Introduction
Renal cell carcinoma (RCC) accounts for 80% of all
primary renal neoplasms and comprises 2–3% of all malignancies
(1). Partial nephrectomy is the most
effective treatment for local clear cell RCC (ccRCC); however, 30%
of patients develop metastatic disease after surgery. Although some
kinase inhibitors have been used in clinical applications,
metastatic RCC remains largely incurable due to the off-target
effects of current agents, and resistance to chemotherapy and
radiation (2,3). Therefore, the identification of novel
markers and targets for the early detection and treatment of RCC is
required (4–7).
Neuron-derived neurotrophic factor (NDNF) is a
glycosylated, disulfide-bonded secretory protein that contains a
fibronectin type III domain. NDNF has been identified as a
neurotrophic factor that can promote neuron migration, growth and
survival, as well as neurite outgrowth (8). Furthermore, NDNF promotes endothelial
cell survival and vessel formation, and has an important role in
the process of revascularization through nitric oxide synthase
3-dependent mechanisms (9). NDNF can
also exert favorable effects on various ischemic cardiovascular
disorders (10); however, the
functional role of NDNF in carcinogenesis has not yet been
identified.
In the present study, the expression and roles of
NDNF in RCC were investigated. The results demonstrated that NDNF
was decreased in ccRCC. In addition, restoring NDNF expression
inhibited the proliferation, migration and invasion of RCC via the
suppression of epithelial-mesenchymal transition (EMT), which
contributes to the progression of cancer by promoting loss of
cell-cell adhesion, leading to a shift in cytoskeletal dynamics
(11). NDNF may therefore be
considered an important regulator of EMT in RCC progression.
Materials and methods
Patient samples
The present study included 69 samples from patients
diagnosed with ccRCC. Renal cancer tissues and paired normal renal
tissues were obtained from each patient and stored at −80°C.
Written informed consent was provided by the patients and their
families for the use of tissue specimens. The present study was
approved by the Ethics Committee of Shenzhen Second People's
Hospital (Shenzhen, China).
Cell culture and lentiviral
infection
The human RCC cell lines ACHN, Caki-1, OS-RC-2,
786-O and 769-P, and the papillary RCC cell line Caki-2, were
purchased from the American Type Culture Collection (Manassas, VA,
USA), ACHN was cultured in MEM (Gibco; Thermo Fisher Scientific,
Inc., Waltham, MA, USA), Caki-1 and Caki-2 were cultured in McCoy's
5A (Gibco; Thermo Fisher Scientific, Inc.), OS-RC-2, 786-O and
769-P were cultured in RPMI 1640 (Gibco; Thermo Fisher Scientific,
Inc.). Cell lines were supplemented with 10% FBS (Gibco; Thermo
Fisher Scientific. Inc.), 100 U/ml penicillin (Gibco; Thermo Fisher
Scientific, Inc.) and 100 µg/ml streptomycin (Gibco; Thermo Fisher
Scientific, Inc.) and were grown in 37°C, 5% CO2
atmosphere. The lentiviral plasmids pLVX-3FLAG (Lenti-Control) and
pLVX-NDNF-3FLAG (Lenti-NDNF) were designed by Shanghai GeneChem
Co., Ltd. (Shanghai, China). The lentiviruses were generated by
cotransfecting the 20 µg lentiviral plasmid, 15 µg pHelper1.0
(gag/pol element) and 10 µg Helper2.0 (VSVG element) into 70–80%
confluence 293T cells (ATCC, Manassas, VA, USA) using
Lipofectamine® 2000 (Invitrogen; Thermo Fisher
Scientific, Inc., Waltham, MA, USA). After 48 h transfection, the
viral supernatants were collected, centrifuged at 70,000 × g at 4°C
for 2 h and filtered. ACHN and 786-O cells were infected with
lentiviruses and 8 µg/ml polybrene (Sigma-Aldrich, Merck KGaA,
Darmstadt, Germany). Infected cells were then exposed to 4 µg/ml
puromycin (cat no. s7417; Selleck Chemicals, Houston, TX, USA) for
4 days. Single-cell clones with stable NDNF expression were
identified by reverse transcription-quantitative polymerase chain
reaction (RT-qPCR) and western blotting.
RT-qPCR
RT-qPCR was carried out as described previously
(12,13). Briefly, total RNA was extracted using
TRIzol® reagent (Invitrogen; Thermo Fisher Scientific,
Inc.) and cDNA was converted from total RNA using the PrimeScript™
RT reagent with gDNA Eraser (Takara Bio, Inc., Otsu, Japan)
according to the manufacturer's protocol. RT-qPCR was performed by
using HotStarTaq DNApolymerase and buffer (Quiagen GmbH, Hilden,
Germany) with an initial denaturation period at 95°C lasting for 15
min, followed by 35 cycles at 94°C for 45 sec, annealing at 60°C
for 30 sec, and extension at 72°C for 45 sec in a thermal cycler
(Applied Biosystems; Thermo Fisher Scientific, Inc.). RT-qPCR was
performed using SYBR Premix Ex Taq™ II (Takara Bio, Inc.) with the
following cycling conditions: Initial denaturation at 95°C or 10
min, followed by 40 cycles of denaturation at 95°C for 45 sec and
annealing at 60°C for 30 sec. The primer sequences were as follows:
NDNF, forward 5′-GGGAAGGCTCAGGTGATCTGGAAC-3′, reverse
5′-AAACCGGATGGGGAACTAGACGAT-3′; and β-actin, forward
5′-GGCACCACACCTTCTACAATGAG-3′, and reverse
5′-GGATAGCACAGCCTGGATAGCA-3′. Each sample was analyzed in
triplicate using the ABI 7300 Real-Time PCR system (Applied
Biosystems; Thermo Fisher Scientific, Inc.). The relative
expression levels of NDNF were calculated using the
2−ΔΔCq method, as previously described (14).
