Introduction
Lung cancer is the most common malignancy and the
leading cause of cancer-related deaths worldwide (1). Globally, approximately 228,150 newly
diagnosed non-small cell lung cancer (NSCLC) cases and 147,510
deaths due to NSCLC are reported annually (2). NSCLC, which is associated with high
morbidity and mortality rates, accounts for 80–85% of all lung
cancer cases (3). NSCLC is
classified into three categories, one with actionable oncogene
mutations including epidermal growth factor receptor (EGFR),
Anaplastic lymphoma kinase (ALK) and ROS proto-oncogene 1,
receptor tyrosine kinase (ROS1), one which responds to
immune checkpoint inhibitors, and others (4,5). Many
patients with NSCLC present with local or distant metastasis at the
time of initial diagnosis, and this is attributed to atypical
clinical manifestations and lack of specific diagnostic techniques
in the early stage of the disease (6). At present, surgical resection and
chemoradiotherapy are applied as first-line therapeutics for NSCLC
(7). Despite considerable
developments in the diagnosis and treatment of NSCLC in the last
decade, including surgical excision, radiotherapy, chemotherapy and
targeted therapy, no noticeable improvements have been made in
patient outcomes, with the 5-year survival rate being only 15%
(8). Tremendous efforts have been
made to elucidate the mechanisms associated with NSCLC pathogenesis
(9,10). Hence, the identification of novel
and promising therapeutic targets is urgent and helpful for the
promotion of NSCLC treatment.
Recently, long noncoding RNAs (lncRNAs) have
received considerable attention in the field of cancer research
(11). These lncRNAs are defined as
a family of transcripts with a characteristic length exceeding 200
nucleotides and the absence of an open reading frame (12). Although these lncRNAs do not encode
proteins, an increasing number of studies have illustrated their
crucial roles as regulators of nearly all cellular processes as
well as of carcinogenesis and cancer progression (13,14).
In particular, reports have described the dysregulation of many
lncRNAs in NSCLC. For example, LINC00514 (15), PVT1 (16), and MIR503HG expression are
upregulated in NSCLC, whereas LINC00261 (17), TOB1-AS1 (18), and NBR2 (19) are expressed at low levels. lncRNAs
may act as tumor suppressors or promoters of the oncogenicity of
NSCLC, and they have been identified in the regulation of multiple
aggressive phenotypes (20,21).
MicroRNAs (miRNAs) are a group of endogenous, highly
conserved, and noncoding RNA molecules comprising 17–24
nucleotides. miRNAs have been implicated in the negative modulation
of gene expression via the inhibition of messenger RNA (mRNA)
translation and/or induction of mRNA degradation through direct
binding to the complementary sites in 3′-untranslated regions of
their target genes (22). lncRNAs
may function as competing endogenous RNAs (ceRNAs) to control gene
expression by competitively binding to miRNA response elements
(23). Therefore, lncRNAs and
miRNAs may exert increasingly crucial functions in terms of the
early diagnosis and molecular-targeted therapy of NSCLC.
Although OSER1 antisense RNA 1 (OSER1-AS1) has been
well studied in hepatocellular carcinoma (24), its expression status, specific
functions, and tumorigenic mechanism in NSCLC remain unknown.
Accordingly, we aimed to assess OSER1-AS1 expression in NSCLC, test
the biological functions of OSER1-AS1 associated with NSCLC, and
illustrate how they affect NSCLC progression.
Materials and methods
Human tissue samples
This study was approved by the Ethics Committee of
Weifang People's Hospital and was performed in accordance with the
principles of the Declaration of Helsinki. In total, 53 patients
with NSCLC (22 male and 31 female patients; age range, 59–73 years)
admitted to Weifang People's Hospital participated in this study
between March 2014 to January 2015 and provided written informed
consent. None of the enrolled patients had previously received
preoperative radiotherapy, chemotherapy, targeted therapy, or any
other anticancer treatments. The standard care of patients with
NSCLC is chemoradiotherapy for stage III and chemotherapy for stage
IV. Previous studies have reported that cytoreductive surgery can
improve the quality of life of patients with advanced or distant
metastasis of lung cancer (25,26);
hence, the 29 NSCLC patients diagnosed with stage III–IV admitted
in our study received cytoreductive surgery. NSCLC tissues and
adjacent normal tissues were obtained after surgical resection and
immediately frozen and conserved in liquid nitrogen.
Cell culture
The human nontumorigenic bronchial epithelial cell
line BEAS-2B was purchased from the Shanghai Academy of Life
Science (Shanghai, China) and cultured in the BEGM™ Bronchial
Epithelial Cell Growth Medium (Lonza/Clonetics Corporation)
supplemented with 0.5 ng/ml epidermal growth factor, 500 ng/ml
hydrocortisone, 0.035 ng/ml bovine pituitary extract, 500 mM
ethanolamine, 500 nM ethanolamine phosphate, 0.01 mg/ml adrenaline,
and 0.1 g/ml retionic acid.
Five human NSCLC cell lines, namely, H522 (human
lung adenocarcinoma cell carcinoma), H460 (human lung large cell
carcinoma), H1703 (human lung squamous cell carcinoma), A549
(NSCLC), and SK-MES-1 (human lung squamous cell carcinoma), were
also obtained from the Shanghai Academy of Life Science. The cell
lines H522, H460, H1703, and A549 were maintained in RPMI-1640
media (Gibco/Thermo Fisher Scientific, Inc.) containing 10% fetal
bovine serum (FBS; Gibco/Thermo Fisher Scientific, Inc.) and 1%
penicillin/streptomycin (Gibco/Thermo Fisher Scientific, Inc.).
SK-MES-1 cells were cultured in Minimal Essential Medium
(Gibco/Thermo Fisher Scientific, Inc.) supplemented with 10% FBS
and 1% penicillin/streptomycin. All cells were cultured at 37°C in
an incubator supplied with 5% CO2.
Transfection
The small interfering RNAs (siRNAs) used to silence
OSER1-AS1 (si-OSER1-AS1#1, si-OSER1-AS1#2 and si-OSER1-AS1#3) and
negative control (NC) siRNA (si-NC) were synthesized by Genepharma.
miR-433-3p mimic, NC miRNA mimic (miR-NC), miR-433-3p inhibitor
(anti-miR-433-3p), and NC miRNA inhibitor (anti-miR-NC) were
obtained from RiboBio. The pcDNA3.1 empty vector and pcDNA3.1-Smad2
overexpression vector (pc-Smad2) were designed and produced by
GeneChem. Transient transfection was conducted using the
Lipofectamine 2000™ reagent (Invitrogen/Thermo Fisher Scientific,
Inc.).
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
Total RNA was extracted from tissue samples or cells
using the RNeasy Mini Kit (Qiagen). The purity and concentration of
total RNA were analyzed using the DeNovix DS-11 Spectrophotometer
(DeNovix, Inc.). Reverse transcription of OSER1-AS1 and
Smad2 mRNA was performed using the PrimeScript RT Reagent
Kit (Takara). The synthesized cDNA was subjected to quantitative
polymerase chain reaction (qPCR) using the SYBR Premix Ex Taq
(Takara). To quantify miR-433-3p expression, total RNA was reverse
transcribed into cDNA using the miRcute miRNA First-Strand cDNA
Synthesis Kit (Tiangen Biotech). Next, qPCR was conducted using the
miRcute miRNA qPCR Detection Kit with SYBR Green (Tiangen Biotech).
Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as
the internal control for the normalization of OSER1-AS1 and
Smad2 mRNA expression, whereas U6 small nuclear RNA was used
for the normalization of miR-433-3p expression. Relative gene
expression levels were analyzed using the 2−ΔΔCq method
(27).
Nuclear and cytoplasmic RNA
fractionation
The PARIS Kit (Invitrogen/Thermo Fisher Scientific,
Inc.) was used for nuclear and cytoplasmic RNA fractionation. The
contents of OSER1-AS1 in nuclear and cytoplasmic fractions were
quantified using RT-qPCR. GAPDH and U6 were used as internal
controls to assess fractioning efficiency.
Cell Counting Kit-8 (CCK-8) assay
Transfected cells that had been incubated at 37°C in
an incubator supplied with 5% CO2 for 24 h were
harvested, resuspended in a culture medium, and seeded into 96-well
plates at a density of 2×103 cells/well. Four
post-inoculation time points were set: 0, 24, 48, and 72 h. At each
time point, cell proliferation was assessed by incubating cells
with 10 µl of CCK-8 solution (Sigma-Aldrich; Merck KGaA) at 37°C
for 2 h, after which the optical density was measured at 450-nm
wavelength using a Tecan microplate reader (Tecan Group, Ltd.).
Flow cytometry
An Annexin V-fluorescein isothiocyanate (FITC)
apoptosis detection kit (BioLegend, Inc.) was used to evaluate cell
apoptosis. After 48 h of culture, transfected cells were digested
with an ethylenediaminetetraacetic acid-free trypsin reagent and
centrifuged at 12,000 × g, followed by two washes with ice-cold
phosphate-buffered solution and resuspension in 100 µl of 1X
binding buffer. Next, the cells were stained with 5 µl Annexin
V-FITC and 5 µl propidium iodide (PI) for 15 min at room
temperature in the dark. Then, the cells were analyzed using a flow
cytometer (FACScan; BD Biosciences) equipped with the CellQuest
software (version 2.9; BD Biosciences) to determine the frequency
of cell apoptosis.
