Introduction
Esophageal squamous cell carcinoma (ESCC)
predominates in Asia and Africa, constituting ~90% of malignant
esophageal tumors (1). However,
esophageal adenocarcinoma (EAC) accounts for only 10% of the cases,
but its incidence is increasing in Asia. EAC typically localizes to
the distal third of the esophagus and is closely linked to chronic
acid reflux, leading to the hallmark metaplasia commonly
originating from Barrett's esophagus (BE). BE histopathology
progresses from metaplasia to dysplasia and, without treatment, can
progress to adenocarcinoma. People with BE have a ~0.2%-0.5% annual
rate of developing EAC (2). In
contrast, adenocarcinoma in the proximal third of the esophagus
without BE is extremely rare and arises either from the focus of
the ectopic gastric mucosa or submucosal glands (3).
A gastric inlet patch (GIP) is an ectopic gastric
mucosal lesion usually found in the cervical esophagus and is
considered an incidental finding, with a reported incidence of
~2.5% (3,4). Given the extreme rarity of GIP-derived
EAC, its treatment strategy is notably complex due to its unique
location, histology, and limited treatment precedents. To the
authors' knowledge, no reported advanced GIP-derived EAC case
exists within the cervical esophagus that was treated using a
multidisciplinary treatment approach. This study described a
GIP-derived EAC successfully treated with multidisciplinary
treatment, including chemotherapy, definitive chemoradiotherapy
(CRT), photodynamic therapy (PDT), and salvage surgery.
Case report
Present medical history
A 64-year-old Japanese man with hypertension,
dyslipidemia, and chronic obstructive pulmonary disease with a
chief complaint of swallowing discomfort visited his doctor.
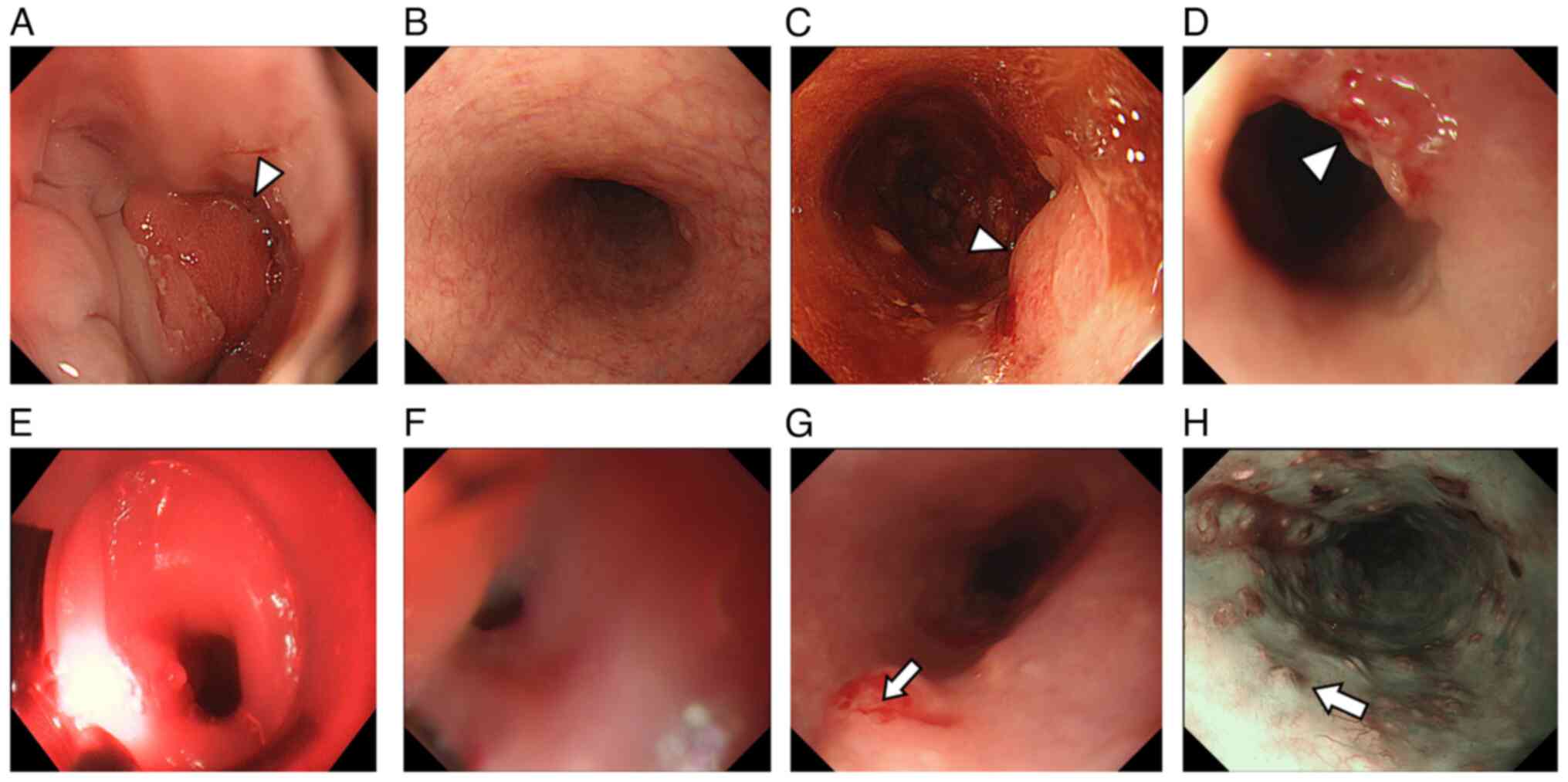
Subsequent esophagogastroduodenoscopy (EGD) identified an apparent
elevated tumor at the ectopic gastric mucosal site of the cervical
esophagus (Fig. 1A). Upon further
examination, he received a diagnosis of advanced cervical EAC
(CePh, 4.5 cm, tub2-por, cT2 N0 M0 IM0, cStage II) and was
subsequently chosen for a multidisciplinary treatment approach.