Western blotting
Proteins were extracted from the cells using 1 ml
radioimmunoprecipitation assay buffer (Thermo Fisher Scientific,
Inc.) containing 10 µl protease inhibitor cocktail (Sigma-Aldrich,
Merck KGaA). Protein concentration was quantified using the
bicinchoninic acid assay kit (Pierce; Thermo Fisher Scientific,
Inc.) Proteins (40 µg/sample) were separated by 10% SDS-PAGE and
transferred onto polyvinylidene difluoride membranes (EMD
Millipore, Billerica, MA, USA). Membranes were blocked in 5%
skimmed milk at room temperature for 1 h and were incubated with
the following primary antibodies diluted in PBS with 5% bovine
serum albumin (cat no. 4240GR100; NeoFROXX GmbH, Hesse, German)
overnight at 4ºC: NDNF (1:1,000 dilution, cat no.
sc-242196; Santa Cruz Biotechnology, Inc., Dallas, TX, USA),
β-tubulin (1:5,000 dilution, cat no. ab6046; Abcam, Cambridge, UK),
N-cadherin (1:1,000 dilution, cat no. sc-7939; Santa Cruz
Biotechnology, Inc.), E-cadherin (1:1,000 dilution, cat no.
sc-31020; Santa Cruz Biotechnology, Inc.), Vimentin (1:1,000
dilution, cat no. sc-7557-R; Santa Cruz Biotechnology, Inc.), MMP-7
(1:1,000 dilution, cat no. sc-8832, Santa Cruz Biotechnology),
Cyclin D1 (1:1,000 dilution, cat no. 2978, Cell Signaling
Technology, Inc., Danvers, MA, USA) and β-catenin (1:1,000
dilution, cat no. sc-7963; Santa Cruz Biotechnology, Inc.). After
three washes with TBST [20 mM Tris-HCl (pH 7.5), 150 mM NaCl, 0.1%
Tween-20], membranes were incubated goat anti-rabbit (1:1,000
dilution, cat no. sc-2004, Santa Cruz Biotechnology, Inc.),
rabbit-anti-goat (1:1,000 dilution, cat no. sc-2922, Santa Cruz
Biotechnology, Inc.), or horse anti-mouse (1:1,000 dilution, cat
no. 7076P2, Cell Signaling Technology, Inc.). At room temperature
for 1 h. Bands were detected using the Alliance Imaging system
(Uvitec, Cambridge, UK) with the Immobilon Western Chemiluminescent
HRP Substrate (EMD Millipore).
Immunofluorescence
Immunofluorescence was carried out as previously
described (11). Briefly,
2×103 cells were fixed in ice-cold 4% paraformaldehyde
containing 0.1% Triton X-100 for 5 min, and incubated with a mouse
anti-flag monoclonal antibody (1:5,000 dilution; cat no.
ABIN3020559, Abgent Biotech Co., Ltd., Suzhou, China) at 4°C
overnight. Cells were then washed three times with PBS and
incubated with a donkey anti-rabbit immunoglobulin G secondary
antibody conjugated to Alexa Fluor® 647 (1:1,000
dilution, cat no. A32795 Thermo Fisher Scientific, Inc.). Cell
nuclei were stained with 4′,6-diamidino-2-phenylindol
(Sigma-Aldrich; Merck KGaA, Darmstadt, Germany). Immunofluorescence
was visualized using a confocal fluorescence microscope.
NDNF ELISA
To detect extracellular NDNF released by ACHN and
786-O cells, 2×106 cells were cultured in MEM (Gibco;
Thermo Fisher Scientific, Inc.) and O RPMI 1640 (Gibco)
supplemented with 1% FBS (Gibco; Thermo Fisher Scientific, Inc.)
for 24 h. The supernatant was collected and concentrated by
ultrafiltration at 1,000 × g at room temperature for 20 min.
Subsequently, 10 µl aliquots were analyzed using the commercially
available NDNF ELISA kit (cat no. SEU433 Hu; Cloud-Clone Corp.,
Katy TX, USA), according to the manufacturer's protocol. Each ELISA
plate was read on a microplate reader at 450 nm absorbance, with a
correction wavelength of 540 nm.
Cell proliferation and colony
formation assays
Cell proliferation was assessed using the Cell
Counting Kit-8 assay (Beyotime Institute of Biotechnology).
Lentivirus-infected cells were grown in a 96-well plate at
2×103 cells/well. After 0, 24, 48, 72 and 96 h, the
CCK-8 reagent (10 µl) was added to each well and incubated at 37°C
for 2 h. Then, the absorbance at 450 nm was detected using a
microplate reader (Bio-Rad Laboratories Inc., Hercules, CA, USA).
With regards to the colony formation assays, 1×103
infected ACHN and 786-O cells were seeded in a 6-well plate and
cultured in MEM (Gibco; Thermo Fisher Scientific, Inc.) and RPMI
1640 (Gibco; Thermo Fisher Scientific, Inc.) for 2 weeks,
respectively. Cells were then stained with 0.1% crystal violet for
5 min after fixation with 4% paraformaldehyde for 20 min at room
temperature. Images of the results were captured, and the number of
colonies was counted. Each experiment was performed in
triplicate.
Cell migration and invasion
assays
Cell migration was measured using 24-well Transwell
chambers (pore size, 8 µm; Corning Incorporated, Corning, NY, USA).
In total, 1×105 cells/well were seeded onto the upper
chamber in serum-free medium. With regards to the cell invasion
assay, ~2×105 cells/well were seeded onto the upper
chamber, which was coated with Matrigel (1:8; 50 µl/well; BD
Biosciences, San Jose, CA, USA). The lower chamber was filled with
MEM (Gibco; Thermo Fisher Scientific, Inc.) medium for ACHN and
RPMI 1640 (Gibco; Thermo Fisher Scientific, Inc.) medium for 786-O
containing 10% FBS. After 48 h, the cells on the surface of the
upper chamber were wiped with cotton buds, whereas the cells on the
bottom chamber were fixed in 4% paraformaldehyde for 15 min and
stained with 0.1% crystal violet for 20 min at room temperature.
Images of cell migration and invasion were captured using an
inverted microscope. Eventually, cells contained in each chamber
were removed using 1 ml 33% acetic acid and seeded into 96-well
plates, after which, the absorbance was measured at 570 nm with a
microplate reader. Each experiment was performed at least three
times.