Cell migration and invasion
assays
Transfected cells were trypsinized, washed,
centrifuged, and collected. The cells were mixed with FBS-free
culture medium to yield a cell suspension at a density of
1×105 cells/ml. Regarding cell migration assay, 100 µl
of the suspension was added to each upper chamber of wells equipped
with 8-µm porous membranes (BD Biosciences), whereas 600 µl of
complete culture medium was added to each lower chamber to induce
migration. After 24 h of incubation at 37°C in an incubator
supplied with 5% CO2, the cells that had not migrated to
the lower chamber were removed with a cotton swab, whereas the
migrated cells were fixed for 20 min with 4% (v/v) paraformaldehyde
and stained for 20 min with 0.1% crystal violet. The number of
migrated cells was counted in five randomly selected fields under
an inverted microscope (magnification, ×100; Olympus Corp.).
Regarding the cell invasion assay, chambers were precoated with
Matrigel (BD Biosciences). All subsequent steps were performed as
described for the cell migration assay.
Tumor xenograft assay
The short hairpin RNAs (shRNAs) targeting OSER1-AS1
(sh-OSER1-AS1) and NC shRNA (sh-NC) were inserted into the pLKO.1
vector. After lentivirus production, H522 cells were injected with
lentivirus expressing sh-OSER1-AS1 or sh-NC. Puromycin selection
was then performed to obtain stably transfected cells.
The protocol for animal experiments was ratified by
the Animal Ethics Committee of the Weifang People's Hospital.
Regarding tumor xenograft assay, a total of 6 4- to 6-week-old male
BALB/c nude mice (20 g; Vital River Laboratory Animal Technology,
Beijing, China) were randomly divided into two groups: sh-OSER1-AS1
(n=3) and sh-NC (n=3). Mice in the sh-OSER1-AS1 and sh-NC groups
were subcutaneously inoculated with 5×106 H522 cells
stably expressing sh-OSER1-AS1 and sh-NC, respectively. All mice
were housed under specific pathogen-free conditions at 25°C with
50% humidity, with a 10/14-h light/dark cycle and ad libitum
food/water access. The width and length of each tumor xenograft
were measured weekly beginning at postinjection 7 days, and tumor
volume (V) was calculated using the following formula: V=(length ×
width2)/2. At postinjection 5 weeks, mice were
euthanized via cervical dislocation. All tumor xenografts were
dissected, photographed, and weighed using an analytical balance.
Subsequently, total RNA and protein were isolated from the tumor
xenografts for RT-qPCR and western blotting, respectively,
according to the aforementioned protocols.
lncRNA-miRNA interactions
Putative miRNAs that directly interact with
OSER1-AS1 were predicted using the public database StarBase 3.0
online tool (http://starbase.sysu.edu.cn/) (28).
RNA immunoprecipitation (RIP)
assay
RIP was performed using the Magna RIP RNA-binding
protein immunoprecipitation kit (Millipore) in accordance with the
product instructions. Briefly, cells were incubated with RIP lysis
buffer supplemented with a protease inhibitor cocktail and an RNase
inhibitor. Then, the cell extract was incubated with magnetic beads
conjugated with anti-Argonaute (anti-Ago2) or anti-IgG antibodies
(dilution 1:5,000; both from cat. no. 03-110; Millipore). IgG was
used as NC. After overnight incubation at 4°C, coprecipitated RNA
was extracted and subjected to RT-qPCR to determine the enrichment
of OSER1-AS1 and miR-433-3p.
Luciferase reporter assay
The OSER1-AS1 fragments carrying predicted wild-type
miR-433-3p-binding sequences or mutant OSER1-AS1 fragments were
amplified and inserted into the psiCHECK-2 plasmid (Promega Corp.).
The resulting luciferase reporter plasmids were labeled as
OSER1-AS1-wt and OSER1-AS1-mut, respectively. Cells were seeded in
24-well plates at a density of 1.5×105 cells per well
and transfected with OSER1-AS1-wt or OSER1-AS1-mut in combination
with miR-433-3p mimic or miR-NC using the Lipofectamine 2000™
reagent. After 48 h of incubation at 37°C in an incubator supplied
with 5% CO2, the transfected cells were rinsed with
phosphate-buffered saline. Luciferase activity was measured using
the Dual-Luciferase Reporter Assay System (Promega Corp.).
Renilla luciferase activity was used to normalize the level
of firefly luciferase activity.
Western blotting
Concentrations of proteins in cells lysed in RIPA
buffer (Beyotime Institute of Biotechnology) were determined using
a BCA protein kit (Beyotime Institute of Biotechnology). Equal
amounts of protein (30 µg) were loaded into each lane of a 10%
sodium dodecyl sulfate polyacrylamide gel, electrophoresed, and
transferred onto polyvinylidene difluoride membranes. The membranes
were blocked for 2 h in 5% fat-free milk at room temperature and
incubated with primary antibodies against Smad2 (cat. no. ab40855;
1:1,000 dilution; Abcam) or GAPDH (cat. no. ab181602; 1:1,000
dilution; Abcam) overnight at 4°C. Subsequently, the membranes were
incubated for 2 h with a horseradish peroxidase-conjugated
immunoglobulin G secondary antibody (cat. no. ab205718; 1:5,000
dilution; Abcam) at room temperature. Enhanced chemiluminescence
western blotting detection reagent (GE Healthcare Life Sciences,
Little Chalfont, UK) was applied to the membranes for visualization
of the protein signals. The densitometry of protein signals was
analyzed with Quantity One software version 4.62 (Bio-Rad
Laboratories, Inc.).
Statistical analysis
All statistical analyses were performed using the
SPSS 18.0 statistics software (SPSS, Inc.). All data are presented
as mean ± standard deviation of three independent replicates.
Correlations between the OSER1-AS1 level and clinicopathological
factors in patients with NSCLC were examined using the
χ2 test. Differences among ≥3 groups were evaluated
using one-way analysis of variance followed by Tukey's test.
Comparisons of data between two groups were performed using
Student's t-test. Correlations between the expression levels of
OSER1-AS1 and miR-433-3p in the NSCLC tissues were assessed using
Spearman's correlation analysis. Overall survival curves were
plotted using the Kaplan-Meier method and compared using the
log-rank test. A P-value of <0.05 was considered to indicate
statistical significance.
Results
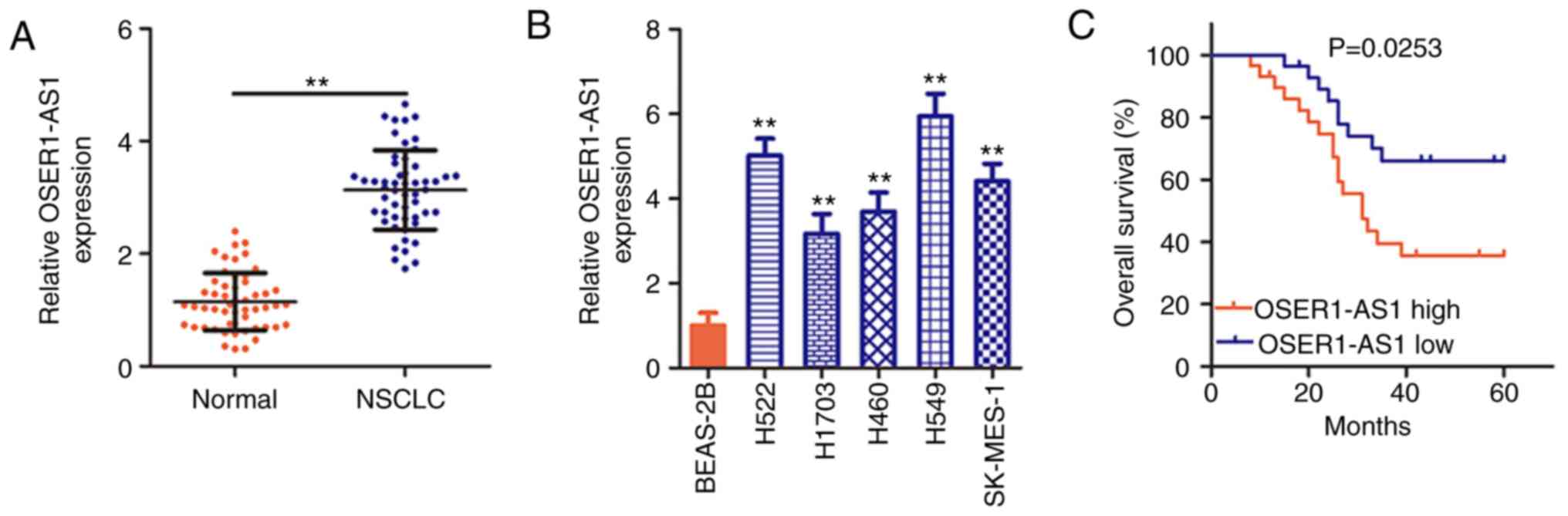
OSER1-AS1 is induced in NSCLC tissues
and cell lines
To determine the OSER1-AS1 expression status in
NSCLC, its expression was evaluated in 53 pairs of NSCLC tissues
and adjacent normal tissues using RT-PCR. OSER1-AS1 expression was
significantly upregulated in the NSCLC tissues compared with that
in the adjacent normal tissues (Fig.
