Introduction
In 1836, the membranous structure between the rectum
and the seminal vesicles was named Denonvilliers’ fascia after
Charles-Pierre Denonvilliers (1).
Denonvilliers’ fascia has been a surgical landmark that urologists
must consider during intrapelvic surgery (2,3).
Denonvilliers’ fascia consists of a single layer arising from the
fusion of the two walls of the embryological peritoneal cul-de-sac,
under embryological and anatomical review (4). In our institution, a urologic
intrapelvic surgery was performed, utilizing Denonvilliers’ fascia.
However, experienced urologists should not disregard cases where
Denonvilliers’ fascia is identified in surgery, and cases where
Denonvilliers’ fascia is not noted in radical prostatectomy. The
incidence of such differences is frequent. To assess these
differences, we reviewed the intraoperative findings and the
excised specimens obtained during a radical prostatectomy, as well
as fixed specimens obtained during total pelvic exenteration.
Materials and methods
Between April 2008 and March 2009, at our
institution, the clinical anatomy of perioperative regions and
excised specimens were reviewed macroscopically for 62 cases of
antegrade radical prostatectomy. Not all cases had a treatment
history for prostate cancer preoperatively. We observed the
relationship between the deferent duct and the surrounding tissue
after bladder neck transection, macroscopically. We also observed
the detachment between the prostate and rectum, following
deferential ligation and ablation, as well as the seminal vesicle
detachment, macroscopically. In addition, we observed the dorsal
surface of the excised specimen macroscopically. Finally, we
histologically examined the region between the prostate and rectum
using fixed specimens from three cases that underwent total pelvic
exenteration performed during the same time period.
Results
Observed relationship between the
deferent duct, following bladder neck transection, and the
surrounding tissue, macroscopically
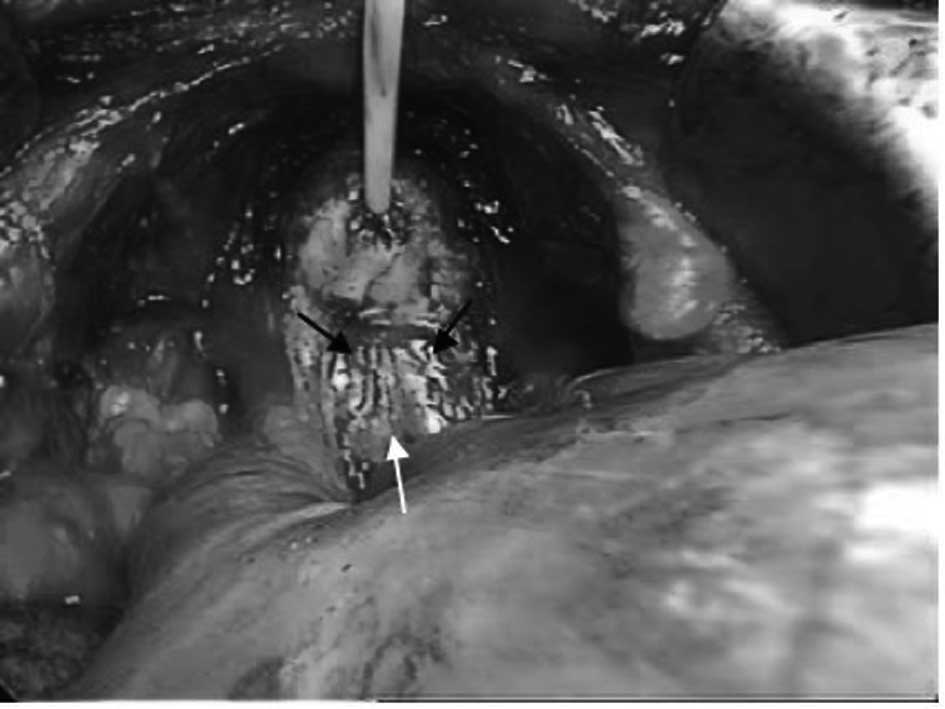
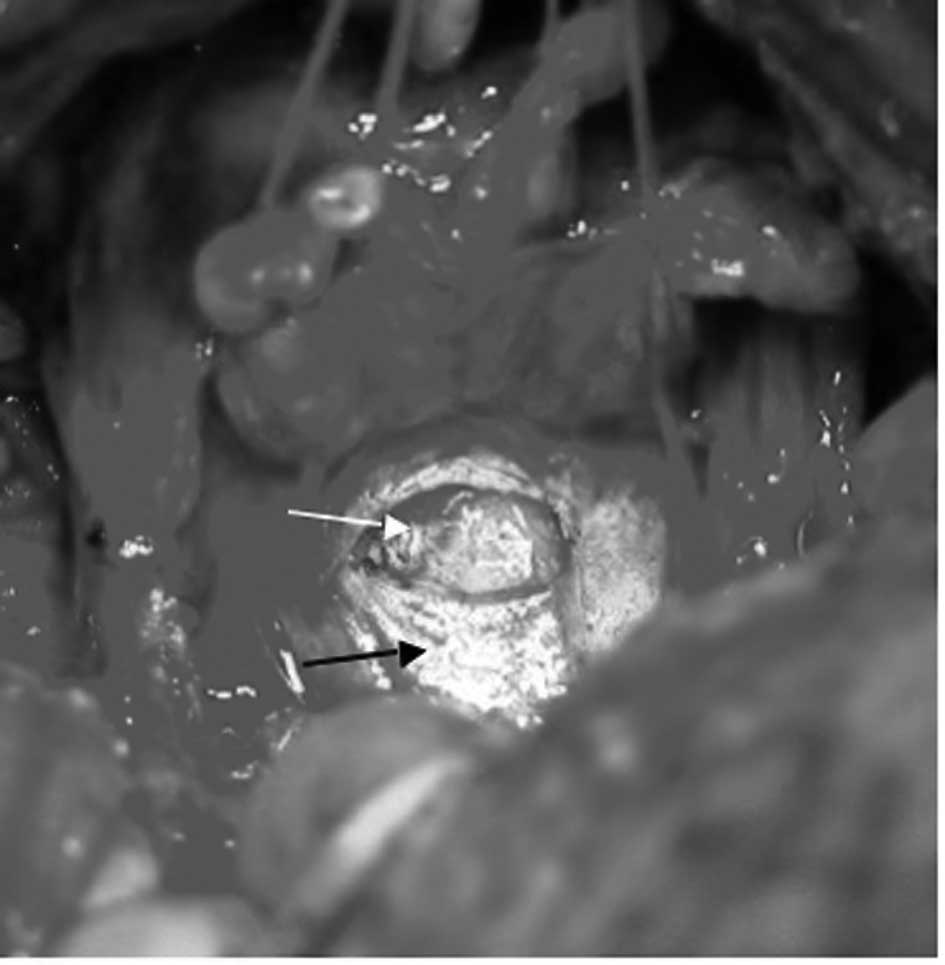
Observations were dorsal in nature after bladder
neck transection. The deferent duct was observed in the connective
tissue (Fig. 1). When the surgeon
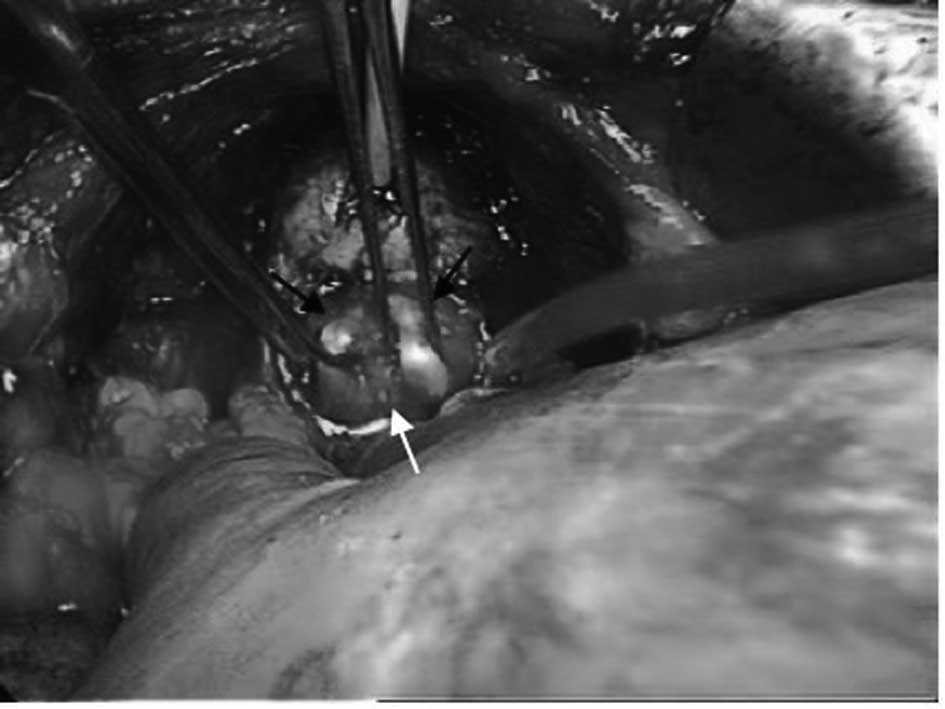
grasped a deferent duct, he was able to confirm that an intricate
connective tissue clung to the surrounding region (Fig. 2). In addition, it was confirmed that
this connective tissue continued to Denonvilliers’ fascia from the
dorsum to the area where the deferent duct was ligated and
separated (Fig. 3).
