Introduction
Gastric cancer is the fifth most common cancer and
the third most common cause of cancer-associated mortality
worldwide (1,2). The proportion of locally advanced or
metastatic diseases is ≥80% of all patients with gastric cancer in
mainland China (3). Carbohydrate
antigen (CA)19-9 may be expressed in gastrointestinal cancer cells
and is regarded as a gastrointestinal cancer-associated antigen.
CA19-9 is also observed in the pancreas, bile duct, salivary gland
and other normal epithelial tissue (4). CA19-9 expression is significantly
increased in patients with malignant tumors, being markedly
increased compared with that in the cases of patients with
inflammatory diseases or normal subjects (5). CA19-9 is an effective diagnostic marker,
commonly used for digestive tract tumor diagnosis, efficacy
evaluation, prognosis and postoperative monitoring (6–8).
In the serum of patients with endometrial carcinoma
and digestive tract malignant tumor, the expression level of CA125
can also be detected. Previous clinical trials have demonstrated
that serum CA125 may be used as indicators of gastric cancer
recurrence; predicting poor prognosis and biological behavior
(9,10). Another study revealed that combined
determination of serum carcinoembryonic antigen (CEA), CA19-9 and
CA72-4 is more sensitive for predicting the risk of recurrence, and
for prognosis compared with each protein alone (11). A number of previous studies have
investigated the association or correlation between serum
biomarkers and the prognosis and rate of recurrence in gastric
cancer; however, the association between prognosis and recurrence,
serum biomarkers and cellular receptors has not been studied
thoroughly. Cellular receptors, including epidermal growth factor
receptor 1, human epidermal growth factor receptor 2 (HER2) and
transforming growth factor β 1 have been identified to serve an
active function in the progression of intestinal-type gastric
adenocarcinoma (12). HER2 has been
recognized as a marker for targeting therapy with trastuzumab used
to treat metastatic gastric cancer (13). The expression of HER2 is upregulated
in >20% of patients with metastatic gastric cancer; however,
there are currently no serological approaches to predict the
expression level of HER2 among patients with locally advanced
gastric cancer (14). Conventional
serum tumor biomarkers, including CA19-9 and CA125, are potentially
associated with the detection and prognosis of gastric cancer. The
aim of the present study was to investigate the associations
between HER2 expression and the level of CA19-9 and CA125, and to
correlate these with the prognosis of patients with gastric
cancer.
Materials and methods
Patients and tissue samples
A total of 256 patients from The Affiliated Hongqi
Hospital of Mudanjiang Medical University (Mudanjiang, China) were
enrolled between January 2014 and December 2014. A total of 157
patients (61.3%) were male and 99 patients (38.7%) were female. The
median age was 63 years, with a range between 28 and 83 years. Data
on patient age, sex and tumor-node-metastasis (TNM) stages were
collected from the hospital medical records. The TNM staging system
used in this research was adopted from the 2010 publication by the
International Union Against Cancer and the American Joint Committee
on Cancer (15). All patients
included in the study provided written inform consent for
participation in the study. The study was approved by the
Biomedical Ethical Committee of Affiliated Hongqi Hospital of
Mudanjiang Medical University. The preoperative and postoperative
gastrointestinal tumor markers were tested. All patients had not
received chemotherapy or surgical treatment of gastric cancer prior
to surgery. The serum CA19-9 and serum CA125 were performed using a
Roche E601 automatic immunoassay analyzer (Roche Diagnostics GmbH,
Mannheim, Germany). The tissue samples were collected by endoscopic
and intraoperative biopsy.
Immunohistochemistry
An optical microscope (magnification, ×200) was used
in this study. Gastric cancer tissues were collected from
endoscopic and intraoperative biopsy and fixed with 10% formic acid
solution for 2 h at 60°C. Subsequently, conventional paraffin
embedding was performed, and the sample was sectioned in to 4 µm
sections. HercepTest Kits (Agilent Technologies, Inc., Santa Clara,
CA, USA) was used for HER2 staining. The kits solution was placed
into a water bath at 95–99°C, and incubated in the water for 40±1
min according to the manufacturer's protocol.