1A). OSER1-AS1 expression was also evaluated in various NSCLC
cell lines using RT-qPCR, with BEAS-2B cells used as the control.
Notably, OSER1-AS1 expression was significantly higher in all five
NSCLC cell lines than in the BEAS-2B cells (Fig. 1B). In summary, OSER1-AS1 expression
is upregulated in both NSCLC tissues and cell lines.
Clinical value of OSER1-AS1 in
patients with NSCLC
Based on the median OSER1-AS1 expression value in
NSCLC tissues, all 53 NSCLC tissues collected from the enrolled
patients were classified into either a low (n=26) or high (n=27)
OSER1-AS1 expression group. χ2 test revealed obvious
correlations between OSER1-AS1 expression and tumor size (P=0.024),
TNM stage (P=0.028), and lymph node metastasis (P=0.045) in the 53
NSCLC tissues (Table I).
Additionally, Kaplan-Meier analysis verified that the
high-OSER1-AS1 expression group had a shorter overall survival than
the low-OSER1-AS1 expression group (Fig. 1C, P=0.0253). The abovementioned data
imply that OSER1-AS1 plays important roles during NSCLC
tumorigenesis and progression.
 | Table I.Association between the
clinicopathological factors of the NSCLC patients and OSER1-AS1
expression. |
Table I.
Association between the
clinicopathological factors of the NSCLC patients and OSER1-AS1
expression.
|
| OSER1-AS1
expression (n=53) |
|
|---|
|
|
|
|
|---|
| Clinicopathological
factors | High (n=27) | Low (n=26) | P-value |
|---|
| Sex |
|
| 0.782 |
|
Male | 12 | 10 |
|
|
Female | 15 | 16 |
|
| Age (years) |
|
| 0.264 |
|
<65 | 8 | 12 |
|
|
≥65 | 19 | 14 |
|
| Classification |
|
| 0.553 |
|
Adenocarcinoma | 11 | 9 |
|
|
Squamous | 12 | 15 |
|
| Large
cell | 4 | 2 |
|
|
Differentiation |
|
| 0.782 |
| Well +
Moderate | 10 | 11 |
|
|
Poor | 17 | 15 |
|
| Tumor size
(cm) |
|
| 0.024 |
|
<3 | 12 | 20 |
|
| ≥3 | 15 | 6 |
|
| TNM stage |
|
| 0.028 |
|
I–II | 8 | 16 |
|
|
III–IV | 19 | 10 |
|
| Lymph node
metastasis |
|
| 0.045 |
|
Negative | 13 | 20 |
|
|
Positive | 14 | 6 |
|
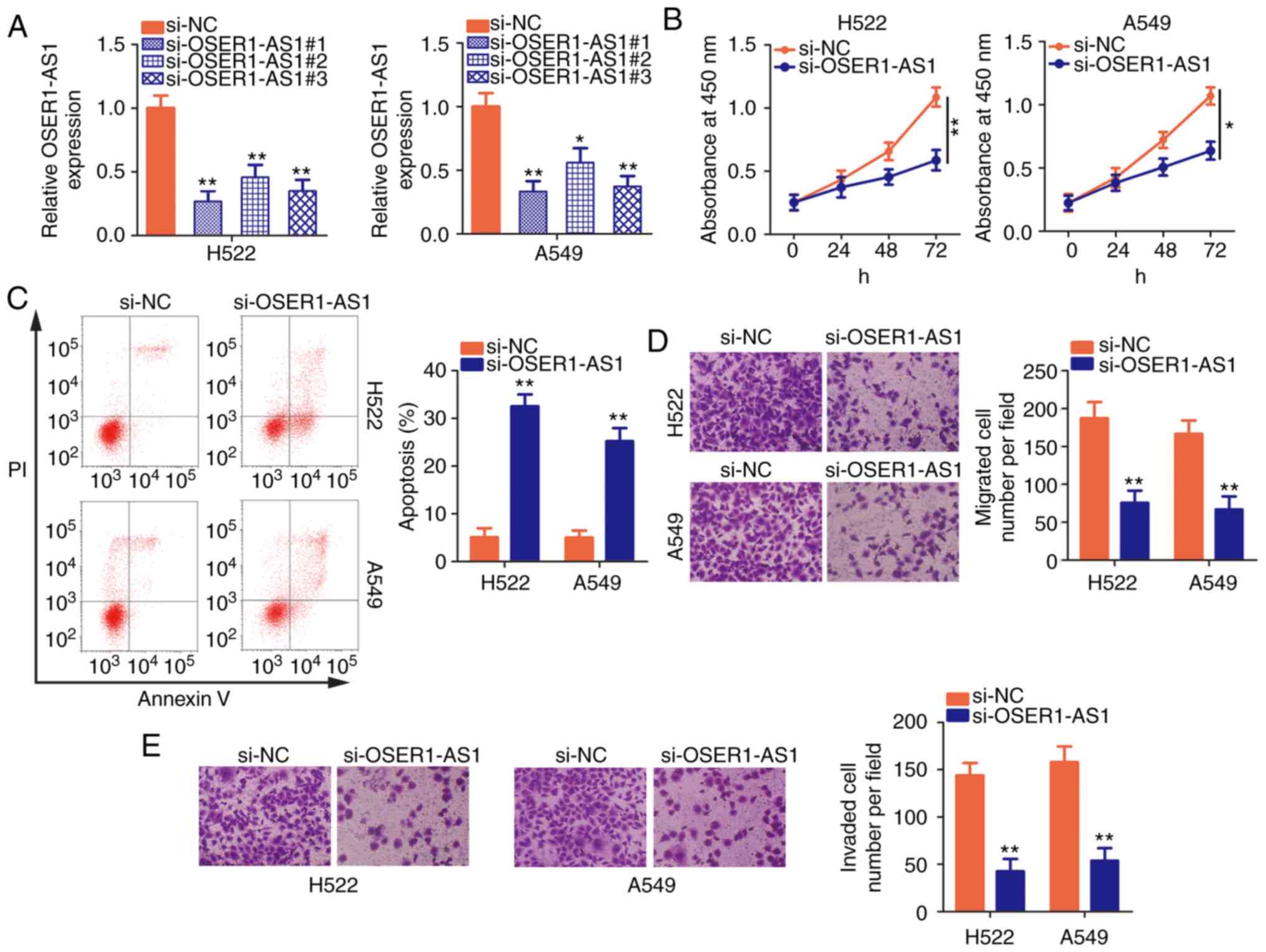
Silencing of OSER1-AS1 restrains NSCLC
cell proliferation, migration, and invasion and promotes cell
apoptosis
H522 and A549 cells exhibited the highest OSER1-AS1
expression among the five NSCLC cell lines; therefore, they were
selected for further functional studies of the roles of OSER1-AS1
in NSCLC. Three siRNAs were designed to disturb OSER1-AS1
expression in H522 and A549 cells, and interference efficiency was
assessed using RT-qPCR. Notably, si-OSER1-AS1#1 (renamed as
si-OSER-AS1) most efficiently silenced OSER1-AS1 expression in H522
and A549 cells (Fig. 2A) and was
chosen for endogenous knockdown experiments. Using the CCK-8 assay,
we demonstrated that OSER1-AS1 knockdown significantly decreased
the proliferative abilities of H522 and A549 cells (Fig. 2B). In addition, si-OSER1-AS1
transfection significantly increased the apoptosis rate in both
cell lines (Fig. 2C), as evidenced
by flow cytometry. Furthermore, the migratory and invasion
abilities of these cells were assessed by cell migration and
invasion assays, respectively. The numbers of migrated (Fig. 2D) and invaded (Fig. 2E) H522 and A549 cells were
significantly reduced after si-OSER1-AS1 transfection, thus
confirming that OSER1-AS1 silencing suppressed the migratory and
invasion abilities of these cells. Taken together, these data
indicate that OSER1-AS1 exerts cancer-promoting actions in NSCLC
progression.
OSER1-AS1 functions as a molecular
sponge for miR-433-3p in NSCLC cells
The specific roles of lncRNAs are determined by
their localization (29). To
illustrate the mechanism by which OSER1-AS1 regulates the
biological behaviors of NSCLC, we first analyzed the distribution
of expression of this lncRNA in NSCLC cells. Nuclear and
cytoplasmic RNA fractionation indicated that OSER1-AS1 was mostly
localized in the cytoplasm of H522 and A549 cells (Fig. 3A), suggesting that this lncRNA
functions as a ceRNA or molecular sponge for certain miRNAs.
