Introduction
Colorectal cancer (CRC) is the third most common
type of cancer and the second leading cause of cancer-associated
mortality worldwide. In 2017, 1,688,780 new cancer cases and
600,920 cancer-associated deaths were projected to occur in the
United States (1). CRC represents
8.4% of all novel cancer cases and has emerged as a public health
concern due to its high morbidity and mortality rates. According to
estimates from the National Cancer Institute, there are 145,600
novel cases of colon and rectal cancer every year, and nearly
51,020 Americans die per year as a result of CRC. Despite this, the
5-year relative survival rate of CRC has increased from 48.6 to
64.4% over past decades, potentially due to increased awareness of
risk factors and widespread implementation of endoscopic screening
(1). Researchers are working to
advance understanding of how to prevent, detect and treat CRC. To
the best of our knowledge, however, the precise aetiology of CRC
and the underlying molecular mechanism remain unclear. Previous
studies have shown that disturbance of sphingosine-1-phosphate
(S1P) signalling and metabolism is associated with the inflammatory
response and numerous types of disease, including the development
and progression of cancer (2–10).
S1P is a membrane-derived bioactive phospholipid synthesized by
sphingosine kinases (SPHKs) 1 and 2 from sphingosine and degraded
by S1P lyase in mammalian cells (11). S1P is exported from cells and
serves as a signal transducer via G protein-coupled S1P receptors
(S1PRs) 1–5 or activates intracellular targets directly to regulate
cell homeostasis and biological functions, including cell
proliferation, migration, invasion and control of immune cell
trafficking and angiogenesis (12). The aforementioned steps, which make
up the S1P axis, might protect patients from colon cancer (13). Data from human tumour cell lines
and tissue show that aberrant expression of S1P-regulating enzymes
and receptors is a key initiating event for malignant
transformation and colon cancer progression (14,15).
S1PR1, S1PR2 and S1PR3 are ubiquitously expressed
S1PRs that mediate diverse functions of S1P in multiple types of
cell, including regulating proliferation and migration, whereas
S1PR4 and S1PR5 are restricted to the immune and nervous systems,
respectively (16). S1P binds to
different subtypes of S1PRs to elicit biological responses
(9). These receptors are
exclusively coupled to heterotrimeric G proteins and Rho or Rac to
control various effector systems, including adenylate cyclase,
phospholipases C and D and extracellular-regulated p38
mitogen-activated protein, c-Jun N-terminal and non-receptor
tyrosine kinase (17–19). S1PR1 and S1PR3 mediate potent
stimulation of migration and invasion of tumour cells by inducing
ERK activation, whereas S1PR2 exhibits an inhibitory effect via Rho
activation and Rac inhibition (20–23).
Therefore, the role of S1PRs in cancer remains controversial.
Studies have shown expression or mutation of S1PRs
in cancer (24–27). Yoshida et al (24) demonstrated that downregulated
expression of S1PR1 increases proliferative activity to enhance the
malignancy of tumours, resulting in poor survival in patients with
glioblastoma. Kothapalli et al (25) reported that the S1P5 gene is
overexpressed in large granular lymphocyte leukaemia and may
protect against apoptosis of these cancer cells. Flori et al
(27) suggested that S1PR2 is a
novel tumour suppressor and survival prognosticator in activated B
cell-like diffuse large B-cell lymphoma. To the best of our
knowledge, however, little is known about the biological function
of S1PR2 and its potential role in CRC. To determine the molecular
pathogenesis of CRC, the present study performed functional
analysis of sphingolipids, focusing on the role of S1PR2 in colon
cancer cell line proliferation and migration. Expression of S1P,
its target receptor S1PR1-5 and its synthetase SPHKs in human
colorectal carcinoma tissue was assessed and the association
between expression of S1PR2 and clinical variables was determined.
The study findings may provide new insight into the development of
new anti-CRC drugs through regulation of the S1P/S1PR2 axis.
Materials and methods
Human specimens
Fresh colon cancer (n=55) and matched non-tumourous
colon tissue (NT) (n=55) were collected following surgical
resection at the First Affiliated Hospital of Nanjing Medical
University (Nanjing, China) from 2010 to 2015. The NT tissue was
obtained at the resection margin, located at a distance of >5 cm
away from the primary tumour. The mean age at diagnosis was
64.00±11.91 years. Plasma was collected from patients with colon
cancer (n=55) and sex- and age-matched healthy donors (n=55; mean
age, 74.51±8.42 years; 25 male and 20 female). Tissues were washed
in ice-cold PBS and a portion of tissues were formalin-fixed and
paraffin-embedded. The rest of the tissue and plasma samples were
stored at −40°C. All patients had a histological diagnosis of colon
cancer and none had received therapy. All specimens were obtained
with verbal consent from the patients and healthy donors, and the
study was approved by the Institutional Ethics Committee of Nanjing
Medical University (approval no. 2016-SR-217).
Immunohistochemical (IHC)
staining
As 5 of the specimens were not large enough, 50
cases were included in the IHC assay. The tissues were fixed with
4% paraformaldehyde (PFA) at room temperature for 20 min and
embedded into paraffin. Tissue paraffin blocks were serially
sectioned at 4 µm. Sections were deparaffinized at 60°C for 30 min
and washed in xylene, and rehydrated in a descending alcohol
series. Tissue sections were placed in a repair box filled with
EDTA antigen repair buffer (pH 9.0) (Wuhan Goodbio Technology, Co.,
Ltd.; cat. no. G12036), and antigen retrieval was performed by
microwaving for 15 min, endogenous peroxidase was blocked at room
temperature with 3% hydrogen peroxide in methanol for 25 min and
tissue was blocked with normal rabbit serum (Gibco; Thermo Fisher
Scientific, Inc.; cat. no. 16120107; 1:10) for 30 min at room
temperature. CRC tissues were incubated with anti-S1PR2 primary
antibody (Santa Cruz Biotechnology, Inc.; cat. no. sc-365589;
1:500) overnight at 4°C. Sections were washed with PBS and
incubated with an HRP-conjugated secondary antibody (Invitrogen;
Thermo Fisher Scientific, Inc.; cat. no. A16160; 1:1,000) at room
temperature for 50 min. After being washed with PBS, the target
protein was stained with 1% 3,3′-diaminobenzidine(DAKO; Shanghai Li
Min Industrial Co., Ltd.; cat. no. K5007) at room temperature for 5
min, and cell nuclei were counterstained blue with haematoxylin at
room temperature for 3 min. The slides were scanned using the
Pannoramic Midi II histological scanner 1.18.1 (3DHISTECH Ltd.).
Next, using the Pannoramic Viewer Program 1.15.3 (3DHISTECH Ltd.),
representative areas were selected. A light microscope (IX73;
Olympus) was used for imaging at ×100 and ×200 magnification. The
staining score for tumour and adjacent normal tissue (0, negative;
1+, weak; 2+, moderate and 3+, strong) was recorded separately. The
estimated proportion of positive tumour cells was calculated as a
percentage. To assess the mean degree of staining within a tumour,
three representative regions were analysed and ≥100 tumour cells
were assessed. S1PR2 expression was assessed by histochemistry
(H)-score system as follows: H-score=∑(I xPi), where I=intensity of
staining and Pi=percentage of stained tumour cells, producing a
score ranging from 0 to 300. The scoring was independently assessed
by two investigators who were blinded to the patient
characteristics and outcomes.
Reverse transcription-quantitative
(RT-q)PCR
Total RNA was extracted from tissue with an RNA
Extraction kit (Takara Biotechnology Co., Ltd.; cat. no. 9767)
according to the manufacturer's protocol. Total RNA was quantified
using a spectrophotometer (Nanodrop ND-1000; Thermo Fisher
Scientific, Inc.). Only RNA with a 260/280 ratio of 1.8-2.0 was
used for cDNA synthesis and reverse transcribed into cDNA using
PrimeScript™ RT Master Mix (Takara Biotechnology Co.,
Ltd.; cat. no. RR036A) at 37°C for 15 min. mRNA levels of target
genes were detected by qPCR using SYBR premix Ex Taq™
(Takara Biotechnology Co., Ltd.; cat. no. 638319) on a
StepOnePlus™ Real-Time PCR System (Applied Biosystems;
Thermo Fisher Scientific, Inc.). The following thermocycling
conditions were used for qPCR: Initial denaturation at 95°C for 30
sec, followed by 40 cycles of denaturation at 95°C for 5 sec,
annealing at 60°C for 30 sec and extension at 60°C for 1 min.