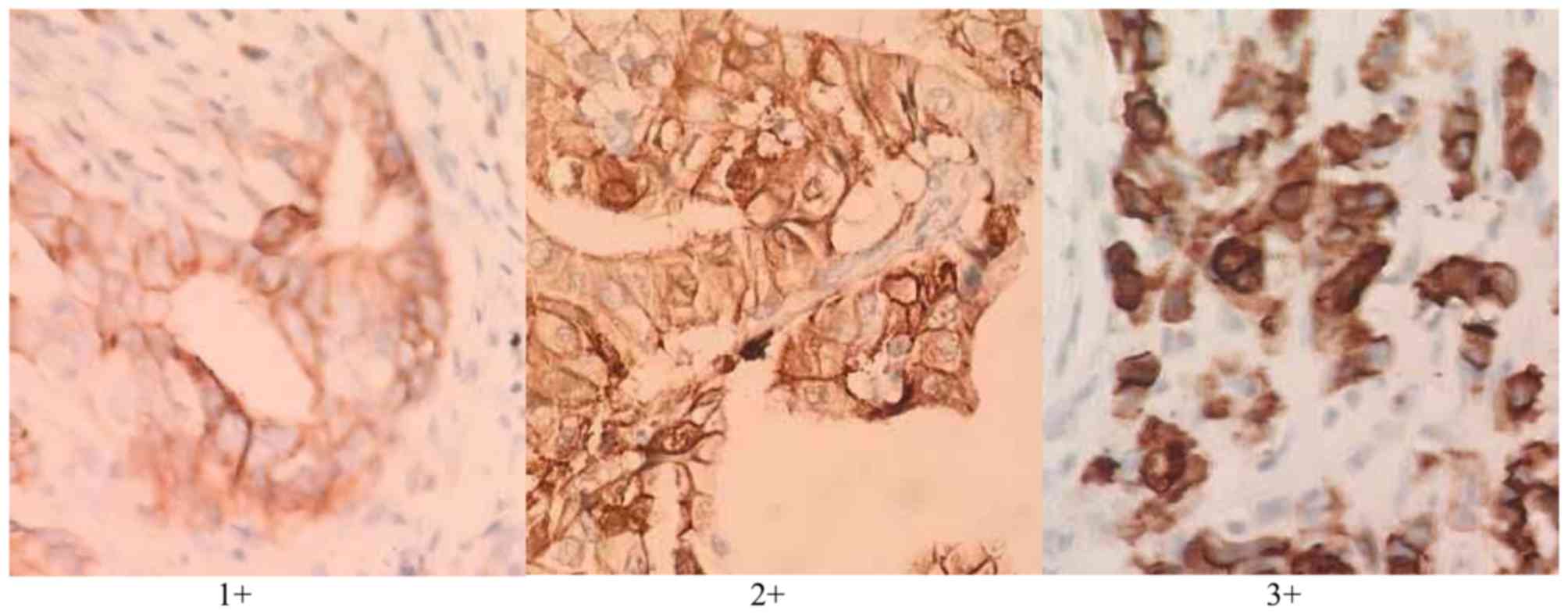
HER2-positive staining was located in the cell
membrane, and the HercepTest modified score was used to evaluate
the immunohistochemical staining results (16): (1+) indicates that >10% of cells
have mild cell membrane staining; (2+) indicates that >10% of
cells have weak complete or basolateral lateral staining; (3+)
means that >10% of cells have medium to strong complete or
basolateral lateral staining (0; meaning negative staining) and
(1+) were classed as negative, and (2+) and (3+) were classed as
positive.
Follow-up
In the present study, the date of surgery for
patients was considered the first diagnosis date and where
observations started. The deadline for observation of mortalities
was the date of mortality, the deadline for observation of
surviving cases was October 31, 2016, and the survival time was
measured in days. The survival time of the patients with lost
follow-up was given as the deadline. Follow-up began in August 2013
and was performed every 3 months with the last visit being October
2016. The follow-up was performed primarily with phone calls. The
medication regimens of individual patients were confirmed; and
subsequent follow-up appointments were scheduled. Instructions of
future treatment were also provided. Patients' overall health,
medication status, any minor or major complains and the follow-up
statuses were recorded. The status of the patients was recorded
using Excel 2010 (Microsoft Corporation, Redmond, WA, USA).
Statistical analysis
Through single factor analysis on the 3-year
survival rate of gastric cancer-associated factors, relevant
factors were introduced into Cox's multivariate regression model to
evaluate the independent prognostic factors of gastric cancer. SPSS
19.0 (IBM Corp., Armonk, NY, USA) was used for statistical
analysis. Homogeneity of variance tests and one-way analysis of
variance was used with the Student-Newman-Keuls post-hoc method if
variance was consistent, or Games-Howell variance method if
variance was not consistent. The association between serum
indicator and the expression of HER2 was analyzed with Pearson's
correlation coefficient test. The survival analyses were conducted
according to the Kaplan-Meier method and survival characteristics
were compared using log-rank tests. P<0.05 was considered to
indicate a statistically significant difference. P<0.01 was
considered to indicate a highly statistically significant
difference.
Results
Patient clinicopathological
features
Differences between the positive rates of CA19-9,
CA125 and HER2 are presented in Table
I. The prevalence of serum CA19-9 and CA125 were 25.78 and
24.22%, respectively. The prevalence of HER2 (2+/3+) was 16.01%.
The prevalence of HER2 (2+/3+) between I/II and III/IV TNM stage
patients with gastric cancer were significantly different
(P<0.05), with III/IV exhibiting increased rates of positive
HER2. The prevalence of HER2 (2+/3+) was also associated with tumor
differentiation (P<0.01). No significant association was
demonstrated between serum CA19-9, CA125 and HER2 positive rates,
and sex or age group (P>0.05). Serum CA125 also exhibited
statistically significant differences in different tumor
differentiation and different TNM stage patients (P<0.05).
 | Table I.Differences between positive rates of
CA19-9, CA125 and HER2 for different clinicopathological features
in patients with gastric carcinoma. |
Table I.
Differences between positive rates of
CA19-9, CA125 and HER2 for different clinicopathological features
in patients with gastric carcinoma.
