Introduction
The importance of chemical carcinogens in the
development of bladder cancer has been well established and
generally accepted. This carcinogenesis process occurs as a
multistep process. Preneoplastic lesions, differently progressed,
but with clonally related genomes, exist prior to in situ
and invasive carcinoma, and may locally contribute to the
independent formation of tumours (1).
Animal models of cancer have been fundamental for
the demonstration of this multistep nature (2). Several rodent models have been
established to study these different stages of urinary bladder
chemical carcinogenesis, namely chemical initiation and promotion,
and the stages of progression (3,4). The
administration of N-butyl-N-(4-hydroxybutyl)-nitrosamine (BBN) to
mice (5–7) or rats (7–11) is
one of the most widely used chemical carcinogens.
The resultant induced tumours in these experimental
models have been histologically well studied. However, little is
known regarding the biological features and genetic similarities
between rodent and human bladder cancer models.
This study aimed to identify biological similarities
between human bladder carcinogenesis and rat chemical-induced
bladder carcinogenesis by assessing DNA content alterations and p53
and Ki-67 immunoexpression, in order to use this preclinical animal
model in future therapeutic experimental studies.
Materials and methods
In this study, we compared biological data collected
from our previous studies, in which human (12) and rat (10) bladder samples with premalignant and
malignant lesions and adjacent mucosa were studied with regards to
histopathology, DNA content, p53 immunoexpression and the Ki-67
labelling index.
Human bladder samples
Consecutive radical cystectomy specimens from 49
patients with previous bladder cancer (non-invasive and invasive
urothelial cell carcinomas), consecutively admitted and treated at
the Instituto Português de Oncologia do Porto, Portugal, between
1989 and 1996, with available archival material for the image
cytometric study and patient consent, were included in the present
study. Only high-risk non-invasive urothelial cell carcinomas
received prior intravesical Bacillus Calmette-Guérin treatment.
Clinical data were collected from the patient records and the
paraffin blocks from the Department of Pathology. All of the
lesions present in each cystectomy specimen were studied, including
the tumour area and the adjacent mucosa (12).
Rat bladder samples
Fifty-three female Fisher 344 rats were obtained at
the age of 5 weeks from Harlan (Amsterdam, The Netherlands).
Details concerning the animals, diet, carcinogens and experimental
protocol were approved by the Portuguese Ethics Committee for
Animal Experimentation (Direcção Geral de Veterinária, approval no.
520/000/000/2003) and are fully described in our previously
published study (10). Briefly,
animals were randomly separated in five groups. BBN was
administered in drinking water at a concentration of 0.05% during a
period of 20 weeks to all animal groups, with the exception of
group 1, which served as the control and was not given any chemical
supplement. After this BBN exposure, animals were maintained on
normal tap water until the end of the experiment. Groups 2 and 3
were exposed only to BBN, and animals were sacrificed after an
intraperitoneal administration of sodium pentobarbital 1 week
(group 2) and 7 weeks (group 3) after BBN exposure. One week after
BBN exposure, groups 4 and 5 began intravesical instillations with
physiological saline solution (PSS) and mitomycin C (MMC),
respectively, once a week for 6 weeks. After this treatment period,
the animals were sacrificed. The tumours and adjacent mucosa were
studied for each bladder specimen.
For intravesical immunotherapy, the dosing schedules
were based on those commonly used in clinical work; 300 μl of MMC
solution (1 mg/1 ml) were used (10). The preparation procedures of the
bladder for macroscopic examination and tissue processing were
previously described (10).
Histopathological analysis
The diagnostic pathology slides were re-evaluated by
experienced uropathologists (T.A. for the human series, and C.L.
and P.O. for the rat specimens). Pathological staging was performed
according to the American Joint Committee on Cancer Staging
(13). The histological
classification and grading were performed using the criteria from
the 2004 World Health Organization (WHO) guidelines (14,15).