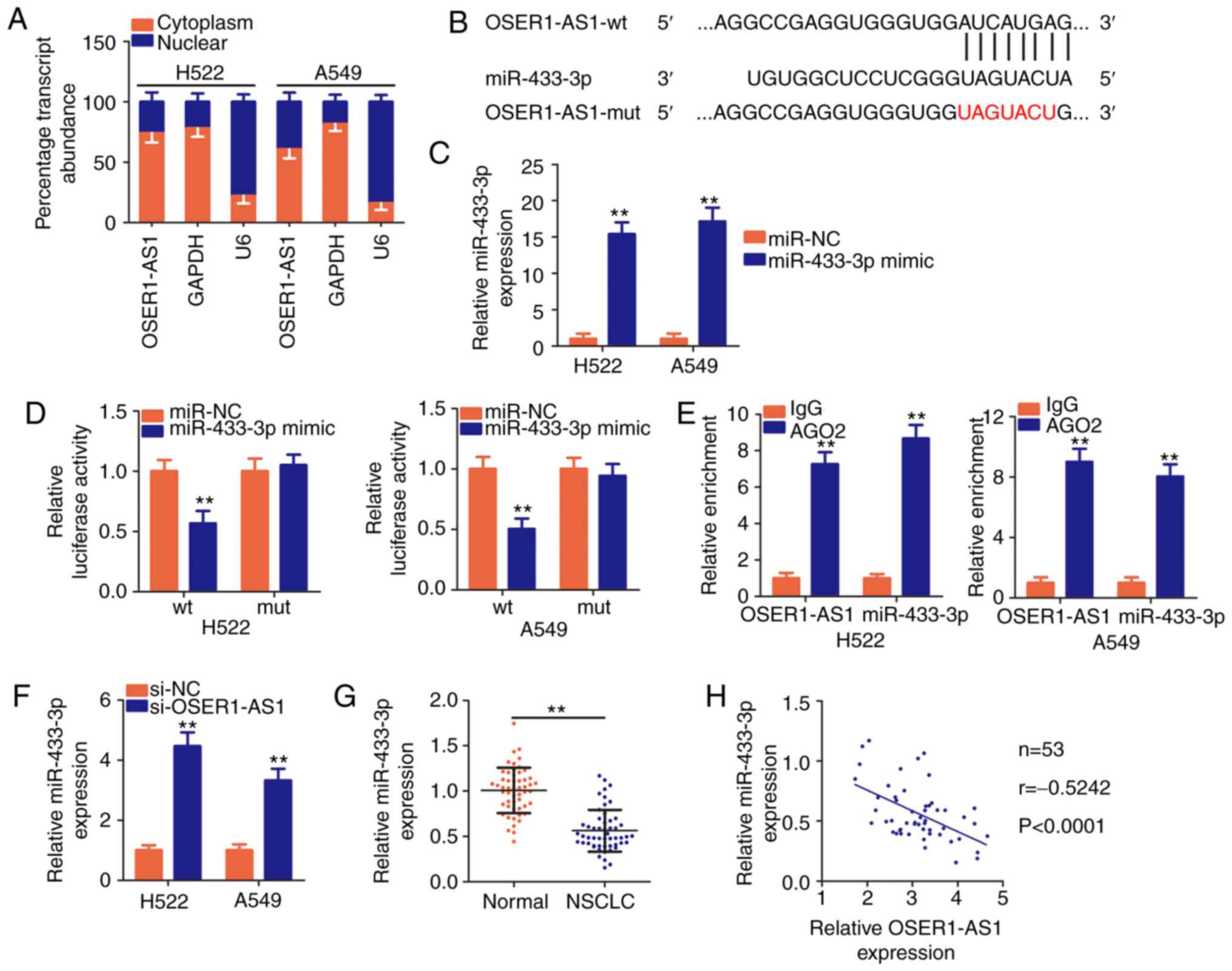 | Figure 3.The long noncoding RNA OSER1-AS1 acts
as a molecular sponge for miR-433-3p in NSCLC cells. (A) Nuclear
and cytoplasmic RNA fractionation to determine the localization of
OSER1-AS1 in H522 and A549 cells. (B) A graphical representation of
wild-type (wt) and mutant (mut) miR-433-3p-binding sites on
OSER1-AS1. (C) Determination of miR-433-3p expression in H522 and
A549 cells transfected with miR-433-3p mimic or miR-NC.
**P<0.01, compared with the miR-NC group. (D) Detection of the
binding interaction between miR-433-3p and OSER1-AS1 in H522 and
A549 cells by luciferase reporter assay. H522 and A549 cells were
cotransfected with miR-433-3p mimic or miR-NC and OSER1-AS1-wt or
OSER1-AS1-mut plasmids. Transfected cells were harvested after 48 h
and subjected to luciferase activity assay. **P<0.01, compared
with the miR-NC group. (E) Evaluation of the enrichment of
miR-433-3p and OSER1-AS1 expression in H522 and A549 cells by RNA
immunoprecipitation assay with Ago2 or IgG antibodies and
subsequent RT-qPCR. **P<0.01, compared with the IgG group. (F)
Quantification of miR-433-3p in OSER1-AS1-depleted H522 and A549
cells using RT-qPCR. **P<0.01, compared with the si-NC group.
(G) Detection of miR-433-3p expression in 53 pairs of NSCLC tissues
and adjacent normal tissues using RT-qPCR. **P<0.01, compared
with adjacent normal tissues. (H) Spearman's correlation analysis
of the correlation between OSER1-AS1 and miR-433-3p expression in
53 NSCLC tissues (r=−0.5242, P<0.0001). OSER1-AS1, RNA OSER1
antisense RNA 1; miR, microRNA; NSCLC, non-small cell lung cancer;
RT-qPCR, reverse transcription-quantitative polymerase chain
reaction. |
According to Starbase3.0, OSER1-AS1 harbors base
pairs complementary to miR-433-3p (Fig.
3B), and the latter miRNA was chosen for subsequent
experimental identification because of its role as a cancer
inhibitor during NSCLC progression (30). Luciferase reporter and RIP assays
were performed to validate the true interaction between OSER1-AS1
and miR-433-3p in NSCLC cells. First, the efficiency of the
miR-433-3p mimic was confirmed through RT-qPCR, and the results
indicated that transfection with the mimic significantly increased
miR-433-3p expression in H522 and A549 cells (Fig. 3C). Luciferase reporter assay
revealed an evident decrease in the luciferase activity of
OSER1-AS1-wt in H522 and A549 cells by upregulation of miR-433-3p
expression compared with that in the miR-NC group, whereas no
significant change was observed in OSER1-AS1-mut reporter plasmid
activity after miR-433-3p overexpression (Fig. 3D). In addition, RIP assay
demonstrated notable abundance of OSER1-AS1 and miR-433-3p in
Ago2-containing beads compared with that in the IgG controls,
suggesting that miR-433-3p can directly bind to OSER1-AS1 in NSCLC
cells (Fig. 3E).
The effects of OSER1-AS1 expression on miR-433-3p
expression in NSCLC cells were evaluated using RT-qPCR. miR-433-3p
expression was significantly increased in the H522 and A549 cells
upon OSER1-AS1 knockdown (Fig. 3F).
Furthermore, RT-qPCR data revealed that miR-433-3p was weakly
expressed in the NSCLC tissues compared with that noted in the
adjacent normal tissues (Fig. 3G).
Concordantly, Spearman's correlation analysis revealed an inverse
correlation between the expression of OSER1-AS1 and miR-433-3p in
the 53 NSCLC tissues (Fig. 3H;
r=−0.5242, P<0.0001). In general, these results suggest that
OSER1-AS1 acts as a ceRNA and molecular sponge for miR-433-3p in
NSCLC cells.
OSER1-AS1 positively controls Smad2
expression in NSCLC cells by sponging miR-433-3p
Given that OSER1-AS1 functions as a ceRNA in NSCLC,
we hypothesized that it modulates the expression of mRNAs targeted
by miR-433-3p. Because a previous study identified Smad2 as
a direct target of miR-433-3p in NSCLC cells (30), we next evaluated Smad2 mRNA
and protein expression in OSER1-AS1-depleted H522 and A549 cells.
RT-qPCR and western blotting revealed that silencing of OSER1-AS1
expression decreased Smad2 expression in the H522 and A549
cells at both the mRNA (Fig. 4A)
and protein (Fig. 4B) levels. To
further assess the relationship between OSER1-AS1 and miR-433-3p
expression in NSCLC cells, we performed RT-qPCR to measure
Smad2 expression in 53 pairs of NSCLC tissues and adjacent
normal tissues. Smad2 expression was significantly higher in
the NSCLC tissues than that noted in the adjacent normal tissues
(Fig. 4C). Furthermore,
Smad2 mRNA expression was positively correlated with
OSER1-AS1 expression in the 53 NSCLC tissues (Fig. 4D; r=0.6175, P<0.0001).
Furthermore, higher Smad2 mRNA expression was observed in
the high-OSER1-AS1 expression group than in the low-OSER1-AS1
expression group (Fig. 4E).