| Clinicopathological
feature | Total, n | CA19-9, n (%) | CA125, n (%) | HER2 2+/3+, n
(%) |
|---|
| Sex |
|
|
|
|
| Male | 157 | 42 (26.75) | 34 (21.66) | 22 (14.01) |
|
Female | 99 | 24 (24.24) | 28 (28.28) | 19 (19.19) |
|
χ2 |
| 0.09 | 1.11 | 0.85 |
|
P-value |
| 0.76 | 0.29 | 0.35 |
| Age, years |
|
|
|
|
| ≤60 | 149 | 36 (24.16) | 34 (22.82) | 20 (13.42) |
|
>60 | 107 | 30 (28.04) | 28 (26.17) | 21 (19.63) |
|
χ2 |
| 0.30 | 0.22 | 1.35 |
|
P-value |
| 0.57 | 0.63 | 0.24 |
| Tumor
differentiation |
|
|
|
|
|
Well/moderate | 211 | 52 (24.64) | 45 (21.33) | 10 (4.74) |
| Poor | 45 | 14 (31.11) | 17 (37.78) | 31 (68.89) |
|
χ2 |
| 0.50 | 4.60 | 108.76 |
|
P-value |
| 0.47 | 0.03 | <0.0001 |
| TNM stage |
|
|
|
|
| I/II | 108 | 31 (28.7) | 19 (17.6) | 9 (8.3) |
|
III/IV | 148 | 35 (23.65) | 43 (29.05) | 32 (21.62) |
|
χ2 |
| 0.59 | 3.86 | 7.23 |
|
P-value |
| 0.44 | 0.04 | <0.001 |
Serum CA19-9, CA125 and HER2 positive
rate in patients with distinct TNM staging prior to surgery
CA19-9 and CA125 concentration were obtained from
clinical examination and HER2 positive rates were obtained from the
immunohistochemical analysis of postoperative gastric cancer
tissue. A total of 57 cases were classified as stage I, 51 cases
were stage II, 142 cases were stage III and 6 cases were stage IV.
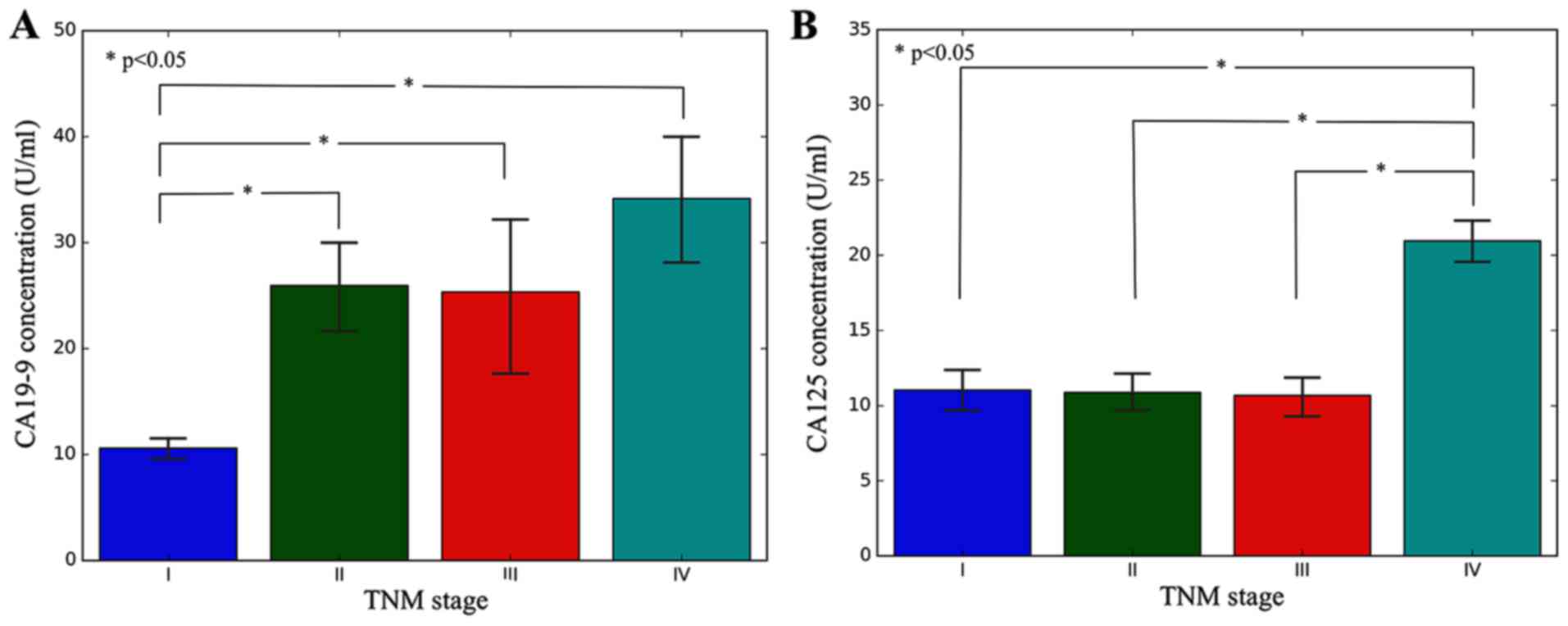
The concentration of CA19-9 and CA125, and the positive rates of
HER2 were compared with distinct TNM stages. The results are
presented in Table II. The results
demonstrated in Table II and
Fig. 1 revealed the positive
expression rates of CA19-9, CA125 and HER2 in patients with
different TNM staging gastric cancer.
 | Table II.Positive expression rates of CA19-9,
CA125 and HER2 in patients with different TNM staging gastric
cancer. |
Table II.
Positive expression rates of CA19-9,
CA125 and HER2 in patients with different TNM staging gastric
cancer.
| TNM stage | CA19-9,
U/mla | CA125,
U/mla | HER2, % |
|---|
| I | 10.9±88.04 | 10.21±1.47 | 5.21 |
| II | 26.92±35.18 | 10.40±9.8 | 19.52 |
| III | 32.91±58.02 | 12.45±10.99 | 40.32 |
| IV | 33.52±46.46 | 21.08±12.3 | 46.34 |
| t/χ2
value | 21.980 | 23.184 | 2.73 |
| P-value | <0.01 | <0.01 | <0.01 |
As can be noted from Table II, the positive rate of CA19-9 and
CA125 in stages III and IV was significantly increased compared
with that in stages I, and II. In addition, the positive rate of
HER2 in different stages of gastric cancer was also statistically
different.