DNA content analysis
The DNA content quantification was performed using
image cytometric analysis and a CAS 200 Image Analysis System (Cell
Analysis Systems, Inc., Elmhurst, IL, USA). All methodological and
DNA histogram analysis procedures were previously described in
detail (6). Briefly, after the
calibration of the image system using a control slide with rat
hepatocytes with a known quantity of DNA, 20–30 lymphocytes and a
minimum of 100 intact non-overlapping urothelial nuclei were
measured and analysed for each case. The G0/G1 peak was visually
identified in each DNA histogram, and the mean, standard deviation
(SD) and coefficient of variation (CV) values were calculated. The
DNA index (DI) describes the relative DNA content of the study
population and was defined as the ratio of mean DNA content of the
urothelial G0/G1 peak divided by the mean DNA content of the
resting diploid lymphocyte G0/G1 peak. The 5cER was also evaluated
and defined as the percentage of cells with values >5n. Lesions
were considered aneuploid only if a separate G0/G1 peak was
distinguishable on the histogram and it differed from the reference
lymphocyte population by >2 SD. A DNA diploid lesion showed a
single distinct G0/G1 peak with a DI within 2 SD of the control
lymphocytes and usually with <1% 5cER.
Immunohistochemical analysis
Human specimens
For the immunohistochemical analysis a standard
avidin-biotin peroxidase method was used, as described in a
previous study (16). The
immunoexpression of p53 and Ki-67 was evaluated with the primary
antibodies D07 (1:50 dilution; Dako®) and MIB1 (1:50
dilution; Novocastra®), respectively.
Paraffin-embedded tissues, known to express nuclear
p53 (colon carcinoma) and have a high proliferation (lymphoma),
were used for titration and positive controls. Negative controls
were performed replacing the primary antibody with 2.5% bovine
serum albumin in PBS.
Rat specimens
A three-step streptavidin-biotin immunoperoxidase
method was used, as described in a previous study (10). The primary antibodies AB-1 (1:50
dilution; Neomarkers-Labvision) for p53 expression and Ki-67 (1:20
dilution; Dako) for proliferative activity, were used. Paraffin
sections from colon and breast cancer with known immunoreactivity
to p53 and Ki-67 antigens, respectively, were used as positive
controls. Negative controls were carried out by replacing the
primary antibodies with PBS.
Slides were re-evaluated by two independent
observers (T.A. and L.S. for the human cases and C.L. and P.O. for
the rat cases) in a blinded fashion, without knowledge of the
clinical data. The evaluation method was also described previously
(16). The entire lesion was
screened to find the region with the maximum fraction of positive-
and contiguous-stained nuclei for p53 and the region with the
maximum fraction of positivity for Ki-67. The percentage of
positive-stained nuclei was scored in this region using a ×40
objective. Whenever possible, at least 100 cells were scored in
each histological lesion. In the human series, the cut-off values
used to distinguish positivity for p53 and Ki-67 were >18 and
>32%, respectively, based on studies reported previously
(16). In the rat samples, the p53
and Ki-67 immunoexpression was calculated as the percentage of
positive nuclei divided by the total number of cells examined
(10).
Statistical analysis
The statistical analysis was carried out using the
SPSS 15.0 statistical package for Windows (SPSS Inc.). A
descriptive analysis was performed for the variables studied. The
incidences of differences in molecular alterations between the
histological groups were evaluated by the Pearson’s Chi-square and
the Fisher’s exact (when n<5) tests. The mean percentage of
positivity of p53 and Ki-67 of the histological types was compared
by analysis of variance (ANOVA). P<0.05 was defined as
statistically significant.
Results
Evaluation of human carcinogenesis
Of the 49 patients included in the present study, 19
cases (38.8%) were classified as high-grade papillary urothelial
carcinoma (HGP) and 30 (61.2%) as invasive urothelial carcinoma
(INV). For all of the tumours the adjacent mucosa was evaluated and
showed differential histological patterns dependent on the adjacent
tumour. Morphological normal urothelium (n=13, 68.5%), hyperplasia
(n=3, 15.8%) and carcinoma in situ (CIS) (n=3, 15.7%)
lesions were found in the HGP adjacent mucosa, while the INV
adjacent mucosa showed morphological normal urothelium (n=14,
46.7%), hyperplasia (n=6, 19.9%), dysplasia (n=2, 6.7%) and CIS
(n=8, 26.7%) lesions.
