Introduction
Uterine cervical cancer (UCC) is the most commonly
diagnosed cancer, and a leading cause of cancer-associated
mortality for women in developing countries, including China
(1). Despite improvements in
diagnostics and therapeutic strategies, the prognosis of UCC
remains poor (2). The World Health
Organization estimates that the prevalence of UCC annually is
~530,000 cases worldwide, and ~50% of patients succumb to the
disease (1). The pathophysiology of
UCC is complex because of multiple risk factors including viral
infection, premature childbirth, inflammation and high estrogen
levels (3). Therefore, an improved
understanding of the initiation and progression of UCC is required
for a timely diagnosis, prevention and therapeutic intervention
(4).
Interleukins (ILs) are produced by many types of
cells and collectively they cause a variety of biological effects,
which are exerted on the immune response and include maturation,
activation and proliferation of immune cells (5). Previous studies have demonstrated that
ILs also contribute an important function in tumor development and
metastasis via autocrine and/or paracrine signaling (6,7).
Interleukin-8 [IL-8 or CXC chemokine ligand 8 (CXCL8)], a cytokine
family member, has previously been demonstrated to contribute to
chronic inflammation and cancer development via several signaling
transduction pathways (8,9). IL-8 derived from cancer cells is able to
promote the capacity of cellular proliferation, adhesion,
migration, invasion, chemoresistance and angiogenesis by binding to
its corresponding receptor in several types of carcinoma,
indicating that IL-8 may serve as a therapeutic target for
malignant tumors (10,11). However, the precise function of IL-8
and its clinical significance in malignancy remain unclear.
In the present study, levels of IL-8 and its
receptors IL-8 receptor A (IL-8RA) and IL-8 receptor B (IL-8RB) in
HeLa cells were investigated, and the differences in the expression
levels of IL-8 in normal uterine cervical tissues and UCC tissues
were compared. The effect of exogenous IL-8 on the proliferation
and migration in HeLa cells, and the exact mechanism by which IL-8
enhanced the oncogenic potential of UCC was also investigated. The
aim of the present study was to clarify the function of IL-8 and
its receptors in UCC, and therefore provide a novel biomarker for
the diagnosis and treatment of UCC.
Materials and methods
Cell culture
The human cervical carcinoma immortalized cell line
HeLa was obtained from the American Type Culture Collection
(Manassas, VA, USA). Cells were maintained in RPMI-1640 medium
(Gibco; Thermo Fisher Scientific, Inc., Waltham, MA, USA) and
supplemented with 10% fetal bovine serum (FBS; Gibco; Thermo Fisher
Scientific, Inc.) and penicillin-streptomycin (100 units/ml) under
standard culture conditions (12).
mRNA expression of genes in HeLa cells
using the reverse transcription-polymerase chain reaction
(RT-PCR)
Following isolation of total RNA from HeLa cells
using TRIzol reagent (Invitrogen; Thermo Fisher Scientific, Inc.),
cDNA was prepared using 2 µg total RNA reverse-transcribed using a
Quant Reverse Transcriptase kit (Tiangen Biotech Co., Ltd.,
Beijing, China), according to the manufacturer's protocol. PCR
primer sequences are presented in Table
I, and annealing and thermocycling conditions are presented in
Table II. The denaturation for each
gene was performed at 94°C for 1 min, and extension was performed
at 72°C for 1 min. Relative mRNA levels were calculated based on
the ratios of detected gene/β-actin using Quantity One®
Software (version 4.2; Bio-Rad Laboratories, Inc., Hercules, CA,
USA).
 | Table I.Primer sequences for genes used in
reverse transcription-polymerase chain reaction experiments. |
Table I.
Primer sequences for genes used in
reverse transcription-polymerase chain reaction experiments.
| Gene | Forward primer | Reverse primer |
|---|
| β-actin |
5′-AGAAAATCTGGCACCACACC-3′ |
5′-CTCCTTAATGTCACGCACGA-3′ |
| IL-8 |
5′-ACATACTCCAAACCTTTCCACC-3′ |
5′-AAAACTTCTCCACAACCCTCTG-3′ |
| IL-8RA |
5′-GACCTACTCTTTGCCCTGAC-3′ |
5′-AACACCATCCGCCATTTT-3′ |
| IL-8RB |
5′-GGAAACTCCCTCGTGATG-3′ |
5′-CAGGAATGTGCCAAAAAT-3′ |
| ERK |
5′-CTTCTCGCCTCAGTTCGC-3′ |
5′-CTCCTGGATGCTTGTCTGGTAA-3′ |
| NUMB |
5′-AAACGCCAACTATCCCTACGC-3′ |
5′-AGCACCAGAAGATTGACCC-3′ |
 | Table II.Specific thermocycling conditions
used for reverse transcription-polymerase chain reactions. |
Table II.