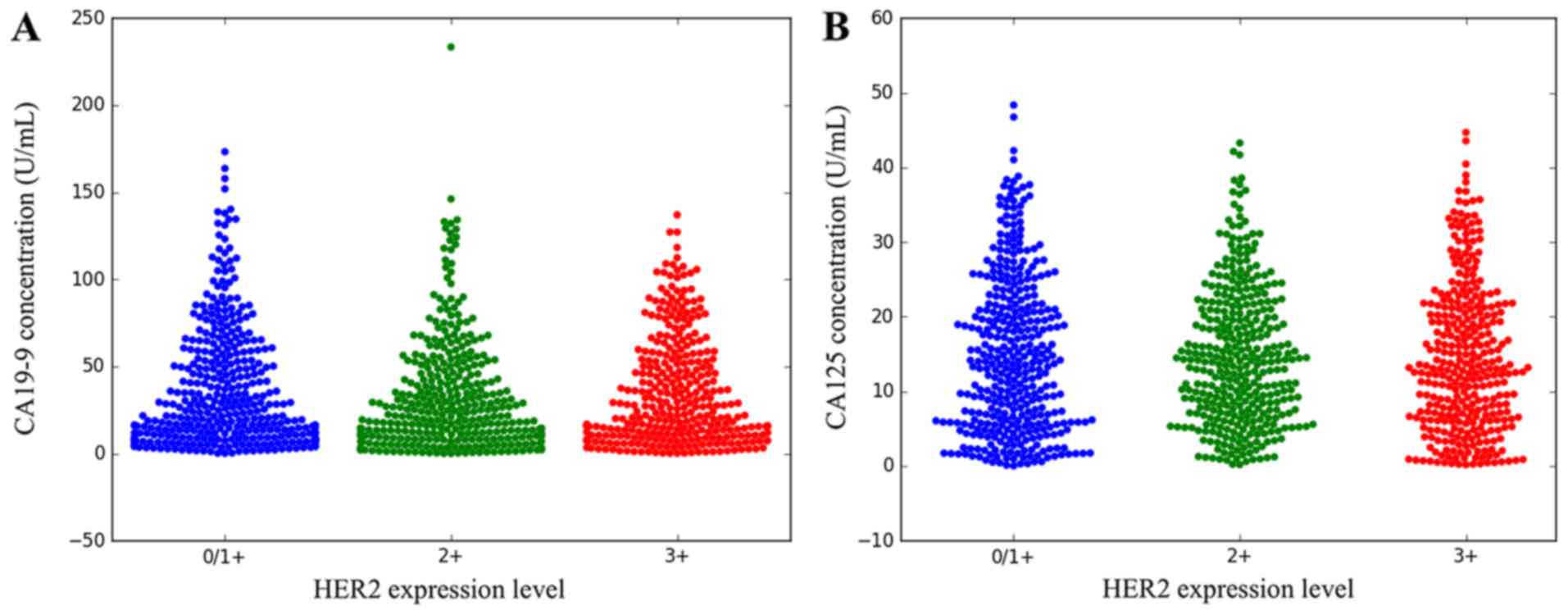
Correlation between HER2 and CA19-9,
CA125
The correlation of serum CA19-9 and CA125 and the
expression of HER2 were analyzed using Pearson's correlation
coefficient. The results indicate that there was no correlation
between the serum CA19-9/CA125 level and the level of HER2
expression (P<0.05). The logistic regression results are
presented in Table III. The
different HER2 expression levels are presented in Fig. 2. The distribution of CA19-9 and CA125
and the expression of HER2 are presented in Fig. 3.
 | Table III.Association between CA19-9 and CA125,
and HER2 positive expression. |
Table III.
Association between CA19-9 and CA125,
and HER2 positive expression.
|
|
| HER2 (2+) | HER2 (3+) | HER2 (2+/3+) |
|---|
|
|
|
|
|
|
|---|
| Marker | HER2 (−) OR | OR (95% CI) |
P-valuea | OR (95% CI) |
P-valuea | OR (95% CI) |
P-valuea |
|---|
| CA19-9 | 1 | 1.36
(0.67–2.63) | 0.21 | 1.24
(0.41–1.76) | 0.35 | 1.42
(0.85–1.38) | 0.25 |
| CA125 | 1 | 1.31
(0.22–8.01) | 0.79 | 1.85
(0.73–1.74) | 0.43 | 1.37
(0.37–1.87) | 0.63 |
CA19-9, CA125 and HER2 as prognosis
markers in patients with gastric cancer
Association between CA19-9, CA125 and
HER2, and recurrence and metastasis
A total of 219 patients with gastric cancer with
follow-up results within 3 years were divided into
recurrence/metastasis group and non-recurrence/metastasis group,
and the expression of serum tumor markers in the preoperative
recurrent group and non-recurrent group were analyzed. There was no
significant difference in the results of postoperative recurrence
and metastasis between the tumor and metastasis positive group, and
negative group. The results are presented in Table IV. Loss of follow-up data of 37
patients was primarily due to the lack of mobile phone connection,
a reason why the access to postoperative survival status
information was not possible.
 | Table IV.Association between CA19-9, CA125 and
HER2, and recurrence and metastasis. |
Table IV.