Each one of these histological patterns found in the
adjacent mucosa showed the same molecular profile, in regards to
the biological variables studied, independently of the adjacent
tumour.
Considering each histological type, we compared the
distribution of the molecular alterations for the biological
variables studied. Statistical significant differences were
observed according to the potential of aggressiveness of the lesion
(Table I).
 | Table IFrequency of molecular alterations for
each histological type observed in the human series. |
Table I
Frequency of molecular alterations for
each histological type observed in the human series.
| Variables | Normal | HYP | DYS | CIS | HGP | INV | p-value |
|---|
| n=27 | n=9 | n=2 | n=11 | n=19 | n=30 | |
|---|
| n (%) | n (%) | n (%) | n (%) | n (%) | n (%) | |
|---|
| DNA ploidy | | | | | | | <0.001 |
| Diploid | 26 (96.3) | 7 (77.8) | 1 (50) | 1 (9.1) | 7 (36.8) | 2 (6.7) | |
| Aneuploid | 1 (3.7) | 2 (22.2) | 1 (50) | 10 (90.9) | 12 (63.2) | 28 (93.3) | |
| 5cER | | | | | | | <0.001 |
| Without | 26 (96.3) | 7 (77.8) | 1 (50) | 1 (9.1) | 9 (47.4) | 2 (6.7) | |
| With | 1 (3.7) | 2 (22.2) | 1 (50) | 10 (90.9) | 10 (52.6) | 28 (93.3) | |
| p53 | | | | | | | 0.001 |
| Negative | 26 (96.3) | 8 (88.9) | 1 (50) | 7 (63.6) | 14 (73.7) | 15 (50.0) | |
| Positive | 1 (3.7) | 1 (11.1) | 1 (50) | 4 (36.4) | 5 (26.3) | 15 (50.0) | |
| Ki-67 | | | | | | | <0.001 |
| Negative | 26 (96.3) | 9 (100) | 2 (100) | 4 (36.4) | 7 (36.8) | 8 (26.7) | |
| Positive | 1 (3.7) | 0 (0) | 0 (0) | 7 (63.6) | 12 (63.2) | 22 (73.3) | |
Evaluation of rat carcinogenesis
The histopathological lesions observed in each
experimental group of rats are described in Table II. No histopathological alterations
in urothelial cells were observed in the control group (group 1).
The frequency of lesions increased with a longer observation period
after BBN exposure. A higher number of HGP and INV cases was
observed in the animal group exposed to BBN and submitted to the
intravesical instillation of physiological saline solution (group
4). CIS lesions were detected only in animals in group 5, exposed
to BBN and treated with MMC.
 | Table IIIncidence of urothelial
histopathological lesions in rats exposed to
N-butyl-N-(4-hydroxybutyl)-nitrosamine and treated with Mitomycin C
and physiological saline solution. |
Table II
Incidence of urothelial
histopathological lesions in rats exposed to
N-butyl-N-(4-hydroxybutyl)-nitrosamine and treated with Mitomycin C
and physiological saline solution.
| Group (n) | Normal | HYP | DYS | CIS | LGP | HGP | INV |
|---|
| n=10 | n=6 | n=41 | n=7 | n=40 | n=30 | n=10 |
|---|
| n (%) | n (%) | n (%) | n (%) | n (%) | n (%) | n (%) |
|---|
| 1 (10) | 10 (100) | 0 (0.0) | 0 (0.0) | 0 (0) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| 2 (12) | 0 (0) | 2 (16.6) | 10 (87.3) | 0 (0) | 10 (83.3) | 2 (16.6) | 2 (16.6) |
| 3 (10) | 0 (0) | 2 (20.0) | 10 (100.0) | 0 (0) | 10 (100.0) | 9 (90.0) | 1 (10.0) |
| 4 (11) | 0 (0) | 1 (9.0) | 11 (100.0) | 0 (0) | 11 (100.0) | 11 (100.0) | 5 (45.4) |
| 5 (10) | 0 (0) | 1 (10.0) | 10 (100.0) | 7 (70) | 9 (90.0) | 8 (80.0) | 2 (20.0) |
The molecular alterations found in each histological
type are presented in Table III.