Later, he underwent induction chemotherapy using the DCF regimen
(docetaxel, cisplatin, and fluorouracil), followed by CRT
comprising cisplatin and fluorouracil, delivering 70 Gy over 35
sessions. This approach was in line with his strong preference for
larynx preservation. Remarkably, he achieved complete response
within 6 months after CRT completion (Fig. 1B). A year later, EGD identified a
local EAC recurrence in the cervical esophagus (Fig. 1C). Therefore, he was referred to the
Department of Gastroenterology at our hospital for endoscopic
treatment using PDT. Nevertheless, a viable tumor persisted despite
PDT procedures (Fig. 1D). After the
second PDT procedure by gastroenterological physicians, a local
recurrence with severe stenosis was identified in the cervical
esophagus (Fig. 1E). As a result,
he was referred to the Department of Gastrointestinal Surgery for
salvage surgery aimed at treating the residual lesions. However,
EGD revealed an elevated lesion at the cervical esophagus, 17 cm
from the incisor, and severe stenosis (Fig. 1F). Endoscopy could be successfully
conducted after balloon dilatation. During further EGD examination,
multiple submucosal tumor-like lesions with vascular atypia were
observed in the upper to middle esophagus, 22 to 28 cm from the
incisor (Fig. 1G and H). Biopsy
revealed that all lesions were adenocarcinomas with suspected
multiple intramural metastases of the esophagus. Subsequent
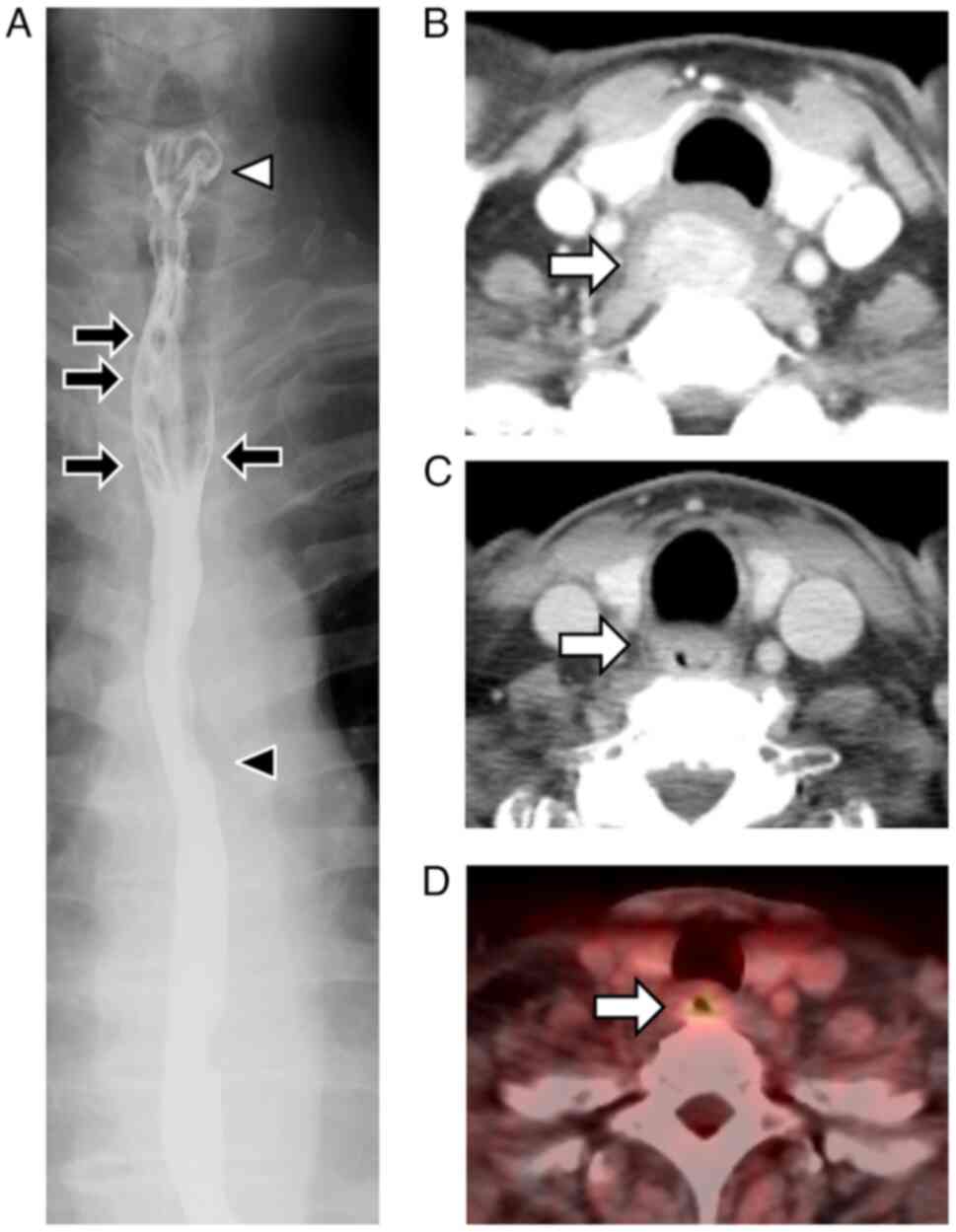
esophageal fluoroscopy revealed a 4.2 cm circumferential stricture
from the entrance of the cervical esophagus (Fig. 2A). Computed tomography (CT) before
treatment showed an apparent elevated tumor occupying the lumen of
the cervical esophagus and no clear lymphadenopathy around the
esophagus (Fig. 2B). Consequently,
CT and positron emission tomography after PDT procedures showed a
wall thickness with abnormal 18F-fluorodeoxyglucose
uptake remaining in the cervical esophagus (Fig. 2C and D). However, no obvious lymph
node or distant metastasis was suspected. As a result, he was
diagnosed with recurrent advanced cervical EAC (CePh, 4.5 cm, por,
CRT-cT2 N0 M0 IM1, CRT-cStage II), and a radical operation was
recommended as a necessary intervention. The surgery included
esophagectomy with extensive mediastinal lymph node dissection and
laryngopharyngectomy. Therefore, robot-assisted minimal invasive
esophagectomy (RAMIE) with laryngopharyngectomy and cervical
lymphadenectomy as radical salvage surgery were performed 4 months
after the second PDT procedure.