Following normalization using β-actin as an internal control, the
data were analysed using the ΔΔCq method and expressed as target
gene/internal ratio [2−ΔCq(target gene-internal)]
(28). The primers were as
follows: S1PR1 forward, 5′-TCCTCGCCATCGCCATTG-3′ and reverse,
5′-GAGAGCAGAAGCAGAGTGAAG-3′; S1PR2 forward,
5′-CAAGGTCCAGGAACACTATA-3′and reverse, 5′-AACAGAGGATGACGATGAA-3′;
S1PR3 forward, 5′-CTACGCACGCATCTACTTCC-3′ and reverse,
5′-CACGCTCACCACAATCACC-3′; S1PR4 forward, 5′-GCTGAAGACGGTGCTGATG-3′
and reverse, 5′-CTGCTGCGGAAGGAGTAG-3′; S1PR5 forward,
5′-CTTCCTGCTGCTGTTGCTC-3′ and reverse, 5′-GCCACTCGGGTCTCTGC-3′;
SPHK1 forward, 5′-TGTGTAGCCTCCCAGCAG-3′ and reverse,
5′-CCCAGACGCCGATACTTC-3′; SPHK2 forward, 5′-ACTGCCCTCACCTGTCTG-3′
and reverse, 5′-TTCTGTCGTTCTGTCTGGATG-3′ and β-actin forward,
5′-TGACGTGGACATCCGCAAAG-3′ and reverse,
5′-CTGGAAGGTGGACAGCGAGG'.
Quantification of S1P
Quantification of S1P from serum was performed by
ELISA using a Human S1P Assay kit (Nanjing SenBeiJia Biological
Technology Co., Ltd.; cat. no. SBJ-H2060-96T) according to the
manufacturer's instructions.
Cell culture and treatment
CRC SW480 and LOVO cell lines, purchased from
Shanghai Institutes for Biological Sciences, Chinese Academy of
Sciences, were maintained in DMEM supplemented with 10% foetal
bovine serum (Invitrogen; Thermo Fisher Scientific, Inc.), 100 U/ml
penicillin G and 100 mg/ml streptomycin (Sigma-Aldrich; Merck
KGaA). All cells were cultured at 37°C with 5% CO2.
Cells were grown in 100-mm dishes and incubated at 37°C in DMEM for
24 h. The small interfering RNA (siRNA) oligonucleotide duplexes
(si-S1PR2: 5′-CCUUCGUAGCCAAUACCUUTT-3′; negative control,
5′-UUCUCCGAACGUGUCACGUTTACGUGACACGUUCGGAGAATT-3′; Shanghai
GenePharma Co., Ltd.) were transfected into cells using
Lipofectamine® 2000 (cat. no. 11668019; Invitrogen;
Thermo Fisher Scientific, Inc.). S1PR2 siRNA (120 pmol) and
transfection reagent (6 µl) were mixed in 300 µl serum-free DMEM,
left to stand for 5 min and then mixed again. Following incubation
at room temperature for 20 min, the mix was added to serum-starved
cells and incubated at 37°C for 4 h. CRC cells were plated the day
before transfection at 400,000 cells/well in 6-well plates in
complete growth medium (Gibco; Thermo Fisher Scientific, Inc.).
S1P, JTE013 and FTY720 were purchased from Cayman Chemical Company
and dissolved in dimethyl sulfoxide (DMSO; Sigma-Aldrich; Merck
KGaA). Cells were pre-treated at 37°C with S1PR2 antagonist JTE013
(5 µM), S1PR1,3,4,5 antagonist FTY720 (5 µM) or DMSO (1%) for 1 h
before stimulation in the presence or absence of S1P (1 µM) at 37°C
for 1 h. DMSO was applied as vehicle. Cells were harvested 24 h
later for migration and invasion assay.
Cell viability assay
Human CRC SW480 cells (5×103 cells/well)
were seeded into 96-well plates, allowed to adhere and
serum-starved overnight at 37°C in 95% air and 5% CO2.
Next, cells were transfected with S1PR2-specific siRNA and treated
with S1P (1 µM) or DMSO. Following 24 h incubation at 37°C, cells
were stained with sterile MTT for 4 h at 37°C, the supernatant was
collected by centrifugation at 13,225 × g for 2 min at 4°C and
removed, and the crystals formed were dissolved using DMSO. The
absorbance at 490 nm was examined.
Wound healing assay
SW480 or LOVO cells were plated at a density of
5×105 cells/well in 6-well culture dishes and allowed to
form a monolayer (70–90% confluence). Following serum-starvation
for 24 h, the cells were scratched with a sterile pipette tip
(200-µl), washed with PBS to remove floating and detached cells and
photographed (time, 0 h) under the 10× objective of an Olympus 1X71
light microscope (Olympus Corporation). Cells were pre-treated with
JTE-013 (5 µM), FTY720 (5 µM) or DMSO for 1 h and treated with S1P
(1 µM) or DMSO. After 24 h, the wound area was. The assay was
performed three times. ImageJ v1.8.0 software (National Institutes
of Health) was used to analyse the cell-free areas in images. A
total of 6–8 horizontal lines were drawn on each image and the
width was calculated.
Transwell cell invasion assay
After being starved overnight, SW480 cells
(6×103 cells/well) were seeded in a Matrigel-coated
upper polycarbonate chamber (8-µm pore size; BD Biocoat Matrigel
Invasion Chamber). The chambers were precoated with 20% Matrigel
for 30 min at 37°C. The upper chamber was filled with serum-free
DMEM and the lower chamber was filled with medium containing 10%
FBS (Gibco-BRL, Rockville, MD, USA). Cells were pre-treated with
JTE-013 (5 µM), FTY720 or vehicle DMSO for 1 h, then treated with
S1P (1 µM) or DMSO and incubated at 37°C in 95% air and 5%
CO2. After 24 h, non-invasive cells were washed off
twice with PBS. The cells that had migrated to the lower surface
were fixed for 30 min with 3.7% paraformaldehyde at 25°C and
stained for 20 min using 0.1% crystal violet solution at 25°C, and
then observed and images captured at ×200 magnification on a light
microscope. For each replicate (n=3), migrated cells were manually
counted and expressed as the average cell number from 4 random
fields. The cell number indicated the cell migration capability and
invasiveness.
Western blotting
Tissues samples were harvested and lysed in ice-cold
lysis buffer (1% Nonidet P-40, 0.5% sodium cholate, 1 mM EDTA, 1 mM
EGTA, 150 mM NaCl, 20 mM HEPES, 3 mM MgCl, 1 mM PMSF, 20 mM
b-glycerophosphate, 1 mM NaF and 1 mM sodium orthovanadate; pH 7.4)
and sonicated (20 kHz; 0°C; 30 sec). Following centrifugation at
13,225 × g for 20 min at 4°C, protein concentrations were assayed
using Bradford protein assay reagent (Bio-Rad Laboratories, Inc.)
with BSA as a standard. Total protein (30 µg) was separated on a
10% SDS-PAGE gel and transferred to a PVDF membrane (Millipore).
The membranes were blocked with 5% non-fat milk in TBST [10 mM Tris
(pH 7.4), 100 mM NaCl and 0.5% Tween 20] for 1 h at room
temperature and incubated with the primary antibody S1PR2 (Santa
Cruz Biotechnology, Inc.; cat. no. sc-365589; 1:1,000). Protein
bands were normalized to GAPDH and detected with anti-GAPDH (Santa
Cruz Biotechnology, Inc.; cat. no. sc-47724; 1:1,000).
Immunoreactive bands were detected using rabbit anti-mouse IgG
HRP-conjugated secondary antibody (Santa Cruz Biotechnology, Inc.;
cat. no. sc-358914; 1:5,000) and SuperSignal Chemiluminescent
Substrate (Thermo Fisher Scientific, Inc.). The densities of bands
were analysed using Image Lab software.
Statistical analysis
Experiments were repeated ≥3 times and data are
presented as the mean and standard error of the mean (SEM).
Statistical analysis for multiple comparisons was performed by
one-way ANOVA followed by post hoc Bonferroni's correction. The
differences in levels of S1P between the CRC group and healthy
donor group were analysed using an unpaired Student's t-test. S1PR2
expression in tumour and matched normal tissues were compared using
Wilcoxon's signed-rank test. P<0.05 was considered to indicate a
statistically significant difference. All statistical analysis was
performed using GraphPad Prism 8.0 (GraphPad Software, Inc.)
software.
