Introduction
Although systemic therapy has improved outcomes in
advanced hepatocellular carcinoma (HCC), which accounts for the
vast majority of primary liver cancers and remains among the
leading causes of cancer-related death worldwide, durable disease
control remains limited for many patients, likely due to underlying
cirrhosis/liver dysfunction and tumor heterogeneity with an
immunosuppressive microenvironment that promotes primary or
acquired resistance (1). Thermal
ablation (TA) techniques, such as radiofrequency ablation (RFA) and
microwave ablation (MWA), are widely accepted for treating HCC,
especially for small liver cancers (2–5), and
offer a feasible alternative for patients with advanced HCC
(1,2,5). MWA
has been increasingly adopted due to its practical advantages,
including shorter ablation times and larger ablation zones, with
local control and overall survival (OS) broadly comparable to RFA
(6). MWA has also been reported to
induce abscopal effects, with untreated lesions showing regression
(7,8). However, similar to other local
treatments, recurrence and metastasis after MWA treatment remain
notable concerns for patients with HCC.
Immunotherapy can enhance antitumor immune
responses, restore immune recognition of tumor cells and provide
durable clinical benefit in a subset of patients with HCC,
particularly those who achieve an objective response or sustained
disease stabilization with immune checkpoint blockade (1,9,10). It
can induce long-lasting antitumor effects through immune memory,
achieving durable tumor control (9). Previous research suggests that
destruction of HCC lesions can induce or enhance systemic antitumor
immune responses (11–13). This response arises from the
tumor-associated antigens (TAAs) processed and presented by
antigen-presenting cells, followed by the amplification or
stimulation of antitumor T cell responses (13,14).
After TA, specific T cell reactions can be triggered, leading to an
increase in the proportion of circulating activated T cells and
natural killer cells, as well as the production of tumor-specific
antibodies (15,16). Accordingly, combining MWA with
immunotherapy may enhance clinical activity by reducing tumor
burden while potentially augmenting antitumor immunity.
Previous studies have shown that MWA can enhance the
body's immune response against tumors by releasing more TAAs,
particularly 3–8 days after treatment (15,17–19).
As ablation-associated immune modulation may be transient (18), combination strategies pairing MWA
with immunotherapy have been explored with the aim of improving
tumor control and clinical outcomes compared with MWA alone. Prior
translational and early clinical studies have suggested that local
ablation may augment antitumor immunity and provide a rationale for
combination strategies with immunotherapy (11,20).
This approach has attracted increasing interest in HCC, due to its
potential to enhance both local and systemic immune responses
(21).
Although prior studies have suggested potential
synergy between thermal ablation and immunotherapy in advanced HCC,
optimal management remains uncertain for patients who develop
limited progression or oligometastatic disease during systemic
therapy and remain technically eligible for local ablation
(11,20). Importantly, this clinical scenario
differs from prior reports that primarily evaluated ablation as an
upfront local therapy or in broader advanced-stage populations
without explicitly focusing on limited progression/oligometastatic
disease during systemic treatment (2–5,20). In
routine practice, these patients represent a distinct clinical
subset whom clinicians often consider treating with local
cytoreduction of a small number of escaping lesions while
continuing immunotherapy, with the intent to prolong systemic
disease control. However, real-world evidence specifically
evaluating MWA as a targeted salvage local intervention (rather
than upfront debulking) in this context remains limited. Therefore,
a retrospective cohort study was conducted to assess the efficacy
and safety of ICIs plus CT-guided MWA applied to ≤2 technically
feasible lesions vs. ICIs alone in advanced HCC patients presenting
with limited progression/oligometastatic disease.
Materials and methods
Participants
The present study screened consecutive patients with
advanced HCC treated at the Cancer Center of the Second People's
Hospital of Neijiang (Neijiang, China) between January 2022 and
January 2024. Eligible patients were those who, after prior
locoregional and/or systemic therapies, developed limited
progression/oligometastatic disease that was considered technically
amenable to CT-guided ablation and then received ICIs with or
without MWA. In the present study, limited
progression/oligometastatic disease was defined as the appearance
or growth of ≤3 intrahepatic and/or extrahepatic lesions, with the
largest lesion ≤5.0 cm, and no radiographic evidence of
macrovascular invasion [(including portal vein tumor thrombus
(PVTT)] at the time of treatment decision. According to the China
Liver Cancer (CNLC) staging system, included patients were stage
IIa, IIb or IIIb (generally corresponding to Barcelona Clinic Liver
Cancer stage B/C) (5). Prior
treatments commonly included transarterial chemoembolization and
hepatic arterial infusion chemotherapy. Baseline demographic,
laboratory and available imaging variables were extracted from
medical records. The overall cohort comprised 52 patients,
including 46 males (88.5%) and 6 females (11.5%), with a median age
of 58.5 years (range, 47–71 years). Screening confirmed that none
of the 52 patients had radiographic macrovascular invasion at the
time of treatment decision. Eligibility also required a largest
lesion ≤5.0 cm. While eligibility (largest lesion ≤5.0 cm and
site-level assessment) was verified by clinicians during the
initial screening of radiology reports and clinical notes to ensure
protocol compliance, the exact continuous numerical measurements
for all lesions and standardized categorical coding for each
specific extrahepatic site were not consistently recorded as
structured variables in the retrospective database (52/52 patients
for each variable). Consequently, these granular descriptors were
not available for formal quantitative comparison between groups.
The present study was approved by the Ethics Committee of the
Second People's Hospital of Neijiang (approval no. 2026RP-0106-03).
The committee approved retrospective use of de-identified clinical
data collected during routine care and waived the requirement for a
study-specific informed consent form. The present study is
registered with ClinicalTrials.gov (trial no. NCT06581497).
Eligibility criteria
Patients were included using the following
eligibility criteria: i) Had advanced HCC (CNLC IIa-IIIb); ii)
developed limited progression/oligometastatic disease defined as
the appearance or growth of ≤3 intrahepatic and/or extrahepatic
lesions with the largest lesion ≤5.0 cm; iii) had no radiographic
macrovascular invasion, including PVTT, at the time of treatment
decision; and iv) received PD-1-based ICIs with or without
CT-guided MWA as salvage local therapy. Patients were excluded if
they had diffuse progression beyond the oligometastatic definition,
uncontrolled comorbidities or contraindications to
ablation/immunotherapy per institutional practice, or insufficient
follow-up to assess outcomes.