Surgical procedure
The surgical procedure was performed by a
multidisciplinary team consisting of gastrointestinal surgeons and
otorhinolaryngologists. First, thoracoscopic esophagectomy and
mediastinal lymphadenectomy were performed with robot assistance as
described previously (5). Second,
to confirm whether the larynx can be preserved, the cervical
esophagus on the anal side of the hypopharynx was cut by cervical
manipulation, and the stump was submitted for intraoperative rapid
pathological diagnosis. As a result, malignant cells were clearly
detected in the oral-side stump, so it was judged that larynx
preservation was impossible. Therefore, additional
laryngopharyngectomy and cervical lymphadenectomy were performed by
otorhinolaryngologists. Third, reconstruction of the digestive
tract via the posterior mediastinal route by pharyngogastric
anastomosis using gastric conduit was performed. Finally, a
permanent tracheostomy was created and the cervical wound was
closed. The overall intraoperative time and the amount of
intraoperative bleeding were 650 min and 300 g, respectively.
Histological findings of the resected
specimens
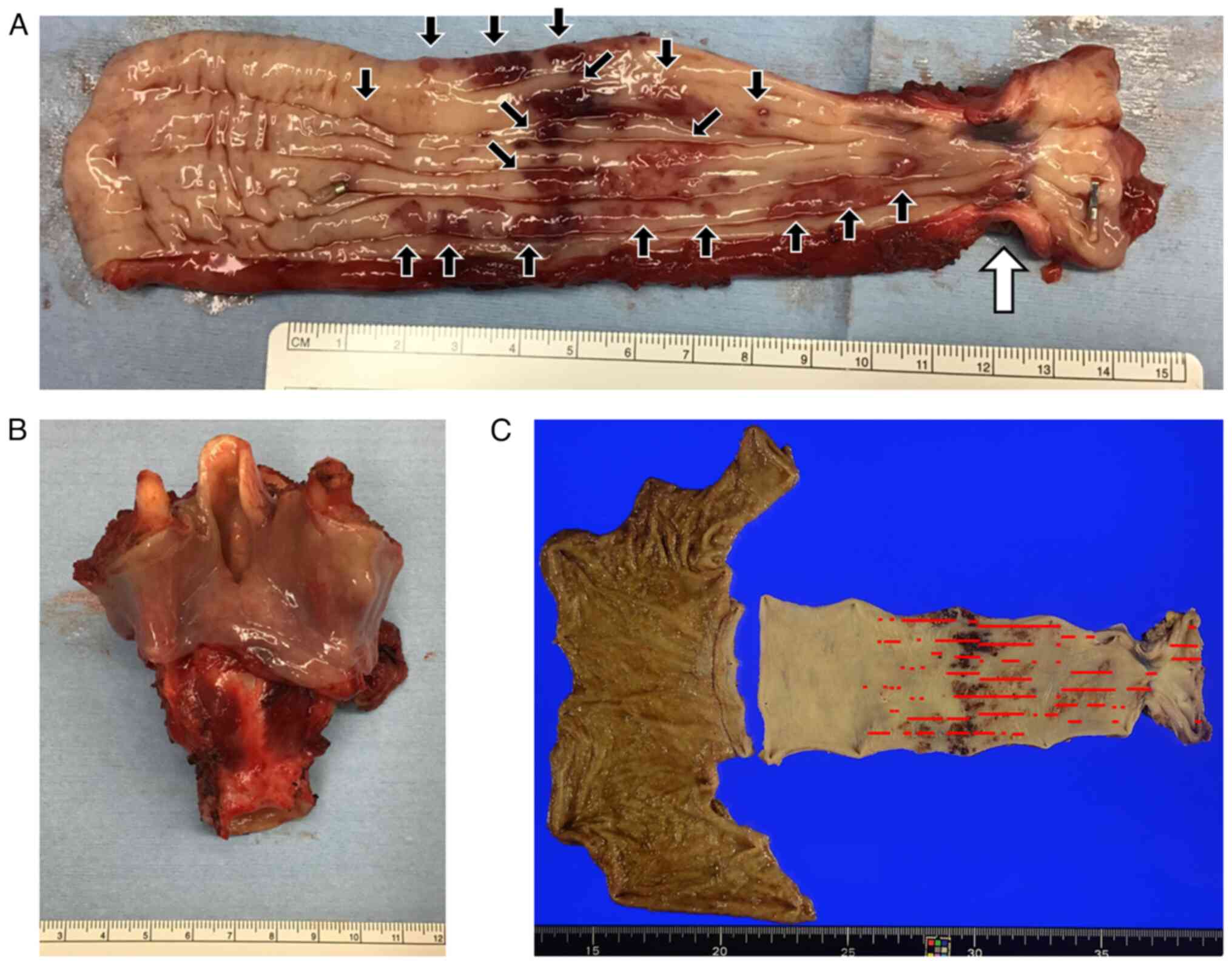
Macroscopically, the proximal side of the esophagus
exhibited noticeable wall hardening and constriction. Furthermore,
numerous submucosal tumors were noticed near the primary tumor
(Fig. 3A). However, the
laryngopharyngectomy specimens showed no evidence of tumor cells
(Fig. 3B).