Results
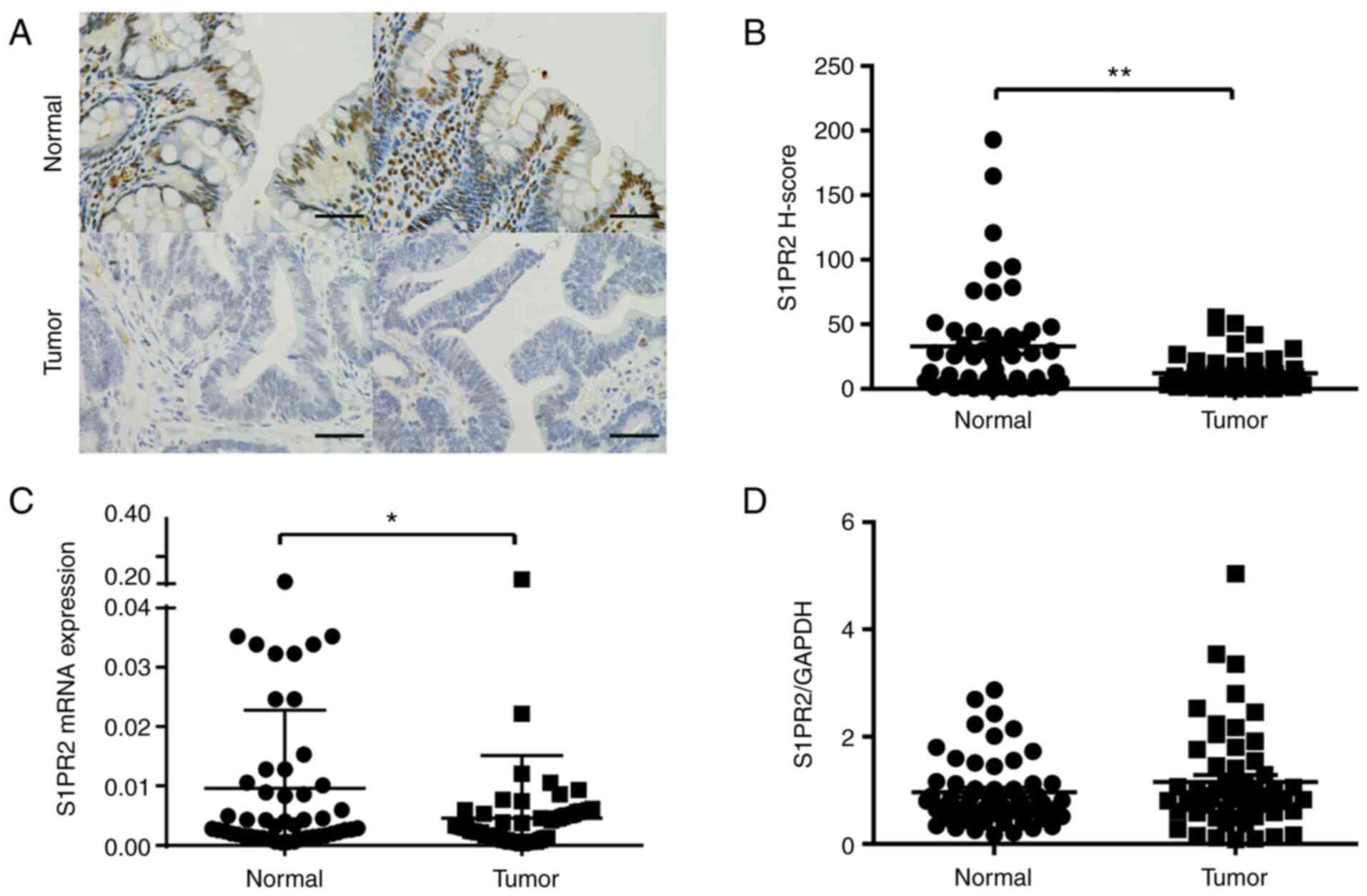
Expression of S1PR2 is downregulated
in human CRC
To test whether S1PR2 serves a role in human CRC,
IHC analysis of S1PR2 in human colon tissue was performed. As 5 of
the specimens were not large enough, 50 cases were included in the
IHC assay. CRC specimens (n=50) had notably lower levels of S1PR2
than normal human colon tissue (n=50), which exhibited strong
S1PR2-positive staining in colon cells (Fig. 1A). IHC staining was performed to
evaluate expression of S1PR2 in tumour and adjacent normal tissue
of patients with CRC using H-score as a continuous variable. Mean
H-score was higher in normal compared with tumour tissue (33.11 vs.
12.23; P<0.001). S1PR2 expression in human CRC tissue was
significantly lower in comparison with that in paired normal colon
tissue (n=50; Fig. 1B). Next,
expression levels of S1PR2 mRNA in tumour and adjacent
non-tumourous tissue of patients with CRC were assessed using
RT-qPCR (n=55). S1PR2 was significantly downregulated in colon
cancer compared with non-tumourous tissue, with a log2 expression
ratio of −3.46 (tumour vs. normal; Fig. 1C). The protein level of S1PR2 in
colon tissue was determined using western blotting (Fig. 1D). Among patients with CRC (n=55),
downregulated S1PR2 was observed in 32 (58%) cases; however; this
was not significantly different (P=0.45; Wilcoxon's signed-rank
test). The mean ratio of S1PR2/GAPDH was slightly higher in tumour
compared with normal tissue (1.15 vs. 0.96) but this was not
significantly different. Taken together, these data showed that
S1PR2 was downregulated in CRC cells and low S1PR2 expression may
serve a role in the development of colon cancer.
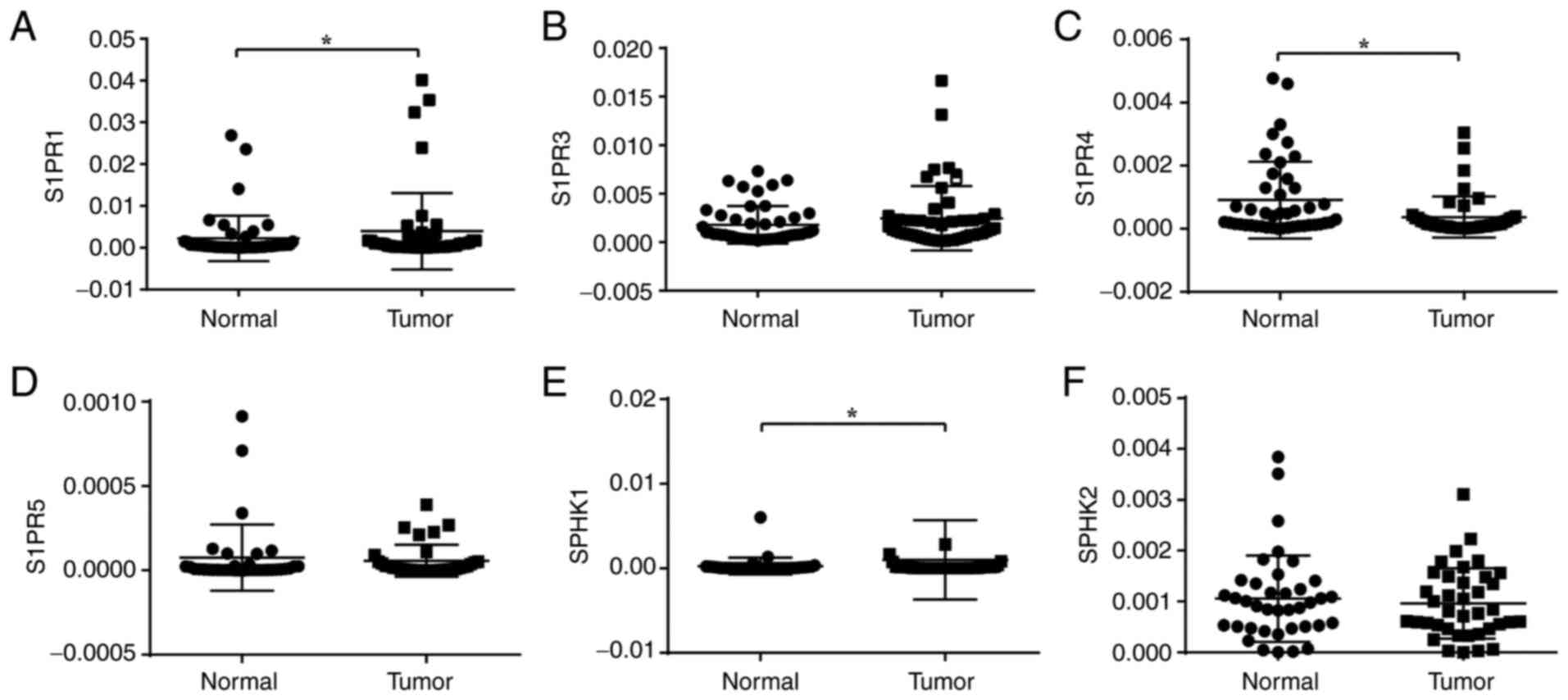
Expression of S1PRs and SPHKs in human
CRC
To confirm the role of the S1P axis in colon cancer,
mRNA expression levels of S1PR1, S1PR3, S1PR4 and S1PR5 were
measured in the tumour and adjacent non-tumourous tissue of
patients with CRC using RT-qPCR (Fig.