Specific thermocycling conditions
used for reverse transcription-polymerase chain reactions.
| Gene | Annealing
temperature, °C | Number of
cycles |
|---|
| β-actin | 54 | 25 |
| IL-8 | 57 | 38 |
| IL-8RA | 55 | 33 |
| IL-8RB | 58 | 33 |
| ERK | 55.6 | 30 |
| NUMB | 59 | 35 |
Protein expression of IL-8 receptors
in HeLa cells using western blot analysis
Total protein extracts from HeLa cells were prepared
with a cell lysis buffer [50 mM Tris-HCl (pH 7.5), 150 mM NaCl, 1
mM sodium-EDTA, 1 mM EDTA, 1% (v/v) TritonX-100, 2.5 mM sodium
pyrophosphate, 1 mM β-glycerophosphate, 1 mM Na3VO4, 1 mM NaF, 1
µg/ml leupeptin and 1 mM PMSF] described previously (13). Following quantification of protein
concentration using the BCA assay kit (Wuhan Boster Biological
Technology, Ltd., Wuhan, China), 40 µg of total proteins per lane
were resolved by 12% SDS-PAGE and transferred onto a polyvinylidene
fluoride membrane (EMD Millipore, Billerica, MA, USA). Following
blocking with commercial blocking buffer (Wuhan Boster Biological
Technology, Ltd.) at 37°C for 1 h, the membranes were probed with
rabbit anti-human IL-8RA/B monoclonal antibody (cat. no. sc-30008;
dilution, 1:500; Santa Cruz Biotechnology, Inc., Dallas, TX, USA)
or mouse anti-β-actin monoclonal antibody (cat. no. sc-130300;
dilution, 1:3,000; Santa Cruz Biotechnology, Inc.) at 4°C
overnight. Following washing with TBST, the membranes were
incubated with goat anti-rabbit IgG (cat. no. BA1054; dilution,
1:1,000; Wuhan Boster Biological Technology, Ltd.) or goat
anti-mouse IgG (cat. no. BA1050; dilution, 1:1,000; Wuhan Boster
Biological Technology, Ltd.) at 37°C for 1 h. Signals were detected
using Western Bright ECL reagent (cat. no. 151030-16; Advansta,
Menlo Park, CA, USA) and Blotting System (Bio-Rad Laboratories,
Inc.). Band densities were subsequently analyzed using Quantity
One® Software (version, 4.2; Bio-Rad Laboratories,
Inc.).
Expression levels of IL-8 in a
cervical tissue microarray using immunohistochemistry
A tissue microarray (Alenabio, Xi'an, China)
containing 40 normal uterine cervical tissue samples and 40 UCC
tissue samples were performed to determine the expression of IL-8.
In the UCC group, all the cancer samples were confirmed as squamous
cell carcinoma. Expression levels of IL-8 in the tissue microarray
were investigated according to a protocol described previously
(14). Briefly, after deparaffinizing
and rehydrating, endogenous peroxidase in the microarray was
deactivated using 3% dilution hydrogen peroxide at room temperature
for 15 min. Antigen retrieval was performed by incubating the
microarray with 10 mM citrate buffer (pH 6.0; cat. no. AR0024;
Wuhan Boster Biological Technology, Ltd.) for 30 min at low power
microwave, and nonspecific binding sites were blocked with normal
rabbit-serum (cat. no. C1404; Shanghai Westang Bio-Tech Co., Ltd.)
at 37°C for 20 min. Subsequently, the tissue microarray was
incubated with mouse anti-IL-8 monoclonal antibody (1:50; Santa
Cruz Biotechnology, Inc.) at 4°C overnight, and was then treated
with a Streptavidin-Biotin Complex kit (cat. no. SA1050; Wuhan
Boster Biological Technology, Ltd.). To quantify the protein level,
the average optical density in a high-power field (magnification,
×400) was detected using BI-2000 Medicine Image Analysis Software
(Chengdu Taimeng Technology Co., Ltd., Chengdu, China). Strong and
weak immunostaining were defined, respectively, as average optical
values of ≥210 and ≤180.
Effect of exogenous IL-8 on the
proliferation of HeLa cells
HeLa cells were seeded in 96-well plates
(1.5×103 cells/well) and incubated in RPMI-1640 medium
supplemented with 10% FBS overnight at 37°C in a 5% CO2
incubator. Subsequently, cells were divided into five groups by
removing old culture medium and replacing it with different
conditioning medium containing 0.1% bovine serum albumin (Wuhan
Boster Biological Technology, Ltd.) and IL-8 (PeproTech, Inc.,
Rocky Hill, NJ, USA) at final concentrations of 0, 20, 40, 60 and
80 ng/ml, respectively. Following a 72-h incubation period, Cell
Counting kit-8 (CCK-8; Boster Biological Technology, Pleasanton,
CA, USA) was added to each well (10 µl per well). Following
incubation in a 5% CO2 incubator at 37°C for 1 h,
absorbance was detected at 450 nm using a microplate reader (BioTek
Instruments, Inc., Winooski, VT, USA).
Effects of exogenous IL-8 on migration
of HeLa cells
A Transwell migration assay was performed using a
24-well chamber (Corning Incorporated, Corning, NY, USA). Briefly,
HeLa cells in Transwell units were divided into three groups and
5×104 cells/well were plated onto the top surface of the
upper chamber containing 100 µl RPMI-1640 medium without FBS. In
the lower chamber, RPMI-1640 medium with 5% FBS and IL-8
(PeproTech, Inc.) at final concentrations of 0, 40 and 60 ng/ml for
the three groups were added, respectively. Following a 24 h
incubation period at 37°C in a 5% CO2 incubator,
non-migratory cells were removed with a cotton swab and filters
containing migratory cells were fixed and stained with 0.1% crystal
violet for 30 min at room temperature. Migratory cells were counted
in ≥5 randomly selected fields using brightfield microscopy
(magnification, ×100).
Statistical analysis
All data are expressed as the mean ± standard
deviation. Data were analyzed using one-way analysis of variance
and q-test. P<0.05 was considered to indicate a statistically
significant difference.