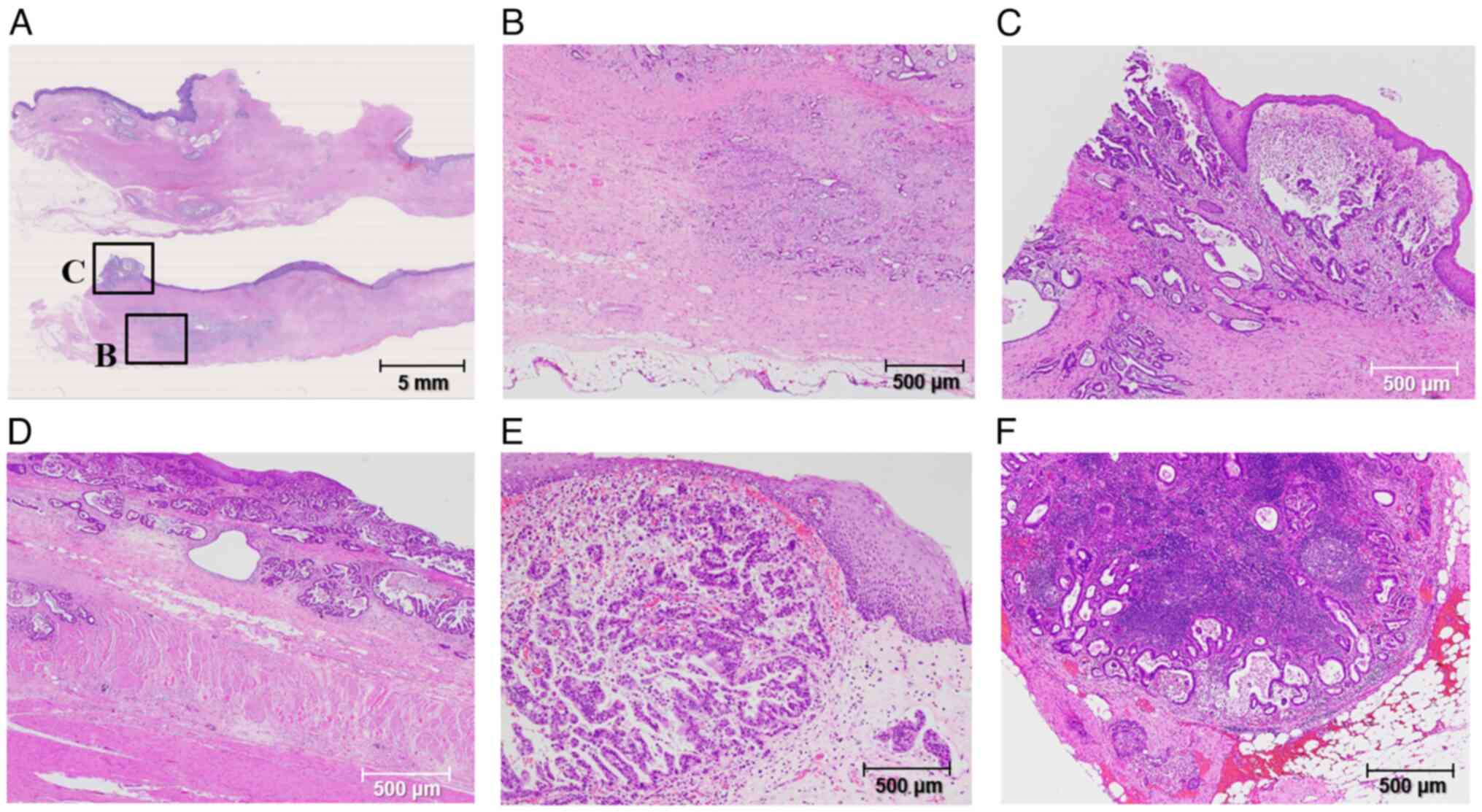
Histologically, viable adenocarcinoma cells remained
with fibrosis and necrosis in the cervical esophagus, and multiple
intramural metastases were distributed in the upper to middle
esophagus (Figs. 3C and 4A). The deepest part of the cancer cells
invaded the muscularis propria of the esophagus (Fig. 4B). Cancer cells were also observed
in the proximal stump of the esophageal resection specimen
(Fig. 4C). Adenocarcinoma cells
were mainly moderate to poorly differentiated and distributed in
the mucosa and lamina propria (Fig.
4D). However, multiple intramural metastases and vascular
invasions were frequently observed in the anal side of the primary
tumor (Fig. 4E). Multiple
metastatic lymph node metastases were evident in mediastinal and
intraabdominal lymph nodes (Fig.
4F). Finally, the pathological diagnosis was advanced cervical
GIP-derived EAC [CePh, 4.5 cm, circ, moderately to poorly
differentiated adenocarcinoma, INFc, ly3, v2, pIM1, pPM0, pDM0,
pRM0, CRT-pT3, pN4 (10/44, #105×1, #106recLx2, #107×1, #110×1,
#112aoAx4, #3ax1) M0, CRT-pStage IVA, D3, Cur B].
Postoperative clinical course
An anastomotic leakage on the 13th day after surgery
was successfully resolved through conservative management.