Anti-programmed cell death protein 1
(PD-1) therapy and ablation combination procedures
In the present retrospective study, the ICIs used
were PD-1 inhibitors, including pembrolizumab (22), camrelizumab (23), toripalimab (24), tislelizumab (25) and sintilimab (26). ICI dosing and administration
followed the manufacturers' prescribing information. Evaluation of
treatment responses was conducted every 6–8 weeks using the
modified Response Evaluation Criteria in Solid Tumors (mRECIST)
(27). According to mRECIST,
complete response (CR) is defined as the disappearance of any
intratumoral arterial enhancement in all target lesions; partial
response (PR) is characterized by a ≥30% reduction in the sum of
diameters of viable (enhancing) target lesions from baseline; and
progressive disease (PD) is identified by an increase of ≥20% from
baseline. MWA was performed under CT guidance within 10–14 days
following the baseline imaging assessment to ensure feasibility.
Subsequent immunotherapy was administered or resumed within 3–7
days after ablation. The ablation procedure was intended to achieve
complete ablation of the target lesions (up to two lesions selected
based on technical feasibility), whether intrahepatic or
extrahepatic, in a single session. If up to three lesions met the
oligometastatic definition, MWA was prioritized for the
dominant/progressing lesions considered most technically feasible,
and the remaining lesions continued to be managed with ongoing
systemic therapy and/or other clinically indicated locoregional
approaches per the discretion of the treating physician.
Safety and efficacy
Adverse events (AEs) were monitored throughout
immunotherapy and up to 90 days after the last administered dose
and were graded according to the Common Terminology Criteria for
Adverse Events (CTCAE; version 5.0) (28). Continuous safety evaluations were
conducted. If a patient experienced Grade ≥3 AEs or intolerable
Grade 2 treatment-related AEs, treatment interruption and
supportive management were implemented according to institutional
practice and the prescribing information of the ICIs. Treatment was
resumed once AEs improved to Grade 1–2 when clinically appropriate;
permanent discontinuation was considered in cases of severe
toxicity or substantial clinical disease progression. In the
combination group, switching to an alternative ICI agent was
permitted at the discretion of the treating physician.
Procedure-related complications of ablation were assessed during
the immediate post-procedure period (0–24 h) and the
post-procedural period (1–30 days). Tumor response was evaluated by
imaging using mRECIST. Disease control rate (DCR) was defined as
the proportion of patients achieving CR, PR or stable disease (SD)
as their best overall response according to mRECIST. The waterfall
plot was based on mRECIST target-lesion response assessments
recorded for routine efficacy evaluation, rather than on uniformly
extractable baseline whole disease tumor burden descriptors;
therefore, these target lesion measurements cannot substitute for
standardized baseline maximum lesion diameter or metastatic site
coding.
Collection of data and study
objectives
The follow-up process included regular phone calls
or outpatient visits. All patients underwent postoperative imaging
and a comprehensive evaluation within 1 month after treatment.
Follow-up visits occurred every 3 weeks, during which blood tests
[a-fetoprotein (AFP), total bilirubin and alanine aminotransferase]
were performed. Contrast-enhanced CT or magnetic resonance imaging
(MRI) scans were conducted every 3 months.
Statistical analysis
The statistical analysis was performed using IBM
SPSS Statistics (version 26.0; IBM Corp.). As this was a
retrospective, single-center study with strict eligibility criteria
(limited progression/oligometastatic disease amenable to ablation),
the cohort size (n=52) reflected the number of consecutive eligible
patients treated during the study period rather than an a priori
sample-size calculation. The analysis should be regarded as
exploratory and hypothesis-generating. Effect sizes [hazard ratios
(HRs) with 95% CIs] are reported alongside P-values; findings
should be interpreted cautiously due to the limited sample size and
number of outcome events.
Propensity score matching (PSM) and inverse
probability of treatment weighting (IPTW) were considered; however,
they were not implemented because the small cohort size, limited
number of events and incomplete availability of key baseline
tumor-burden variables which could yield unstable matches or
extreme weights. The sample size is comparable to other
single-center real-world retrospective cohorts evaluating
locoregional therapy combined with immunotherapy in advanced HCC,
which commonly include several dozen patients (16,17).
Categorical variables are presented as frequencies
and percentages. Group comparisons were performed using Pearson's
χ2 test or Fisher's exact test, as appropriate based on
the expected frequencies [for example Pearson's χ2 test
for DCR and Fisher's exact test for objective response rate (ORR)
and other variables with small cell counts]. Continuous variables
were compared using the Mann-Whitney U test. OS and
progression-free survival (PFS) were estimated using the
Kaplan-Meier method and compared using the log-rank test. Median OS
and PFS with 95% CIs were calculated using the Brookmeyer-Crowley
method. HRs and 95% CIs were estimated using univariable and
multivariable Cox proportional hazards models to identify
independent prognostic factors. A two-sided P<0.05 was
considered to indicate a statistically significant difference. No
adjustment was applied for multiple comparisons; P-values are
therefore reported for descriptive inference. As the cohort size
and event counts were small, multivariable Cox estimates may be
unstable and should be interpreted as exploratory. The adjusted
models were therefore interpreted alongside the Kaplan-Meier curves
and univariable Cox results.
Results
Patient demographic and baseline
clinical characteristics
From January 2022 to January 2024, a total of 52
eligible patients meeting the inclusion and exclusion criteria were
enrolled in the present study. Among them, 31 patients underwent
ICI monotherapy, while 21 patients received a combination therapy
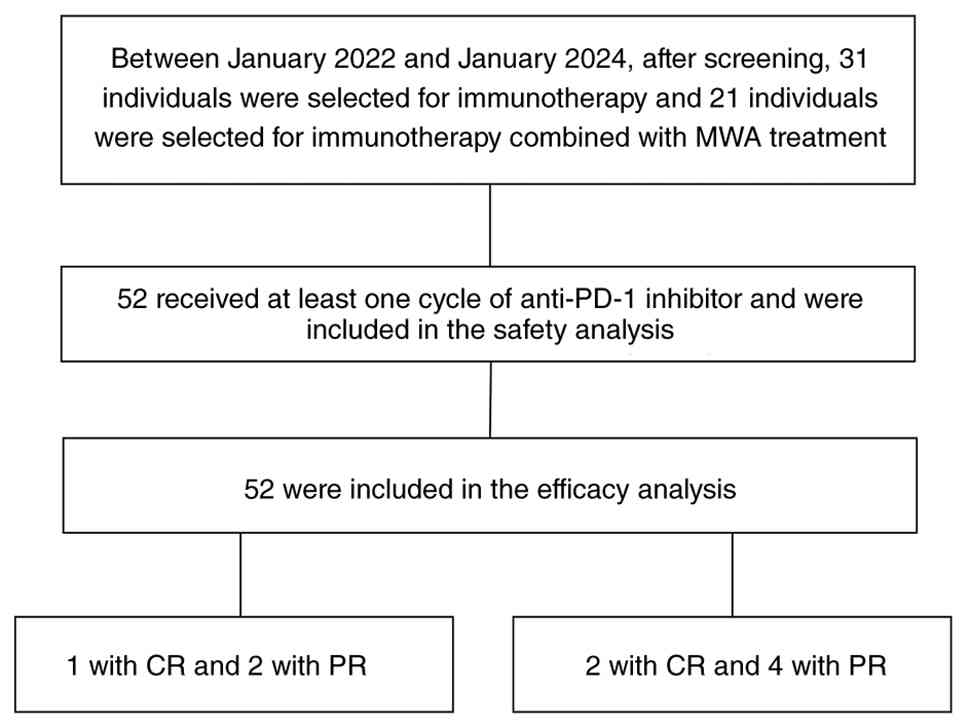
involving MWA and ICIs (Fig. 1).