Observed detachment between the prostate
and rectum, following deferential ligation/ablation and seminal
vesicle detachment macroscopically
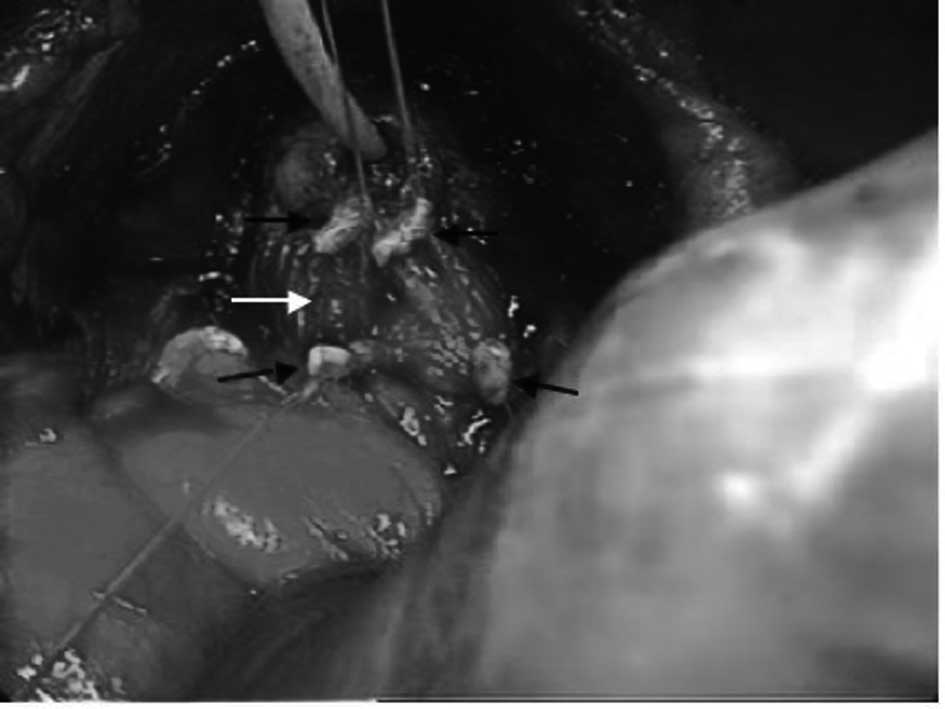
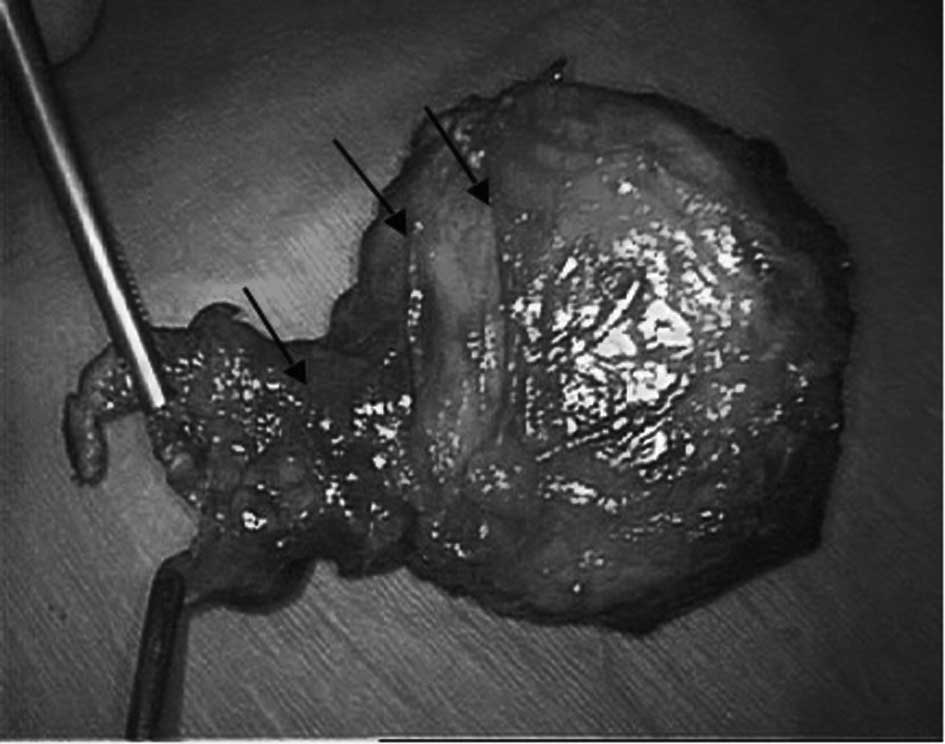
When the prostate was pulled ventrally,
Denonvilliers’ fascia was observed. It extended from the anterior
prostate to the dorsal rectum, and was present in the form of a
sheet of membrane (Fig. 4). This
membrane did not differ from that which continued to Denonvilliers’