Results
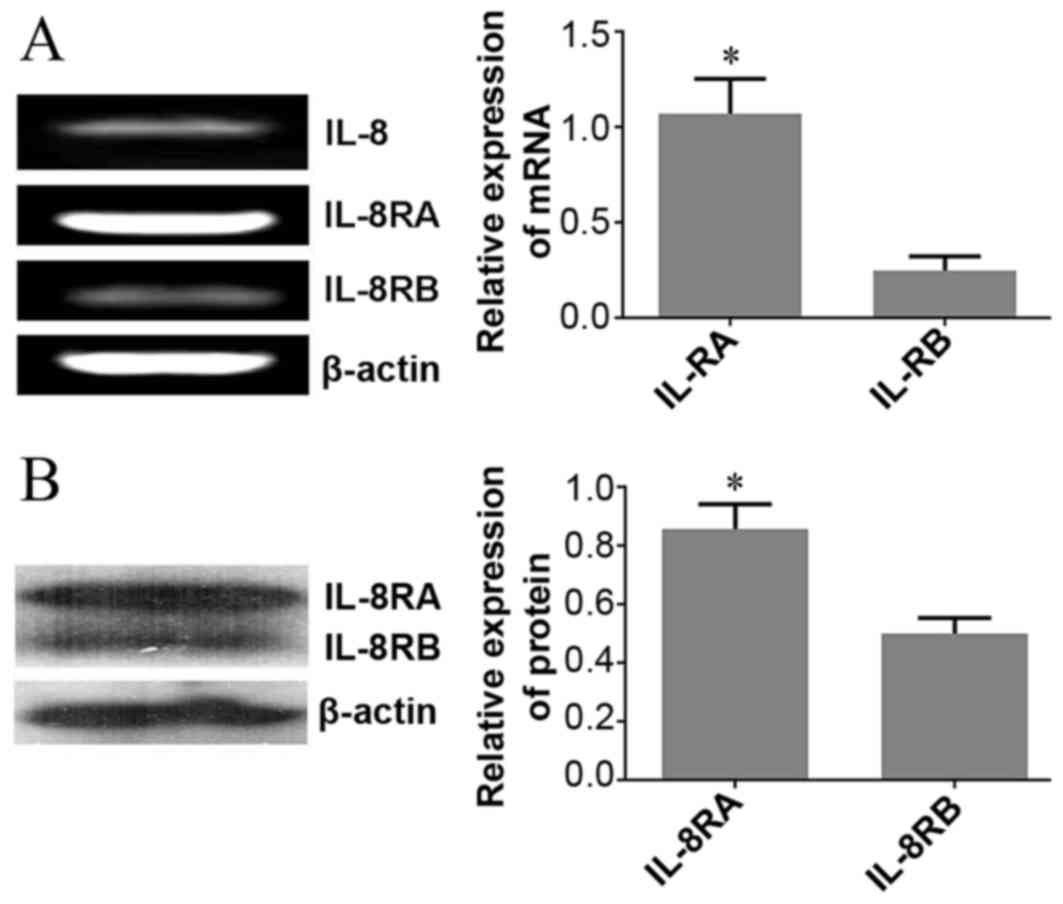
mRNA expression of IL-8 and its
receptors in HeLa cells determined using RT-PCR
To explore the potential function of IL-8 in HeLa
cells, the present study determined IL-8 mRNA expression and its
receptors using RT-PCR. Results demonstrated that IL-8 and its
receptors were expressed in HeLa cells, and IL-8RA mRNA levels were
significantly upregulated in HeLa cells compared with those of
IL-8RB (Fig. 1A).
Protein expression of IL-8 receptors
in HeLa cells determined using western blot analysis
On the basis of results obtained from RT-PCR,
western blot analysis was utilized to determine the expression
levels of IL-8 receptors (IL-8RA/RB) in HeLa cells. Consistent with
RT-PCR results, there was a statistically significant increase in
the protein expression of IL-8RA compared with that of IL-8RB
(Fig. 1B).