Despite the period of observation and type of intravesical
instillation, the frequency of DNA aneuploidy, 5c aneuploid cells,
p53 overexpression and abnormal Ki-67 immunoexpression increased
with the degree of aggressiveness of the urothelial lesion.
 | Table IIIFrequency of molecular alterations
for each histological type observed in the rat series. |
Table III
Frequency of molecular alterations
for each histological type observed in the rat series.
| Variables | Normal | HYP | DYS | CIS | LGP | HGP | INV | p-value |
|---|
| n (%) | n (%) | n (%) | n (%) | n (%) | n (%) | n (%) | |
|---|
| DNA ploidy | | | | | | | | <0.001 |
| Diploid | 10 (100.0) | 4 (80.0) | 9 (56.3) | 0 (0.0) | 17 (70.8) | 0 (0.0) | 0 (0.0) | |
| Aneuploid | 0 (0.0) | 1 (20.0) | 7 (43.8) | 7 (100.0) | 7 (29.2) | 23 (100.0) | 3 (100.0) | |
| 5cER | | | | | | | | <0.001 |
| Without | 8 (80.0) | 4 (80.0) | 9 (56.3) | 0 (0.0) | 19 (79.2) | 2 (8.7) | 0 (0.0) | |
| With | 2 (20.0) | 1 (20.0) | 7 (43.8) | 7 (100.0) | 5 (20.8) | 21 (91.3) | 3 (100.0) | |
| p53
(positivity) |
| mean value | 10 (0.0) | 6 (20.7) | 17 (35.5) | 7 (37.4) | 34 (36.0) | 26 (44.0) | 4 (38.9) | <0.001 |
| Ki-67
(positivity) |
| mean value | 10 (0.0) | 6 (16.8) | 17 (23.5) | 7 (29.3) | 34 (23.3) | 26 (34.0) | 4 (23.9) | <0.001 |
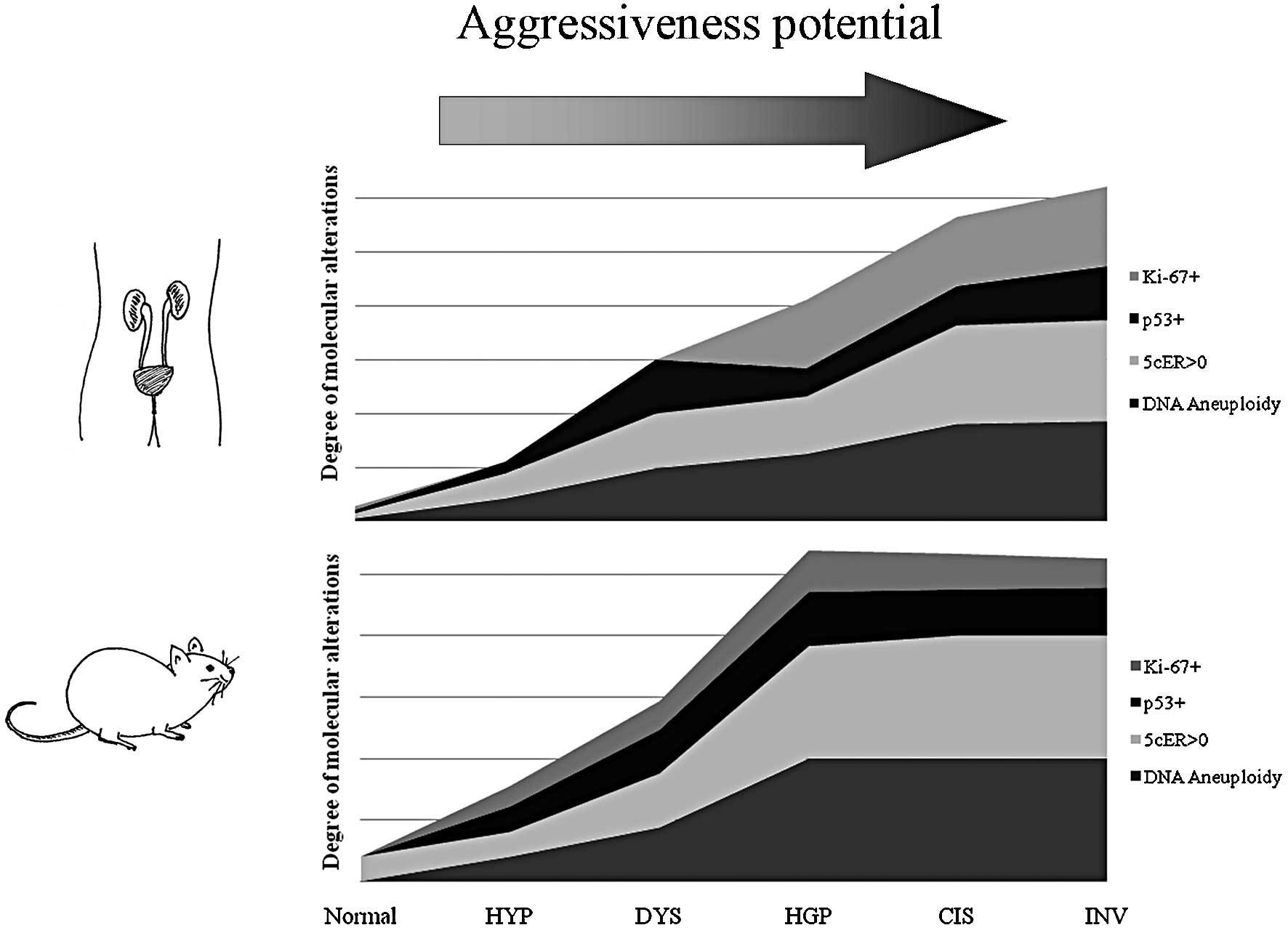
The comparison between the two cancer models, in
regards to the molecular alterations found in each histological
pattern, is illustrated in Fig. 1.