fascia (Fig. 3). A membrane was
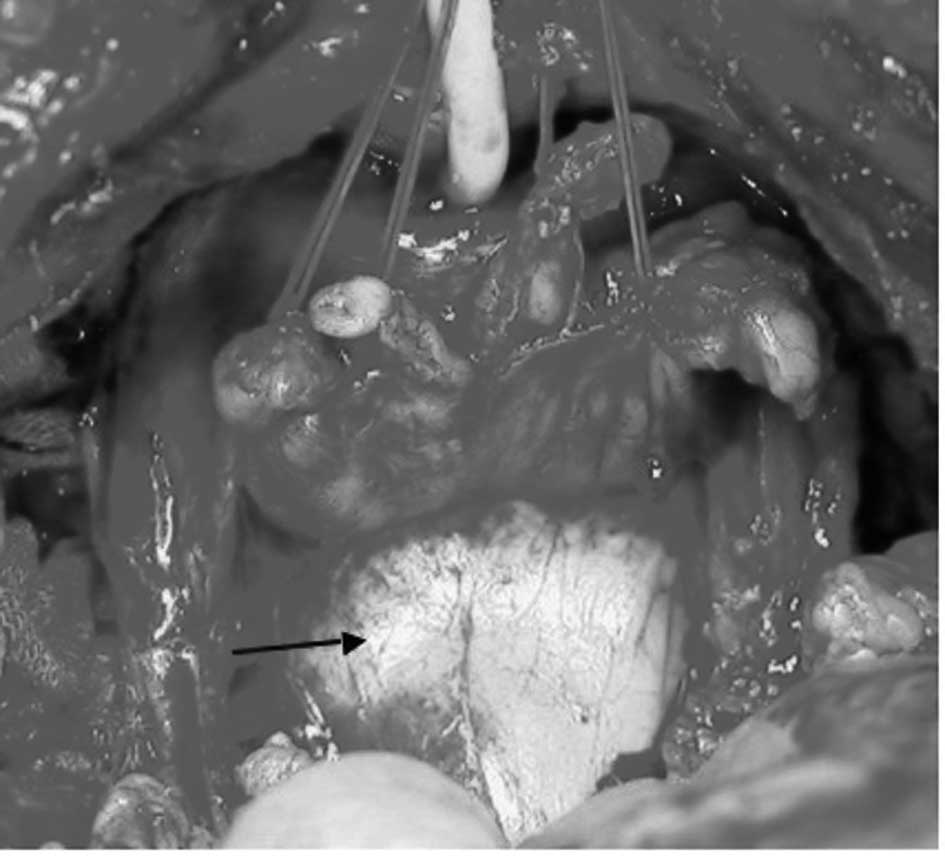
observed when the prostate was pulled more ventrally and the
bladder was pulled more dorsally. The webbed connective tissue was
observed more distally when the membranous structure was sharply
cut open (Fig. 5). In addition, the
webbed connective tissue was identified as a new membranous
structure when tension was applied to the tissue. A series of
repeated operations was possible as stated above.
