the Children's Oncology Group. Pediatr Blood Cancer.
61:1754–1760. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Petitprez A, Poindessous V, Ouaret D,
Regairaz M, Bastian G, Guérin E, Escargueil AE and Larsen A:
Acquired irinotecan resistance is accompanied by stable
modifications of cell cycle dynamics independent of MSI status. Int
J Oncol. 42:1644–1653. 2013.PubMed/NCBI
|
|
25
|
Groh T, Hrabeta J, Khalil MA, Doktorova H,
Eckschlager T and Stiborova M: The synergistic effects of
DNA-damaging drugs cisplatin and etoposide with a histone
deacetylase inhibitor valproate in high-risk neuroblastoma cells.
Int J Oncol. 47:343–352. 2015.PubMed/NCBI
|