2A-D). S1PR1 and S1PR3 were highly expressed S1PR subtypes in
human colon tissue, whereas S1P4 and S1P5 were barely detectable.
Transcript levels for S1PR1 gene were significantly higher in
tumour than in normal tissue, while those of S1PR4 gene were lower
(P<0.05). Transcripts of S1PR3 gene were higher in tumour tissue
but there was no significant difference (P=0.0748). The results
were consistent with previous studies that suggested that S1PR1 and
S1PR3 are upregulated in numerous types of cancer (27). mRNA expression levels of SPHK1 and
SPHK2 in colon tissue were measured (Fig. 2E and F). SPHK1 gene expression was
significantly upregulated in tumour tissue, which is consistent
with previous data (29). These
results demonstrated variation in expression of S1PR and
sphingolipid metabolism enzymes in colon cancer.
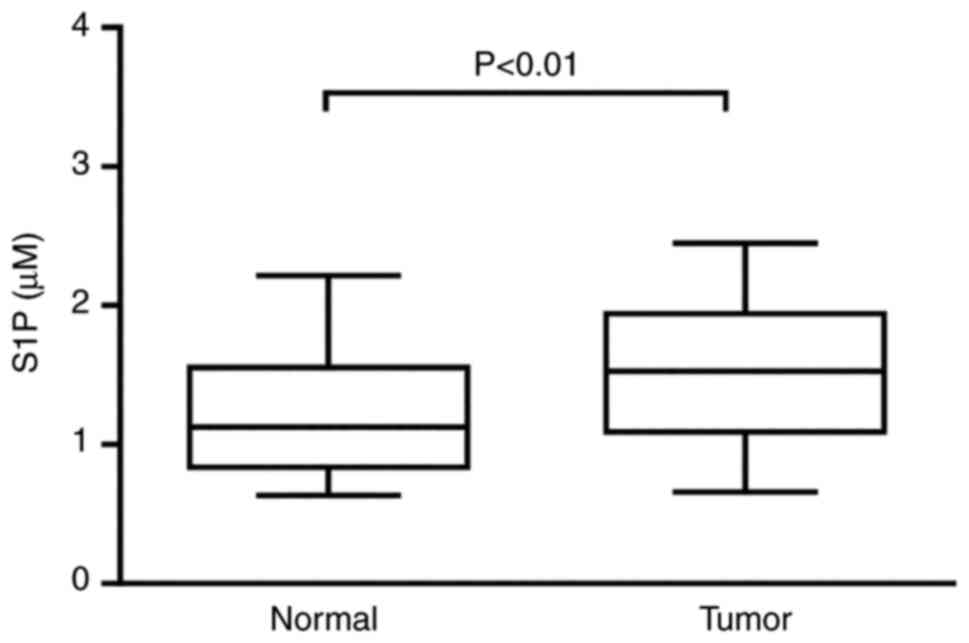
Level of S1P is higher in patients
with CRC
Activation of SPHK1 leads to synthesis and secretion
of S1P, which has been shown to influence cellular function via
interactions with specific receptors, including S1PR2 (30). Expression of S1P in the peripheral
blood of patients with CRC may be altered via the SPHK/S1P/S1PR
signalling pathway. Therefore, S1P levels were measured in serum
from patients with CRC (n=55) and healthy individuals (n=55) using
ELISA. S1P level were higher in patients with CRC (Fig. 3).
Migration of human colon cell lines is
increased by inhibition of S1PR2
S1P has been shown to regulate cellular
physiological functions, including proliferation and migration, in
different types of cancer (4). To
determine the effect of exogenous S1P on the colon cancer cell
lines SW480 and LOVO in vitro, wound healing assay was
performed to investigate the role of S1P in colon cancer cell
migration. LOVO cells treated with 1 µM S1P showed a significant
increase in migration distance, whereas that of SW480 cells treated
with 1 µM S1P was significantly inhibited (Fig. 4A). JTE013, a specific S1PR2
antagonist, further promoted migration of SW480 and LOVO cells
stimulated by S1P (Fig. 4A and C).
To confirm the role of S1PR2 in cell migration, siRNA was used to
specifically silence S1PR2 expression. Relative to the vehicle
control, knockdown of S1PR2 by siRNA significantly promoted
migration of SW480 and LOVO cells, which is consistent with the
aforementioned results (Fig. 4B and
D). Inhibition of S1PR2 by JTE013 or S1PR2-siRNA significantly
promoted migration of SW480 and LOVO cells. These data indicated
that expression of S1PR2 may inhibit the migration of colon cancer
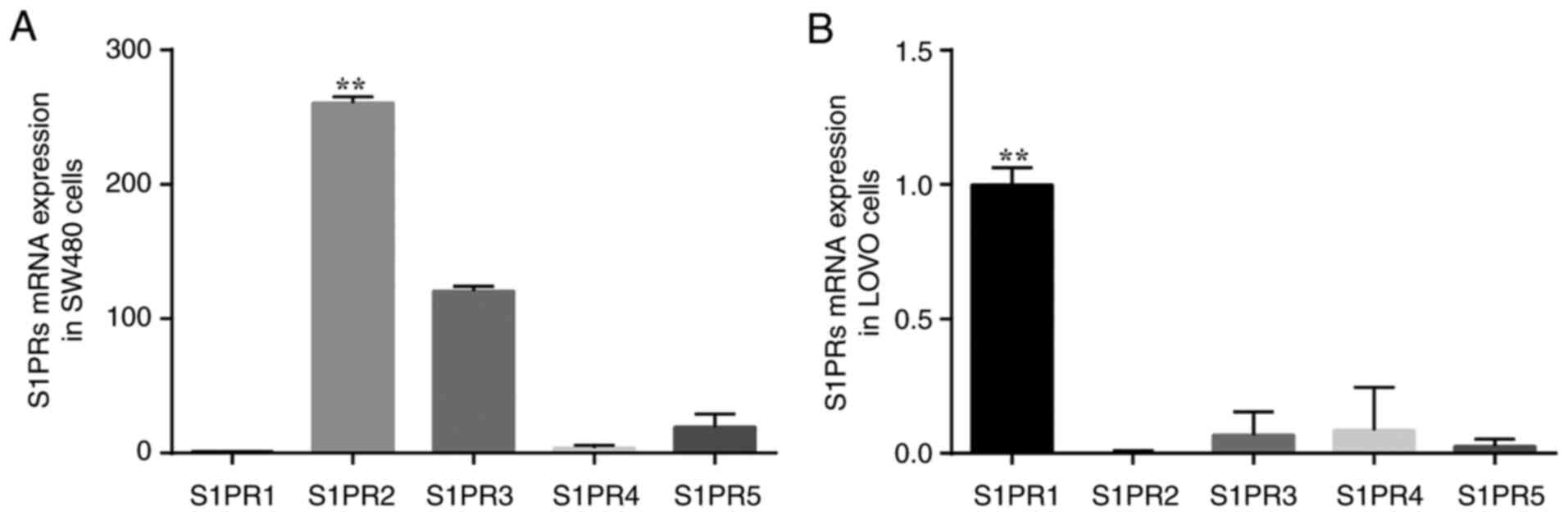
cells. Expression levels of S1PR in SW480 and LOVO cells were
determined. Transcripts for S1PR genes were expressed at varying
levels in colon cancer cells (Fig.
5). The mRNA expression levels of S1PR2 and S1PR3 were high in
SW480 cells (Fig. 5A), whereas
S1PR2 was expressed only at low levels in LOVO cells (Fig. 5B). Therefore, the effect of S1P on
migration of different colon cancer cell lines may depend on S1PR
subtypes expressed on the cell surface.