The recorded baseline characteristics were broadly similar between
groups (Table I). The majority of
the study population was male (88.5%), and hepatitis B virus (HBV)
infection was identified as the primary cause of HCC in 88.5% of
the cases. Importantly, all patients testing positive for HBV
surface antigen received antiviral treatment, utilizing either
entecavir or tenofovir.
 | Table I.Patient baseline demographic and
clinical characteristics. |
Table I.
Patient baseline demographic and
clinical characteristics.
| Variables | ICIs (n=31) | MWA + ICIs
(n=21) | P-value |
|---|
| Median age, years
(range) | 59 (48–71) | 58 (47–69) | 0.555 |
| Age category,
yearsa |
|
|
|
|
<65 | 21 (67.7) | 15 (71.4) |
|
|
≥65 | 10 (32.3) | 6 (28.6) |
|
| Sex |
|
| 0.380 |
|
Male | 26 (83.9) | 20 (95.2) |
|
|
Female | 5 (16.1) | 1 (4.8) |
|
| ECOG performance
status |
|
| 0.282 |
| 0 | 13 (41.9) | 12 (57.1) |
|
| 1 | 18 (58.1) | 9 (42.9) |
|
| Hepatitis B surface
antigen |
|
| 0.672 |
|
Negative | 3 (9.7) | 3 (14.3) |
|
|
Positive | 28 (90.3) | 18 (85.7) |
|
| Hepatitis C virus
antibody |
|
| 0.672 |
|
Negative | 28 (90.3) | 18 (85.7) |
|
|
Positive | 3 (9.7) | 3 (14.3) |
|
| AFP |
|
| 0.327 |
| <400
ng/ml | 13 (41.9) | 6 (28.6) |
|
| ≥400
ng/ml | 18 (58.1) | 15 (71.4) |
|
| CNLC stage |
|
| 0.895 |
|
IIa | 5 (16.1) | 3 (14.3) |
|
|
IIb | 12 (38.7) | 7 (33.3) |
|
|
IIIb | 14 (45.2) | 11 (52.4) |
|
| Total
bilirubin |
|
| 0.943 |
| ≤20
µmol/l | 13 (41.9) | 9 (42.9) |
|
| >20
µmol/l | 18 (58.1) | 12 (57.1) |
|
| Alanine
aminotransferase |
|
| 0.236 |
| <50
U/l | 17 (54.8) | 8 (38.1) |
|
| ≥50
U/l | 14 (45.2) | 13 (61.9) |
|
| Number of active
tumors |
|
| 0.822 |
| 1 | 23 (74.2) | 15 (71.4) |
|
|
2-3 | 8 (25.8) | 6 (28.6) |
|
Treatment efficacy
Short-term therapeutic efficacy
Among the 31 patients receiving ICIs, 1 (3.2%)
achieved CR, 2 (6.5%) achieved PR, 13 (41.9%) had SD, 13 (41.9%)
had PD and 2 (6.5%) were not assessable. By contrast, among the 21
patients treated with MWA plus ICIs, 2 (9.5%) achieved CR, 4
(19.0%) achieved PR, 11 (52.4%) had SD, 3 (14.3%) had PD and 1
(4.8%) was not assessable (Table
II). The ORR was 28.6% (6/21) in the combination group vs. 9.7%
(3/31) with ICIs alone, and the DCRs were 81.0% (17/21) vs. 51.6%
(16/31), respectively. Based on the distribution of expected
frequencies, the improvement in DCR reached statistical
significance (P=0.031; Pearson's χ2 test). ORR was
higher in the combination group but was not statistically
significant (P=0.133; Fisher's exact test). A total of 3 patients
had non-assessable responses due to atypical post-treatment imaging
findings and/or insufficient imaging data that precluded
classification as CR, PR, SD or PD according to mRECIST; these
cases were recorded as ‘not assessable’ in Table II. Fig.
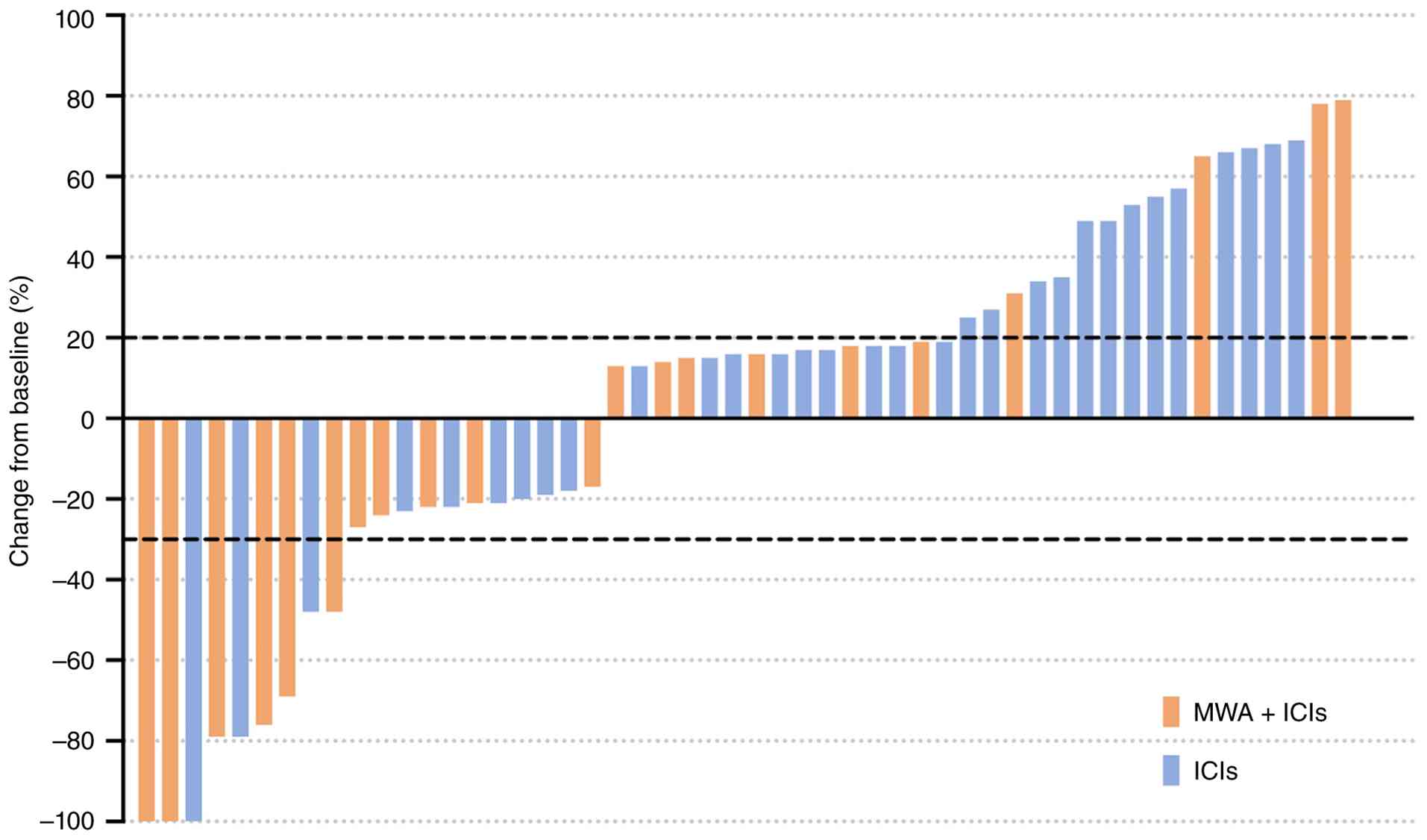
2 shows that a greater proportion of patients in the MWA plus
ICI group experienced tumor shrinkage of the target lesions and
fewer patients had marked target-lesion growth compared with the
ICI-alone group, consistent with the higher DCR observed in the
combination cohort. Fig. 3 further
demonstrates that responses and disease stabilization were
generally more durable in the MWA plus ICI group, whereas earlier
progression events were more frequent with ICI monotherapy.