Database mining
The Oncomine database (15) (https://www.oncomine.org/) was used for retrieving the
alterations in NDNF mRNA levels in RCC tissues and corresponding
disease-free normal and/or normal adjacent tissues. Data from Gumz
et al (16), (C) Lenburg et
al (17) and (D) Yusenko et
al (18) were retrieved using
standard settings.
Statistical analysis
All the data are presented as the means ± standard
deviation obtained from three separate experiments. Statistical
analysis was conducted using GraphPad Prism 5.0 (GraphPad Software,
San Diego, CA, USA) and SPSS 22.0 software (SPSS, Chicago, IL,
USA). Paired t-tests were used to analyze the expression levels of
NDNF in ccRCC compared with in adjacent normal kidney tissues, and
nonpaired t-tests were used to determine the associations between
NDNF expression in ccRCC compared to that in normal kidney tissues.
Nonpaired t-tests were used to determine the effects of ectopic
expression on cell function. P<0.05 was considered to indicate a
statistically significant difference.
Results
NDNF expression is reduced in
ccRCC
In our previous study, deep sequencing analysis
demonstrated that NDNF expression is decreased in ccRCC compared
with in paired nontumor tissues (19). To investigate whether NDNF expression
was altered during RCC carcinogenesis, NDNF expression in 69 ccRCC
and paired nontumor tissues was analyzed. Results demonstrated that
NDNF expression was silenced or strongly decreased in 65 of 69
ccRCC samples (P<0.001; Fig. 1A).
An analysis of the Oncomine database also revealed that NDNF
expression was significantly lower in RCC tissues compared with in
normal renal tissues in three independent studies (Fig. 1B-D) (16–18), thus
indicating that NDNF was reduced in ccRCC. NDNF protein was also
examined by western blotting in 16 RCC clinical specimens and
paired nontumor tissues. NDNF expression in RCC was markedly
reduced compared with in paired nontumor tissues (Fig. 1E). These data revealed that the loss
of NDNF expression may be associated with RCC carcinogenesis. Since
the NDNF antibody used for western blotting was not suitable for
immunofluorescence, NDNF expression and subcellular location in
renal tissues were not determined. However, after the infection of
ACHN cells with NDNF-3Flag expression lentiviruses,
immunofluorescence analysis revealed the cytoplasmic location of
NDNF-3Flag (Fig. 1F).
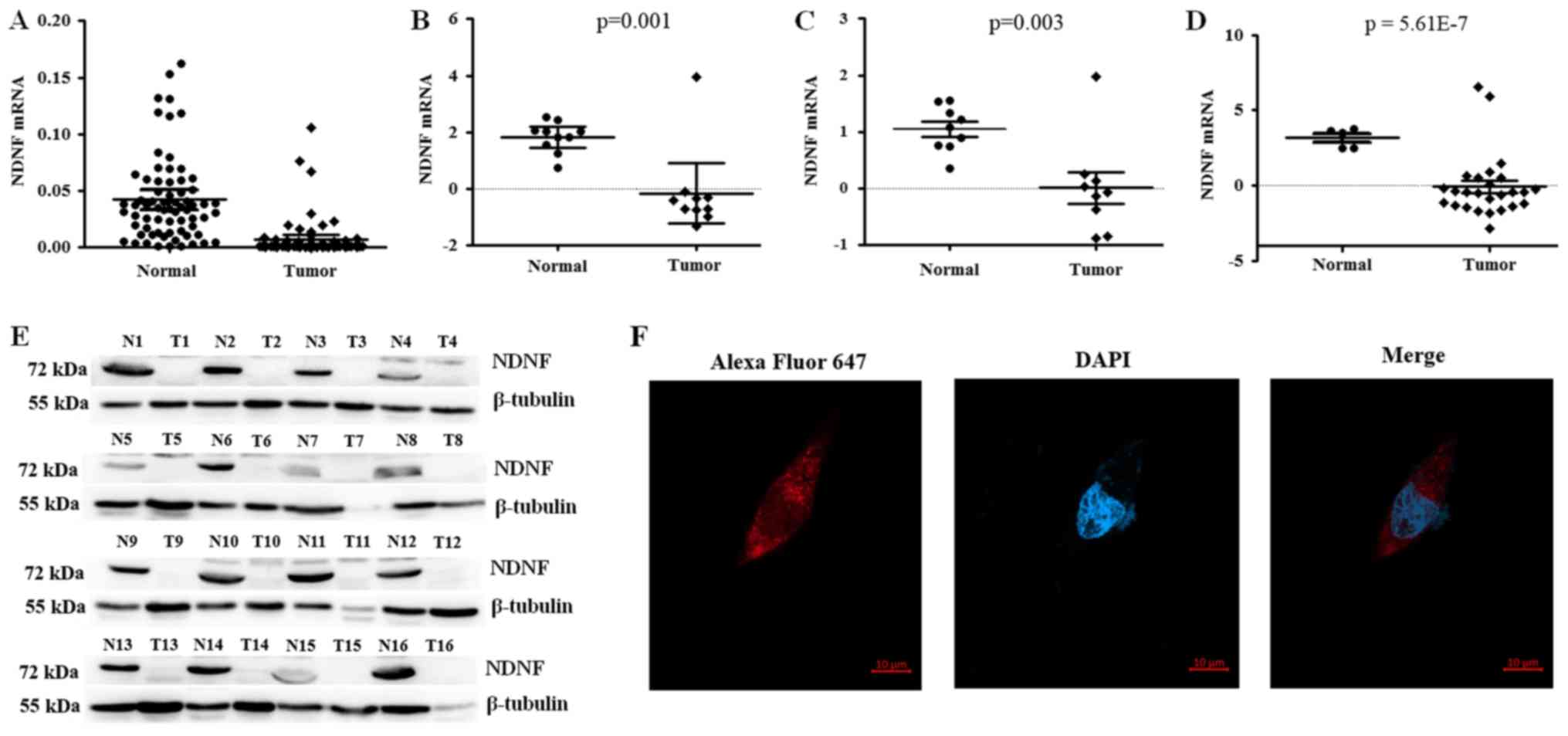 | Figure 1.NDNF expression is reduced in ccRCC.