Subsequently, he was transferred to another hospital for
rehabilitation on the 45th day after the operation. Given the
substantial risk of recurrence, he underwent adjuvant chemotherapy
with oral S-1 (a prodrug of 5-fluorouracil) for 1 year after the
prescribed protocol for adjuvant chemotherapy in gastric cancer
(6,7). Although the quality of life
deteriorated due to the loss of vocal function, he has spent his
daily life without significant deterioration in his general
condition or body weight. Fortunately, the patient remained
relapse-free with the assessment using EGD and CT, achieving a
3-year survival after the salvage surgery.
Discussion
This is a cervical EAC case that developed in the
cervical ectopic gastric mucosa. EAC risk factors include
gastroesophageal reflux disease (GERD), BE, obesity, and smoking.
BE histopathology progresses from metaplasia to dysplasia and,
without treatment, can progress to adenocarcinoma. People with BE
have a ~0.2%-0.5% annual rate of developing EAC. Although alcohol
consumption is not associated with EAC risk, other exposures, such
as physical activity, nutrition, and medication use, require
further studies. Genetic variants are also associated with EAC
risk, but their overall contribution is low (2). Additionally, he did not drink alcohol
and had a heavy smoking history. There was also a family history of
gastric cancer, but the presence of Helicobacter pylori was
not investigated in detail. Generally, the prognosis in EAC has
been reported to be poor because of the late presentation of
symptoms and the aggressiveness of the tumor; appropriate treatment
strategy, screening, and surveillance trials of high-risk
individuals are needed (1,2).
The previously reported prevalence of GIP in the
proximal esophagus ranges from 0.18 to 14% in endoscopic studies
(8–11). However, adenocarcinoma incidence
among patients with cervical ectopic gastric mucosa is 0–1.56%
(12). Orosey et al
(13) reported only 5 (1.3%)
patients with ectopic gastric mucosa among 398 EAC diagnosed over
14 years, and only 3 (0.8%) patients had ectopic gastric mucosa
within the proximal esophagus. Only 58 EAC and ectopic gastric
mucosa were reported between 1950 and 2015 worldwide, and most were
from Japan.
The pathogenesis of adenocarcinoma within an ectopic
gastric mucosa might comprise a metaplastic-dysplastic pathway,
leading to intestinal metaplasia and an intestinal-type
adenocarcinoma or the development of adenocarcinoma within
gastric/foveolar cells in an ectopic gastric mucosa (3). Tang et al (14) reported that GERD and BE are
significantly more common in cervical ectopic gastric mucosa,
suggesting that acid reflux is involved in their development
(14–16). This case was not associated with BE
and had no history of GERD treatment.
Several case reports of EAC derived from ectopic
gastric mucosa have been reported (17–23).
Kitasaki et al (17)
reported a case of repeated local recurrence at the same site
despite multiple radical endoscopic resections. Ito et al
(18) reported that ESD-pT1a (MM)
EAC derived from ectopic gastric mucosa in the neck developed lymph
node recurrence. In our case, although the primary tumor was
relatively mild, lymphatic and vascular invasion was significant,
there were more lymph node metastases than preoperatively
diagnosed, and there were widespread intramural esophageal
metastases. These results suggested that EAC in the cervical or
upper thoracic esophagus, rich in lymphatic chains and vascular
networks, may be difficult to treat and have a poor prognosis
(23). The basic treatment strategy
for EAC is local control through resection, and some reports have
shown that endoscopic resection can be expected to treat cervical
esophageal lesions in early-stage cancers (19–22).
Tanaka et al (22) reported
a case in which complete resection was achieved with
larynx-preserving surgery, indicating that larynx-preserving
surgery is possible for localized lesions that do not extend to the
hypopharynx as long as negative margins are ensured. In contrast,
von Rahden et al (24)
reported a treated case of EAC derived from the heterotopic gastric
mucosa by definitive CRT. Surgery after neoadjuvant chemotherapy
(NAC) and CRT for esophageal cancer is frequently performed in
Europe and the United States (25–27).
This study first selected induction DCF therapy and subsequent
definitive CRT as a radical treatment strategy because the patient
strongly desired to preserve the larynx.
In a lesion extending to the pharynx, combined
resection of the pharynx and laryngopharynx is unavoidable,
resulting in the loss of vocalization and swallowing functions. In
our case, the primary lesion not only extended to the pharynx but
was also accompanied by extensive multiple intramural metastases in
the anal esophagus and the necessity of mediastinal lymph node
dissection. Finally, laryngopharyngectomy was unavoidable for
radical resection without residual tumor. Indeed, in superior
aerodigestive airway cancer with rare histology like EAC or
sarcoma, the protocols have to be individualized and made by a
multidisciplinary team to obtain a better prognosis (28). In the present cases, a
multidisciplinary treatment was subsequently performed by a
multidisciplinary team consisting of gastroenterological
physicians, radiologists, gastrointestinal surgeons, and
otorhinolaryngologists. In recent years, thoracoscopic and
robot-assisted surgeries have become increasingly popular for
minimally invasive esophagectomy (MIE). The same applies to EAC
treatment. Warner et al (29) reported that MIE is an acceptable
surgical therapy for advanced-stage esophageal malignancies after
neoadjuvant CRT without evidence for increased morbidity or
mortality. Tagkalos et al (30) reported the usefulness of RAMIE vs.
conventional MIE for EAC. Therefore, this study elected RAMIE for
thorough and less invasive mediastinal lymph node dissection and
esophagectomy.
DCF therapy as induction chemotherapy has been
positioned as a standard treatment for NAC for advanced ESCC after
the results of the JCOG1109 trial due to its potency (31). The usefulness of DCF therapy as NAC
for adenocarcinoma cases has not been sufficiently demonstrated
(32). In contrast, several reports
on the usefulness of NAC for gastroesophageal adenocarcinoma have
been recently reported (33–37).