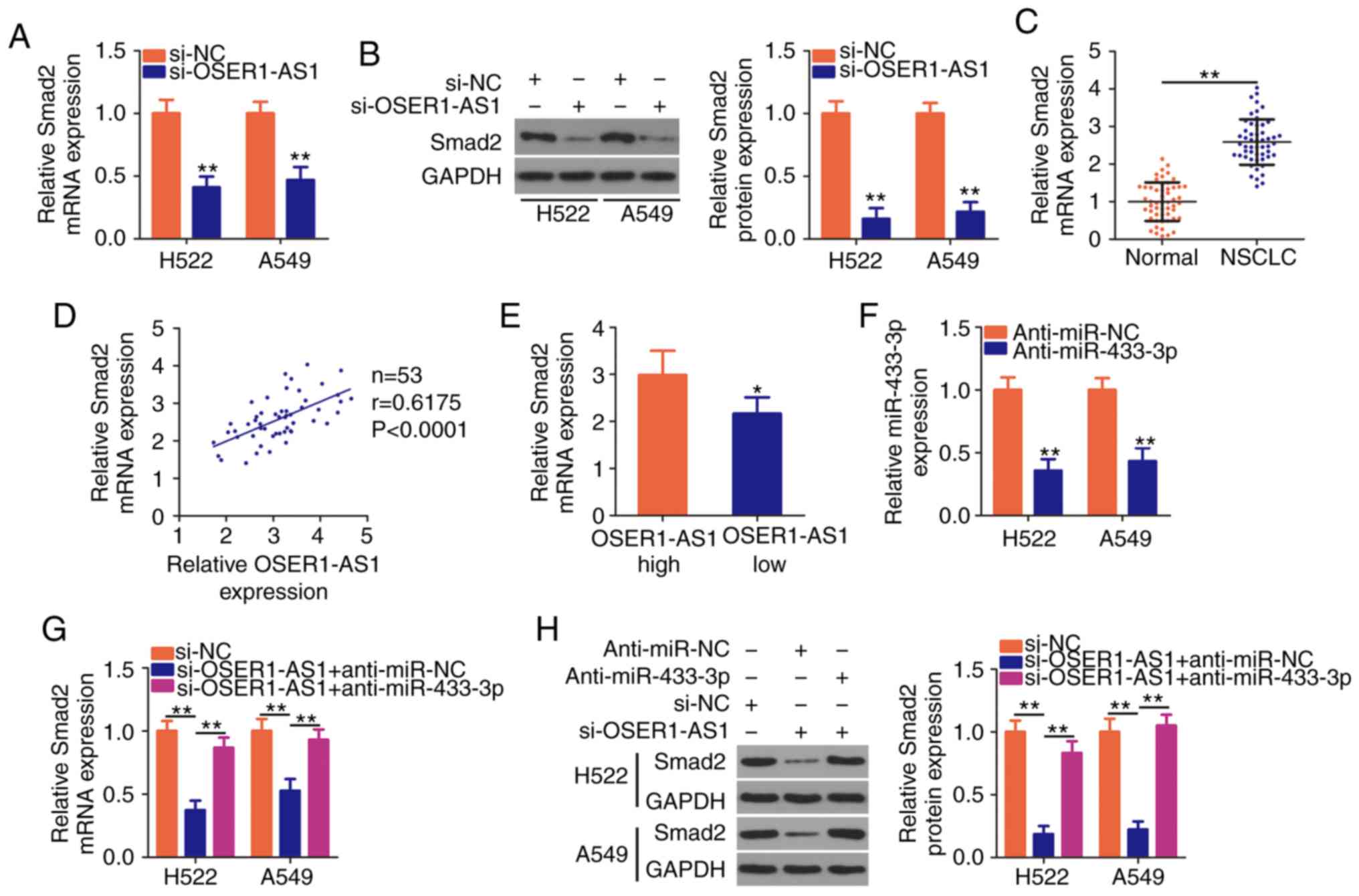 | Figure 4.OSER1-AS1 knockdown suppresses
Smad2 expression in NSCLC cells. (A and B) Detection of
Smad2 mRNA and protein expression in OSER1-AS1-deficient
H522 and A549 cell lines by RT-qPCR and western blotting,
respectively. **P<0.01, compared with the si-NC group. (C)
Determination of Smad2 mRNA expression in 53 pairs of NSCLC
tissues and adjacent normal tissues using RT-qPCR. **P<0.01. (D)
Spearman's correlation analysis of the correlation between
OSER1-AS1 and Smad2 mRNA expression in the 53 NSCLC tissues
(r=0.6175, P<0.0001). (E) Comparison of Smad2 expression
in NSCLC tissues derived from patients in the high- and
low-OSER1-AS1 expression groups. *P<0.05, compared with the
OSER1-AS1 high group. (F) miR-433-3p expression in H522 and A549
cells transfected with anti-miR-433-3p or anti-miR-NC using
RT-qPCR. **P<0.01, compared with the anti-miR-NC group. (G and
H) Cotransfection of OSER1-AS1-deficient H522 and A549 cells with
anti-miR-433-3p or anti-miR-NC and detection of Smad2 protein
expression using RT-qPCR and western blotting, respectively.
**P<0.01. OSER1-AS1, RNA OSER1 antisense RNA 1; Smad2,
mothers against decapentaplegic homolog 2; miR, microRNA; NSCLC,
non-small cell lung cancer; RT-qPCR, reverse
transcription-quantitative polymerase chain reaction. |
Rescue assays were performed to determine whether
OSER1-AS1 acts as a regulator of Smad2 expression in NSCLC
cells by sponging miR-433-3p. We first determined the transfection
efficiency of a miR-433-3p inhibitor (anti-miR-433-3p) using
RT-qPCR (Fig. 4F). Subsequently,
anti-miR-433-3p or anti-miR-NC was introduced together with
si-OSER1-AS1 into H522 and A549 cells, after which RT-qPCR and
western blotting were performed to determine any changes in
Smad2 mRNA and protein expressions, respectively. Notably,
OSER1-AS1 knockdown led to a considerable decrease in Smad2
mRNA (Fig. 4G) and protein
(Fig. 4H) expression, whereas the
inhibitory impact of the knockdown was reversed by miR-433-3p
inhibition. In general, OSER1-AS1 acts as a ceRNA for miR-433-3p
and thereby increases Smad2 expression in NSCLC cells.
miR-433-3p inhibition or Smad2
restoration counteracts the suppressive effects of OSER1-AS1
knockdown on NSCLC cell proliferation, migration, and invasion and
promoted cell apoptosis