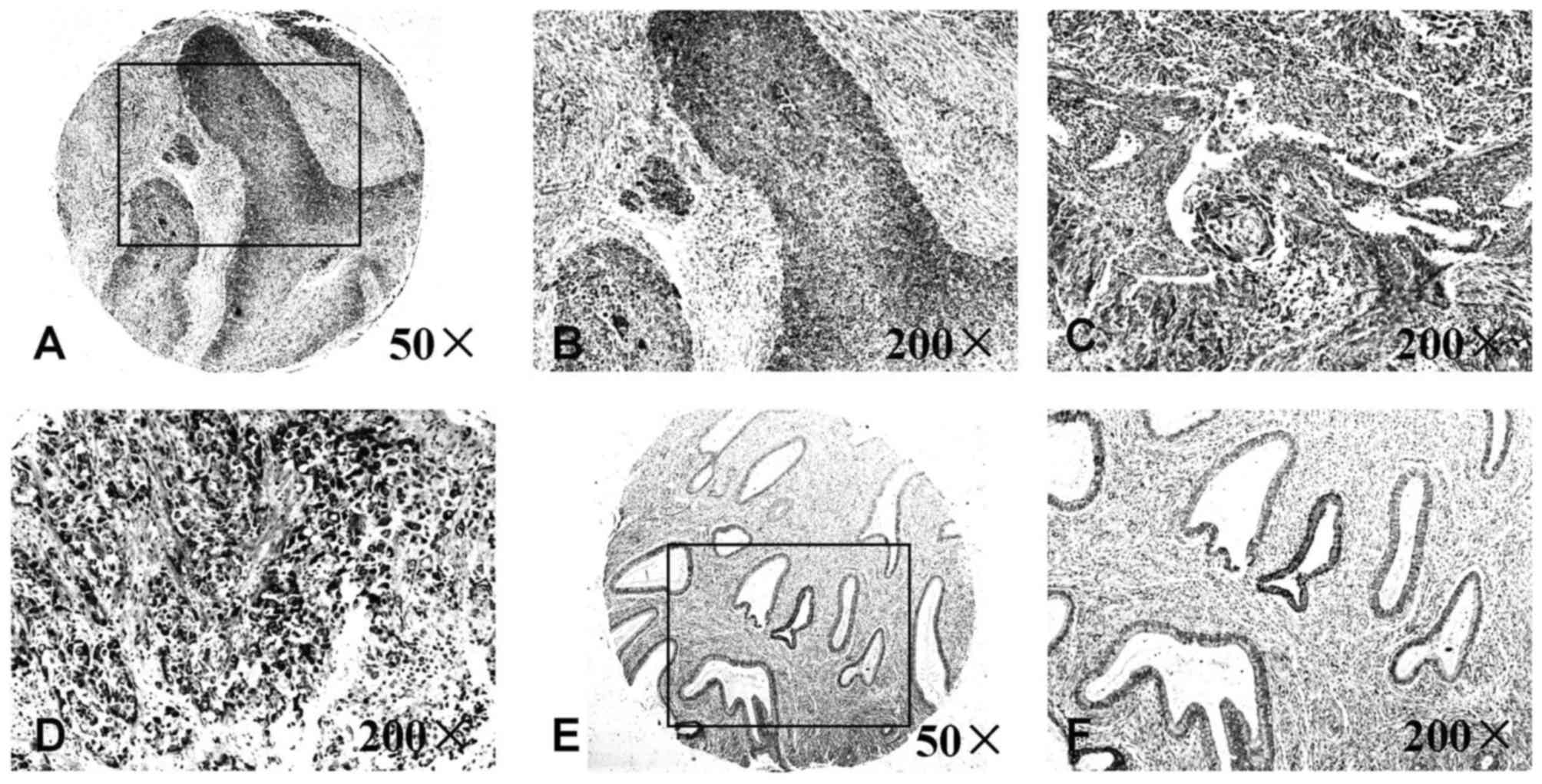
Expression levels of IL-8 determined
using a human uterine cervical tissue microarray
To further investigate the clinical significance of
IL-8 in UCC, a tissue microarray was used to compare the expression
of IL-8 in normal uterine cervical tissues and UCC tissues
(Fig. 2). Compared with the normal
uterine cervical tissues, the expression levels of IL-8 in UCC
tissues were increased in terms of average optical values. No
association between the increased expression of IL-8 and age or
clinical stage of UCC was identified using the assay (Table III).
 | Table III.Expression of IL-8 in a cervical
tissue microarray. |
Table III.
Expression of IL-8 in a cervical
tissue microarray.
| Clinical
parameter | Average optical
density | P-value |
|---|
| Cervical cancer
tissues | 202.60±7.51 | <0.01 |
| Normal cervical
tissues | 181.23±12.46 |
|
| TNM stage |
| >0.05 |
| I | 202.54±6.77 |
|
| II | 202.01±7.88 |
|
|
III | 203.14±7.39 |
|
| Age, years |
| >0.05 |
|
≤50 | 202.58±7.35 |
|
|
≥51 | 202.62±7.76 |
|
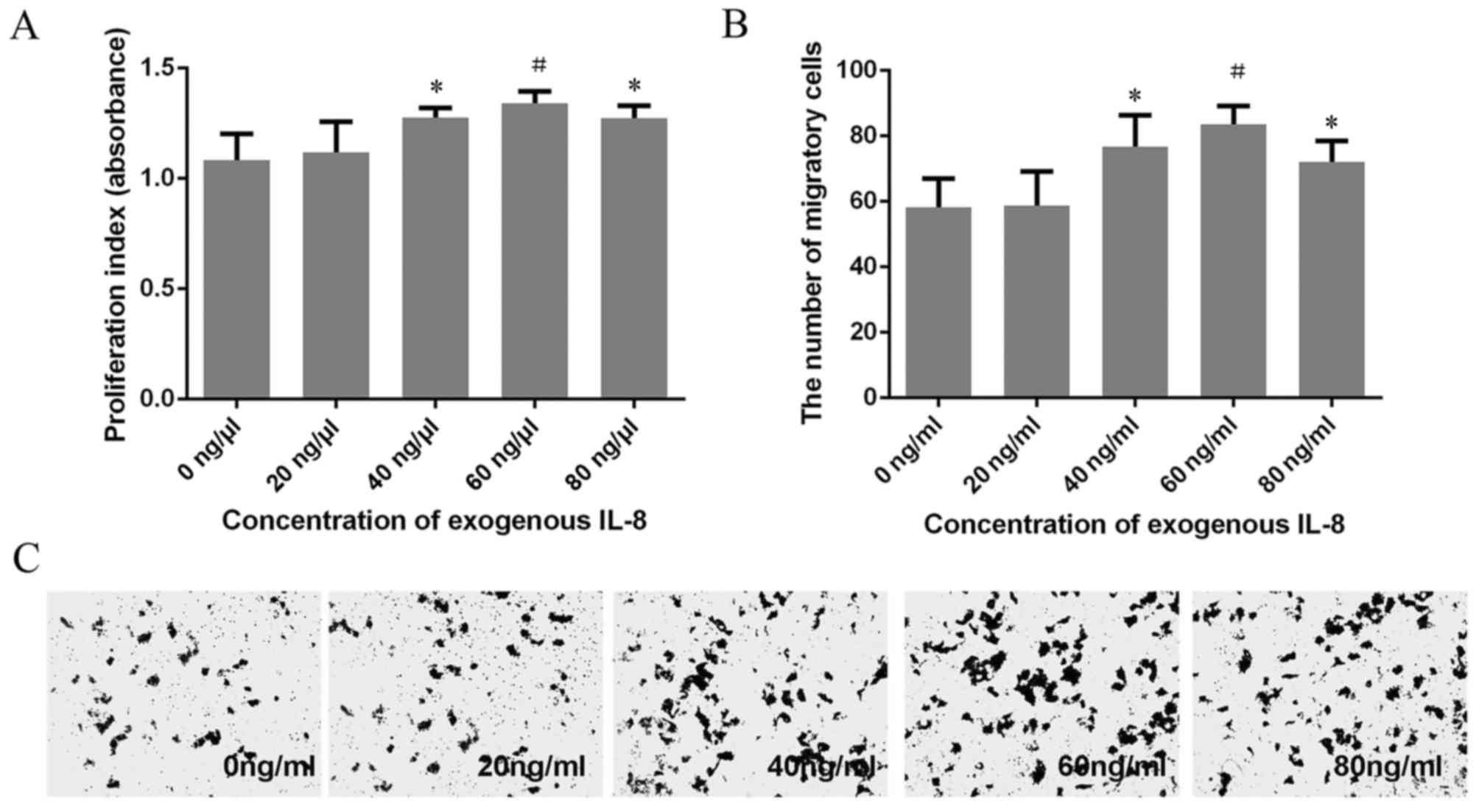
Effect of exogenous IL-8 on the
proliferation of HeLa cells determined using the CCK-8 assay
A CCK-8 assay was utilized to explore the
proliferation of HeLa cells treated with different concentrations
of exogenous IL-8. Results demonstrated that the proliferation of
HeLa cells were markedly enhanced as the concentration of IL-8
increased (40, 60 and 80 ng/ml). The absorbance reached a maximum
(1.341±0.056) when the concentration of IL-8 was 60 ng/ml. The