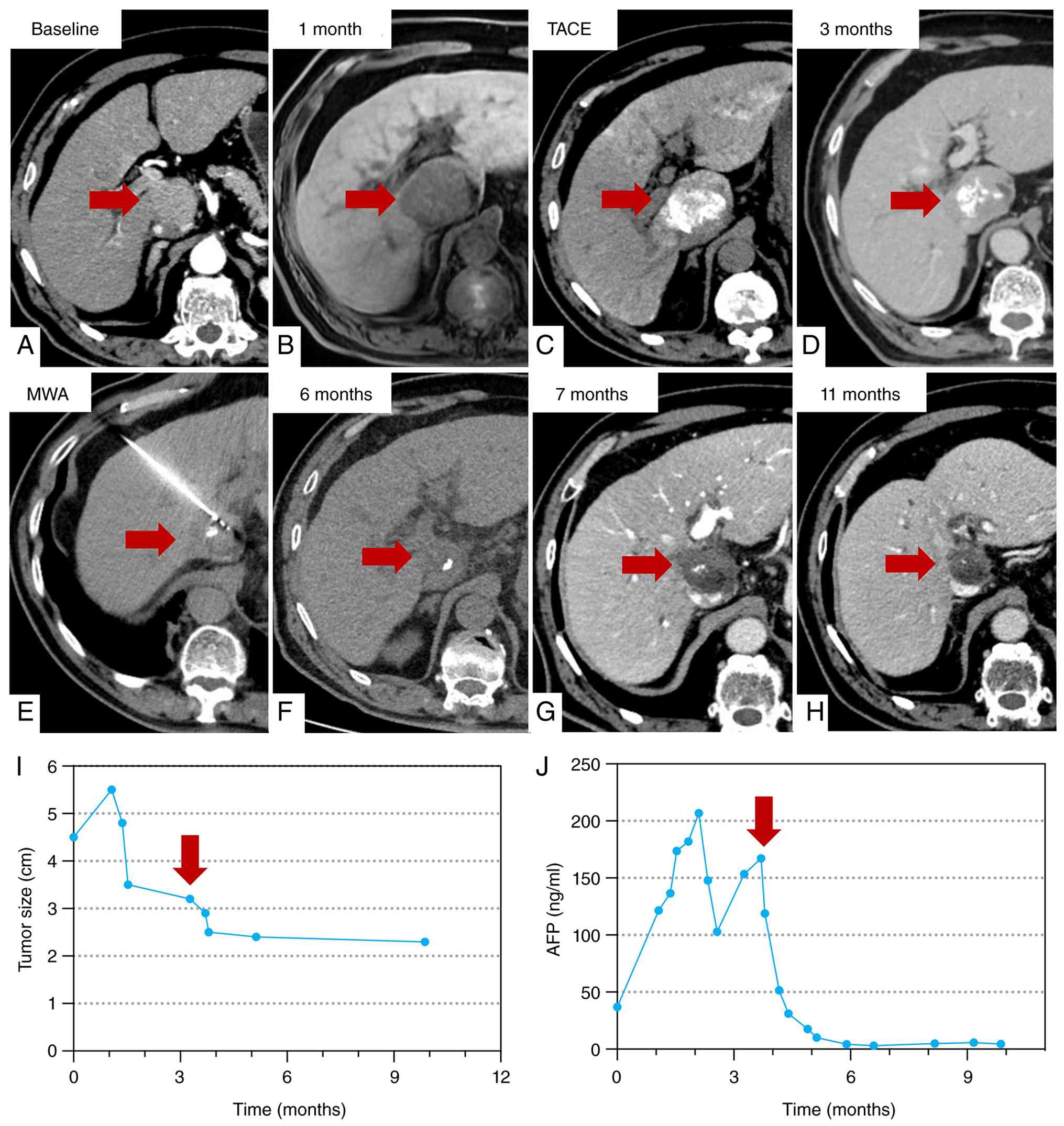
Fig. 4, Fig. 5, Fig.
6 provide representative longitudinal imaging examples from the
combination group, illustrating complete ablation of the treated
lesions with loss of arterial enhancement on follow-up CT/MRI,
accompanied by decreasing target-lesion size over time and
corresponding declines in serum AFP in responding patients.
 | Table II.Short-term efficacy. |
Table II.