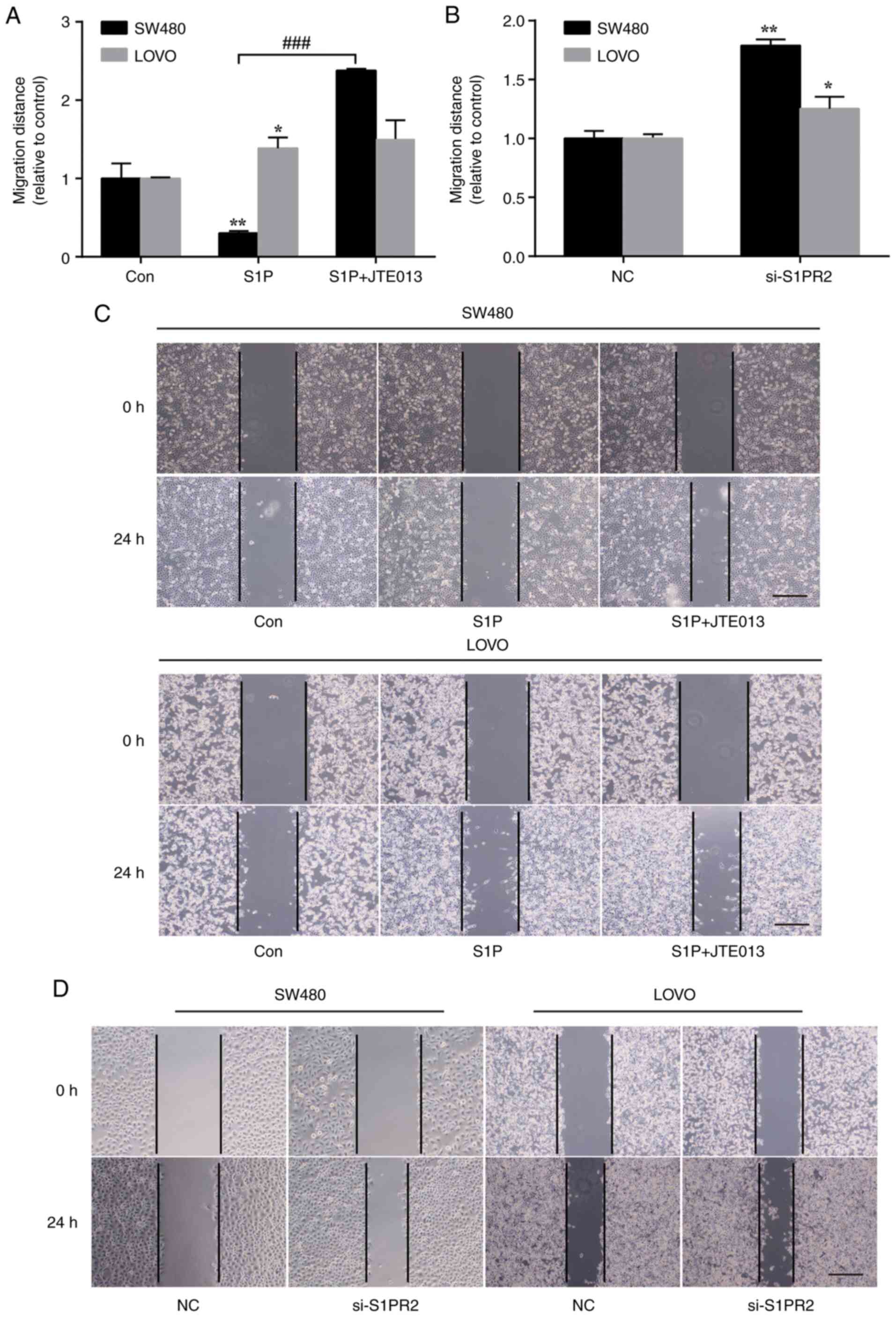 | Figure 4.Effect of S1PR2 on cell migration.
(A) Wound healing assay was performed in the presence or absence of
1 µM S1P and 5 µM JTE013. *P<0.05, **P<0.01, Con vs. S1P;
###P<0.001, S1P vs. S1P + JTE013. (B) Wound healing
assays were performed in cells transfected with S1PR2-specific
siRNA or control. n=3. *P<0.05, **P<0.01, NC vs. si-S1PR2.
(C) Representative images of the wound healing assay. Wound healing
assay was performed in the presence or absence of 1 µM S1P and 5 µM
JTE013. Scale bar, 200 µm. (D) Representative images of the wound
healing assay. Wound healing assays were performed in cells
transfected with S1PR2-specific siRNA or control. Scale bar, 200
µm. S1PR, S1PR, sphingosine-1-phosphate receptor; Con, control; NC,
negative control. |
S1PR2 mediates the effect of S1P on
migration and invasion of SW480 cells
To determine the role of S1PR2 in the migration and
invasion of SW480 cells, wound healing and Transwell assays were
performed using SW480 cells pre-treated in the presence or absence
of JTE-013 and FTY720 for 1 h before S1P stimulation. There was no
significant difference between the S1P and the control group with
regard to the wound healing of SW480 cells (Fig. 6A). Treatment with 5 µM JTE-013
increased SW480 cell migration but this was not significant
(Fig. 6A and C). Quantification of
cell migration width confirmed a significant increase in cell
migration following treatment with JTE013 + S1P compared with
JTE013-alone (Fig. 6A; P<0.05).
Co-treatment with S1P and 5 µM FTY720, an inhibitor of other S1PRs
(S1PR1, 3, 4 and 5), significantly attenuated migration compared
with the JTE013 + S1P group (Fig.
6A; P<0.01). S1P significantly enhanced invasion of SW480
cells in Transwell assay compared with the control group (Fig. 6B; P<0.01). Pre-treatment with
JTE013 further promoted SW480 cell invasion, and invasion was
higher in the FTY720 + S1P group than the Con + S1P group (Fig. 6B; P<0.01). These results
suggested that S1PR2 inhibited migration of cancer. Following
transfection with S1PR2-specific siRNA, SW480 cell proliferation
was significantly increased (Fig.
6E; P<0.05 and P<0.001, Con vs. si-S1PR and Con + S1P vs.
si-S1PR + S1P, respectively), while there was no significant
difference in SW480 cell viability following S1P stimulation.
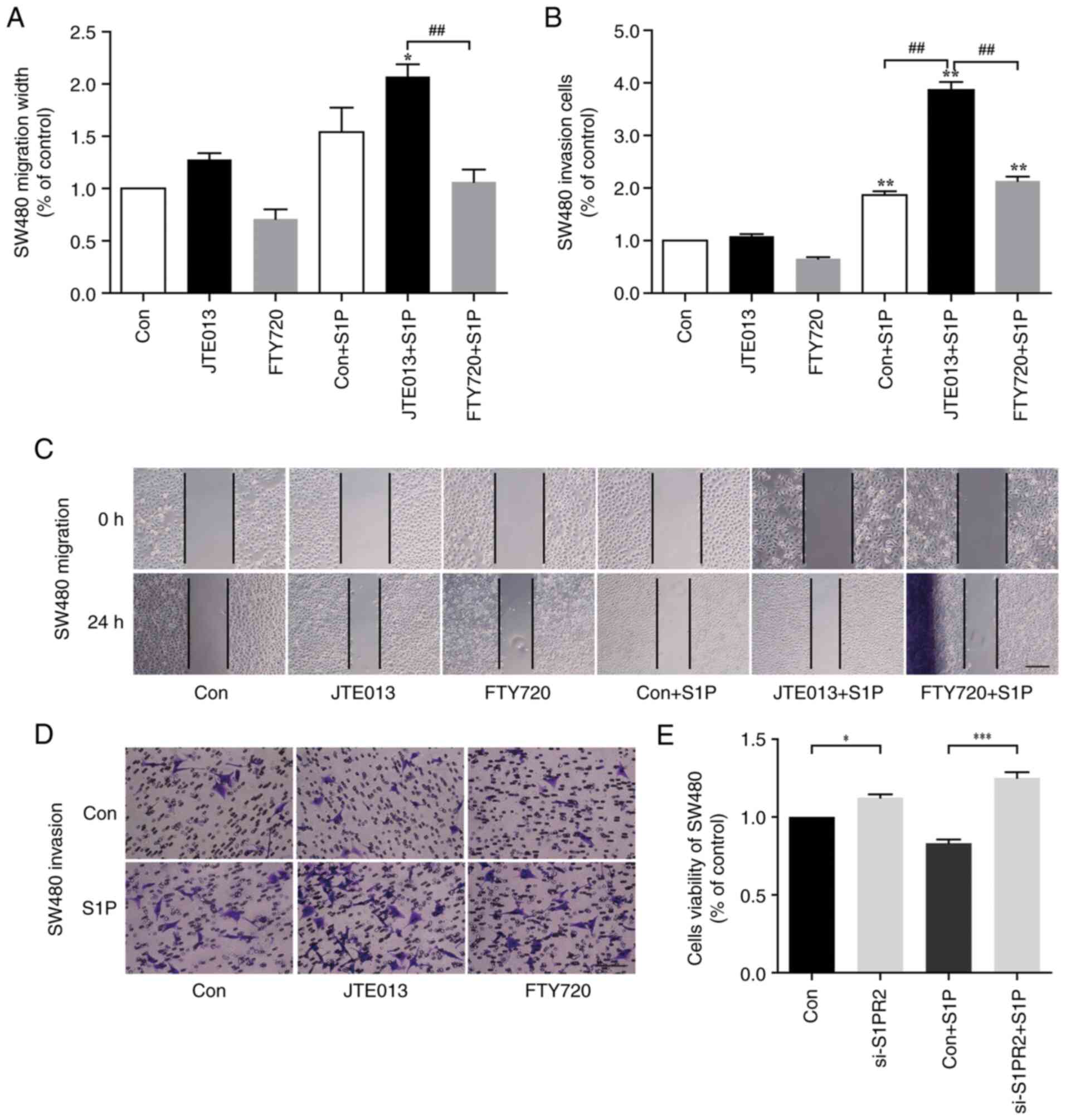 | Figure 6.Role of S1PR2 in cell migration,
invasion and proliferation. (A) Wound healing assay was performed
in the presence or absence of 1 µM S1P, 5 µM JTE013 and/or 5 µM
FTY720. *P<0.05, JTE013 vs. JTE013 + S1P;
##P<0.01, JTE013 + S1P vs. FTY720 + S1P. (B)
Transwell assay was performed in the presence or absence of 1 µM
S1P, 5 JTE013 and/or 5 µM FTY720. Data are expressed as the
percentage of invading cells relative to untreated control.