proliferation rate decreased in HeLa cells treated with 40 and 80
ng/ml exogenous IL-8, compared with HeLa cells treated with 60
ng/ml exogenous IL-8. However, no statistically significant
difference was identified (Fig.
3A).
Effect of exogenous IL-8 on migration
of HeLa cells determined using a Transwell assay
Analysis of migration indicated that HeLa cells
treated with 40, 60 and 80 ng/ml exogenous IL-8 migrated faster
compared with untreated HeLa cells. The migratory efficiency of
HeLa cells treated with 60 ng/ml exogenous IL-8 was the greatest;
however, no significant difference between HeLa cells treated with
40, 60 and 80 ng/ml exogenous IL-8 was identified (Fig. 3B and C). These results were in
agreement with the CCK-8 proliferation assay.
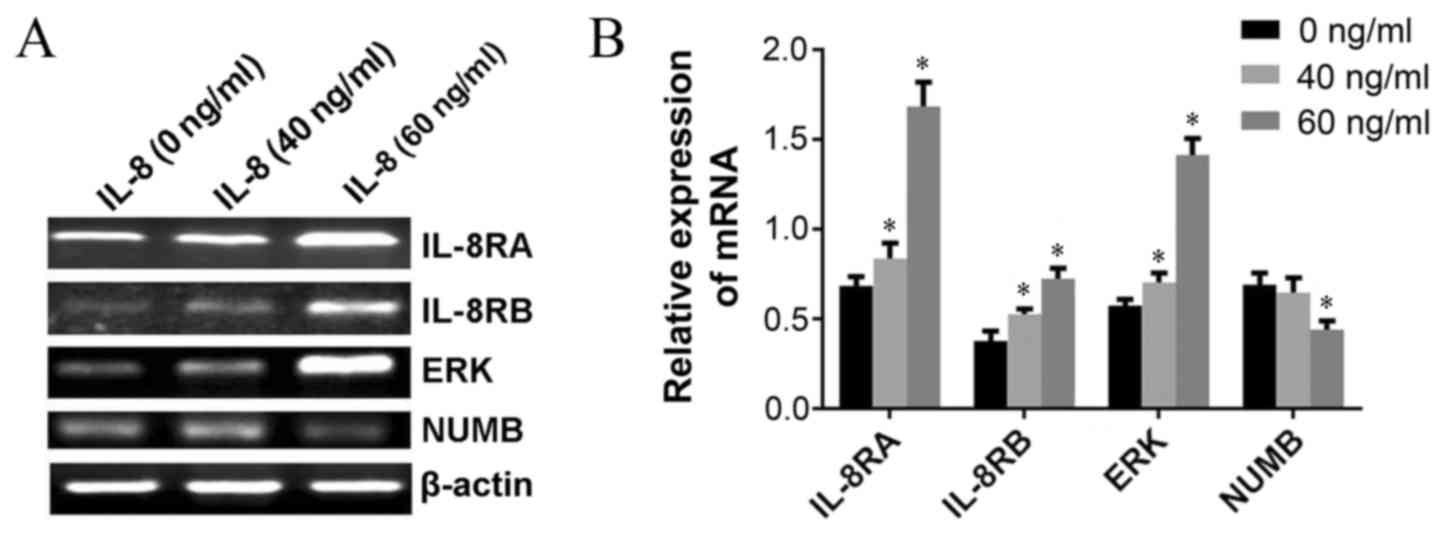
Exogenous IL-8 regulates the
expression of carcinogenesis-associated genes in HeLa cells
determined using RT-PCR
To elucidate the underlying molecular mechanism(s)
of IL-8-associated tumorigenesis in UCC, RT-PCR was performed in
order to investigate whether IL-8 regulated the expression of
carcinogenesis-associated genes in HeLa cells. Results demonstrated
that the expression of NUMB at the mRNA level were markedly
inhibited following administration of 60 ng/ml exogenous IL-8, and
the expression of IL-8RA, IL-8RB and extracellular-signal-regulated
kinases (ERKs) were upregulated when cells were treated with 40 and
60 ng/ml exogenous IL-8 compared with untreated cells (Fig. 4A and B).