Dorsal macroscopic observation in the
prostatectomy specimen
The dorsal side of the specimen appeared to be
covered by the thin membrane. Furthermore, several regions that
continued along this thin membrane were noted. These regions
appeared macroscopically as a stump (Fig. 6).
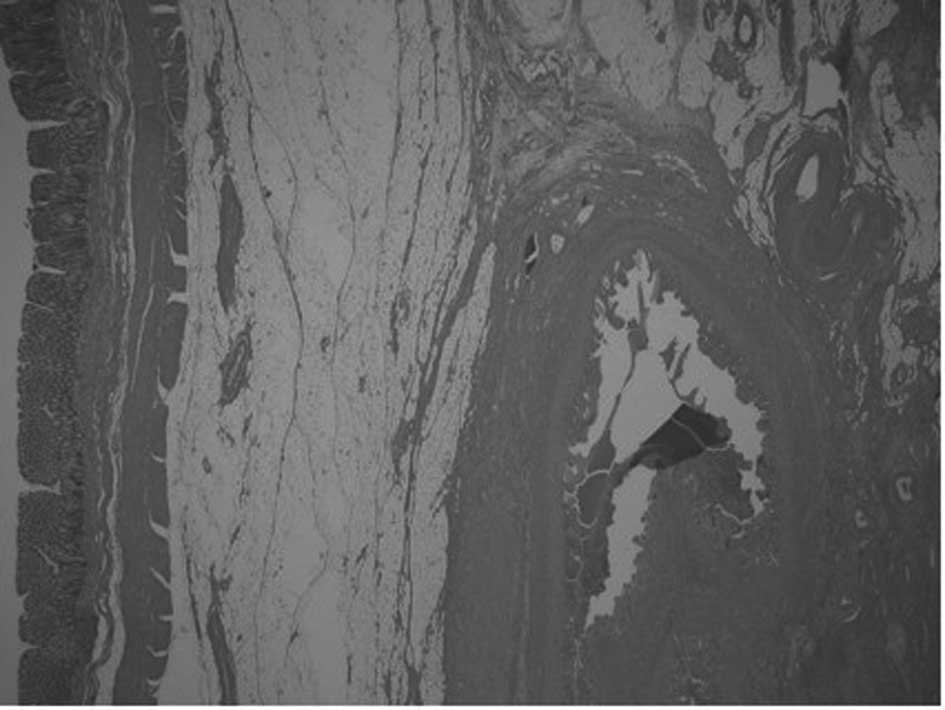
Histological examination of the fixed
specimen removed in the total pelvic exenteration
Intricate connective tissue and fat were present
between the prostate and rectum in the 62 cases. These tissues
continued to the connective tissue around the seminal vesicles and
prostate (Fig. 7).
Discussion
Although Denonvilliers’ fascia is present between
the prostate and rectum anatomically, Denonvilliers’ fascia cannot
be identified in all surgical cases. Urologic surgeons may
accomplish surgery more securely and safely when they can identify
Denonvilliers’ fascia without becoming perplexed by the differences
present in each sugical case.
We firstly observed Denonvilliers’ fascia during a
an antegrade radical prostatectomy, macroscopically. To solve such
contradictions, five findings were focused on. It was noted that i)
a connective tissue clung to the deferent duct following bladder
neck transection and that the connective tissues were separated
from the deferent duct; ii) the connective tissues that clung to
the deferent duct continued to Denonvilliers’ fascia; iii)
Denonvilliers’ fascia was identified as a membranous structure
surgically; iv) the webbed connective tissue was observed more
distally when the membranous structure was sharply cut open and v)
the webbed connective tissue was identified as a membranous
structure again as tension was applied to this webbed connective
tissue.
As a result of these findings, we believe that
Denonvilliers’ fascia may be perceived surgically only as an
intricate connective tissue. In other words, the intricate
connective tissue present in three dimensions gave the appearance
of a membranous structure in two dimensions since tension was
applied to this connective tissue in a definite direction, and this
new membranous structure was recognized merely as a membrane
surgically.