High similarities were observed between the rodent urothelium
carcinogenesis process and the corresponding process in humans, in
regards to histopathological features and the profile of biological
alterations: DNA aneuploidy, p53 overexpression and high
proliferative index measured by Ki-67 immunoexpression. Despite
these similarities, a higher frequency of alterations was observed
in earlier stages in rat chemical-induced carcinogenesis, namely in
5c aneuploid cells, p53 overexpression and higher Ki-67 labelling
index.
Discussion
Cancer involves abnormal cellular growth and appears
to have a common molecular basis. Comparing the genetic signatures
of human and murine bladder carcinogenesis, William and colleagues
(7) found that cell cycle-related
genes are involved in the two processes, supporting our options to
select DNA content, p53 alterations and Ki-67 labelling index as
surrogate markers of bladder carcinogenesis.
The present study found compelling similarities
between the rodent urothelium carcinogenesis process and the
corresponding process in humans, in regards to histopathological
features and the profile of biological alterations: DNA aneuploidy,
p53 overexpression and high proliferative index measured by Ki-67
immunoexpression.
The histopathological alterations observed by our
group in rat chemical-induced bladder carcinogenesis, namely
hyperplasia, dysplasia, low- and high-grade papillary urothelial
cell carcinoma, CIS and INV were also observed in natural human
bladder carcinogenesis (14,15).