Discussion
IL-8, also referred to as CXCL8, is a cytokine of
the CXC chemokine family that is associated with inflammatory and
immune responses, and is also an important biomarker for a range of
diseases including various types of cancer. In the present study,
the function of IL-8 in UCC was investigated and novel results
included: IL-8 and its receptors may be involved in the development
and progression of UCC, exogenous IL-8 promotes the carcinogenic
potential of HeLa cells by upregulating the expression of its
receptors IL-8RA, IL-8RB and ERK, and downregulating the expression
of NUMB.
IL-8 exerts its biological effects via
G-protein/adenylate cyclase and G-protein/phospholipase C signaling
pathways by binding with its receptors IL-8RA and IL-8RB (4,15). IL-8 RA
and IL-8RB exhibit marked affinity for IL-8, and the presence of
IL-8 and its receptors during tumor development and progression is
a relatively common occurrence (16).
IL-8RA has an increased specificity for IL-8 and a decreased
affinity for other cytokines, whereas IL-8RB is less specific for
IL-8 but is able to bind with other cytokines including CXCL3 and
CXCL5 (17). Therefore, this may be
the primary reason for the increase in the expression levels of
IL-8RA compared with IL-8RB in HeLa cells. The IL-8/IL-8 receptor
axis has been identified to be associated with various types of
human cancer including pilomatricoma and hepatoma (18–20).
However, the underlying molecular mechanisms by which IL-8 and its
receptors are involved in UCC remain unclear. In the present study,
results obtained from in vitro and in vivo
experiments demonstrated that IL-8 was expressed in HeLa cells and
UCC tissues, and the expression levels of IL-8 in UCC tissue were
significantly increased compared with those in normal uterine
cervical tissue; however, no association between overexpression of
IL-8 with patient age, stage or clinical classification of UCC was
identified in the present study. In addition, IL-8 receptors were
expressed at the mRNA and protein levels in HeLa cells. Therefore,
results demonstrated that IL-8 and its receptors may contribute to
the development of UCC. Previous reports demonstrated that the
overexpressed IL-8 and its receptors produced by tumor cells and
the tumor microenvironment promoted tumor development by
stimulating angiogenesis and increasing the capacity of cellular
adhesion, migration and proliferation, and causing chemotherapy
resistance (21–23). In the present study, a CCK-8 assay and
Transwell assay were performed to study the effects of different
concentrations of exogenous IL-8 on the malignant behavior of HeLa
cells. As HeLa cells were exposed to increasing concentrations of
IL-8, there was also an increase in the proliferation and
migration. In addition, increasing levels of IL-8RA and IL-8RB were
observed, and this further confirmed the carcinogenic potential of
the IL-8/IL-8 receptor axis in UCC.
The ERK signaling pathway transmits mitogenic
signals and regulates numerous cellular functions, including
proliferation and differentiation (24,25). In
order to adapt to internal and external environmental stimuli, the
ERK cascade must be strictly regulated in order to produce the
correct biological response (26,27). An
aberrant ERK cascade is commonly observed in human tumors, and is
accompanied by the deregulation of proliferation and malignant
transformation. Recent studies have identified that the ERK
signaling pathway inhibits the apoptosis of tumor cells by
influencing the mitochondrial apoptosis pathway, for example, the
ERK signaling pathway can exert its anti-apoptotic control by
non-activation of certain pro-apoptotic kinases, including Janus
kinase and p38 mitogen activated protein kinase. The increased ERK
activity is associated with invasion and metastasis in
cholangiocarcinoma and breast carcinoma (28,29).
However, conflicting results suggest that the activation of ERK may
exert diverse effects in a different microenvironment, for example,
ERK may be able to promote cell apoptosis by enhancing the
activities of the apoptotic proteins caspase-9 and caspase-3
(30,31). Clinically, therapeutic agents
targeting the ERK signaling pathway may also lead to the
development of drug resistance (11,32). The
results of the present study demonstrated that exogenous IL-8
increased proliferation and migration in HeLa cells, and
upregulated the expression of ERK simultaneously.