However, which neoadjuvant treatment is best for patients with
gastroesophageal junction (GEJ) tumors remains controversial. The
FLOT4 trial showed a significant overall survival benefit of the
perioperative triplet regimen (fluorouracil + leucovorin,
oxaliplatin, and docetaxel) plus surgery compared to the
ECF/ECX-MAGIC regimen (fluorouracil or capecitabine + cisplatin and
epirubicin) for resectable gastric or GEJ adenocarcinoma (38). In Europe and the United States,
preoperative CRT has better results than NAC and has become the
standard treatment for advanced GEJ adenocarcinoma. In the CROSS
study, long-term follow-up results of neoadjuvant CRT combined with
surgery compared to surgery alone demonstrated more profound
survival benefits in patients with squamous cell carcinoma than in
those with GEJ adenocarcinoma (35). Unfortunately, cure by induction DCF
therapy and subsequent definitive CRT was not obtained in our case,
and salvage surgery was unavoidable.
PDT is an effective treatment for postradiotherapy
residual tumors and local recurrence, but it is not originally
indicated for cervical lesions due to the risk of esophageal
stricture, injury, and perforation (39). Conversely, Hayashi et al
(40) reported that PDT is also
effective for cervical lesions and is a treatment expected to
expand its indications in the future. In this case, the cause of
stenosis remains unclear whether it was caused by cauterization by
PDT or CRT. Moreover, PDT is the approved curative treatment for
high-grade dysplasia and EAC in BE by the U.S. Food and Drug
Administration and provides favorable and comparable long-term
outcomes to esophagectomy (41,42).
Because this patient was resistant to various anticancer
treatments, such as chemotherapy, CRT, and PDT, and multiple
intramural metastases later appeared, it was considered that
salvage surgery was inevitable for a radical cure. However, no
remaining columnar epithelial components were observed around the
cervical EAC of the resected specimen. Although cervical ectopic
gastric mucosa indeed existed before treatment, it was thought that
it had been cauterized by the effects of CRT or PDT during the
clinical course of the treatment for cervical EAC.
In summary, this is a rare multidisciplinary
treatment approach for advanced cervical GIP-derived EAC of the
cervical esophagus, which was resistant to chemotherapy, CRT, and
PDT and radically resected by salvage surgery. For refractory EAC,
a long-term prognosis can be expected by aiming to eradicate the
tumor through multimodal treatment tailored to the disease
state.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
KO, NI and IN designed the study and drafted the
manuscript. KO, TY, TA and IN performed the surgery and therapeutic
management of the patient. TH performed the endoscopic treatment of
the patient. TO performed the postoperative management and managed
adjuvant chemotherapy. KO, TY, TA, TH, TO and NI obtained medical
images. DK, TM, TH, HF, NI, SK, IN and HT contributed to analysis
of the patient's data and the editing of the report. KO and NI
confirm the authenticity of all the raw data. All authors have read
and approved the final version of the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Written informed consent was obtained from the
patient for the publication of this case report and the
accompanying images.
Authors' information
KO, NI and IN are specialists and instructors of the
Japanese Society of Gastroenterological Surgery and the Japanese
Esophageal Society, and serve as councilors of the Japanese
Esophageal Society.
Use of artificial intelligence tools
During the preparation of this work, AI tools were
used to improve the readability and language of the manuscript, and
subsequently, the authors revised and edited the content produced
by the AI tools as necessary, taking full responsibility for the
ultimate content of the present manuscript.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
GIP
|
gastric inlet patch
|
|
EAC
|
esophageal adenocarcinoma
|
|
PDT
|
photodynamic therapy
|
|
ESCC
|
esophageal squamous cell carcinoma
|
|
BE
|
Barrett's esophagus
|
|
CRT
|
chemoradiotherapy
|
|
EGD
|
esophagogastroduodenoscopy
|
|
CT
|
computed tomography
|
|
RAMIE
|
robot-assisted minimal invasive
esophagectomy
|
|
GERD
|
gastroesophageal reflux disease
|
|
NAC
|
neoadjuvant chemotherapy
|
|
MIE
|
minimally invasive esophagectomy
|
|
GEJ
|
gastroesophageal junction
|
References
|
1
|
Rubenstein JH and Shaheen NS:
Epidemiology, diagnosis, and management of esophageal
adenocarcinoma. Gastroenterology. 149:302–317.e1. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Lagergren J, Smyth E, Cunninham G and
Lagergren P: Oesophageal cancer. Lancet. 390:2383–2396. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Riddiough GE, Hornby ST, Asadi K and Aly
A: Gastric adenocarcinoma of the upper oesophagus: A literature
review and case report. Int J Surg Case Rep. 30:205–214. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Dziadkowiec KN, Sánchez-Luna SA, Stawinski
P and Proenza J: Adenocarcinoma arising from a cervical esophageal
inlet patch: The malignant potential of a small lesion. Cureus.