Short-term efficacy.
| Response | ICIs (n=31) | ICIs + MWA
(n=21) | P-value |
|---|
| CR | 1 (3.2) | 2 (9.5) | - |
| PR | 2 (6.5) | 4 (19.0) | - |
| SD | 13 (41.9) | 11 (52.4) | - |
| PD | 13 (41.9) | 3 (14.3) | - |
| Not assessable | 2 (6.5) | 1 (4.8) | - |
| ORRa, % | 9.7 | 28.6 | 0.133 |
| DCRb, % | 51.6 | 81.0 | 0.031 |
Long-term efficacy
The data cutoff was June 2025; median OS for the
overall cohort was 15.1 months and median PFS was 6.0 months
(Fig. 7A and B). Compared with the
ICI monotherapy group, the MWA plus ICIs group achieved a longer
median OS (11.0 vs. 16.8 months; log-rank P=0.008; Fig. 7C). The median PFS was also longer in
the combination group (4.0 vs. 9.0 months; log-rank P<0.001;
Fig. 7D). HRs with 95% CIs were
estimated using Cox proportional hazards models and are provided in
Tables III and IV; due to the small number of events, the
adjusted OS HR should be interpreted together with the Kaplan-Meier
curves and the more conservative univariable estimate.
 | Table III.Univariate and multivariate analysis
of overall survival in patients with advanced hepatocellular
carcinoma. |
Table III.
Univariate and multivariate analysis
of overall survival in patients with advanced hepatocellular
carcinoma.
|
| Univariate
analysis | Multivariate
analysis |
|---|
|
|
|
|
|---|
| Variables | HR | P-value | HR | P-value |
|---|
| Age (per year) | 1.040
(0.994–1.089) | 0.090 | 1.025
(0.972–1.082) | 0.363 |
| Sex
(male/female) | 1.091
(0.324–3.669) | 0.888 | 0.772
(0.185–3.225) | 0.723 |
| ECOG performance
status (0/1) | 1.777
(0.755–4.18) | 0.188 | 1.592
(0.544–4.658) | 0.395 |
| Hepatitis B surface
antigen | 0.778
(0.231–2.615) | 0.685 | 0.435
(0.101–1.871) | 0.263 |
| AFP (<400/≥400
ng/ml) | 2.558
(1.011–6.467) | 0.047 | 5.912
(1.669–20.941) | 0.006 |
| CNLC stage |
|
|
|
|
|
IIb | 1.396
(0.608–3.207) | 0.432 | 0.903
(0.214–3.799) | 0.889 |
|
IIIb | 0.728
(0.314–1.688) | 0.459 | 1.604
(0.38–6.761) | 0.520 |
| Total
bilirubin | 1.447
(0.633–3.31) | 0.381 | 2.154
(0.783–5.924) | 0.137 |
| Alanine
aminotransferase | 0.928
(0.406–2.122) | 0.860 | 1.093
(0.411–2.908) | 0.858 |
| Number of active
tumors | 1.044
(0.411–2.651) | 0.928 | 1.046
(0.315–3.477) | 0.942 |
| Treatment (MWA +
ICIs vs. ICIs) | 0.585
(0.155–0.992) | 0.046 | 0.176
(0.048–0.642) | 0.008 |
 | Table IV.Univariate and multivariate analysis
of PFS in patients with advanced HCC. |
Table IV.
Univariate and multivariate analysis
of PFS in patients with advanced HCC.
|
| Univariate
analysis | Multivariate
analysis |
|---|
|
|
|
|
|---|
| Variables | HR | P-value | HR | P-value |
|---|
| Age (per year) | 1.000
(0.971–1.03) | 0.997 |
1.010(0.976–1.045) | 0.578 |
| Sex
(male/female) | 0.691
(0.289–1.656) | 0.408 | 0.398
(0.134–1.184) | 0.098 |
| ECOG performance
status (0/1) | 1.060
(0.573–1.96) | 0.853 | 0.779
(0.36–1.685) | 0.527 |
| Hepatitis B surface
antigen | 0.943
(0.368–2.416) | 0.903 | 0.510
(0.169–1.543) | 0.233 |
| AFP(<400/≥400
ng/ml) | 2.276
(1.188–4.361) | 0.013 | 4.327
(1.853–10.106) | 0.001 |
| CNLC stage |
|
|
|
|
|
IIb | 1.082
(0.582–2.013) | 0.802 | 0.803
(0.274–2.357) | 0.690 |
|
IIIb | 0.843
(0.453–1.569) | 0.589 | 1.368
(0.504–3.713) | 0.539 |
| Total
bilirubin | 1.023
(0.557–1.878) | 0.943 | 0.891
(0.45–1.763) | 0.740 |
| Alanine
aminotransferase | 1.068
(0.585–1.949) | 0.831 | 0.790
(0.389–1.605) | 0.515 |
| Number of active
tumors | 1.266
(0.619–2.59) | 0.519 | 1.512
(0.588–3.887) | 0.391 |
| Treatment (MWA +
ICIs vs. ICIs) | 0.386
(0.198–0.753) | 0.005 | 0.270
(0.122–0.595) | 0.001 |
In patients with AFP ≥400 ng/ml, median OS (9.0 vs.
17.0 months; log-rank P=0.042; Fig.
8A) and median PFS (2.5 vs. 8.0 months; log-rank P<0.001;
Fig. 8B) were longer in the MWA
plus ICIs group. In the subgroup with AFP <400 ng/ml, no
statistically significant differences in OS or PFS were observed
between groups (Fig. 8C and D).
Analysis of prognostic factors
The Cox univariate analysis revealed that OS and PFS
were influenced by factors such as MWA combined with ICIs treatment
and AFP concentration. Elevated AFP concentration was identified as
a risk factor associated with decreased OS (P=0.047) and PFS
(P=0.013). Conversely, receipt of MWA plus ICIs was associated with
improved OS (HR=0.585; 95% CI, 0.155–0.992; P=0.046) and PFS
(HR=0.386; 95% CI, 0.198–0.753; P=0.005) in univariable Cox models.
These associations remained significant in multivariable Cox models
adjusting for available covariates for both OS (HR=0.176; 95% CI,
0.048–0.642; Wald P=0.008) and PFS (HR=0.270; 95% CI, 0.122–0.595;
Wald P=0.001) (Tables III and
IV). The adjusted OS HR was
notably smaller than the univariable estimate and may reflect an
unstable model in this small dataset. The OS trend is therefore
more informative than the exact adjusted OS point estimate.
Notably, the adjusted HR for AFP in the OS model (HR=5.912; 95% CI,
1.669–20.941) showed a wide confidence interval, suggesting
potential sparse-data bias or model overfitting due to the small
number of events; therefore, this covariate estimate should be
interpreted with caution.
Safety profile
Safety analyses were descriptive only. The present
study did not observe a clear unexpected safety pattern, but it was
underpowered for formal between-group comparisons (Table V). Any-grade hepatic laboratory
abnormalities were common in both groups. Numerically, some
laboratory abnormalities were more frequent in the combination
cohort (such as Grade 3–4 transaminitis was 33.3 vs. 0.0%), which
is consistent with the transient acute liver injury expected
following thermal ablation, but between-group comparisons were
underpowered and should be interpreted descriptively.