(A) NDNF expression in 69 ccRCC and paired noncancerous tissues was
determined by reverse transcription-quantitative polymerase
reaction (P<0.0001). The Oncomine database was used to analyze
NDNF expression in previously published RCC datasets: (B) Gumz
et al (16), (C) Lenburg et
al (17) and (D) Yusenko et
al (18) using standard settings.
(E) NDNF protein in ccRCC and paired healthy tissues, as determined
by western blotting. (F) Immunofluorescence staining captured by
fluorescence microscopy, including Alexa Fluor® 647,
DAPI and merged images. The NDNF protein was observed mainly in the
cytoplasm. Scale bar, 10 µm. ccRCC, clear cell RCC; DAPI,
4′,6-diamidino-2-phenylindol; NDNF, neuron-derived neurotrophic
factor; RCC, Renal cell carcinoma. |
Analysis of NDNF expression in human
RCC cell lines
The expression of NDNF in five RCC cell lines (ACHN,
OS-RC-2, Caki-1, 786-O and 769-P) and in the Caki-2 papillary RCC
cell line was determined. The results revealed that Caki-1 and
Caki-2 cell lines expressed high NDNF mRNA expression levels,
whereas no NDNF mRNA was detected in ACHN, OS-RC-2, 786-O and 769-P
cell lines (Fig. 2A). NDNF protein
expression levels were also examined in the RCC cell lines by
western blotting. Notably, no NDNF protein was detected in the six
cell lines while NDNF protein is high in normal kidney tissues,
although Caki-1 and Caki-2 cells exhibited high NDNF mRNA levels
(Fig. 2B). It has been reported that
NDNF is a glycosylated, disulfide-bonded secretory protein and was
detected in cell supernatants (8).
However, no NDNF was detected in any of the RCC cell line
supernatants (data not shown).
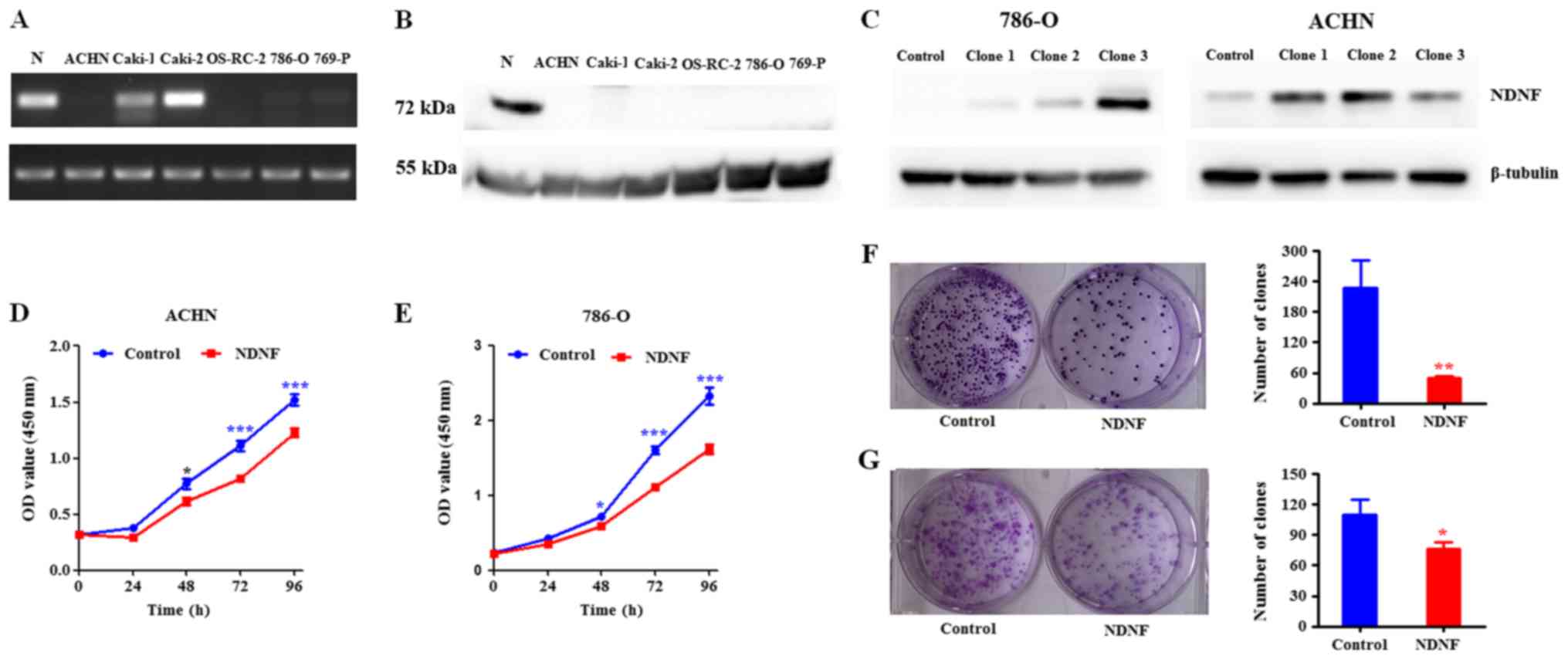 | Figure 2.NDNF suppresses the proliferation of
RCC cells. (A) Expression levels of NDNF mRNA in normal kidney
tissues and various RCC lines (ACHN, Caki-1, Caki-2, 769-P, OS-RC-2
and 786–0) were determined by RT-PCR. (B) NDNF protein expression
in RCC cell lines was determined by western blotting. (C) NDNF
protein expression in single-cell clones infected with NDNF, and in
control cells, was determined by western blotting. β-tubulin was
used as an internal control. (D and E) Effects of ectopic NDNF
expression on ACHN (clone 2) and 786-O (clone 3) cells;
proliferation was detected using the Cell Counting Kit-8 assay.