12:e92842020.PubMed/NCBI
|
|
5
|
Ninomiya I, Okamoto K, Yamaguchi T, Saito
H, Terai S, Moriyama H, Kinoshita J and Fushida S: Optimization of
robot-assisted thoracoscopic esophagectomy in the lateral decubitus
position. Esophagus. 18:482–488. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Sakuramoto S, Sasako M, Yamaguchi T,
Kinoshita T, Fujii M, Nashimoto A, Furukawa H, Nakajima T, Ohashi
Y, Imamura H, et al: Adjuvant chemotherapy for gastric cancer with
S-1, an oral fluoropyrimidine. N Engl J Med. 357:1810–1820. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Sasako M, Sakuramoto S, Katai H, Kinoshita
T, Furukawa H, Yamaguchi T, Nashimoto A, Fujii M, Nakajima T and
Ohashi Y: Five-year outcomes of a randomized phase III trial
comparing adjuvant chemotherapy with S-1 versus surgery alone in
stage II or III gastric cancer. J Clin Oncol. 29:4387–4393. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Peitz U, Vieth M, Evert M, Arand J,
Roessner A and Malfertheiner P: The prevalence of gastric
heterotopia of the proximal esophagus is underestimated, but
preneoplasia is rare-correlation with Barrett's esophagus. BMC
Gastroenterol. 17:872017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yin Y, Li H, Feng J, Zheng K, Yoshida E,
Wang L, Wu Y, Guo X, Shao X and Qi X: Prevalence and clinical and
endoscopic characteristics of cervical inlet patch (heterotopic
gastric mucosa): A systematic review and meta-analysis. J Clin
Gastroenterol. 56:e250–e262. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ciocalteu A, Popa P, Ionescu M and Gheonea
DI: Issues and controversies in esophageal inlet patch. World J
Gastroenterol. 25:4061–4073. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Akbayir N, Alkim C, Erdem L, Sökmen HM,
Sungun A, Başak T, Turgut S and Mungan Z: Heterotopic gastric
mucosa in the cervical esophagus (inlet patch): Endoscopic
prevalence, histological and clinical characteristics. J
Gastroenterol Hepatol. 19:891–896. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Rusu R, Ishaq S, Wong T and Dunn JM:
Cervical inlet patch: New insights into diagnosis and endoscopic
therapy. Frontline Gastroenterol. 9:214–220. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Orosey M, Amin M and Cappell MS: A 14-year
study of 398 esophageal adenocarcinomas diagnosed among 156,256
EGDs performed at two large hospitals: An inlet patch is proposed
as a significant risk factor for proximal esophageal
adenocarcinoma. Dig Dis Sci. 63:452–465. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Tang P, McKinley MJ, Sporrer M and Kahn E:
Inlet patch: Prevalence, histologic type, and association with
esophagitis, Barrett esophagus, and antritis. Arch Pathol Lab Med.
128:444–447. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Coleman HG, Xie SH and Lagergren J: The
epidemiology of esophageal adenocarcinoma. Gastroenterology.
154:390–405. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Sawas T and Katzka DA: Esophageal
adenocarcinoma phenotypes and risk factors. Curr Opin
Gastroenterol. 38:423–427. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kitasaki N, Hamai Y, Yoshikawa T, Emi M,
Kurokawa T, Hirohata R, Ohsawa M and Okada M: Recurrent esophageal
adenocarcinoma derived from ectopic gastric mucosa: A case report.
Thorac Cancer. 13:876–879. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ito M, Dobashi A, Komori M, Sugimura S,
Aizawa D, Takahashi K, Tanishima Y and Sumiyama K: Lymph node
metastasis after endoscopic submucosal dissection of a superficial
esophageal adenocarcinoma arising from the ectopic gastric mucosa
of the cervical esophagus: A case report. DEN Open. 3:e2142023.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Nakao E, Fujisaki J, Nakano K, Kawachi H,
Narimiya N, Suzuki S, Namikawa K, Tokai Y, Yoshimizu S, Horiuchi Y,
et al: Early esophageal adenocarcinoma with non-Barrett's columnar
epithelium origin: Two case reports and a literature review. Intern
Med. 62:1939–1946. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ohki D, Tsuji Y, Yamazawa S, Ushiku T and
Tateishi K: Gastrointestinal: Esophageal adenocarcinoma arising
from circumferential ectopic gastric mucosa: A case report. J
Gastroenterol Hepatol. 37:472022. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ikeda R, Hirasawa K, Ozeki Y, Sawada A,
Nishio M, Fukuchi T, Sato C and Maeda S: Cervical esophageal
adenocarcinoma of intestinal type in ectopic gastric mucosa. DEN
Open. 3:e1412022. View
Article : Google Scholar : PubMed/NCBI
|
|
22
|
Tanaka M, Ushiku T, Ikemura M, Junji
Shibahara J, Seto Y and Fukayama M: Esophageal adenocarcinoma
arising in cervical inlet patch with synchronous Barrett's
esophagus-related dysplasia. Pathol Int. 64:397–401. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Stiles BM, Mirza F, Port JL, Lee PC, Paul
S, Christos P and Altorki NK: Predictors of cervical and recurrent
laryngeal lymph node metastases from esophageal cancer. Ann Thorac
Surg. 90:1805–1811. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
von Rahden BHA, Stein HJ, Becker K and
Siewert RJ: Esophageal adenocarcinomas in heterotopic gastric
mucosa: Review and report of a case with complete response to
neoadjuvant radiochemotherapy. Dig Surg. 22:107–112. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Sjoquist KM, Burmeister BH, Smithers BM,
Zalcberg JR, Simes RJ, Barbour A and Gebski V; Australasian
Gastro-Intestinal Trials Group, : Survival after neoadjuvant
chemotherapy or chemoradiotherapy for resectable oesophageal
carcinoma: An updated meta-analysis. Lancet Oncol. 12:681–692.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Ronellenfitsch U, Schwarzbach M, Hofheinz
R, Kienle P, Kieser M, Slanger TE, Burmeister B, Kelsen D,
Niedzwiecki D, Schuhmacher C, et al: Preoperative
chemo(radio)therapy versus primary surgery for gastroesophageal
adenocarcinoma: Systematic review with meta-analysis combining
individual patient and aggregate data. Eur J Cancer. 49:3149–3158.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wang DB, Zhang X, Han HL, Xu YJ, Sun DQ
and Shi ZL: Neoadjuvant chemoradiotherapy could improve survival
outcomes for esophageal carcinoma: A meta-analysis. Dig Dis Sci.