The biological history of bladder cancer shows that
DNA aneuploidy is a reflection of genomic instability and
chromosomal derangement, and a high aneuploidy level was observed
in human advanced bladder cancer (17). TP53 genetic alterations are
associated with tumour stage and grade, and there is a significant
association with patient outcome (16,18,19). A
high proliferative index is now accepted as a prognostic factor in
human bladder cancer improving identification of patients who are
at increased risk for disease progression after radical cystectomy
(20).
In the experimental orthotopic model described
herein, we observed the entire spectrum of lesions that is
described in the dual-track pathway of human bladder
carcinogenesis: the papillary and non-papillary pathways (21). We also observed that the variation
in the frequency of genetic alterations was similar between the
rodent (rat) and human cancer models. Thus, the rate of DNA
aneuploidy, p53 immunoexpression and Ki-67 labelling index was
higher in more aggressive lesions. However, a biological profile
similar to that in human invasive tumours was observed in early
tumour stages in rats suggesting that this murine model is a
beneficial model with which to study the invasive carcinogenesis
pathway. William and colleagues also observed that induced murine
tumours exhibited more similarities in gene expression to human
muscle invasive tumours than non-invasive tumours. Thus, rodent
(rat) tumours provide an accurate mechanistic study of genes
putatively involved in invasive and metastatic bladder cancer
(7).
We also observed a high frequency of biological
alterations in high-risk non-muscle invasive tumours, similar to
those that occur in human counterparts. This result indicates that
this model is also suitable for the study of these particular
tumours.
The present and previous results obtained by our
group (10,11) emphasize that we now have a
technically suitable and highly reproducible model of bladder
urothelial carcinogenesis. This model resembles human disease both
histologically and in biological behaviour, particularly for
high-risk non-muscle invasive and invasive urothelial bladder
cancer. Therefore, it is a reliable model with which to identify
new molecular targets and evaluate the feasibility and tumour
response of new cytotoxic drugs or drug combinations in the field
of urologic oncology.
Acknowledgements
This research study was supported by a grant from
Associação Portuguesa de Urologia, 2008. The authors are also
grateful to Eng. D. Sanders, Portuguese Institute of Oncology, for
the English version.
References
|
1
|
Höglund M: Bladder cancer, a two phased
disease? Semin Cancer Biol. 17:225–232. 2007.
|
|
2
|
Crallan RA, Georgopoulos NT and Southgate
J: Experimental models of human bladder carcinogenesis.
Carcinogenesis. 27:374–381. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Oliveira PA, Colaço A, De la Cruz PLF and
Lopes C: Experimental bladder carcinogenesis-rodent models. Exp
Oncol. 28:2–11. 2006.PubMed/NCBI
|
|
4
|
Arentsen HC, Hendricksen K, Oosterwijk E
and Witjes JÁ: Experimental rat bladder urothelial cell carcinoma
models. World J Urol. 27:313–317. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Grubbs CJ, Lubet RA, Koki AT, Leahy KM,
Masferrer JL, Steele VE, Kelloff GF, Hill DL and Seibert K:
Celecoxib inhibits N-butyl-N-(4-hydroxybutyl)-nitrosamine-induced
urinary bladder cancers in male B6D2F1 mice and female Fischer-344
rats. Cancer Res. 60:5599–5602. 2000.PubMed/NCBI
|
|
6
|
Oliveira PA, Palmeira C, Lourenço LM and
Lopes CA: Evaluation of DNA content in preneoplastic changes of
mouse urinary bladder induced by N-butyl-N-(4-hydroxybutyl)
nitrosamine. J Exp Clin Cancer Res. 24:609–616. 2005.PubMed/NCBI
|
|
7
|
Williams PD, Lee JK and Theodorescu D:
Molecular credentialing of rodent bladder carcinogenesis models.
Neoplasia. 10:838–846. 2008.PubMed/NCBI
|
|
8
|
Druckrey H, Preussmann R, Ivankovic S,
Schmidt CH, Mennel HD and Stahl KW: Selective induction of bladder
cancer in rats by dibutyl-and
N-butyl-n-(4-hydroxybutyl)nitrosamine. Z Krebsforsch. 66:280–290.