Experiments were repeated three times. Results are presented as the
means ± SD (*P<0.05, ***P<0.001). (F and G) Effects of
ectopic NDNF expression on the colony formation of ACHN (clone 2)
and 786-O (clone 3) cells. NDNF-stable cells and control cells were
seeded at low density and cultured for 2 weeks. Experiments were
repeated three times. Results are presented as the means ± SD (n=3,
*P<0.05, **P<0.01). NDNF group: Lenti-NDNF infected cells;
Control group: Lenti-Control infected cells. NDNF, neuron-derived
neurotrophic factor; OD, optical density; RCC, Renal cell
carcinoma; SD, standard deviation. |
NDNF suppresses proliferation and
colony formation of RCC cells
Since ACHN and 786-O RCC cell lines exhibited the
lowest mRNA and protein expression levels (Fig. 2A and B), NDNF expression was
reintroduced into human ACHN and 786-O cell lines. RCC infection
with NDNF-bearing lentiviral or empty vectors (control) was
performed. The infected cells were subsequently selected with
puromycin, and single-cell clones were selected to establish stable
cell lines. The ectopic expression of exogenous NDNF was confirmed
by RT-qPCR analysis (data not shown) and western blotting (Fig. 2C). Only a very small amount of NDNF
was detected in the cell culture supernatant (data not shown).
Clones 2 and 3 for 786-O, and clones 1 and 2 for ACHN presented
high NDNF expression, and were therefore selected for further
analysis.
The effects of NDNF on RCC were then assessed by
measuring cell proliferation and colony formation. The results
revealed that ectopic expression of NDNF significantly reduced the
proliferation of ACHN (clone 2) and 786-O (clone 3) in a
time-dependent manner (P<0.05) (Fig.
2D and E). The colony formation assay also revealed that
ectopic expression of NDNF significantly decreased ACHN (clone 2)
(Fig. 2F) and 786-O (clone 3)
(Fig. 2G) colony formation. Similar
results were obtained from ACHN (clone 1) and 786-O (clone 2) cells
(data not shown).
NDNF inhibits the migration and
invasion of RCC cells
To further investigate whether NDNF affects RCC cell
migration and invasion, Transwell migration and invasion assays
were performed. Compared with control cells, ectopic expression of
NDNF significantly suppressed the migratory abilities of ACHN
(clone 2) and 786-O (clone 3) cells (Fig.
3A-D). The Boyden chamber tests demonstrated that the invasive
abilities of ACHN (clone 2) and 786-O (clone 3) cells transfected
with NDNF were decreased compared with control cells (Fig. 3E-H). Similar results were obtained
from ACHN (clone 1) and 786-O (clone 2) cells (data not shown).
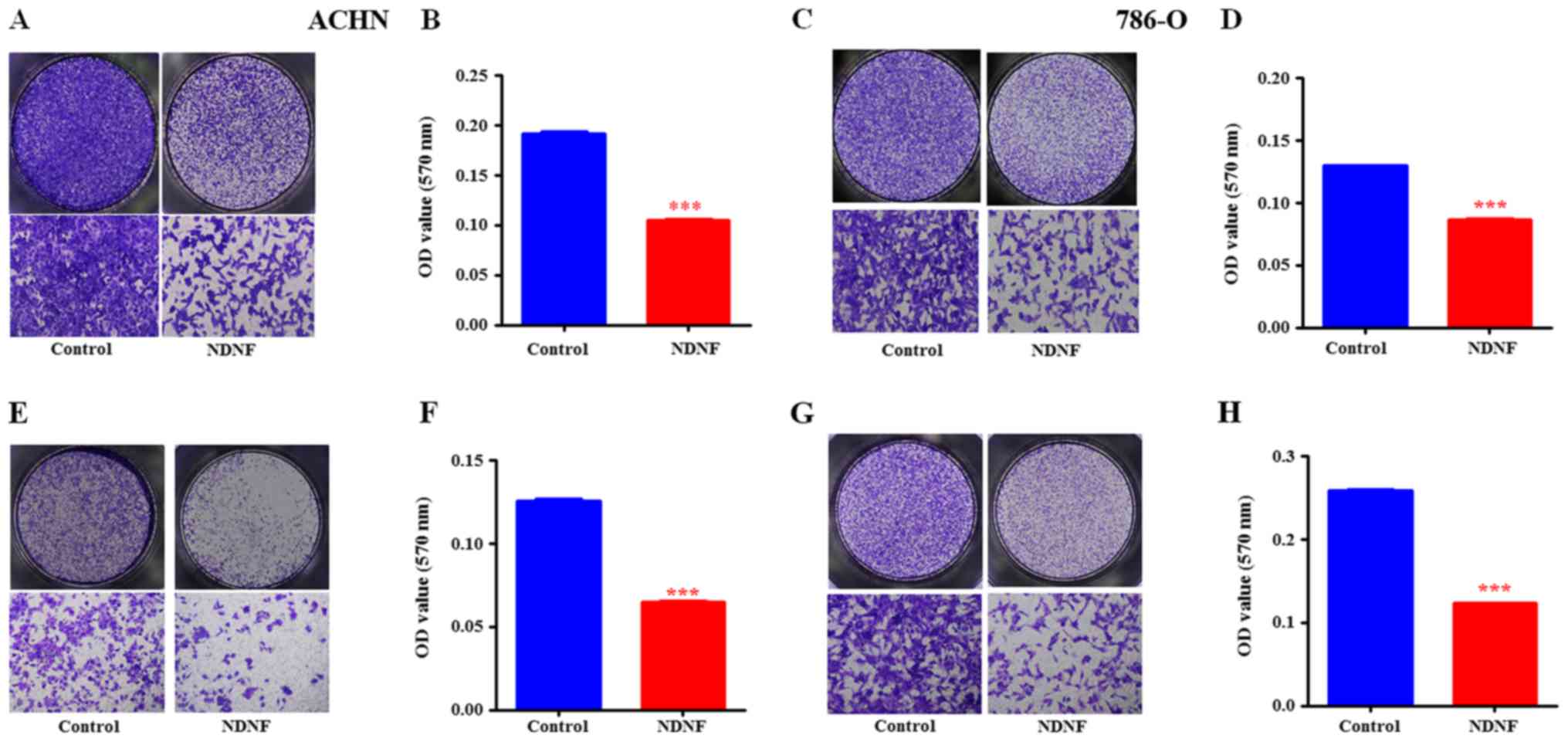 | Figure 3.NDNF inhibits the migration and
invasion of ACHN (clone 2) and 786-O (clone 3) RCC cells. (A)
Representative whole well (×20) and local magnification (×100)
image of effects of NDNF on the migration of ACHN cells, as
determined by Transwell migration assays. (B) The quantitative
analysis of the migrated ACHN cells. (C) Representative whole well
(×20) and local magnification (×100) image of effects of NDNF on
the migration of 786-O cells, as determined by Transwell migration
assays. (D) The quantitative analysis of the migrated 786-O cells.