Association between CA19-9, CA125 and
HER2, and recurrence and metastasis.
|
| CA19-9 | CA125 | HER2 |
|---|
|
|
|
|
|
|---|
| Expression | Recurrence
metastasis | No recurrence
metastasis | Recurrence
metastasis | No recurrence
metastasis | Recurrence
metastasis | No recurrence
metastasis |
|---|
| Positive | 29 | 8 | 34 | 11 | 32 | 13 |
| Negative | 88 | 94 | 89 | 85 | 84 | 90 |
| χ2 | 11.836 | 10.378 | 6.595 |
| P-value | 0.001 | 0.001 | 0.010 |
As observed from Table
IV, CA19-9, CA125 and HER2-positive patients with gastric
cancer recurrence and metastasis were increased compared with
negative patients. The analyzed results of experimental data were
statistically significant (P<0.01). The HER2
immunohistochemistry scores of gastric cancer tissues are presented
in Fig. 2.
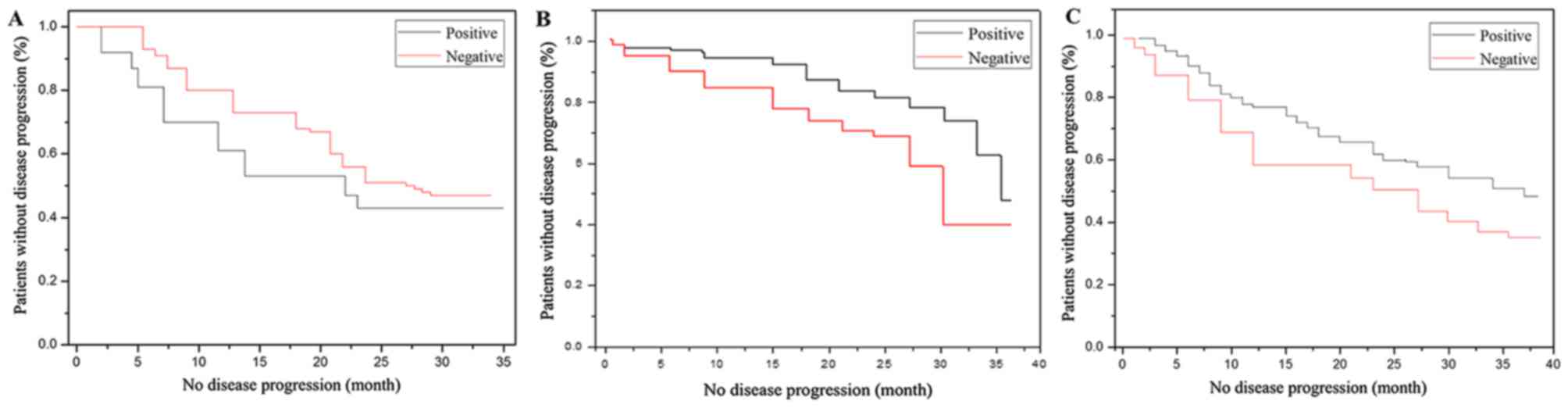
Kaplan Meier analysis of recurrence
and metastasis of gastric cancer
Survival analysis was performed in patients with
stage I, II and III tumors, without disease progression. The
comparisons were performed on the preoperative CA19-9, CA125,
HER2-negative and -positive groups. The three indicators were
compared separately. The results demonstrated that HER2-positive
patients had recurrence and metastasis earlier compared with the
negative patients. CA19-9, CA125 and HER2-positive patients without
disease progression experiences survival times of 26 months on
average (Table V). Negative patients
without disease progression experienced survival times of 35 months
on average. Gastric cancer recurrence and metastasis time curves
are presented in Fig. 4.
 | Table V.Recurrence and metastasis time;
Log-Rank test results. |
Table V.
Recurrence and metastasis time;
Log-Rank test results.
| Tumor markers | χ2 | df | P-value |
|---|
| CA19-9 | 23.919 | 1 | <0.0001 |
| CA125 | 10.4055 | 1 | 0.011 |
| HER2 | 15.981 | 1 | 0.002 |
Univariate analysis of 3-year survival
rate of patients with gastric cancer
The association between sex, age, stage, tumor
markers and 3-year survival rate of patients with gastric cancer
were used for single factor analysis. The patients <60 years of
age experienced a higher 3-year survival rate compared with
patients >60 years of age (P<0.05). The 3-year survival rates
were significantly different for distinct stages (P<0.05). The
3-year survival rate of stage I was increased compared with that of
stage II and III (P<0.05). The 3-year survival rate of patients
with positive tumor markers was significantly decreased compared
with that of negative patients (P<0.05). Age, stage and
preoperative tumor markers were associated with the 3-year survival
rate of gastric cancer (P<0.05), as shown in Table VI.
 | Table VI.Univariate analysis of 3-year
survival rate of patients with gastric cancer. |
Table VI.
Univariate analysis of 3-year
survival rate of patients with gastric cancer.