Treatment-emergent AEs were defined as events occurring during ICI
therapy and up to 90 days after the last dose; procedure-related
events following MWA were additionally assessed within 0–24 h and
1–30 days post-procedure. The final AE tabulation applied the same
prespecified safety window uniformly across both cohorts to
minimize misclassification. Between-group comparisons were
descriptive and used Fisher's exact test because of small cell
counts, without adjustment for multiple comparisons. Transaminitis
and hyperbilirubinemia were the most common AEs in both groups. The
high incidence of any-grade hyperbilirubinemia in the combination
cohort (85.7%) may, at least in part, reflect transient
post-ablation liver injury rather than prolonged systemic drug
toxicity. In the combination cohort, Grade 3–4 transaminitis was
observed in 7 patients (33.3%) and Grade 3–4 hyperbilirubinemia in
5 patients (23.8%). In the ICIs alone cohort, Grade 3–4
transaminitis was not observed, although Grade 3–4
hyperbilirubinemia occurred in 3 patients (9.7%).
 | Table V.Comparison of complications between
the two groups. |
Table V.
Comparison of complications between
the two groups.
|
| Grade 3–4 | Any
gradea |
|---|
|
|
|
|
|---|
| AEs | ICIs + MWA
(n=21) | ICIs (n=31) | P-value | ICIs + MWA
(n=21) | ICIs (n=31) | P-value |
|---|
| Fatigue | 0 | 0 | - | 0 | 2 (6.5) | 0.509 |
| Transaminitis | 7 (33.3) | 0 | 0.001 | 7 (33.3) | 11 (35.5) | >0.999 |
| Fever | 0 | 0 | - | 4 (19.0) | 2 (6.5) | 0.207 |
| Diarrhea | 0 | 0 | - | 8 (38.1) | 13 (41.9) | >0.999 |
| Pneumonitis | 0 | 0 | - | 4 (19.0) | 2 (6.5) | 0.207 |
|
Hyperbilirubinemia | 5 (23.8) | 3 (9.7) | 0.244 | 18 (85.7) | 20 (64.5) | 0.118 |
| Hypothyroidism | 0 | 0 | - | 0 | 2 (6.5) | 0.509 |
| Pruritus | 0 | 0 | - | 2 (9.5) | 11 (35.5) | 0.050 |
| Rash | 0 | 0 | - | 2 (9.5) | 11 (35.5) | 0.050 |
|
Hyperthyroidism | 0 | 0 | - | 0 | 1 (3.2) | >0.999 |
|
Hypoalbuminemia | 0 | 0 | - | 13 (61.9) | 11 (35.5) | 0.090 |
| Nausea | 0 | 0 | - | 0 | 1 (3.2) | >0.999 |
Regarding the categorization of AEs, while all
events were monitored, immune-related AEs (irAEs) were not
prospectively tracked as a distinct, strictly defined category
separate from other treatment-emergent or procedure-related AEs in
the present retrospective dataset. These events were managed with
temporary ICI interruption when indicated, hepatoprotective therapy
and corticosteroids when an immune-related mechanism was clinically
suspected, and they generally improved to Grade 1–2 without
permanent organ dysfunction. Treatment discontinuation due to
toxicity occurred in 3/21 patients (14.3%) in the combination group
and 5/31 patients (16.1%) in the monotherapy group, whereas
discontinuation due to disease progression occurred in 11/21
(52.4%) and 19/31 (61.3%) patients, respectively.
With respect to MWA-related local reactions, events
were primarily consistent with post-ablation syndrome (such as
transient fever and localized pain) and resolved within a few days
with conservative symptomatic care. No major procedure-related
complications (severe hemorrhage, bile duct injury, liver abscess
or treatment-related mortality) occurred in the combination
cohort.
Discussion
MWA is widely used as a locoregional treatment for
HCC and is recommended as a first-line option for local therapy in
the 2022 Chinese Society of Clinical Oncology guidelines (29). The high temperatures generated
during MWA lead to coagulation and necrosis of HCC tissue, reducing
tumor burden and disrupting the immunosuppressive environment of
the tumor. This process exposes TAAs, promotes the synthesis of
heat shock proteins and effectively activates antitumor immune
responses (14,21). Despite its potential, the efficacy
of MWA may be limited by high postoperative recurrence and
metastasis rates.
Immunosuppression serves a key role in cancer
recurrence and metastasis (30–34).
Immunotherapy aims to boost the body's immune system to TAAs and
induce tumor cell death. When combined with other treatments,
immunotherapy can reduce the risk of both local and distant
recurrence. However, tumor-induced immune suppression often weakens
the effectiveness of immunotherapy. MWA helps to overcome this by
eliminating tumor cells and releasing antigens from apoptotic and
necrotic tissue. Evidence from animal models and clinical studies
suggests that MWA induces immunogenic tumor cell death, releasing
tumor-associated antigens and damage-associated molecular patterns
(such as high mobility group box 1/ATP/heat-shock proteins) that
promote dendritic-cell activation and antigen cross-presentation,
thereby priming cytotoxic CD8+ T cell responses via type
I interferon-linked pathways (including cyclic GMP-AMP
synthase-stimulator of interferon genes), while also modulating the
tumor microenvironment and checkpoint signaling (such as programmed
death ligand 1 upregulation), providing a rationale for combination
with ICIs (1,11,14).
However, this response tends to be short-lived, and the post-MWA
window may be the optimal time to enhance antitumor immunity.
Accordingly, combining MWA with immunotherapy has
been proposed as a potentially effective treatment strategy
(8,15,20,21).
However, while earlier proof-of-concept and retrospective reports
have established the rationale for this combination, data
specifically focusing on its use as a rescue therapy for patients
with limited progression remain relatively scarce. The present
study builds on prior reports by evaluating this clinical scenario
in a real-world setting. In the selected retrospective cohort, MWA
as a targeted intervention for limited progression was associated
with longer mPFS (9.0 vs. 4.0 months) and mOS (16.8 vs. 11.0
months) than ICI monotherapy. The direction of association was
consistent in the Kaplan-Meier analyses and in both univariable and
multivariable Cox models, but the adjusted OS effect size should be
interpreted cautiously in view of model instability.
The outcomes of the present study should not be
directly compared with external trials because of differences in
patient selection, line of therapy and study design. For instance,
in the KEYNOTE-224 trial evaluating second-line pembrolizumab
monotherapy, the ORR was 17%, with an mPFS of 4.9 months and an mOS
of 12.9 months (22). The present
ICIs monotherapy group demonstrated comparable baseline outcomes
(ORR of 9.7%, mPFS of 4.0 months and mOS of 11.0 months). By
contrast, the present combination cohort achieved a significantly
improved DCR of 81.0% and a numerically higher ORR of 28.6%,
alongside extended mPFS (9.0 months) and mOS (16.8 months). The
between-group difference in ORR did not reach significance, which
may reflect limited power in this small cohort. These findings are
broadly consistent with prior retrospective reports of
ablation-immunotherapy combinations, which have reported median PFS
values in the range of 5–8 months (17,20).