(E) Representative whole well (×20) and local magnification (×100)
image of effects of NDNF on the invasion ability of ACHN cells, as
determined by Matrigel-coated Transwell invasion assays. (F) The
quantitative analysis of the invaded ACHN cells. (G) Representative
whole well (×20) and local magnification (×100) image of effects of
NDNF on the invasion ability of 786-O cells, as determined by
Matrigel-coated Transwell invasion assays. (H) The quantitative
analysis of the invaded 786-O cells. All experiments were performed
in triplicate. Data are presented as the means ± SD (n=3,
***P<0.001). NDNF group; Lenti-NDNF-infected cells; Control
group, Lenti-Control-infected cells. NDNF, neuron-derived
neurotrophic factor; OD, optical density; RCC, Renal cell
carcinoma; SD, standard deviation. |
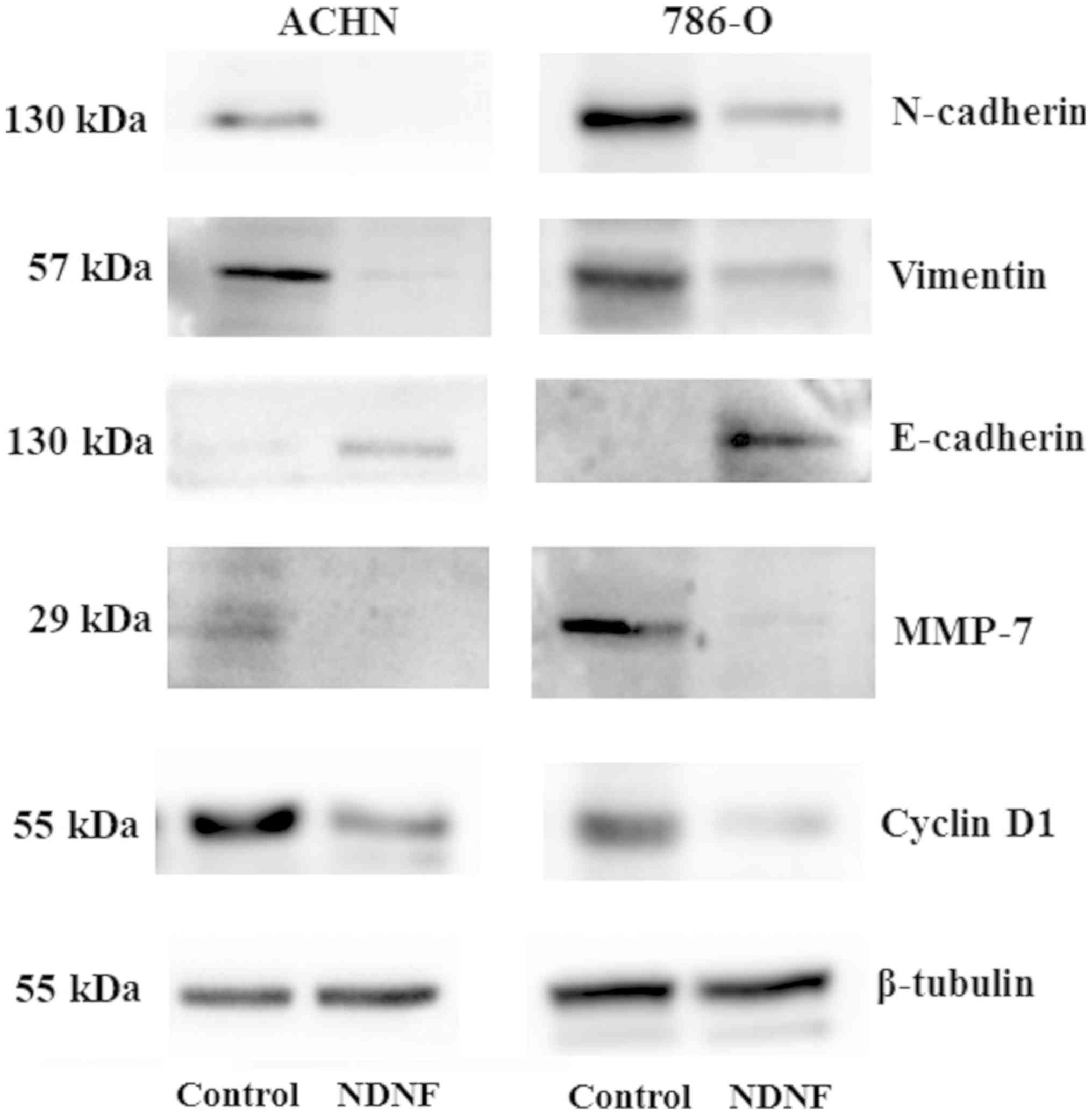
NDNF alters the expression of EMT
markers in RCC cells
EMT serves a critical role in tumor progression by
transforming epithelial cells into a mesenchymal state (11). To determine whether NDNF inhibits RCC
invasion via regulating EMT, the expression levels of the
epithelial marker E-cadherin, and the mesenchymal markers
N-cadherin and vimentin, were examined. Western blotting
demonstrated that ectopic expression of NDNF induced a marked
increase expression in E-cadherin in ACHN and 786-O cells; however,
N-cadherin and Vimentin expression was decreased (Fig. 4).