| Characteristic | Total, n | Mortalities, n | 3-year cumulative
survival ratea | P-value |
|---|
| Sex |
|
|
|
|
|
Male | 134 | 67 | 0.501 (67/134) | 0.725 |
|
Female | 85 | 40 | 0.532 (45/85) |
|
| Age, years |
|
|
|
|
|
≥60 | 105 | 56 | 0.463 (49/105) | 0.034 |
|
<60 | 114 | 49 | 0.572 (65/114) |
|
| TNM stage |
|
|
|
|
| I | 42 | 8 | 0.821 (34/42) | 0.007 |
| II | 54 | 18 | 0.672 (36/54) |
|
|
III | 123 | 79 | 0.354 (44/123) |
|
| CA19-9 |
|
|
|
|
|
Negative | 131 | 57 | 0.562 (74/131) | 0.014 |
|
Positive | 88 | 66 | 0.245 (22/88) |
|
| CA125 |
|
|
|
|
|
Negative | 155 | 62 | 0.603 (93/155) | 0.007 |
|
Positive | 64 | 44 | 0.309 (20/64) |
|
| HER2 |
|
|
|
|
|
Negative | 123 | 46 | 0.625 (77/123) | 0.011 |
|
Positive | 96 | 63 | 0.345 (33/96) |
|
Multivariate analysis of 3-year
survival rate of patients with gastric cancer
The 3-year cumulative survival rate was used as the
dependent variable, and age, sex, CA19-9, CA125 and HER2 were used
as independent variables by univariate analysis of statistically
significant differences (P<0.05). Multivariate logistic
regression analysis was performed. The results demonstrated that
HER2 [odds ratio (OR)=2.55] and CA19-9 (OR=1.22) were independent
prognostic factors in patients with gastric cancer. The risk of
preoperative HER2 positive mortality was 2.55 times higher compared
with that of preoperative negative patients. The risk of
preoperative CA19-9 positive mortality was 1.22 times higher
compared with that of preoperative-negative patients, as shown in
Table VII.
 | Table VII.Multivariate analysis of 3-year
survival rate of patients with gastric cancer. |
Table VII.
Multivariate analysis of 3-year
survival rate of patients with gastric cancer.
| Characteristic | Regression
coefficient | SE of partial
regression coefficient | P-value | Relative risk | Relative risk | 95% CI |
|---|
| Age | −0.287 | 0.165 | 0.082 | 0.761 | 0.553 | 1.045 |
| Sex | 0.024 | 0.073 | 0.842 | 1.027 | 0.895 | 1.207 |
| CA19-9 | 0.193 | 0.147 | 0.194 | 1.222 | 0.997 | 1.565 |
| CA125 | 1.176 | 0.191 | 0.383 | 1.190 | 0.828 | 1.712 |
| HER2 | 0.920 | 0.449 | 0.046 | 2.553 | 1.529 | 3.771 |
Discussion
In 1979, scientists identified antibodies against
colon cancer in mice with colon cancer, and termed them 1165NS199.
These antibodies recognized the body's gastrointestinal
cancer-associated carbohydrate antigen, and later this antigen was
termed the sugar chain antigen 19-9 (4). Previous studies demonstrated that CA
19-9 is associated with the prognosis of patients with gastric
cancer, and that it could be used as a prognostic indicator and
postoperative monitoring index of gastric cancer (17). Sougioultzis et al (18) conducted a retrospective analysis of
114 cases of gastric cancer, and demonstrated that serum CA 19-9
level is significantly increased in stage IV patients compared with
that in stages I, II and III, and preoperative CA19-9 expression is
associated with recurrence and metastasis. Marrelli et al
(19) monitored the level of CEA and
CA19-9 in recurrent and non-recurrence groups following radical
gastrectomy, and revealed that the recurrence group was positive
for least one tumor marker. Compared with the non-recurrence group,
the recurrence group CA19-9 was significantly increased (20). During the follow-up period following
surgery, elevated tumor markers indicate a high risk of recurrence
(21). The serum levels of CEA,
CA72-4 and CA19-9 in 102 patients with gastric cancer were measured
by Mihmanli et al (20), in
which single factor analysis was performed on recurrence and
metastasis. Their results demonstrated that sex, CA72-4 and
abnormal CA9-9 levels were associated with a worse prognosis. In
addition, multivariate regression analysis was performed on the
recurrence and metastasis data, which indicated that CA72-4 and
CA19-9 were risk factors for recurrence (22).
CA125 is a glycoprotein that was detected in 1983 as
an epithelial ovarian cancer antigen, which binds to the monoclonal
antibody OC125 (21). In 80% of
patients with ovarian cancer, an elevated serum level of CA 125 was
detected. Therefore, CA 125 may be used as an indicator of
differential diagnosis of ovarian benign and malignant tumors. CA
125 is the most sensitive marker for the detection of epithelial
ovarian cancer and is important for diagnosing ovarian cancer, as
well as curative effect and prognosis of ovarian cancer (22). CA125 may also be detected in the serum
of patients with endometrial and gastrointestinal cancer. Previous
clinical trials revealed that serum CA 125 may be used as an
indicator to detect the recurrence of gastric cancer, and predict
the prognosis and poor biological behavior. The majority of studies
on the association between CA 125 and gastric cancer suggest that
elevated serum CA 125 is associated with peritoneal metastasis
(23–25). Byström et al (26) demonstrated that CA 125 is primarily
distributed throughout the ovaries and fallopian tube epithelium,
however, it is also identified in the peritoneum, pleura and
pericardium of the mesothelial cells. The peritoneal metastasis in
those tissues may be a result of peritoneal examination or
adhesion, leading to CA 125 antigen content being increased
significantly (26).
HER-2 is a member of the human epidermal growth
factor receptor family, located on the human chromosome 17q21. This
protein has cell membrane glycoproteins with tyrosine kinase
activity, which belongs to the tyrosine kinase type I receptor
family (27). The HER-2 gene is a
proto-oncogene. Amplification of the gene induces overexpression of
the protein in the cell membrane leading to malignant cells. HER-2
is normally only expressed in human fetuses, and is expressed at
low levels in a small number of adult tissues that are involved in
cell division, growth and reproduction regulation (27). It is associated with cell motility,
cell viability enhancement and cancer cell migration (27). HER-2 is overexpressed in a variety of
human tumors, such as breast cancer, ovarian cancer, endometrial
cancer, lung adenocarcinoma and primary renal cell carcinoma. The
overexpression of HER-2 is associated with tumor invasion,
metastasis, chemotherapy resistance and poor prognosis. HER-2 has
been demonstrated to be involved in the proliferation,
differentiation, metastasis and anti-apoptotic effect of cancer
cells (28). Overexpression of HER-2
may be indicative of a higher degree of malignancy, rapid
progression, sort remission period following chemotherapy, and
increased resistance to chemotherapy and endocrine therapy, leading
to poor prognosis and decreased survival rates of the patients
(29).