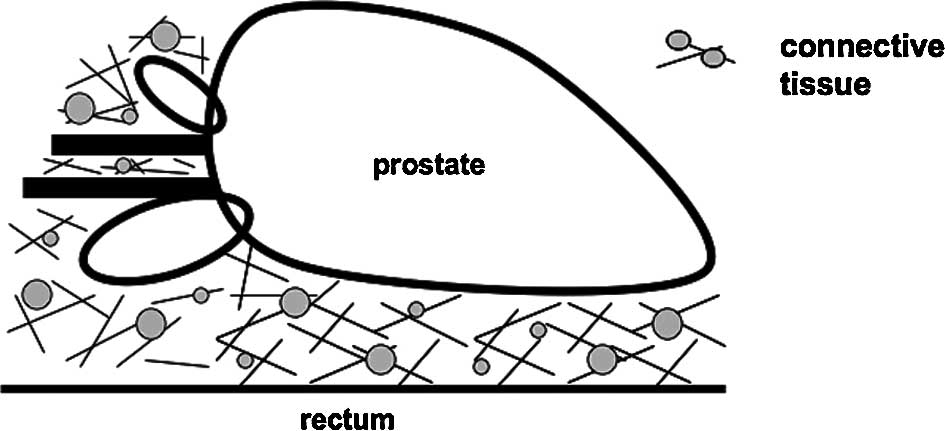
We represented this structure schematically to fully
explain the misleading two-dimensional appearance of this
connective tissue. As a result of these findings, we conclude that
the connective tissue between the prostate and rectum is complex.
Moreover, this connective tissue continues with the connective
tissue around the deferent duct and seminal vesicles (Fig. 8). As the seminal vesicles, deferent
duct and prostate are pulled ventrally to develop an operative
field, tension can be applied to this connective tissue. This
connective tissue changes to a membranous structure and may be
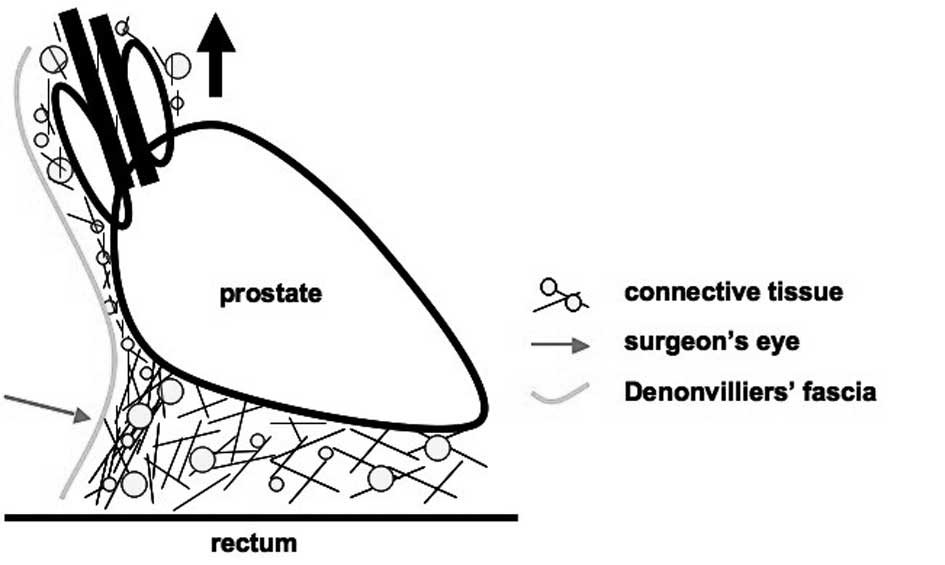
identified as Denonvilliers’ fascia (Fig. 9). While tension was applied to the
tissue, an incision was made to Denonvilliers’ fascia, and this
membranous structure mitigated the tension (arrowheads). In
addition, the prostate was pulled more ventrally and tension was
applied to the connective tissue which was noted more distally.
Consequently, the connective tissue was identified as a new
membranous structure. Thus, the above-mentioned series of
operations may lead to the identification of the membranous
structure, Denonvilliers’ fascia. This identification occurred
while tension was applied to the connective tissue when it was in a
state of no tension due to previous perioperative operations
(Fig. 10).