**P<0.01, Con vs. Con + S1P, JTE013 vs. JTE013 + S1P, FTY720 vs.
FTY720 + S1P. ##P<0.01, JTE013 vs. JTE013 + S1P vs.
FTY720 + S1P. (C) Representative images of the wound healing assay.
Scale bar, 200 µm. (D) Representative images of the Transwell
assay. Scale bar, 100 µm. (E) MTT assay was performed to detect
human CRC cell viability. SW480 cells were transfected with
S1PR2-specific siRNA and treated in the presence or absence of 1 µM
S1P. n=3. *P<0.05, Con vs. si-S1PR2, ***P<0.001, Con +S1P vs.
si-S1PR2 + S1P. S1PR, S1PR, sphingosine-1-phosphate receptor; si,
small interfering; Con, control. |
Discussion
CRC is associated with mortality and its 5-year
survival rate is associated with clinical stage (1). The early discovery and diagnosis of
aggressive carcinoma is key to improving survival rate. There is a
need to identify novel molecular therapeutic targets for diagnosis
and treatment of CRC. S1P is synthesized by SPHKs and binds S1PR1-5
to elicit downstream responses, thus regulating cellular processes,
including proliferation, migration and metastasis (9,19,31–34).
Previous studies have suggested that aberrant expression of S1P
signalling is involved in inflammation, angiogenesis and lymphocyte
trafficking (35–37). The synthesis and breakdown of S1P
are dysregulated in multiple types of cancer, such as
hepatocellular carcinoma and ovarian cancer (7,38,39).
To the best of our knowledge, however, the underlying mechanisms
are not fully understood. The present study aimed to investigate
the role of SPHK/S1P/S1PR signalling in colon cancer.
SPHK1 and SPHK2 are key regulators of
sphingolipid-mediated functions (19,35).
Previous studies have shown that expression of SPHK1 is upregulated
in breast cancer and is associated with drug resistance and poor
prognosis (40–43). The present results suggested that
SPHK1 expression in colorectal tissue was upregulated during
malignant transformation of human CRC. This was consistent with
previous reports that peripheral circulation and tumour S1P levels
are increased in colon cancer (44,45).
The present results demonstrated that synthesis of S1P was
dysregulated in CRC, which lead to increased production of S1P. The
bioactive lipid S1P serves a key role in cancer by promoting cell
proliferation and invasion and angiogenesis (16,46–48).
S1P levels in plasma from patients with CRC and healthy individuals
were measured using ELISA to determine whether circulating S1P
levels were a useful biomarker; levels of S1P were significantly
elevated in patients with CRC patients. These results are
consistent with a previous report that S1P is upregulated in
patients with other types of cancer, liver or neural system tumours
(49).
S1P exerts its signalling function in an autocrine
or paracrine manner via S1PR1-5 (11). The results of the present study
suggested that expression levels of S1PR2 and S1PR4 were
significantly downregulated in CRC, while expression of S1PR1 was
upregulated, which is consistent with previous studies on
glioblastoma and hepatocellular carcinoma (24,30).
Wound healing assay demonstrated that LOVO cells treated with
exogenous S1P showed a significant increase in cell migration,
whereas migration of SW480 cells was inhibited. The effect of S1P
on the migration of different colon cancer cell lines may depend on
receptor subtype distribution on the cell surface. S1P transduces
signals by binding plasma membrane G-protein-coupled receptors. The
expression of S1PR is tissue-specific and S1P signalling produces
different cellular outcomes (11).
The present study demonstrated that expression levels of S1PR
subtypes were different in the two cell lines, which may contribute
to the different results. mRNA expression levels of S1PR2 and S1PR3
were high in SW480 cells, whereas S1PR2 was expressed at low levels
in LOVO cells. The effect of S1P on migration was based on the
expression of S1PR2. This was consistent with the increased
migration following inhibition of S1PR2. Expression of S1PR2 mRNA
was increased in CRC tissue but decreased in colon cancer cells,
suggesting that expression of different types of S1PR was
associated with malignancy potential and confirming that SW480 has
a low metastatic ability.
S1PR2 levels have previously been reported to be
decreased in colon and prostate cancer compared with normal tissue
and are associated with poor patient prognosis (15,50).
To the best of our knowledge, however, no correlation has
previously been found between S1PR2 expression and disease-free
survival in human CRC. Therefore, it was hypothesized that S1PR2
may also be involved in the progression and development of CRC.
Here, expression of S1PR2 in 55 patients with CRC was detected at
the protein and mRNA levels by western blotting, IHC and RT-qPCR.
Expression of S1PR2 was downregulated in CRC, as evident in 32/55
cases in western blotting and 33/50 cases in IHC. The gene
expression of S1PR2 was significantly downregulated in tumour
tissue compared with normal surgical margin tissue.
S1PR2 serves a key role in cancer progression
(16). Salas et al
(34) suggested that systemic
SPHK1/S1P-regulated metastatic potential via inhibition of S1PR2
signalling further elevated breast cancer metastasis suppressor 1
level and suppressed lung tumour metastasis. Here, S1PRs were
expressed at different levels in colon cancer cell lines. Treatment
of cells with S1PR2 antagonist JTE013 in the presence of S1P
enhanced promotion of cell migration and invasion compared with the
control group. Moreover, treatment with S1PR2 siRNA significantly
enhanced cell proliferation compared with the control group.
Therefore, the promoting effect of S1P was enhanced by inhibition
of S1PR2 expression, which confirmed the hypothesis that loss of
S1PR2 in colon cancer may contribute to cell migration and
invasion.
FTY720 (also named Fingolimod) is an
immunosuppressant agent used to treat multiple sclerosis and has
shown anticancer activity (13,51)
against glioblastoma and haematological malignancy (37). FTY720 decreases cell viability and
survival and inhibits cancer progression via downregulation of key
nutrient transport proteins, selectively starving cancer cells to
death (52). FTY720 interferes
with the SPHK1/S1P/S1PR1 axis and suppresses the NF-κB/IL-6/Stat3
amplification loop and colitis-associated cancer in mice (21). Here, treatment with 5 µM FTY720
inhibited promotion of cell migration and invasion induced by
JTE013. FTY720, a sphingosine analogue, primarily acts via S1PR1,
3, 4 and 5 to cause receptor endocytosis, which leaves S1P binding
to S1PR2 alone, thus enhancing the role of S1PR2 in the inhibition
of cell migration and invasion. This suggests that FTY720 may be
useful for treating CRC in humans.
The present study had certain limitations. The
present preliminary clinical study primarily focused on the role of
S1PR2 in the proliferation and migration of colon cancer cell
lines; due to the limited sample size, expression levels of the
other four S1PRs were only measured by RT-qPCR rather than using
the same methods as for S1PR2. The association between S1PRs and
S1P was not fully elucidated; further investigation is required to
determine the role of the SPHK/S1P/S1PR signalling pathway in colon
cancer. Although the present study revealed that secretion of SPHK1
was upregulated in CRC tissue and S1P levels in serum of patients
with CRC were higher than those in healthy individuals, the
potential correlation between them was not assessed. Further
experiments are required to verify whether CRC cells secreted S1P.