We next performed rescue assays to confirm whether
OSER1-AS1 affected the malignant properties of NSCLC cells by
regulating the miR-433-3p/Smad2 axis. Here, we introduced
si-OSER1-AS1 with anti-miR-433-3p or anti-miR-NC into H522 and A549
cells. CCK-8 assay revealed that cotransfection with
anti-miR-433-3p neutralized the inhibitory effect of OSER1-AS1
interference on H522 and A549 cell proliferation (Fig. 5A). Additionally, OSER1-AS1-deficient
H522 and A549 cells exhibited increased apoptosis, which was
reversed after miR-433-3p inhibition as demonstrated by flow
cytometry (Fig. 5B). Furthermore,
the reintroduction of anti-miR-433-3p abolished the effects of
OSER1-AS1 knockdown on the migration (Fig. 5C) and invasiveness (Fig. 5D) of H522 and A549 cells.
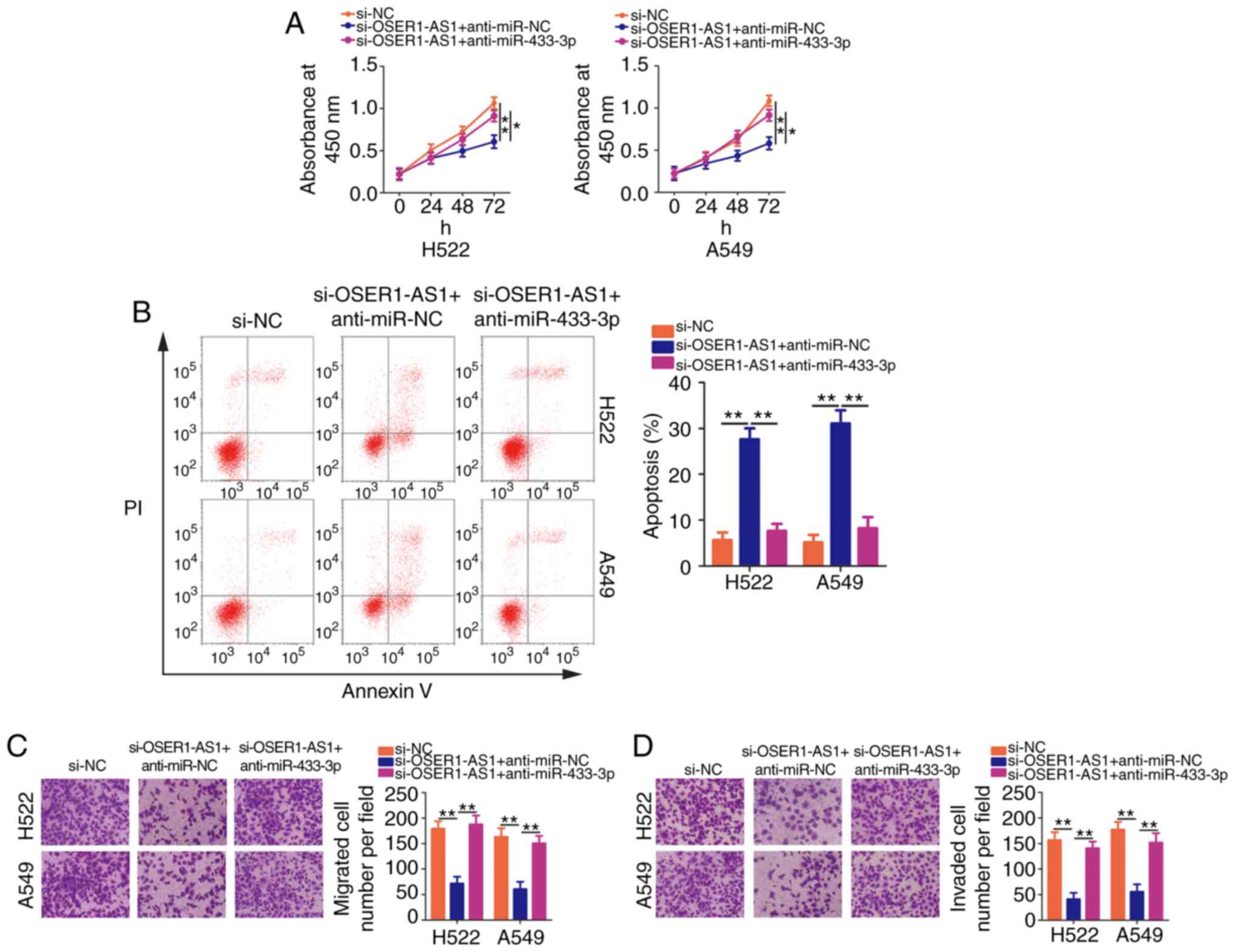 | Figure 5.miR-433-3p knockdown abrogates the
effects of OSER1-AS1 depletion on the malignant phenotypes of NSCLC
cells. H522 and A549 cell lines were cotransfected with
si-OSER1-AS1 and anti-miR-433-3p or anti-miR-NC. (A) Cell
proliferation, (B) apoptosis, (C) migration, and (D) invasion of
the cotransfected cells were determined using Cell Counting Kit-8
assay, flow cytometry, and cell migration assay, and cell invasion
assay, respectively. *P<0.05 and **P<0.01. miR, microRNA;
OSER1-AS1, RNA OSER1 antisense RNA 1; NSCLC, non-small cell lung
cancer; PI, propidium iodide. |
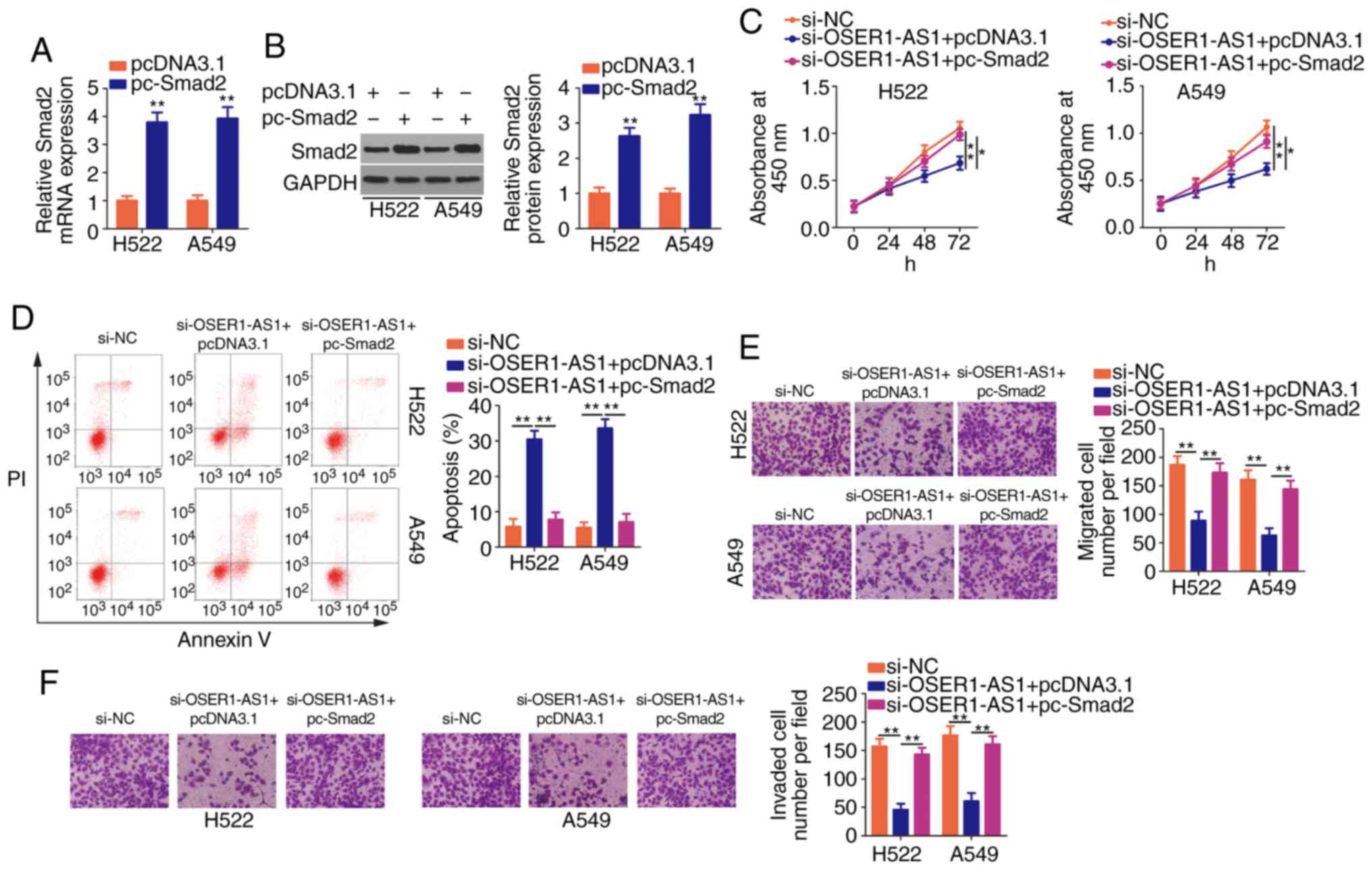
Transfection with pc-Smad2 led to a noticeable
increase in Smad2 mRNA (Fig.
6A) and protein (Fig. 6B)
expression in the H522 and A549 cells, as revealed using RT-qPCR
and western blotting, respectively. Additionally, rescue assays
were performed on H522 and A549 cells after cotransfection with
si-OSER1-AS1 and pc-Smad2 or pcDNA3.1. Similarly, the restoration
of Smad2 expression in H522 and A549 cells reversed the
suppressed proliferation (Fig. 6C),
promoted apoptosis (Fig. 6D) as
well as impaired migration (Fig.
6E) and invasion (Fig. 6F)
induced by OSER1-AS1 knockdown. In summary, OSER1-AS1 exerts its
pro-oncogenic roles in NSCLC cells by regulating the
miR-433-3p/Smad2 axis.
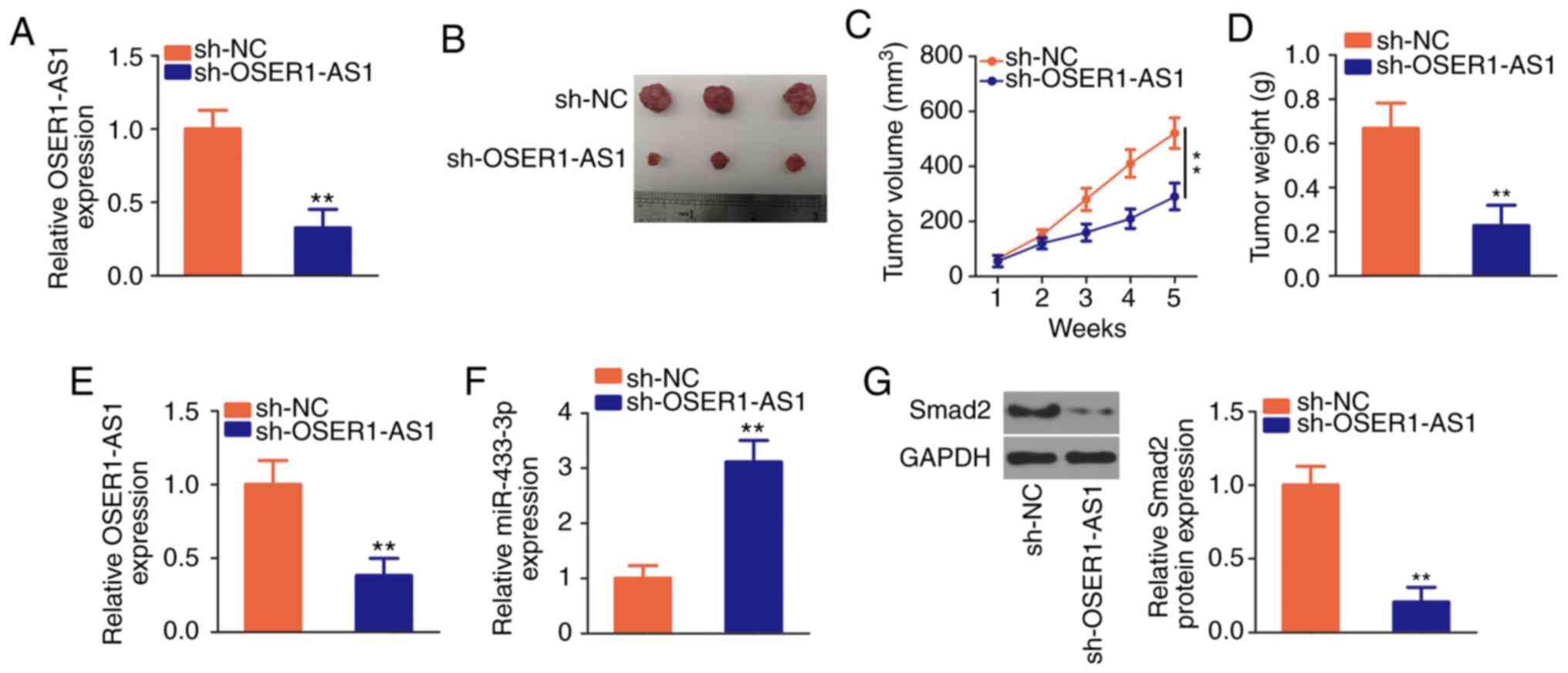
OSER1-AS1 knockdown restrains the
growth of NSCLC cells in vivo
The effects of OSER1-AS1 inhibition on NSCLC growth
in vivo were further investigated by tumor xenograft assay.