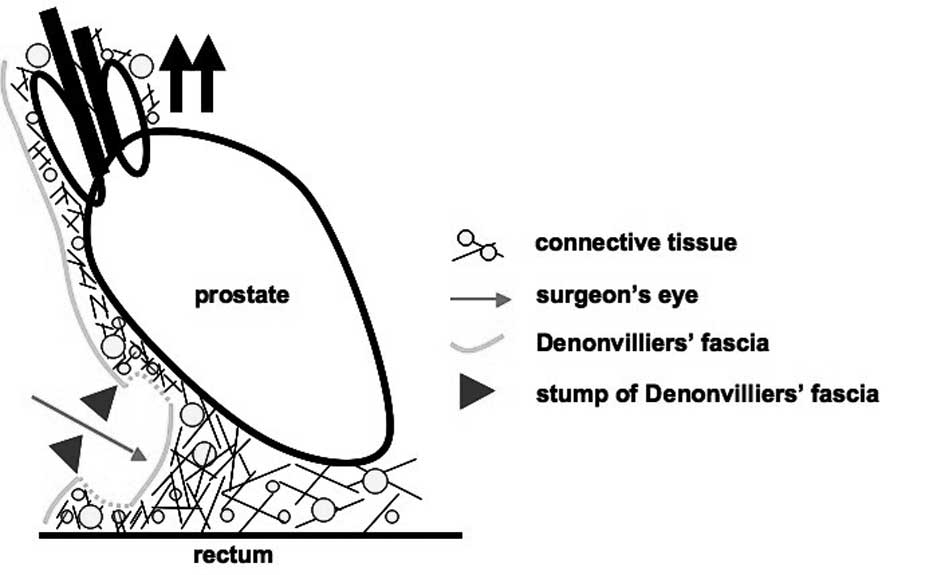
When we observed macroscopically the dorsum of a
specimen from a radical prostatectomy, the dorsal surface of the
prostate appeared to be covered with a thin membrane that we
considered to be Denonvilliers’ fascia. It was noted in several
parts of the area which continued with Denonvilliers’ fascia, but
which were judged macroscopically to be a stump (Fig. 6). This stump which continued along
Denonvilliers’ fascia was different in number. The stump also
varied with respect to the region and the thickness in each
prostatectomy specimen. Denonvilliers’ fascia, which changed shape
from a connective tissue to a membranous structure upon the
application of tension, released tension upon perioperative
operation. Therefore, the issue that arises is whether the stumps
of Denonvilliers’ fascia, which represent a congregation of
connective tissue, is present in various forms in different
specimens.
Finally, we examined three cases of fixed specimens
between the prostate and rectum in the pelvic exenteration. We
noted that the fat and connective tissue were present intricately
at several levels between the prostate and rectum in the 62 cases.
These fixed specimens of the pelvic exenteration cases are thought
to be in an approximate state to the living body (Fig. 7). It was thought that these results
also supported our observations of Denonvilliers’ fascia.
Therefore, the reason for the surgical understanding
of the Denonvilliers’ fascia differing in each case may be that the
quantity of connective tissue identified as Denonvilliers’ fascia
is dissimilar and that its tension varies. It is beneficial that a
surgeon performing urological intrapelvic surgery understand that
Denonvilliers’ fascia is identified surgically as a structure
consisting of connective tissue by a detachment operation. In
addition, the fascia itself consists of a complex dense connective
tissue. Moreover, it can be speculated that retroperitoneal fascia
is similar to Denonvilliers’ fascia in its constitution. We
consistently discussed the membranous structure in the surgical
procedure, but we did not discuss the structure either
embryologically or anatomically.
In conclusion, intricate connective tissue was
anatomically present between the prostate and the rectum. A surgeon
separated and applied tension to this connective tissue and in the
process created a membranous structure. This structure was
surgically identified as Denonvilliers’ fascia.
Acknowledgements
The authors thank Mr. Brian Quinn for the linguistic
comments and help with the manuscript.
References
|
1
|
Denonvilliers CPD: Anatomie du perinee.
Bull Soc Anat. 11:1051836.
|
|
2
|
Young HH: The early diagnosis and radical
cure of carcinoma of the prostate. Being a study of 40 cases and a
presentation of a radical operation, which was carried out in 4
cases. Johns Hopkins Hosp Bull. 16:3151905.
|
|
3
|
Young HH: The cure of cancer of the
prostate by radical perineal prostatectomy (prostato-seminal
vesiculectomy): history, literature and statistics of Young’s
operation. J Urol. 143:11661990.
|
|
4
|
Van Ophoven A and Roth S: The anatomy and
embryological origins of the fascia of Denonvilliers: a
medico-historical debate. J Urol. 157:3–9. 1997.PubMed/NCBI
|