Furthermore, the results of the present study also
demonstrated a decrease in NUMB mRNA expression in HeLa cells
treated with exogenous IL-8. The endocytic adaptor protein NUMB, an
intrinsic regulator of cell fate determination, inhibits the Notch
signaling pathway and the p53/p21 axis, contributing to the control
of asymmetric cell division, maintaining stem cell compartments and
representing a mechanism of tumor suppression (33–35). NUMB
expression is frequently decreased in tumors, and NUMB dysfunction
or loss leads to differentiation defects and serves a crucial
function in mammary tumorigenesis (36,37). The
present study therefore determined that the downregulation of NUMB
expression by exogenous IL-8 was one explanation for the
improvement of migration and proliferation in HeLa cells.
In conclusion, the results of the present study
demonstrated that the underlying molecular mechanisms which
regulate the expression of ERK and NUMB are affected by exogenous
IL-8, and are associated with an increase in the migration and
proliferation of HeLa cells. These results demonstrate that
inhibition of the IL-8/IL-8 receptor axis may be a specific target
for the treatment of UCC.
Acknowledgements
The present study was supported by the National
Nature Science Foundation of China (grant no. 81272854), Nature
Science Foundation of Heilongjiang Province (grant no. D201129),
Science and Innovation Team Foundation of Education Department of
Heilongjiang Province (grant no. cxtd-2016-03), President
Innovation and Entrepreneurship Foundation of Jiamusi University
(grant no. xzyf2014-12) and the Innovation and Entrepreneurship
Training Program for College Students of Heilongjiang Province
(grant no. 201410222036).
References
|
1
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Miyamoto Y, Nakagawa S, Wada-Hiraike O,
Seiki T, Tanikawa M, Hiraike H, Sone K, Nagasaka K, Oda K, Kawana
K, et al: Sequential effects of the proteasome inhibitor bortezomib
and chemotherapeutic agents in uterine cervical cancer cell lines.
Oncol Rep. 29:51–57. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Hernández-Hernández DM, Apresa-García T
and Patlán-Pérez RM: Epidemiological overview of uterine cervical
cancer. Rev Med Inst Mex Seguro Soc. 53 Suppl 2:S154–S161.
2015.PubMed/NCBI
|
|
4
|
Jiang WG, Sanders AJ, Ruge F and Harding
KG: Influence of interleukin-8 (IL-8) and IL-8 receptors on the
migration of human keratinocytes, the role of PLC-γ and potential
clinical implications. Exp Ther Med. 3:231–236. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Stronach EA, Cunnea P, Turner C, Guney T,
Aiyappa R, Jeyapalan S, de Sousa CH, Browne A, Magdy N, Studd JB,
et al: The role of interleukin-8 (IL-8) and IL-8 receptors in
platinum response in high grade serous ovarian carcinoma.
Oncotarget. 6:31593–31603. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Siegel R, Ma J, Zou Z and Jemal A: Cancer
statistics, 2014. CA Cancer J Clin. 64:9–29. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ferlay J, Steliarova-Foucher E,
Lortet-tieulent J, Rosso S, Coebergh JW, Comber H, Forman D and
Bray F: Cancer incidence and mortality patterns in Europe:
Estimates for 40 countries in 2012. Eur J Cancer. 49:1374–1403.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Brat DJ, Bellail AC and Van Meir EG: The
role of interleukin-8 and its receptors in gliomagenesis and
tumoral angiogenesis. Neuro Oncol. 7:122–133. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Singh RK and Lokeshwar BL: The
IL-8-regulated chemokine receptor CXCR7 stimulates EGFR signaling
to promote prostate cancer growth. Cancer Res. 71:3268–3277. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Russo RC, Garcia CC, Teixeira MM and
Amaral FA: The CXCL8/IL-8 chemokine family and its receptors in
inflammatory diseases. Expert Rev Clin Immunol. 10:593–619. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Araki S, Omori Y, Lyn D, Singh RK,
Meinbach DM, Sandman Y, Lokeshwar VB and Lokeshwar BL:
Interleukin-8 is a molecular determinant of androgen independence
and progression in prostate cancer. Cancer Res. 67:6854–6862. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Sims LB, Curtis LT, Frieboes HB and
Steinbach-Rankins JM: Enhanced uptake and transport of
PLGA-modified nanoparticles in cervical cancer. J
Nanobiotechnology. 14:332016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Shen L, Wen N, Xia M, Zhang YU, Liu W, Xu
YE and Sun L: Calcium efflux from the endoplasmic reticulum
regulates cisplatin-induced apoptosis in human cervical cancer HeLa
cells. Oncol Lett. 11:2411–2419. 2016.PubMed/NCBI
|
|
14
|
Gui SL, Teng LC, Wang SQ, Liu S, Lin YL,
Zhao XL, Liu L, Sui HY, Yang Y, Liang LC, et al: Overexpression of
CXCL3 can enhance the oncogenic potential of prostate cancer. Int
Urol Nephrol. 48:701–709. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Tsai YJ, Hao SP, Chen CL and Wu WB:
Thromboxane A2 regulates CXCL1 and CXCL8 chemokine expression in
the nasal mucosa-derived fibroblasts of chronic rhinosinusitis
patients. PLoS One. 11:e01584382016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Liu Q, Li A, Tian Y, Wu JD, Liu Y, Li T,
Chen Y, Han X and Wu K: The CXCL8-CXCR1/2 pathways in cancer.
Cytokine Growth Factor Rev. 31:61–71. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Singh JK, Simões BM, Howell SJ, Farnie G
and Clarke RB: Recent advances reveal IL-8 signaling as a potential
key to targeting breast cancer stem cells. Breast Cancer Res.