57:3226–3233. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Vrînceanu D, Dumitru M, Ştefan AA,
Mogoantă CA and Sajin M: Giant pleomorphic sarcoma of the tongue
base-a cured clinical case report and literature review. Rom J
Morphol Embryol. 61:1323–1327. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Warner S, Chang YH, Paripati H, Ross H,
Ashman J, Harold K, Day R, Stucky CC, Rule W and Jaroszewski D:
Outcomes of minimally invasive esophagectomy in esophageal cancer
after neoadjuvant chemoradiotherapy. Ann Thorac Surg. 97:439–445.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Tagkalos E, van der Sluis PC, Berlth F,
Poplawski A, Hadzijusufovic E, Lang H, van Berge Henegouwen MI,
Gisbertz SS, Müller-Stich BP, Ruurda JP, et al: Robot-assisted
minimally invasive thoraco-laparoscopic esophagectomy versus
minimally invasive esophagectomy for resectable esophageal
adenocarcinoma, a randomized controlled trial (ROBOT-2 trial). BMC
Cancer. 21:10602021. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Nakamura K, Kato K, Igaki H, Ito Y,
Mizusawa J, Ando N, Udagawa H, Tsubosa Y, Daiko H, Hironaka S, et
al: Three-arm phase III trial comparing cisplatin plus 5-FU (CF)
versus docetaxel, cisplatin plus 5-FU (DCF) versus radiotherapy
with CF (CF-RT) as preoperative therapy for locally advanced
esophageal cancer (JCOG1109, NExT study). Jpn J Clin Oncol.
43:752–755. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Ajani JA, Moiseyenko VM, Tjulandin S,
Majlis A, Constenla M, Boni C, Rodrigues A, Fodor M, Chao Y, Voznyi
E, et al: Clinical benefit with docetaxel plus fluorouracil and
cisplatin compared with cisplatin and fluorouracil in a phase III
trial of advanced gastric or gastroesophageal cancer
adenocarcinoma: The V-325 study group. J Clin Oncol. 25:3205–3209.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Walsh TN, Noonan N, Hollywood D, Kelly A,
Keeling N and Hennessy TP: A comparison of multimodal therapy and
surgery for esophageal adenocarcinoma. N Engl J Med. 335:462–467.
1996. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Ychou M, Boige V, Pignon JP, Conroy T,
Bouché O, Lebreton G, Ducourtieux M, Bedenne L, Fabre JM,
Saint-Aubert B, et al: Perioperative chemotherapy compared with
surgery alone for resectable gastroesophageal adenocarcinoma: An
FNCLCC and FFCD multicenter phase III trial. J Clin Oncol.
29:1715–1721. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Shapiro J, van Lanschot JJB, Hulshof MCCM,
van Hagen P, van Berge Henegouwen MI, Wijnhoven BPL, van Laarhoven
HWM, Nieuwenhuijzen GAP, Hospers GAP, Bonenkamp JJ, et al:
Neoadjuvant chemoradiotherapy plus surgery versus surgery alone for
oesophageal or junctional cancer (CROSS): Long-term results of a
randomised controlled trial. Lancet Oncol. 16:1090–1098. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Grizzi G, Petrelli F, Di Bartolomeo M,
Viti M, Texeira Moraes M, Luciani A, Passalacqua R, Ghidini M,
Tomasello G, Baiocchi GL and Celotti A: Preferred neoadjuvant
therapy for gastric and gastroesophageal junction adenocarcinoma: A
systematic review and network meta-analysis. Gastric Cancer.
25:982–987. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Kim S, Paget-Bailly S, Messager M, Nguyen
T, Mathieu P, Lamfichekh N, Fein F, Fratté S, Cléau D, Lakkis Z, et
al: Perioperative docetaxel, cisplatin, and 5-fluorouracil compared
to standard chemotherapy for resectable gastroesophageal
adenocarcinoma. Eur J Surg Oncol. 43:218–225. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Al-Batran SE, Homann N, Pauligk C, Goetze
TO, Meiler J, Kasper S, Kopp HG, Mayer F, Haag GM, Luley K, et al:
Perioperative chemotherapy with fluorouracil plus leucovorin,
oxaliplatin, and docetaxel versus fluorouracil or capecitabine plus
cisplatin and epirubicin for locally advanced, resectable gastric
or gastrooesophageal junction adenocarcinoma (FLOT4): A randomised,
phase 2/3 trial. Lancet. 393:1948–1957. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Wu H, Minamide T and Yano T: Role of
photodynamic therapy in the treatment of esophageal cancer. Dig
Endosc. 31:508–516. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Hayashi T, Asahina Y, Nakanishi H,
Terashima T, Okamoto K, Yamada S, Takatori H, Kitamura K, Mizukoshi
E, Ninomiya I and Kaneko S: Evaluation of the efficacy and safety
of salvage photodynamic therapy by talaporfin sodium for cervical
esophageal cancers and lesions larger than 3 cm. Esophagus.
18:645–654. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Komanduri S, Muthusamy VR and Wani S:
Controversies in endoscopic eradication therapy for Barrett's
esophagus. Gastroenterology. 154:1861–1875.e1. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Overholt BF, Wang KK, Burdick JS,
Lightdale CJ, Kimmey M, Nava HR, Sivak MV Jr, Nishioka N, Barr H,
Marcon N, et al: Five-year efficacy and safety of photodynamic
therapy with photofrin in Barrett's high-grade dysplasia.
Gastrointest Endosc. 66:460–468. 2007. View Article : Google Scholar : PubMed/NCBI
|