There are numerous tumor markers associated with the
occurrence and development of gastric cancer. However, due to
various factors, including the sensitivity, monitoring methods and
monitoring cost, there are not many markers widely used in the
clinic. Currently, the most commonly used markers relevant to
gastric cancer include CA19-9, CA125 and HER2 (9,26).
In the present study, the association between the
aforementioned biomarkers and clinicopathological characteristics
of gastric cancer were investigated. Serum tumor markers were
negative or significantly decreased in patients with stage I, II
and III gastric cancer following radical gastrectomy. Serum tumor
marker expression increased alongside progress of the disease,
particularly in patients with recurrence and metastasis; in certain
cases, expression was double or triple the post-surgery value. The
positive rate of CA 19-9, CA 125 and HER2 in patients with gastric
cancer prior to surgery was significantly different from the
positive rate of tumor markers in the first 3 months following
surgery. The results demonstrated that radical gastrectomy removed
the expression of tumor markers in the cancer and the serum tumor
markers were significantly decreased or negative.
In conclusion, the present study investigated the
association between HER2 expression and CA19-9 and CA125, and
explored the association between them and the prognosis of patients
with gastric cancer. From the results of the present study, the
following conclusions may be drawn: CA19-9, CA125 and HER2 may be
used to diagnose the recurrence or metastasis of gastric cancer;
the combined detection is able to improve the sensitivity and
efficiency of predicting the recurrence or metastasis of gastric
cancer; the positive rate of CA19-9 and CA125 in stages III/IV was
increased compared with that in stages I/II; pre-surgery positive
serum CA19-9 and CA125 was associated with poor prognosis in
patients with gastric cancer; and CA19-9 and HER2 were independent
prognostic factors for gastric cancer.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
Conception and design, was the responsibility of HZ
and JC. HX and GH were responsible for the collection and assembly
of data. AD and HX completed data analysis and interpretation and
HZ and AD wrote the manuscript. All authors read and approved the
final manuscript.
Ethics approval and consent to
participate
The study was approved by the Biomedical Ethical
Committee of Affiliated Hongqi Hospital of Mudanjiang Medical
University. All patients included in the study provided written
inform consent for participation in the study.
Consent for publication
Patients provided written informed consent for their
participation in the present study and the publication of any
data.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Massimo R, Fassan M and Graham DY:
Epidemiology of gastric cancerGastric Cancer. Springer; Cham: pp.
23–34. 2015, PubMed/NCBI
|
|
2
|
Miyashiro I, Hiratsuka M, Sasako M, Sano
T, Mizusawa J, Nakamura K, Nashimoto A, Tsuburaya A and Fukushima
N: Gastric Cancer Surgical Study Group (GCSSG) in the Japan
Clinical Oncology Group (JCOG): High false-negative proportion of
intraoperative histological examination as a serious problem for
clinical application of sentinel node biopsy for early gastric
cancer: Final results of the Japan Clinical Oncology Group
multicenter trial JCOG0302. Gastric Cancer. 17:316–323. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Nashimoto A, Akazawa K, Isobe Y, Miyashiro
I, Katai H, Kodera Y, Tsujitani S, Seto Y, Furukawa H, Oda I, et
al: Gastric cancer treated in 2002 in Japan: 200 annual report of
the JGCA nationwide registry. Gastric Cancer. 16:1–27. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bai LY, Chiu CF, Yang HR and Yang TY: 2230
Palliative gastrectomy does not prolong survival of metastatic
gastric cancer patients with both high CEA and high CA19-9 values
at diagnosis. Eur J Cancer. 51:S4102015. View Article : Google Scholar
|
|
5
|
Sun Z and Zhang N: Clinical evaluation of
CEA, CA19-9, CA72-4 and CA125 in gastric cancer patients with
neoadjuvant chemotherapy. World J Surg Oncol. 12:3972014.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
He CZ, Zhang KH, Li Q, Liu XH, Hong Y and
Lv NH: Combined use of AFP, CEA, CA125 and CAl9-9 improves the
sensitivity for the diagnosis of gastric cancer. BMC gastroenterol.
13:872013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Liang Y, Wang W, Fang C, Raj SS, Hu WM, Li
QW and Zhou ZW: Clinical significance and diagnostic value of serum
CEA, CA19-9 and CA72-4 in patients with gastric cancer. Oncotarget.
7:49565–49573. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Yang AP, Liu J, Lei HY, Zhang QW, Zhao L
and Yang GH: CA72-4 combined with CEA, CA125 and CAl9-9 improves
the sensitivity for the early diagnosis of gastric cancer. Clin
Chim Acta. 437:183–186. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Shimada H, Noie T, Ohashi M, Oba K and
Takahashi Y: Clinical significance of serum tumor markers for
gastric cancer: A systematic review of literature by the Task Force
of the Japanese Gastric Cancer Association. Gastric Cancer.
17:26–33. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kim JH, Jun KH, Jung H, Park IS and Chin
HM: Prognostic value of preoperative serum levels of five tumor
markers (Carcinoembryonic Antigen, CA19-9, Alpha-fetoprotein,
CA72-4, and CA125) in gastric cancer. Hepatogastroenterology.