Since membrane-type matrix metalloproteinases
(MT-MMPs) are pivotal regulators of cell invasion, growth and
survival, the effects of NDNF on MT-MMP expression were examined.
The results revealed that ectopic expression of NDNF markedly
inhibited MMP-7 expression (Fig.
4).
Discussion
Although neurons are considered the primary source
of NDNF (8,9), a previous study revealed that NDNF is
expressed in normal renal tissues and is downregulated in ccRCC
(19). In the present study, NDNF
expression was analyzed in 69 ccRCC samples and paired noncancerous
tissues, and in silico gene expression data screening was
conducted using the Oncomine database (16–18). The
present study confirmed that NDNF was decreased in ccRCC. The
molecular weight of the strongest band for NDNF was determined at
72 kD, although additional unspecific bands were visible. Whether
these unspecific bands are modifications of the NDNF protein
remains unknown. In addition, NDNF antibody available was used only
for western blot analysis and is not suitable for immunohistology
staining. The expression of NDNF in patients with RCC cannot be
correlated with clinical data, e.g. tumor status, grading and
survival. The expression of NDNF in a panel of RCC cell lines was
also assessed. No NDNF protein was detected in the RCC cells,
although NDNF mRNA expression was high in Caki-1 and Caki-2 cells.
This lack of protein detection could be due to a frameshift
mutation or deletion of the NDNF gene, translational regulation via
microRNAs or post-translational modifications. These results
suggested that the reduced NDNF expression may be associated with
RCC carcinogenesis.
To investigate the role of NDNF in RCC
carcinogenesis, functional assays were performed. The results
demonstrated that restoring NDNF expression decreased the cell
proliferation rate by ~20%, and inhibited the migration and
invasion rates by >40%. These data demonstrated that restoring
the expression of NDNF markedly suppressed RCC cell proliferation,
migration and invasion in vitro, although they were in
contradiction with the data observed in neurons (8) and hypoxic endothelial cells (9). This observation may reflect the
tissue-specificity of NDFN. It has been reported that some oncogene
proteins in glioma may act as tumor suppressors in other types of
carcinoma (20–22). In addition, it was demonstrated that
restoring NDNF expression could inhibit Cyclin D1 expression in
ACHN and 786-O RCC cells. Cyclin D1 is able to form active
complexes with either cyclin-dependent kinase (CDK) 4 or CDK6,
which, in turn, drives G1 to S phase progression
(23). NDNF may therefore inhibit RCC
cell proliferation by causing G1 to S phase cell cycle
arrest.
The EMT is a normal process associated with organ
development and disease, including tumor growth and aggressiveness
(7,24,25).
During EMT, epithelial cells become more motile and invasive, which
are characteristics of mesenchymal-like cells (25,26). It is
generally accepted that EMT contributes to tumor development and
progression. In the present study, it was demonstrated that
restoring NDNF expression reduced the expression levels of some
hallmarks of mesenchymal cells, including N-cadherin and Vimentin,
but increased E-cadherin. In addition, ectopic NDNF expression
significantly inhibited the expression of MMP-7, which is known to
cleave E-cadherin (27). It has been
reported that MMP-7 is a downstream target gene of Wnt/β-catenin
signaling, and has an important role in early tumor progression and
metastasis (28). Reducing NDNF
expression may therefore induce RCC invasion and stimulate EMT via
the Wnt/β-catenin signaling pathway. However, NDNF ectopic
expression has a minor effect on the nuclear localization of
β-catenin (data not shown). The effects of NDNF on RCC cell
migration and invasion may therefore involve other pathways,
including the Ras signaling pathway; these other pathways should be
investigated in future studies.
In conclusion, NDNF was significantly downregulated
in ccRCC tissues. In addition, restoring NDNF expression inhibited
RCC proliferation, migration and invasion. Ectopic NDNF expression
also inhibited RCC progression by suppressing the EMT process. NDNF
may therefore act as a regulator of EMT in RCC progression and may
represent a novel therapeutic target for the treatment of advanced
RCC.
Acknowledgements
Not applicable.
Funding
This study was supported by grants from the National
Natural Science Foundation of China (grant no. 81472584), the
National Key Scientific Program of China (grant no. 2014CBA02005),
the Guangdong Key Laboratory funds of Systems Biology and Synthetic
Biology for Urogenital Tumors (grant no. 2017B030301015) and the
Shenzhen Project of Science and Technology (grant no.
20160520174730707).
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
ZL conceived and designed the study. LX, YL, HM, YH,
LW and SL developed the methodology and performed experiments; LX
and BN analyzed the data; ZC, XL and ZL reviewed and interpreted
the data; ZL supervised the study and LX and ZL wrote the
manuscript. All authors read and approved the final manuscript.
Ethics approval and consent to
participate
This research was approved by the Shenzhen Second
People's Hospital Ethics Committee and was conducted according to
Institutional Review Committee guidelines. Written informed consent
was obtained from patients.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
NDNF
|
neuron-derived neurotrophic factor
|
|
RCC
|
Renal cell carcinoma
|
|
EMT
|
epithelial-mesenchymal transition
|
References
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2016. CA Cancer J Clin. 66:7–30. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Cohen HT and McGovern FJ: Renal-cell
carcinoma. N Engl J Med. 353:2477–2490. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Das M: Cabozantinib improves clinical
outcomes in renal cancer. Lancet Oncol. 18:e22017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Liu S, Tian Z, Zhang L, Hou S, Hu S, Wu J,
Jing Y, Sun H, Yu F, Zhao L, et al: Combined cell surface carbonic
anhydrase 9 and CD147 antigens enable high-efficiency capture of
circulating tumor cells in clear cell renal cell carcinoma
patients. Oncotarget. 7:59877–59891. 2016.PubMed/NCBI
|
|
5
|
Xu G, Lu X, Huang T and Fan J: ARHGAP24
inhibits cell cycle progression, induces apoptosis and suppresses
invasion in renal cell carcinoma. Oncotarget. 7:51829–51839.