The interference efficiency of sh-OSER1-AS1 in H522 cells was
verified using RT-qPCR (Fig. 7A).
Subsequently, H522 cells that stably expressed sh-OSER1-AS1 or
sh-NC were inoculated subcutaneously into nude mice. The tumor
xenografts in the sh-OSER1-AS1 group were smaller in size and
volume than those in the sh-NC group (Fig. 7B and C). Additionally, the weight of
tumor xenografts was significantly lower in the sh-OSER1-AS1 group
than that in the sh-NC group (Fig.
7D). All mice were euthanized after 5 weeks, and the tumor
xenografts were dissected and analyzed to determine OSER1-AS1,
miR-433-3p, and Smad2 expression. RT-qPCR revealed the
continued suppression of OSER1-AS1 (Fig. 7E) and upregulation of miR-433-3p
(Fig. 7F) expression in the tumor
xenografts of mice injected with H522 cells that stably expressed
OSER1-AS1. Furthermore, Smad2 protein expression was significantly
reduced in the tumor xenografts that originated from H522 cells
stably transfected with sh-OSER1-AS1 (Fig. 7G). These results suggest that
OSER1-AS1 knockdown impedes the growth of NSCLC cells in
vivo by upregulating miR-433-3p and downregulating Smad2
expression.
Discussion
Many recent studies have demonstrated the abnormal
expression of various long noncoding RNAs (lncRNAs) in non-small
cell lung cancer (NSCLC) (31–33).
These dysregulated lncRNAs act as important regulators of the
pathogenesis of NSCLC and have crucial effects on NSCLC
tumorigenesis, progression, and metastasis (34–36).
Therefore, a comprehensive study of the functional involvement of
lncRNAs in NSCLC may yield useful information and help identify
attractive targets for anticancer management. To date, more than
3,000 lncRNAs have been validated in the human genome, but only few
of them have been studied (37). In
this study, we evaluated OSER1 antisense RNA 1 (OSER1-AS1)
expression in NSCLC tissues and cell lines and explored the
clinical value of aberrantly high OSER1-AS1 expression in NSCLC.
Furthermore, we conducted a series of functional experiments to
explore the detailed roles of OSER1-AS1 in NSCLC progression.
Moreover, we thoroughly elucidated the downstream target of
OSER1-AS1 and its underlying mechanisms.
The oncogenic action of OSER1-AS1 in hepatocellular
carcinoma has been well studied. In this tumor type, OSER1-AS1
expression is upregulated and strongly associated with adverse
clinicopathological factors and low disease-free survival and
overall survival (24).
Functionally, interference with OSER1-AS1 expression in
hepatocellular carcinoma cells suppressed their proliferation,
migration, and invasion and promoted their apoptosis (24). To the best of our knowledge, no
previous study has investigated the expression patterns and
detailed functions of OSER1-AS1 in NSCLC. Herein, we collected 53
pairs of NSCLC tissues and adjacent normal tissues as well as
determined OSER1-AS1 expression using RT-qPCR. Our results revealed
strong expression of OSER1-AS1 in the NSCLC tissues. Consistently,
all five tested NSCLC cell lines exhibited relatively higher
OSER1-AS1 expressions than human nontumorigenic bronchial
epithelial BEAS-2B cells. High OSER1-AS1 expression presented an
obvious correlation with tumor size, TNM stage, lymph node
metastasis and shorter overall survival in patients with NSCLC.
Functional research affirmed that OSER1-AS1 knockdown restricted
NSCLC cell proliferation, migration, and invasion in vitro;
promoted cell apoptosis; and restrained tumor growth in
vivo.
The mechanistic events by which lncRNAs participate
in carcinogenesis and cancer progression depend on their
subcellular localization in tumor cells (29). Accordingly, we first determined the
expression distribution of OSER1-AS1 in NSCLC cells through nuclear
and cytoplasmic RNA fractionation. OSER1-AS1 was found to be mostly
located in the cytoplasm of NSCLC cells. Increasing evidence
suggests that cytoplasmic lncRNAs can act as competitive endogenous
RNAs (ceRNAs) and thereby competitively bind to the target sites of
miRNAs, leading to the release of target mRNAs (38,39).
In this study, we used the StarBase 3.0 database to
identify potential miRNAs as targets for sponging by OSER1-AS1.
Bioinformatics analysis indicated that OSER1-AS1 contained base
pairs complementary to miR-433-3p. Next, we conducted a series of
experiments to validate our hypothesis. Luciferase reporter and RIP
assays further verified that OSER1-AS1 bound directly to miR-433-3p
in NSCLC cells. Furthermore, miR-433-3p expression was drastically
downregulated in NSCLC tissues, and this expression pattern
exhibited an inverse correlation with that of OSER1-AS1. Moreover,
OSER1-AS1 knockdown led to increased miR-433-3p expression and
decreased Smad2 (a direct target gene of miR-433-3p)
expression in NSCLC cells, whereas miR-433-3p inhibition
counteracted the regulatory effect of OSER1-AS1 knockdown on
Smad2 expression. Therefore, the oncogenic role of OSER1-AS1
in NSCLC can be explained in the context of a ceRNA pathway
involving OSER1-AS1, miR-433-3p and Smad2.
Fan et al reported that OSER1-AS1 performs
oncogenic roles in hepatocellular carcinoma by regulating the
miR-372-3p/Rab23 axis (24).
miR-372-3p is upregulated in NSCLC and exerts tumor-suppressive
actions on cell growth and metastasis (40). Hence, the miR-372-3p/Rab23 axis
could not be the downstream regulatory mechanism of OSER1-AS1 in
NSCLC. In this study, our new findings revealed different working
mechanisms, which were not consistent with previous studies
regarding OSER1-AS1 (24).
Aberrant expression of miR-433-3p has been observed
in multiple human cancer types including NSCLC (30). In our study, miR-433-3p
overexpression led to obvious decreases in NSCLC cell
proliferation, migration, and invasion in vitro and hindered
tumor growth in vivo. Mechanistically, Smad2 has been
verified as a direct downstream target of miR-433-3p in NSCLC
cells. Smad2, a member of the Smad family, is a key signal
transducer and transcriptional modulator implicated in the
regulation of multiple signaling pathways (41). Smad2 expression is
upregulated in NSCLC, where it modulates various cancer-associated
aggressive processes (42–44). These observations highlight the
importance of the miR-433-3p/Smad2 axis in NSCLC
tumorigenesis and progression. Nevertheless, the upstream mechanism
that regulates this axis remains largely unclear and warrants
further investigation. Our results demonstrated an apparent
increase in Smad2 expression in NSCLC tissues as well as its
positive correlation with OSER1-AS1 expression. Our restoration
experiments revealed that enhanced activity of the
miR-433-3p/Smad2 axis alleviated the inhibitory effects of
OSER1-AS1 knockdown on NSCLC cell proliferation, migration, and
invasion and reversed its apoptosis-enhancing effect. These results
collectively imply that the tumor-promoting actions of OSER1-AS1 in
NSCLC cells may be attributable to the miR-433-3p-induced
upregulation of Smad2 expression.
In the present research, we did not analyze the
driver oncogene mutations in the NSCLC tissues; hence, statistical
analysis could not be performed to test the comparisons between
OSER1-AS1 expression and oncogene mutations in NSCLC. This is a
limitation of our study, and we will resolve the limitation in the
near future.
OSER1-AS1 drives the malignant properties of NSCLC
cells in vitro and in vivo by regulating the
miR-433-3p/Smad2 axis. Identification of the
OSER1-AS1/miR-433-3p/Smad2 pathway offers a novel
perspective on the discovery of effective therapeutic and
diagnostic targets for NSCLC.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
QZ and XL conceived and designed the study. XL, SH
and YG carried out the experiments. QZ and XL wrote the paper. All
authors reviewed and edited the manuscript. All authors read and
approved the manuscript and agree to be accountable for all aspects
of the research in ensuring that the accuracy or integrity of any
part of the work are appropriately investigated and resolved.
Ethics approval and consent to
participate
The present study was approved by the Research
Ethics Committee of Weifang People's Hospital (Weifang, China), and
was performed in accordance with the Declaration of Helsinki and
the guidelines of the Ethics Committee of The Seventh People's
Hospital. Written informed consent was obtained from all patients
for the use of their clinical tissues.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2019. CA Cancer J Clin. 69:7–34. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Shin JY, Yoon JK and Marwaha G: Progress
in the treatment and outcomes for Early-Stage non-small cell lung
cancer. Lung. 196:351–358. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Lagos GG, Izar B and Rizvi NA: Beyond
tumor PD-L1: Emerging genomic biomarkers for checkpoint inhibitor
immunotherapy. Am Soc Clin Oncol Educ Book. 40:1–11.