15:2102013. View
Article : Google Scholar : PubMed/NCBI
|
|
18
|
Cianga CM, Cianga P, Dumitrescu GF and
Sava A: IL-8, IL-8RA (CXCR1) and IL-8RB (CXCR2) expression in
pilomatricoma. Rom J Morphol Embryol. 57:59–64. 2016.PubMed/NCBI
|
|
19
|
Fernando RI, Castillo MD, Litzinger M,
Hamilton DH and Palena C: IL-8 signaling plays a critical role in
the epithelial-mesenchymal transition of human carcinoma cells.
Cancer Res. 71:5296–5306. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Aravalli RN and Greten TF: FoxC1: Novel
regulator of inflammation-induced metastasis in hepatocellular
carcinoma. Gastroenterology. 149:861–863. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wu D, Tao J, Ding J, Qu P, Lu Q and Zhang
W: Interleukin-11, an interleukin-6-like cytokine, is a promising
predictor for bladder cancer prognosis. Mol Med Rep. 7:684–688.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Xiao YC, Yang ZB, Cheng XS, Fang XB, Shen
T, Xia CF, Liu P, Qian HH, Sun B, Yin ZF and Li YF: CXCL8,
overexpressed in colorectal cancer, enhances the resistance of
colorectal cancer cells to anoikis. Cancer Lett. 361:22–32. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Mellado M, Rodríguez-Frade JM, Mañes S and
Martínez-A C: Chemokine signaling and functional responses: The
role of receptor dimerization and TK pathway activation. Annu Rev
Immunol. 19:397–421. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Liu F, Zheng S, Liu T, Liu Q, Liang M, Li
X, Sheyhidin I, Lu X and Liu W: MicroRNA-21 promotes the
proliferation and inhibits apoptosis in Eca109 via activating
ERK1/2/MAPK pathway. Mol Cell Biochem. 381:115–125. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Palena C, Hamilton DH and Fernando RI:
Influence of IL-8 on the epithelial-mesenchymal transition and the
tumor microenvironment. Future Oncol. 8:713–722. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lake D, Corrêa SA and Müller J: Negative
feedback regulation of the ERK1/2 MAPK pathway. Cell Mol Life Sci.
73:4397–4413. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Bastida MF, Sheth R and Ros MA: A BMP-Shh
negative-feedback loop restricts Shh expression during limb
development. Development. 136:3779–3789. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Murphy C, McGurk M, Pettigrew J,
Santinelli A, Mazzucchelli R, Johnston PG, Montironi R and Waugh
DJ: Nonapical and cytoplasmic expression of interleukin-8, CXCR1,
and CXCR2 correlates with cell proliferation and microvessel
density in prostate cancer. Clin Cancer Res. 11:4117–4127. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
MacManus CF, Pettigrew J, Seaton A, Wilson
C, Maxwell PJ, Berlingeri S, Purcell C, McGurk M, Johnston PG and
Waugh DJ: Interleukin-8 signaling promotes translational regulation
of cyclin D in androgen-independent prostate cancer cells. Mol
Cancer Res. 5:737–748. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Venkatakrishnan G, Salgia R and Groopman
JE: Chemokine receptors CXCR-1/2 activate mitogen-activated protein
kinase via the epidermal growth factor receptor in ovarian cancer
cells. J Biol Chem. 275:6868–6875. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Luppi F, Longo AM, de Boer WI, Rabe KF and
Hiemstra PS: Interleukin-8 stimulates cell proliferation in
non-small cell lung cancer through epidermal growth factor receptor
transactivation. Lung Cancer. 56:25–33. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kawamura M, Toiyama Y, Tanaka K, Saigusa
S, Okugawa Y, Hiro J, Uchida K, Mohri Y, Inoue Y and Kusunoki M:
CXCL5, a promoter of cell proliferation, migration and invasion, is
a novel serum prognostic marker in patients with colorectal cancer.
Eur J Cancer. 48:2244–2251. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Tosoni D, Zecchini S, Coazzoli M, Colaluca
I, Mazzarol G, Rubio A, Caccia M, Villa E, Zilian O, Di Fiore PP
and Pece S: The Numb/p53 circuitry couples replicative self-renewal
and tumor suppression in mammary epithelial cells. J Cell Biol.
211:845–862. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Sajadimajd S, Yazdanparast R and Akram S:
Involvement of Numb-mediated HIF-1α inhibition in
anti-proliferative effect of PNA-antimiR-182 in
trastuzumab-sensitive and -resistant SKBR3 cells. Tumour Biol.
37:5413–5426. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Faraldo MM and Glukhova MA: Regulating the
regulator: Numb acts upstream of p53 to control mammary stem and
progenitor cell. J Cell Biol. 211:737–739. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Sheng W, Dong M, Zhou J, Li X, Liu Q, Dong
Q and Li F: Cooperation among Numb, MDM2 and p53 in the development
and progression of pancreatic cancer. Cell Tissue. 354:521–532.
2013. View Article : Google Scholar
|
|
37
|
Rojas A, Liu G, Coleman I, Nelson PS,
Zhang M, Dash R, Fisher PB, Plymate SR and Wu JD: IL-6 promotes
prostate tumorigenesis and progression through autocrine
cross-activation of IGF-IR. Oncogene. 30:2345–2355. 2011.
View Article : Google Scholar : PubMed/NCBI
|