The observed survival differences may reflect, at least in part, a
combination of local cytoreduction by MWA and immunologic effects
related to tumor-associated antigen release, which could
potentially augment systemic ICI activity. In the univariable Cox
analysis, the OS HR was 0.585, whereas in the multivariable model
the adjusted HR shifted further away from the null (HR=1.0) to
0.176, suggesting a stronger apparent association after covariate
adjustment; however, due to the small sample size and limited
number of events, this shift may reflect model
sensitivity/instability and should be interpreted cautiously. In
addition, log-rank and Cox tests are based on different statistics
and may yield different P-values, particularly in small cohorts
with limited events. This discrepancy, together with the wide
confidence interval, suggests possible sparse-data bias or
overfitting in the small-event setting. Therefore, interpretation
should emphasize the consistent direction of association across
Kaplan-Meier and Cox analyses rather than the point estimate of the
adjusted OS HR. For PFS, receipt of MWA plus ICIs was also
associated with improved outcome after adjustment after adjusting
for available covariates, such as baseline AFP levels. The present
study was not powered to establish comparable safety between
groups, and the AE findings should be interpreted
descriptively.
The present study is limited by its retrospective
design and potential selection bias. Patients selected for MWA
necessarily had anatomically feasible lesions, which may have been
associated with a more favorable baseline disease profile than that
of patients treated with ICIs alone. Screening confirmed that none
of the 52 patients had radiographic macrovascular invasion at the
time of treatment decision, and eligibility required a largest
lesion ≤5.0 cm. While eligibility (largest lesion ≤5.0 cm and
site-level assessment) was verified by clinicians during the
initial screening of radiology reports and clinical notes to ensure
protocol compliance, the exact continuous numerical measurements
for all lesions and standardized categorical coding for each
specific extrahepatic site were not consistently recorded as
structured variables in the retrospective database (52/52 patients
for each variable). Consequently, these granular descriptors were
not available for formal quantitative comparison between groups, so
residual confounding in tumor burden cannot be excluded. Due to the
small cohort size and limited number of events, multivariable Cox
estimates, particularly for OS, may be unstable and should be
interpreted as exploratory and alongside the Kaplan-Meier curves
and univariable Cox results. The present study did not use
propensity-based methods (PSM/IPTW) because the key baseline
tumor-burden variables needed for a credible propensity model were
not available, and the small sample could yield unstable matches or
extreme weights.
In the present small retrospective real-world
cohort, MWA plus immunotherapy was feasible and was associated with
improved OS and PFS compared with ICI monotherapy in selected
patients with advanced HCC and limited progression. These
observations are exploratory, and the adjusted OS estimate should
be interpreted cautiously due to the small dataset; confirmation in
larger prospective studies is required.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study are not
publicly available due to institutional and privacy restrictions
but may be requested from the corresponding author.
Authors' contributions
CW and YL conceived and designed the study. CW, SC,
LD, TZ, FH, YW, OJ and YL collected the clinical data from
electronic medical records, organized and structured the variables
into a standardized database for statistical analysis, and verified
the accuracy of the records. CW, OJ and YL performed the
statistical analysis and generated the figures and tables. CW and
YL drafted the manuscript, and all authors critically revised the
manuscript for important intellectual content. OJ and YL supervised
the study. CW and YL confirm the authenticity of all the raw data.
All authors read and approved the final version of the
manuscript.
Ethics approval and consent to
participate
The present retrospective protocol was reviewed and
approved by the Ethics Committee of the Second People's Hospital of
Neijiang (approval no. 2026RP-0106-03) in accordance with the
Declaration of Helsinki. The approval specifically authorized the
retrospective analysis of de-identified clinical data from the
2022–2024 patient cohort and was obtained prior to data extraction
and statistical analysis. Under the hospital's routine admission
process, patients provided generic written consent for the research
use of de-identified clinical data. As the present study was a
retrospective analysis of existing records, the requirement for
study-specific informed consent was waived by the ethics committee.
The present study is registered with ClinicalTrials.gov (trial no.
NCT06581497).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Llovet JM, Kelley RK, Villanueva A, Singal
AG, Pikarsky E, Roayaie S, Lencioni R, Koike K, Zucman-Rossi J and
Finn RS: Hepatocellular carcinoma. Nat Rev Dis Primers. 7:62021.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Heimbach JK, Kulik LM, Finn RS, Sirlin CB,
Abecassis MM, Roberts LR, Zhu AX, Murad MH and Marrero JA: AASLD
guidelines for the treatment of hepatocellular carcinoma.
Hepatology. 67:358–380. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
European Association for the Study of the
Liver, . EASL clinical practice guidelines: Management of
hepatocellular carcinoma. J Hepatol. 69:182–236. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Vogel A, Meyer T, Sapisochin G, Salem R
and Saborowski A: Hepatocellular carcinoma. Lancet. 400:1345–1362.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Xie DY, Ren ZG, Zhou J, Fan J and Gao Q:
2019 Chinese clinical guidelines for the management of
hepatocellular carcinoma: Updates and insights. Hepatobiliary Surg
Nutr. 9:452–463. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Facciorusso A, Di Maso M and Muscatiello
N: Microwave ablation versus radiofrequency ablation for the
treatment of hepatocellular carcinoma: A systematic review and
meta-analysis. Int J Hyperthermia. 32:339–344. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Cerrito L, Pallozzi M, Urbani I, Archilei
S, Miliani S, Creta E, Stella L, Gasbarrini A and Ponziani FR:
Unexpected therapeutic implications: The abscopal effect in the
management of hepatocellular carcinoma. Cancers (Basel).