Cell migration was assessed at only one time point; more time
points need to be analysed and migration velocity should be
determined in future. Animal experiments were not performed to
confirm the present findings. The expression of molecules was only
assessed by western blotting, IHC and RT-qPCR. More studies are
needed to confirm the results of the present study. Basic molecular
research involving downstream signalling pathways is also
required.
In summary, the present study indicated that
expression levels of S1PRs, particularly S1P2, were associated with
migration and invasion of CRC cells. The present study identified a
novel mechanism in which S1P inhibited tumour cell migration and
invasion via the S1PR2-dependent pathway, suggesting that S1PR2 may
be a therapeutic target for treatment of colon cancer.
Acknowledgements
Not applicable.
Funding
The present study was funded by The Science and Technology
Development Fund, Macau SAR (grant nos. 130/2017/A3, 0099/2018/A3
and 0098/2021/A2), The National Natural Science Foundation of China
(grant no. 81570522), Priority Academic Program Development of
Jiangsu Higher Education Institutions and Science and Technology
Planning Project of Guangdong Province (grant no.
2020B1212030008).
Availability of data and materials
The datasets used and/or analysed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
XZ conceptualized and supervised the study,
collected and analysed data and edited the manuscript. JY designed
the methodology, performed the experiments, collected, analysed and
visualized data and wrote the manuscript. QW performed the
experiments, collected and analysed data, edited the manuscript and
supervised the study. LS and YC performed the experiments. XZ and
JY confirm the authenticity of all the raw data. All authors have
read and approved the final manuscript.
Ethics approval and consent to
participate
All specimens were obtained for a previous study and
were used in the present study with verbal consent from the
patients and healthy donors. The study was approved by the
Institutional Ethics Committee of Nanjing Medical University
(approval no. 2016-SR-217).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
Statistics, 2017. CA Cancer J Clin. 67:7–30. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Van Doorn R, Van Horssen J, Verzijl D,
Witte M, Ronken E, Van Het HB, Lakeman K, Dijkstra CD, Van Der Valk
P, Reijerkerk A, et al: Sphingosine 1-phosphate receptor 1 and 3
are upregulated in multiple sclerosis lesions. Glia. 58:1465–1476.
2010. View Article : Google Scholar
|
|
3
|
Zhang G, Yang L, Kim GS, Ryan K, Lu S,
O'Donnell RK, Spokes K, Shapiro N, Aird WC, Kluk MJ, et al:
Critical role of sphingosine-1-phosphate receptor 2 (S1PR2) in
acute vascular inflammation. Blood. 122:443–455. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Baldari CT: The hematopoietic oncoprotein
FOXP1 promotes tumor cell survival in diffuse large B-cell lymphoma
by repressing S1PR2 signaling. Blood. 127:1380–1381. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Green JA and Cyster JG: S1PR2 links
germinal center confinement and growth regulation. Immunol Rev.
247:36–51. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Green JA, Suzuki K, Cho B, Willison LD,
Palmer D, Allen CD, Schmidt TH, Xu Y, Proia RL, Coughlin SR and
Cyster JG: The sphingosine 1-phosphate receptor S1P2
maintains the homeostasis of germinal center B cells and promotes
niche confinement. Nat Immunol. 12:672–680. 2011. View Article : Google Scholar
|
|
7
|
Sánchez DI, González-Fernández B,
San-Miguel B, de Urbina JO, Crespo I, González-Gallego J and Tuñón
MJ: Melatonin prevents deregulation of the sphingosine
kinase/sphingosine 1-phosphate signaling pathway in a mouse model
of diethylnitrosamine-induced hepatocellular carcinoma. J Pineal
Res. 62:e123692017. View Article : Google Scholar
|
|
8
|
Higashi K, Matsuzaki E, Hashimoto Y,
Takahashi-Yanaga F, Takano A, Anan H, Hirata M and Nishimura F:
Sphingosine-1-phosphate/S1PR2-mediated signaling triggers Smad1/5/8
phosphorylation and thereby induces Runx2 expression in
osteoblasts. Bone. 93:1–11. 2016. View Article : Google Scholar
|
|
9
|
Nagahashi M, Yuza K, Hirose Y, Nakajima M,
Ramanathan R, Hait NC, Hylemon PB, Zhou H, Takabe K and Wakai T:
The roles of bile acids and sphingosine-1-phosphate signaling in
the hepatobiliary diseases. J Lipid Res. 57:1636–1643. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Shida D, Inoue S, Yoshida Y, Kodaka A,
Tsuji T and Tsuiji M: Sphingosine kinase 1 is upregulated with
lysophosphatidic acid receptor 2 in human colorectal cancer. World
J Gastroenterol. 22:2503–2511. 2016. View Article : Google Scholar
|
|
11
|
Spiegel S and Milstien S:
Sphingosine-1-phosphate: An enigmatic signalling lipid. Nat Rev Mol
Cell Bio. 4:397–407. 2003. View
Article : Google Scholar
|
|
12
|
Pyne S and Pyne NJ: Sphingosine
1-phosphate signalling in mammalian cells. Biochem J. 349:385–402.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Gonzalezcabrera PJ, Brown S, Studer SM and
Rosen H: S1P signaling: New therapies and opportunities. F1000
Prime Reports. 6:1092014.PubMed/NCBI
|
|
14
|
Pyne NJ and Pyne S: Sphingosine
1-phosphate and cancer. Nat Rev Cancer. 10:489–503. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Pyne NJ, Mcnaughton M, Boomkamp S,
Macritchie N, Evangelesti C, Martelli AM, Jiang HR, Ubhi S and Pyne
S: Role of sphingosine 1-phosphate receptors, sphingosine kinases
and sphingosine in cancer and inflammation. Adv Biol Regul.
60:151–159. 2015. View Article : Google Scholar
|
|
16
|
Sukocheva OA, Furuya H, Ng ML, Friedemann
M, Menschikowski M, Tarasov VV, Chubarev VN, Klochkov SG, Neganova
ME, Mangoni AA, et al: Sphingosine kinase and
sphingosine-1-phosphate receptor signaling pathway in inflammatory
gastrointestinal disease and cancers: A novel therapeutic target.
Pharmacol Ther. 207:1074642020. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Okamoto H, Takuwa N, Gonda K, Okazaki H,
Chang K, Yatomi Y, Shigematsu H and Takuwa Y: EDG1 is a functional
sphingosine-1-phosphate receptor that is linked via a Gi/o to
multiple signaling pathways, including phospholipase C activation,
Ca2+ mobilization, Ras-mitogen-activated protein kinase activation,
and adenylate cyclase inhibition. J Biol Chem. 273:27104–27110.
1998. View Article : Google Scholar
|
|
18
|
Hannun YA and Obeid LM: Principles of
bioactive lipid signalling: Lessons from sphingolipids. Nat Rev Mol
Cell Bio. 9:139–150. 2008. View
Article : Google Scholar
|
|
19
|
Ng ML, Wadham C and Sukocheva OA: The role
of sphingolipid signalling in diabetesassociated pathologies
(Review). Int J Mol Med. 39:243–252. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Arikawa K, Takuwa N, Yamaguchi H, Sugimoto
N, Kitayama J, Nagawa H, Takehara K and Takuwa Y: Ligand-dependent
inhibition of B16 melanoma cell migration and invasion via
endogenous S1P2 G protein-coupled receptor. Requirement of
inhibition of cellular RAC activity. J Biol Chem. 278:32841–32851.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Liang J, Nagahashi M, Kim EY, Harikumar
KB, Yamada A, Huang WC, Hait NC, Allegood JC, Price MM and Avni D:
Sphingosine-1-phosphate links persistent STAT3 activation, chronic
intestinal inflammation, and development of colitis-associated
cancer. Cancer Cell. 23:107–120. 2013. View Article : Google Scholar
|
|
22
|
Du W, Takuwa N, Yoshioka K, Okamoto Y,
Gonda K, Sugihara K, Fukamizu A, Asano M and Takuwa Y: S1P(2), the
G protein-coupled receptor for sphingosine-1-phosphate, negatively
regulates tumor angiogenesis and tumor growth in vivo in mice.
Cancer Res. 70:772–781. 2010. View Article : Google Scholar
|
|
23
|
Watson C, Long JS, Orange C, Tannahill CL,
Mallon E, Mcglynn LM, Pyne S, Pyne NJ and Edwards J: O-56 High
expression of sphingosine 1-phosphate receptors, S1P 1 and S1P 3,
sphingosine kinase 1 and ERK-1/2 is associated with development of
tamoxifen resistance in ER positive breast cancer patients. Eur J
Cancer Suppl. 8:212010. View Article : Google Scholar
|
|
24
|
Yoshida Y, Nakada M, Harada T, Tanaka S,
Furuta T, Hayashi Y, Kita D, Uchiyama N, Hayashi Y and Hamada J:
The expression level of sphingosine-1-phosphate receptor type 1 is
related to MIB-1 labeling index and predicts survival of
glioblastoma patients. J Neurooncol. 98:41–47. 2010. View Article : Google Scholar
|
|
25
|
Kothapalli R, Kusmartseva I and Loughran
TP: Characterization of a human sphingosine-1-phosphate receptor
gene (S1P 5) and its differential expression in LGL leukemia.