1964.PubMed/NCBI
|
|
9
|
Kunze E, Schauer A and Schatt S: Stages of
transformation in the development of
N-butyl-N-(4-hydroxybutyl)-nitrosamine-induced transitional cell
carcinomas in the urinary bladder of rats. Z Krebsforch Klin Onkol
Cancer Res Clin Oncol. 87:139–160. 1976.PubMed/NCBI
|
|
10
|
Oliveira PA, Palmeira C, Colaço A, De La
Cruz PLF and Lopes C: DNA content analysis, expression of Ki-67 and
p53 in rat urothelial lesions induced by N-butyl-N-(4-hydroxybutyl)
nitrosamine and treated with mitomycin C and bacillus
Calmette-Guérin. Anticancer Res. 26:2995–3004. 2006.PubMed/NCBI
|
|
11
|
Palmeira C, Oliveira PA, Arantes-Rodrigues
R, Colaço A, De la Cruz PLF, Lopes C and Santos L: DNA cytometry
and kinetics of rat urothelial lesions during chemical
carcinogenesis. Oncol Rep. 21:247–252. 2009.PubMed/NCBI
|
|
12
|
Palmeira C, Lameiras C, Amaro T, Lima L,
Kock A, Lopes C, Oliveira PA and Santos L: CIS is a surrogate
marker of genetic instability and field carcinogenesis in the
urothelial mucosa. Urol Oncol. Oct 23–2009.(Epub ahead of
print).
|
|
13
|
Greene FL, Page DL, Fleming ID, et al:
AJCC Cancer Staging Manual. 6th edition. Springer Verlag; New York:
2002, View Article : Google Scholar
|
|
14
|
Sauter G, Algaba F, Amin M, et al: Tumours
of the urinary system: non-invasive urothelial neoplasias. WHO
Classification of Tumours of the Urinary System and Male Genital
Organs. Eble JN, Sauter G, Epstein JL and Sesterhenn I: IARCC
Press; Lyon: pp. 110–123. 2004
|
|
15
|
Lopez-Beltran A, Sauter G, Gasser T, et
al: Tumours of the urinary system: infiltrating urothelial
carcinoma. WHO Classification of Tumours of the Urinary System and
Male Genital Organs. Eble JN, Sauter G, Epstein JL and Sesterhenn
I: IARCC Press; Lyon: pp. 93–109. 2004
|
|
16
|
Santos L, Amaro T, Costa C, Pereira S,
Bento MJ, Lopes P, Oliveira J, Criado B and Lopes C: Ki-67 index
enhances the prognostic accuracy of the urothelial supeficial
bladder carcinoma risk group classification. Int J Cancer.
105:267–272. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Palmeira CA, Oliveira PA, Seixas F, Pires
MA, Lopes C and Santos L: DNA image cytometry in bladder cancer:
state of the art. Anticancer Res. 28:443–450. 2008.PubMed/NCBI
|
|
18
|
Cordon-Cardo C: Molecular alterations
associated with bladder cancer initiation and progression. Scand J
Urol Nephrol. 42:S154–S165. 2008. View Article : Google Scholar
|
|
19
|
Lacy S, Lopez-Beltran A, MacLennan GT,
Foster SR, Montironi R and Cheng L: Molecular pathogenesis of
urothelial carcinoma – the clinical utility of emerging new
biomarkers and future molecular classification of bladder cancer.
Anal Quant Cytol Histol. 31:5–16. 2009.
|
|
20
|
Margulis V, Lotan Y, Karakiewicz PI, et
al: Multi-institutional validation of the predictive value of Ki-67
labeling index in patients with urinary bladder cancer. J Natl
Cancer Inst. 101:114–119. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Spiess PE and Czerniak B: Dual-track
pathway of bladder carcinogenesis. Practical implications. Arch
Pathol Lab Med. 130:844–852. 2006.PubMed/NCBI
|