61:863–869. 2014.PubMed/NCBI
|
|
11
|
Yu J, Zhang S and Zhao B: Differences and
correlation of serum CEA, CA19-9 and CA72-4 in gastric cancer. Mol
Clin Oncol. 4:441–449. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Docea AO, Mitruţ P, Cernea D, Georgescu
CC, Olimid D, Mărgăritescu C and Dumitrescu D: Immunohistochemical
expression of EGF, c-erbB-2 and EGFR in intestinal variant of
gastric adenocarcinomas. Rom J Morphol Embryol. 54:545–554.
2013.PubMed/NCBI
|
|
13
|
Docea AO, Mitruţ P, Grigore D, Pirici D,
Călina DC and Gofiţă E: Immunohistochemical expression of TGF beta
(TGF-β), TGF beta receptor 1 (TGFBR1), and Ki67 in intestinal
variant of gastric adenocarcinomas. Rom J Morphol Embryol. 53 3
Suppl:S683–S692. 2012.
|
|
14
|
Liu XH, Sun M, Nie FQ, Ge YB, Zhang EB,
Yin DD, Kong R, Xia R, Lu KH, Li JH, et al: Lnc RNA HOTAIR
functions as a competing endogenous RNA to regulate HER2 expression
by sponging miR-331-3p in gastric cancer. Mol Cancer. 13:922014.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Edge SB and Compton CC: The American Joint
Committee on Cancer: The 7th edition of the AJCC cancer staging
manual and the future of TNM. Ann Surg Oncol. 17:1471–1474. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Hofmann M, Stoss O, Shi D, Büttner R, van
de Vijver M, Kim W, Ochiai A, Rüschoff J and Henkel T: Assessment
of a HER2 scoring system for gastric cancer: Results from a
validation study. Histopathology. 52:797–805. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Chiu CF, Yang HR, Yang MD, Jeng LB, Yang
TY, Sargeant AM and Bai LY: Palliative Gastrectomy Prolongs
Survival of Metastatic Gastric Cancer Patients with Normal
Preoperative CEA or CA19-9 Values: A Retrospective Cohort Study.
Gastroenterol Res Pract. 2016:68460272016. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Sougioultzis S, Syrios J, Xynos ID,
Bovaretos N, Kosmas C, Sarantonis J, Dokou A, Tzivras D, Zografos
G, Felekouras E, et al: Palliative gastrectomy and other factors
affecting overall survival in stage IV gastric adenocarcinoma
patients receiving chemotherapy: A retrospective analysis. Eur J
Surg Oncol. 37:312–318. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Marrelli D, De Stefano A, de Manzoni G,
Morgagni P, Di Leo A and Roviello F: Prediction of recurrence after
radical surgery for gastric cancer: A scoring system obtained from
a prospective multicenter study. Ann Surg. 241:247–255. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Mihmanli M, Dilege E, Demir U, Coskun H,
Eroglu T and Uysalol MD: The use of tumor markers as predictors of
prognosis in gastric cancer. Hepatogastroenterology. 51:1544–1547.
2004.PubMed/NCBI
|
|
21
|
Lordick F and Janjigian YY: Clinical
impact of tumour biology in the management of gastroesophageal
cancer. Nat Rev Clin Oncol. 13:348–360. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Verlato G, Roviello F, Marchet A,
Giacopuzzi S, Marrelli D, Nitti D and de Manzoni G: Indexes of
surgical quality in gastric cancer surgery: Experience of an
Italian network. Ann Surg Oncol. 16:594–602. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kim KM, Bilous M, Chu KM, Kim BS, Kim WH,
Park YS, Ryu MH, Sheng W, Wang J, Chao Y, et al: Human epidermal
growth factor receptor 2 testing in gastric cancer: Recommendations
of an Asia-Pacific task force. Asia Pac J Clin Oncol. 10:297–307.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ieni A, Barresi V, Rigoli L, Caruso RA and
Tuccari G: HER2 status in premalignant, early, and advanced
neoplastic lesions of the stomach. Dis Markers. 2015:2348512015.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Luis M, Tavares A, Carvalho LS,
Lara-santos L, Araújo A and De mello RA: Personalizing therapies
for gastric cancer: Molecular mechanisms and novel targeted
therapies. World J Gastroenterol. 19:6383–6397. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Byström P, Berglund A, Nygren P, Wernroth
L, Johansson B, Larsson A, Einarsson R and Glimelius B: An
explorative study on the clinical utility of baseline and serial
serum tumour marker measurements in advanced upper gastrointestinal
cancer. Oncol Rep. 24:1645–1652. 2010.PubMed/NCBI
|
|
27
|
Moore RG, McMeekin DS, Brown AK,
DiSilvestro P, Miller MC, Allard WJ, Gajewski W, Kurman R, Bast RC
Jr and Skates SJ: A novel multiple marker bioassay utilizing HE4
and CA125 for the prediction of ovarian cancer in patients with a
pelvic mass. Gynecol Oncol. 112:40–46. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ishida M, Kagawa S, Shimoyama K, Takehara
K, Noma K, Tanabe S, Shirakawa Y, Tazawa H, Kobayashi H and
Fujiwara T: Trastuzumab-based photoimmunotherapy integrated with
viral HER2 transduction inhibits peritoneally disseminated
HER2-negative cancer. Mol Cancer Ther. 15:402–411. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Emoto S, Ishigami H, Yamashita H,
Yamaguchi H, Kaisaki S and Kitayama J: Clinical significance of
CA125 and CA72-4 in gastric cancer with peritoneal dissemination.
Gastric Cancer. 15:154–161. 2012. View Article : Google Scholar : PubMed/NCBI
|