Biochim Biophys Acta. 1579:117–123. 2002. View Article : Google Scholar
|
|
26
|
Cattoretti G, Mandelbaum J, Lee N, Chaves
AH, Mahler AM, Chadburn A, Dallafavera R, Pasqualucci L and
Maclennan AJ: Targeted disruption of the S1P2 sphingosine
1-phosphate receptor gene leads to diffuse large B-cell lymphoma
formation. Cancer Res. 69:8686–8692. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Flori M, Schmid CA, Sumrall ET, Tzankov A,
Law CW, Robinson MD and Muller A: The hematopoietic oncoprotein
FOXP1 promotes tumor cell survival in diffuse large B-cell lymphoma
by repressing S1PR2 signaling. Blood. 127:1438–1448. 2016.
View Article : Google Scholar
|
|
28
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Kawamori T, Kaneshiro T, Okumura M,
Maalouf S, Uflacker A, Bielawski J, Hannun YA and Obeid LM: Role
for sphingosine kinase 1 in colon carcinogenesis. FASEB J.
23:405–414. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Bao M, Chen Z, Xu Y, Zhao Y, Zha R, Huang
S, Liu L, Chen T, Li J, Tu H and He X: Sphingosine kinase 1
promotes tumour cell migration and invasion via the S1P/EDG1 axis
in hepatocellular carcinoma. Liver Int. 32:331–338. 2012.
View Article : Google Scholar
|
|
31
|
Terashita T, Kobayashi K, Nagano T, Kawa
Y, Tamura D, Nakata K, Yamamoto M, Tachihara M, Kamiryo H and
Nishimura Y: Administration of JTE013 abrogates experimental asthma
by regulating proinflammatory cytokine production from bronchial
epithelial cells. Respir Res. 17:1462016. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zhang X, Wang W, Ji XY, Ritter JK and Li
N: Knockout of sphingosine kinase 1 attenuates renal fibrosis in
unilateral ureteral obstruction model. Am J Nephrol. 50:196–203.
2019. View Article : Google Scholar
|
|
33
|
Nuno DW and Lamping KG: Dietary fatty acid
saturation modulates sphingosine-1-phosphate-mediated vascular
function. J Diabetes Res. 2019:23542742019. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Salas A, Ponnusamy S, Senkal CE,
Meyers-Needham M, Selvam SP, Saddoughi SA, Apohan E, Sentelle RD,
Smith C, Gault CR, et al: Sphingosine kinase-1 and sphingosine
1-phosphate receptor 2 mediate Bcr-Abl1 stability and drug
resistance by modulation of protein phosphatase 2A. Blood.
117:5941–5952. 2011. View Article : Google Scholar
|
|
35
|
Jozefczuk E, Nosalski R, Saju B, Crespo E,
Szczepaniak P, Guzik TJ and Siedlinski M: Cardiovascular effects of
pharmacological targeting of sphingosine kinase 1. Hypertension.
75:383–392. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Kunkel GT, Maceyka M, Milstien S and
Spiegel S: Targeting the sphingosine-1-phosphate axis in cancer,
inflammation and beyond. Nat Rev Drug Discov. 12:688–702. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Perla AS, Fratini L, Cardoso PS, de Farias
CB, Da CJM and Roesler R: Fingolimod (FTY720) reduces viability and
survival and increases histone H3 acetylation in medulloblastoma
cells. Pediatr Hematol Oncol. 37:170–175. 2020. View Article : Google Scholar
|
|
38
|
Pyne S and Pyne NJ: New Perspectives on
the Role of Sphingosine 1-Phosphate in Cancer. Sphingolipids in
Disease. Handbook of Experimental Pharmacology. Gulbins E and
Petrache I: 216. Springer; Vienna: pp. 55–71. 2013, View Article : Google Scholar
|
|
39
|
Beach JA, Aspuria PJ, Cheon DJ, Lawrenson
K, Agadjanian H, Walsh CS, Karlan BY and Orsulic S: Sphingosine
kinase 1 is required for TGF-beta mediated
fibroblastto-myofibroblast differentiation in ovarian cancer.
Oncotarget. 7:4167–4182. 2016. View Article : Google Scholar
|
|
40
|
Shida D, Takabe K, Kapitonov D, Milstien S
and Spiegel S: Targeting SphK1 as a new strategy against cancer.
Curr Drug Targets. 9:662–673. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Chen MH, Yen CC, Cheng CT, Wu RC, Huang
SC, Yu CS, Chung YH, Liu CY, Chang PM, Chao Y, et al:
Identification of SPHK1 as a therapeutic target and marker of poor
prognosis in cholangiocarcinoma. Oncotarget. 6:23594–23608. 2015.
View Article : Google Scholar
|
|
42
|
Patmanathan SN, Johnson SP, Lai SL, Bernam
SP, Lopes V, Wei W, Ibrahim MH, Torta F, Narayanaswamy P and Wenk
MR: Aberrant expression of the S1P regulating enzymes, SPHK1 and
SGPL1, contributes to a migratory phenotype in OSCC mediated
through S1PR2. Sci Rep. 6:256502016. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Ruckhäberle E, Rody A, Engels K, Gaetje R,
von Minckwitz G, Schiffmann S, Grösch S, Geisslinger G, Holtrich U,
Karn T and Kaufmann M: Microarray analysis of altered sphingolipid
metabolism reveals prognostic significance of sphingosine kinase 1
in breast cancer. Breast Cancer Res Treat. 112:41–52. 2008.
View Article : Google Scholar
|
|
44
|
Furuya H, Tamashiro PM, Shimizu Y, Iino K,
Peres R, Chen R, Sun Y, Hannun YA, Obeid LM and Kawamori T:
Sphingosine Kinase 1 expression in peritoneal macrophages is
required for colon carcinogenesis. Carcinogenesis. 38:1218–1227.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Furuya H, Shimizu Y, Tamashiro PM, Iino K,
Bielawski J, Chan O, Pagano I and Kawamori T: Sphingosine kinase 1
expression enhances colon tumor growth. J Transl Med. 15:1202017.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Weigert A, Olesch C and Brune B:
Sphingosine-1-Phosphate and macrophage biology-how the sphinx tames
the big eater. Front Immunol. 10:17062019. View Article : Google Scholar
|
|
47
|
Andrieu G, Ledoux A, Branka S, Bocquet M,
Gilhodes J, Walzer T, Kasahara K, Inagaki M, Sabbadini RA,
Cuvillier O and Hatzoglou A: Sphingosine 1-phosphate signaling
through its receptor S1P5 promotes chromosome segregation and
mitotic progression. Sci Signal. 10:eaah40072017. View Article : Google Scholar
|
|
48
|
Suh JH and Saba JD:
Sphingosine-1-phosphate in inflammatory bowel disease and
colitis-associated colon cancer: The fat's in the fire. Transl
Cancer Res. 4:469–483. 2015.PubMed/NCBI
|
|
49
|
Wang C, Mao J, Redfield S, Mo Y, Lage JM
and Zhou X: Systemic distribution, subcellular localization and
differential expression of sphingosine-1-phosphate receptors in
benign and malignant human tissues. Exp Mol Pathol. 97:259–265.
2014. View Article : Google Scholar
|
|
50
|
Herr DR, Reolo MJ, Peh YX, Wang W, Lee CW,
Rivera R, Paterson IC and Chun J: Sphingosine 1-phosphate receptor
2 (S1P2) attenuates reactive oxygen species formation and inhibits
cell death: Implications for otoprotective therapy. Sci Rep.
6:245412016. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Brinkmann V, Billich A, Baumruker T,
Heining P, Schmouder R, Francis G, Aradhye S and Burtin P:
Fingolimod (FTY720): Discovery and development of an oral drug to
treat multiple sclerosis. Nat Rev Drug Discov. 9:883–897. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Barthelemy C, Barry AO, Twyffels L and
Andre B: FTY720-induced endocytosis of yeast and human amino acid
transporters is preceded by reduction of their inherent activity
and TORC1 inhibition. Sci Rep. 7:138162017. View Article : Google Scholar : PubMed/NCBI
|