2016.PubMed/NCBI
|
|
6
|
El-Mokadem I, Kidd T, Pratt N, Fleming S
and Nabi G: Tumour suppressor gene (CDKNA2) status on chromosome 9p
in resected renal tissue improves prognosis of localised kidney
cancer. Oncotarget. 7:73045–73054. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Tang H, Chen Y, Liu X, Wang S, Lv Y, Wu D,
Wang Q, Luo M and Deng H: Downregulation of HSP60 disrupts
mitochondrial proteostasis to promote tumorigenesis and progression
in clear cell renal cell carcinoma. Oncotarget. 7:38822–38834.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kuang XL, Zhao XM, Xu HF, Shi YY, Deng JB
and Sun GT: Spatio-temporal expression of a novel neuron-derived
neurotrophic factor (NDNF) in mouse brains during development. BMC
Neurosci. 11:1372010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Ohashi K, Enomoto T, Joki Y, Shibata R,
Ogura Y, Kataoka Y, Shimizu Y, Kambara T, Uemura Y, Yuasa D, et al:
Neuron-derived neurotrophic factor functions as a novel modulator
that enhances endothelial cell function and revascularization
processes. J Biol Chem. 289:14132–14144. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Joki Y, Ohashi K, Yuasa D, Shibata R,
Kataoka Y, Kambara T, Uemura Y, Matsuo K, Hayakawa S, Hiramatsu-Ito
M, et al: Neuron-derived neurotrophic factor ameliorates adverse
cardiac remodeling after experimental myocardial infarction. Circ
Heart Fail. 8:342–351. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Dongre A and Weinberg RA: New insights
into the mechanisms of epithelial-mesenchymal transition and
implications for cancer. Nat Rev Mol Cell Biol. 20–Nov;2018.(Epub
ahead of print).
|
|
12
|
Xie J, Zhu C, Wu J, Li C, Luo L, Xia L, Li
X, Gui Y, Cai Z and Li Z: Down-regulation of C12orf59 is associated
with a poor prognosis and VHL mutations in renal cell carcinoma.
Oncotarget. 7:6824–6834. 2016.PubMed/NCBI
|
|
13
|
Luo L, Wu J, Xie J, Xia L, Qian X, Cai Z
and Li Z: Downregulated ECRG4 is associated with poor prognosis in
renal cell cancer and is regulated by promoter DNA methylation.
Tumour Biol. 37:1121–1129. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Rhodes DR, Kalyana-Sundaram S, Mahavisno
V, Varambally R, Yu J, Briggs BB, Barrette TR, Anstet MJ,
Kincead-Beal C, Kulkarni P, et al: Oncomine 3.0: Genes, pathways,
and networks in a collection of 18,000 cancer gene expression
profiles. Neoplasia. 9:166–180. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Gumz ML, Zou H, Kreinest PA, Childs AC,
Belmonte LS, LeGrand SN, Wu KJ, Luxon BA, Sinha M, Parker AS, et
al: Secreted frizzled-related protein 1 loss contributes to tumor
phenotype of clear cell renal cell carcinoma. Clin Cancer Res.
13:4740–4749. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Lenburg ME, Liou LS, Gerry NP, Frampton
GM, Cohen HT and Christman MF: Previously unidentified changes in
renal cell carcinoma gene expression identified by parametric
analysis of microarray data. BMC Cancer. 3:312003. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yusenko MV, Kuiper RP, Boethe T, Ljungberg
B, van Kessel AG and Kovacs G: High-resolution DNA copy number and
gene expression analyses distinguish chromophobe renal cell
carcinomas and renal oncocytomas. BMC Cancer. 9:1522009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zhou L, Chen J, Li Z, Li X, Hu X, Huang Y,
Zhao X, Liang C, Wang Y, Sun L, et al: Integrated profiling of
microRNAs and mRNAs: microRNAs located on Xq27.3 associate with
clear cell renal cell carcinoma. PLoS One. 5:e152242010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Pickard A and McCance DJ: IGF-binding
protein 2-oncogene or tumor suppressor? Front Endocrinol
(Lausanne). 6(25)2015.PubMed/NCBI
|
|
21
|
Echizen K, Nakada M, Hayashi T, Sabit H,
Furuta T, Nakai M, Koyama-Nasu R, Nishimura Y, Taniue K, Morishita
Y, et al: PCDH10 is required for the tumorigenicity of glioblastoma
cells. Biochem Biophys Res Commun. 444:13–18. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Li Z, Chim JC, Yang M, Ye J, Wong BC and
Qiao L: Role of PCDH10 and its hypermethylation in human gastric
cancer. Biochim Biophys Acta. 1823:298–305. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Musgrove EA, Caldon CE, Barraclough J,
Stone A and Sutherland RL: Cyclin D as a therapeutic target in
cancer. Nat Rev Cancer. 11:558–572. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Galichon P, Finianos S and Hertig A:
EMT-MET in renal disease: Should we curb our enthusiasm? Cancer
Lett. 341:24–29. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Piva F, Giulietti M, Santoni M, Occhipinti
G, Scarpelli M, Lopez-Beltran A, Cheng L, Principato G and
Montironi R: Epithelial to mesenchymal transition in renal cell
carcinoma: Implications for cancer therapy. Mol Diagn Ther.
20:111–117. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Han WQ, Zhu Q, Hu J, Li PL, Zhang F and Li
N: Hypoxia-inducible factor prolyl-hydroxylase-2 mediates
transforming growth factor beta 1-induced epithelial-mesenchymal
transition in renal tubular cells. Biochim Biophys Acta.
1833:1454–1462. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
McGuire JK, Li Q and Parks WC: Matrilysin
(matrix metalloproteinase-7) mediates E-cadherin ectodomain
shedding in injured lung epithelium. Am J Pathol. 162:1831–1843.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Tan RJ, Zhou D, Zhou L and Liu Y:
Wnt/β-catenin signaling and kidney fibrosis. Kidney Int Suppl
(2011). 4:84–90. 2014. View Article : Google Scholar : PubMed/NCBI
|