18:4082026. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Formenti SC and Demaria S: Combining
radiotherapy and cancer immunotherapy: A paradigm shift. J Natl
Cancer Inst. 105:256–265. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
El-Khoueiry AB, Sangro B, Yau T, Crocenzi
TS, Kudo M, Hsu C, Kim TY, Choo SP, Trojan J, Welling THR, et al:
Nivolumab in patients with advanced hepatocellular carcinoma
(CheckMate 040): An open-label, non-comparative, phase 1/2 dose
escalation and expansion trial. Lancet. 389:2492–2502. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Finn RS, Qin S, Ikeda M, Galle PR, Ducreux
M, Kim TY, Kudo M, Breder V, Merle P, Kaseb AO, et al: Atezolizumab
plus bevacizumab in unresectable hepatocellular carcinoma. N Engl J
Med. 382:1894–1905. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Greten TF, Mauda-Havakuk M, Heinrich B,
Korangy F and Wood BJ: Combined locoregional-immunotherapy for
liver cancer. J Hepatol. 70:999–1007. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Shi L, Chen L, Wu C, Zhu Y, Xu B, Zheng X,
Sun M, Wen W, Dai X, Yang M, et al: PD-1 blockade boosts
radiofrequency ablation-elicited adaptive immune responses against
tumor. Clin Cancer Res. 22:1173–1184. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zerbini A, Pilli M, Penna A, Pelosi G,
Schianchi C, Molinari A, Schivazappa S, Zibera C, Fagnoni FF,
Ferrari C and Missale G: Radiofrequency thermal ablation of
hepatocellular carcinoma liver nodules can activate and enhance
tumor-specific T-cell responses. Cancer Res. 66:1139–1146. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ali MY, Grimm CF, Ritter M, Mohr L,
Allgaier HP, Weth R, Bocher WO, Endrulat K, Blum HE and Geissler M:
Activation of dendritic cells by local ablation of hepatocellular
carcinoma. J Hepatol. 43:817–822. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Nobuoka D, Motomura Y, Shirakawa H,
Yoshikawa T, Kuronuma T, Takahashi M, Nakachi K, Ishii H, Furuse J,
Gotohda N, et al: Radiofrequency ablation for hepatocellular
carcinoma induces glypican-3 peptide-specific cytotoxic T
lymphocytes. Int J Oncol. 40:63–70. 2012.PubMed/NCBI
|
|
16
|
Leuchte K, Staib E, Thelen M, Gödel P,
Lechner A, Zentis P, Garcia-Marquez M, Waldschmidt D, Datta RR,
Wahba R, et al: Microwave ablation enhances tumor-specific immune
response in patients with hepatocellular carcinoma. Cancer Immunol
Immunother. 70:893–907. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Li X, Zhang Q, Lu Q, Cheng Z, Liu F, Han
Z, Yu X, Yu J and Liang P: Microwave ablation combined with
apatinib and camrelizumab in patients with advanced hepatocellular
carcinoma: A single-arm, preliminary study. Front Immunol.
13:10239832022. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Bo XW, Sun LP, Yu SY and Xu HX: Thermal
ablation and immunotherapy for hepatocellular carcinoma: Recent
advances and future directions. World J Gastrointest Oncol.
13:1397–1411. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Chu KF and Dupuy DE: Thermal ablation of
tumours: Biological mechanisms and advances in therapy. Nat Rev
Cancer. 14:199–208. 2014. View
Article : Google Scholar : PubMed/NCBI
|
|
20
|
Duffy AG, Ulahannan SV, Makorova-Rusher O,
Rahma O, Wedemeyer H, Pratt D, Davis JL, Hughes MS, Heller T,
ElGindi M, et al: Tremelimumab in combination with ablation in
patients with advanced hepatocellular carcinoma. J Hepatol.
66:545–551. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Dumolard L, Ghelfi J, Roth G, Decaens T
and Macek Jilkova Z: Percutaneous ablation-induced immunomodulation
in hepatocellular carcinoma. Int J Mol Sci. 21:43982020. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhu AX, Finn RS, Edeline J, Cattan S,
Ogasawara S, Palmer D, Verslype C, Zagonel V, Fartoux L, Vogel A,
et al: Pembrolizumab in patients with advanced hepatocellular
carcinoma previously treated with sorafenib (KEYNOTE-224): A
non-randomised, open-label phase 2 trial. Lancet Oncol. 19:940–952.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Xu J, Shen J, Gu S, Zhang Y, Wu L, Wu J,
Shao G, Zhang Y, Xu L, Yin T, et al: Camrelizumab in combination
with apatinib in patients with advanced hepatocellular carcinoma
(RESCUE): A nonrandomized, open-label, phase II trial. Clin Cancer
Res. 27:1003–1011. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
He MK, Liang RB, Zhao Y, Xu YJ, Chen HW,
Zhou YM, Lai ZC, Xu L, Wei W, Zhang YJ, et al: Lenvatinib,
toripalimab, plus hepatic arterial infusion chemotherapy versus
lenvatinib alone for advanced hepatocellular carcinoma. Ther Adv
Med Oncol. 13:175883592110027202021. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Shen L, Guo J, Zhang Q, Pan H, Yuan Y, Bai
Y, Liu T, Zhou Q, Zhao J, Shu Y, et al: Tislelizumab in Chinese
patients with advanced solid tumors: An open-label,
non-comparative, phase 1/2 study. J Immunother Cancer.
8:e0004372020. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Hoy SM: Sintilimab: First global approval.
Drugs. 79:341–346. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Lencioni R and Llovet JM: Modified RECIST
(mRECIST) assessment for hepatocellular carcinoma. Semin Liver Dis.
30:52–60. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
National Cancer Institute (NCI), . Common
terminology criteria for adverse events (CTCAE) version 5.0. Cancer
Therapy Evaluation Program. NCI; Bethesda, MD: 2017
|
|
29
|
Jiang Z, Li J, Chen J, Liu Y, Wang K, Nie
J, Wang X, Hao C, Yin Y, Wang S, et al: Chinese society of clinical
oncology (CSCO) breast cancer guidelines 2022. Transl Breast Cancer
Res. 3:132022. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Chen KJ, Lin SZ, Zhou L, Xie HY, Zhou WH,
Taki-Eldin A and Zheng SS: Selective recruitment of regulatory T
cell through CCR6-CCL20 in hepatocellular carcinoma fosters tumor
progression and predicts poor prognosis. PLoS One. 6:e246712011.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Fu J, Xu D, Liu Z, Shi M, Zhao P, Fu B,
Zhang Z, Yang H, Zhang H, Zhou C, et al: Increased regulatory T
cells correlate with CD8 T-cell impairment and poor survival in
hepatocellular carcinoma patients. Gastroenterology. 132:2328–2339.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Gao Q, Qiu SJ, Fan J, Zhou J, Wang XY,
Xiao YS, Xu Y, Li YW and Tang ZY: Intratumoral balance of
regulatory and cytotoxic T cells is associated with prognosis of
hepatocellular carcinoma after resection. J Clin Oncol.
25:2586–2593. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Moeini A, Torrecilla S, Tovar V, Montironi
C, Andreu-Oller C, Peix J, Higuera M, Pfister D, Ramadori P, Pinyol
R, et al: An immune gene expression signature associated with
development of human hepatocellular carcinoma identifies mice that
respond to chemopreventive agents. Gastroenterology.
157:1383–1397.e11. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Yeung OWH, Lo CM, Ling CC, Qi X, Geng W,
Li CX, Ng KTP, Forbes SJ, Guan XY, Poon RTP, et al: Alternatively
activated (M2) macrophages promote tumour growth and invasiveness
in hepatocellular carcinoma. J Hepatol. 62:607–616. 2015.
View Article : Google Scholar : PubMed/NCBI
|