2020.PubMed/NCBI
|
|
5
|
Travis WD: Lung cancer pathology: Current
concepts. Clin Chest Med. 41:67–85. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Vansteenkiste J, Crino L, Dooms C,
Douillard JY, Faivre-Finn C, Lim E, Rocco G, Senan S, Van Schil P,
Veronesi G, et al: 2nd ESMO Consensus Conference on lung cancer:
Early-stage non-small-cell lung cancer consensus on diagnosis,
treatment and follow-up. Ann Oncol. 25:1462–1474. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Pirker R: Adjuvant chemotherapy in
patients with completely resected non-small cell lung cancer.
Transl Lung Cancer Res. 3:305–310. 2014.PubMed/NCBI
|
|
8
|
Torre LA, Siegel RL and Jemal A: Lung
cancer statistics. Adv Exp Med Biol. 893:1–19. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Brambilla E and Gazdar A: Pathogenesis of
lung cancer signalling pathways: Roadmap for therapies. Eur Respir
J. 33:1485–1497. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kadara H, Scheet P, Wistuba II and Spira
AE: Early events in the molecular pathogenesis of lung cancer.
Cancer Prev Res (Phila). 9:518–527. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Peng WX, Koirala P and Mo YY:
LncRNA-mediated regulation of cell signaling in cancer. Oncogene.
36:5661–5667. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ponting CP, Oliver PL and Reik W:
Evolution and functions of long noncoding RNAs. Cell. 136:629–641.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Huarte M: The emerging role of lncRNAs in
cancer. Nat Med. 21:1253–1261. 2015. View
Article : Google Scholar : PubMed/NCBI
|
|
14
|
Cheetham SW, Gruhl F, Mattick JS and
Dinger ME: Long noncoding RNAs and the genetics of cancer. Br J
Cancer. 108:2419–2425. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ma HP, Wang LX, Li W, Guo HH, Wu Y and Li
XY: Upregulation of LINC00504 is associated with aggressive
progression and poor prognosis in non-small cell lung cancer. Eur
Rev Med Pharmacol Sci. 24:699–703. 2020.PubMed/NCBI
|
|
16
|
Qiu C, Li S, Sun D and Yang S: lncRNA PVT1
accelerates progression of non-small cell lung cancer via targeting
miRNA-526b/EZH2 regulatory loop. Oncol Lett. 19:1267–1272.
2020.PubMed/NCBI
|
|
17
|
Wang Z, Zhang J, Yang B, Li R, Jin L, Wang
Z, Yu H, Liu C, Mao Y and You Q: Long Intergenic noncoding RNA
00261 acts as a tumor suppressor in non-small cell lung cancer via
regulating miR-105/FHL1 Axis. J Cancer. 10:6414–6421. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Shangguan WJ, Liu HT, Que ZJ, Qian FF, Liu
LS and Tian JH: TOB1-AS1 suppresses non-small cell lung cancer cell
migration and invasion through a ceRNA network. Exp Ther Med.
18:4249–4258. 2019.PubMed/NCBI
|
|
19
|
Gao YP, Li Y, Li HJ and Zhao B: LncRNA
NBR2 inhibits EMT progression by regulating Notch1 pathway in
NSCLC. Eur Rev Med Pharmacol Sci. 23:7950–7958. 2019.PubMed/NCBI
|
|
20
|
Wang Y, Luo X, Liu Y, Han G and Sun D:
Long noncoding RNA RMRP promotes proliferation and invasion via
targeting miR-1-3p in non-small-cell lung cancer. J Cell Biochem.
120:15170–15181. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Bian C, Yuan L and Gai H: A long
non-coding RNA LINC01288 facilitates non-small cell lung cancer
progression through stabilizing IL-6 mRNA. Biochem Biophys Res
Commun. 514:443–449. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Calin GA and Croce CM: MicroRNA signatures
in human cancers. Nat Rev Cancer. 6:857–866. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Cesana M, Cacchiarelli D, Legnini I,
Santini T, Sthandier O, Chinappi M, Tramontano A and Bozzoni I: A
long noncoding RNA controls muscle differentiation by functioning
as a competing endogenous RNA. Cell. 147:358–369. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Fan J, Zhang J, Huang S and Li P: lncRNA
OSER1-AS1 acts as a ceRNA to promote tumorigenesis in
hepatocellular carcinoma by regulating miR-372-3p/Rab23 axis.
Biochem Biophys Res Commun. 521:196–203. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Sibio S, Sica GS, Di Carlo S, Cardi M, Di
Giorgio A, Sollazzo BM and Sammartino P: Surgical treatment of
intraperitoneal metastases from lung cancer: Two case reports and a
review of the literature. J Med Case Rep. 13:2622019. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yi E, Kim D, Cho S, Kim K and Jheon S:
Clinical outcomes of cytoreductive surgery combined with
intrapleural perfusion of hyperthermic chemotherapy in advanced
lung adenocarcinoma with pleural dissemination. J Thorac Dis.
8:1550–1560. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Li JH, Liu S, Zhou H, Qu LH and Yang JH:
starBase v2.0: Decoding miRNA-ceRNA, miRNA-ncRNA and protein-RNA
interaction networks from large-scale CLIP-Seq data. Nucleic Acids
Res. 42((Database Issue)): D92–D97. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wang L, Cho KB, Li Y, Tao G, Xie Z and Guo
B: Long Noncoding RNA (lncRNA)-mediated competing endogenous RNA
networks provide novel potential biomarkers and therapeutic targets
for colorectal cancer. Int J Mol Sci. 20:57582019. View Article : Google Scholar
|
|
30
|
Li J, Chen M and Yu B: miR-433 suppresses
tumor progression via Smad2 in non-small cell lung cancer. Pathol
Res Pract. 215:1525912019. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Tang L, Wang T, Zhang Y, Zhang J, Zhao H,
Wang H, Wu Y and Liu K: Long non-coding RNA AWPPH promotes
postoperative distant recurrence in resected non-small cell lung
cancer by upregulating transforming growth factor beta 1 (TGF-β1).
Med Sci Monit. 25:2535–2541. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Bai Y, Zhang G, Chu H, Li P and Li J: The
positive feedback loop of lncRNA DANCR/miR-138/Sox4 facilitates
malignancy in non-small cell lung cancer. Am J Cancer Res.
9:270–284. 2019.PubMed/NCBI
|
|
33
|
Liu X, Lu X, Zhen F, Jin S, Yu T, Zhu Q,
Wang W, Xu K, Yao J and Guo R: LINC00665 induces acquired
resistance to Gefitinib through recruiting EZH2 and activating
PI3K/AKT pathway in NSCLC. Mol Ther Nucleic Acids. 16:155–161.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Zhang Y, Li Y, Han L, Zhang P and Sun S:
SUMO1P3 is associated clinical progression and facilitates cell
migration and invasion through regulating miR-136 in non-small cell
lung cancer. Biomed Pharmacother. 113:1086862019. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Zhang B, Wang H, Wang Q, Xu J, Jiang P and
Li W: Knockout of lncRNA UCA1 inhibits drug resistance to gefitinib
via targeting STAT3 signaling in NSCLC. Minerva Med. 110:273–275.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Li B, Gu W and Zhu X: NEAT1 mediates
paclitaxel-resistance of non-small cell of lung cancer through
activation of Akt/mTOR signalling pathway. J Drug Target.
27:1061–1067. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Hutchinson JN, Ensminger AW, Clemson CM,
Lynch CR, Lawrence JB and Chess A: A screen for nuclear transcripts
identifies two linked noncoding RNAs associated with SC35 splicing
domains. BMC Genomics. 8:392007. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Wang JJ, Huang YQ, Song W, Li YF, Wang H,
Wang WJ and Huang M: Comprehensive analysis of the lncRN-associated
competing endogenous RNA network in breast cancer. Oncol Rep.
42:2572–2582. 2019.PubMed/NCBI
|
|
39
|
Xu F, Zhao Y, Qin G, Huan Y, Li L and Gao
W: Comprehensive analysis of competing endogenous RNA networks
associated with cholangiocarcinoma. Exp Ther Med. 18:4103–4112.
2019.PubMed/NCBI
|
|
40
|
Wang Q, Liu S, Zhao X, Wang Y, Tian D and
Jiang W: MiR-372-3p promotes cell growth and metastasis by
targeting FGF9 in lung squamous cell carcinoma. Cancer Med.
6:1323–1330. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Kamato D and Little PJ: Smad2 linker
region phosphorylation is an autonomous cell signalling pathway:
Implications for multiple disease pathologies. Biomed Pharmacother.
124:1098542020. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Chen T, Zhu J, Cai T, Du W, Zhang Y, Zhu
Q, Liu Z and Huang JA: Suppression of non-small cell lung cancer
migration and invasion by hsa-miR-486-5p via the TGF-β/SMAD2
signaling pathway. J Cancer. 10:6014–6024. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Othman N and Nagoor NH: Overexpression of
miR3615p plays an oncogenic role in human lung adenocarcinoma
through the regulation of SMAD2. Int J Oncol. 54:306–314.
2019.PubMed/NCBI
|
|
44
|
Wang Z, Lu Y, Sheng B, Ding Y and Cheng X:
Catalpol inhibits TGF-β1-induced epithelial-mesenchymal transition
in human non-small-cell lung cancer cells through the inactivation
of Smad2/3 and NF-κB signaling pathways. J Cell Biochem. Sep
11–2018.doi: 10.1002/jcb.27535 (Epub Ahead of Print).
|