Ovarian cancer (OC) is a highly common malignancy of
the female reproductive system that ranks 4th among all causes of
cancer-related mortality among women (1,2). The early
symptoms of OC are atypical, and reliable methods in terms of early
detection are insufficient, with ~70% of cases already presenting
with International Federation of Gynecology and Obstetrics (FIGO)
stage III/IV disease at clinical diagnosis (3). OC is currently treated with full-stage
surgery or tumor cell reduction combined with first-line
chemotherapy drugs (paclitaxel or platinum) as the standard of
care. Chemotherapy results in first remission in ~80% of patients,
whereas the majority of cases experience tumor recurrence and
progressive chemoresistance within 5 years, achieving a 5-year
survival rate of <47.6% (3). Thus,
further investigations are required in order to gain insight into
the pathogenesis OC and design novel therapeutic strategies.
OC is divided into various histopathological
subtypes, including epithelial, germ cell and sex cord-stromal
tumors. Almost 90% of malignant ovarian tumors are epithelial,
originating in the ovarian surface epithelium (OSE). However, over
the past few years, the fimbriae of the fallopian tubes were
considered as the probable site of origin of such malignancies,
particularly high-grade serous OC (HGSOC) (4,5). It is
noteworthy that simple original OSE exhibiting mesenchyme-related
characteristics is characterized by the Müllerian epithelium since
its development towards malignancy. Based on the morphology,
function and antigenic similarity to the Müllerian duct epithelium,
epithelial OC (EOC) is divided into five major subtypes that are
histologically defined as follows: Mucinous (3%), clear cell (10%),
endometrioid (10%), low-grade serous OC (LGSOC; <5%) and HGSOC
(70%) (6–8). The primary origin of HGSOC is the
fimbriated end of the fallopian tube, and its characteristics
include common TP53 mutations and aberrations in genes
involved in cell cycle control [e.g., neurofibromin 1 (NF1),
retinoblastoma 1 (RB1) and cyclin E1 (CCNE1)], or the
inactivation of genes involved in homologous recombination DNA
repair (e.g., BRCA1 and BRCA2) (9,10). LGSOC
may be implicated in the activation of the MAPK pathway via
NRAS, KRAS or BRAF mutations, and is associated with
high levels of estrogen receptor (ER) and progesterone receptor
(PR) expression (11). Endometrioid
and clear cell OC may be associated with endometriosis, whereas
certain lifestyle factors (e.g., smoking) noticeably increase the
risk of developing mucinous OC (12,13).
Moreover, the pathogenesis of OC is also associated with mutations
in the AT-rich interaction domain 1A (ARID1A),
phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit
alpha (PIK3CA) and phosphatase and tensin homolog
(PTEN) genes (14–16). Ovarian germ cell tumors develop from
primordial germ cells of the embryonic gonads, and the most common
germ cell malignancies include yolk sac tumor, teratoma and
dysgerminoma. Sex cord-stromal neoplasms may originate from a wide
range of cell types within the sex cords and gonadal stroma.
Granulosa cell tumors (GCTs) are the most common in this group,
accounting for ~5% of all OC cases.
Despite its major impact on public health, the
factors regulating the development and progression of OC have yet
to be fully elucidated (17,18). In general, women who inherit mutations
in either one of the two breast cancer susceptibility genes
(BRCA1 or BRCA2) are at an increased risk of
developing breast cancer and OC compared with women without a
BRCA gene mutation. The overall lifetime risk for developing
OC is 20–40% for women who have a BRCA1 mutation and 10–20%
for women who have a BRCA2 mutation. Women in the general
population have a <2% risk of developing OC (19). The National Comprehensive Cancer
Network (NCCN) OC guidelines (2021) (20) recommend poly(ADP-ribose) polymerase
(PARP) inhibitors for maintenance therapy in patients with
platinum-sensitive recurrent OC, regardless of the biomarker
status. In addition, the widespread application of hormone therapy
in the clinical treatment of breast and endometrial cancers has
made hormone receptors the primary research direction for a
targeted cure for OC (21,22). It has been clearly stated in the NCCN
OC guidelines (2021) (20) and in the
European Society of Medical Oncology/European Society of
Gynecological Oncology guidelines (23) that hormonal therapy may be used to
treat patients with platinum-resistant and recurrent OC.
Epidemiological studies have demonstrated that steroid hormones
released through the hypothalamic/pituitary/ovarian axis can
stimulate or suppress OC progression: Gonadotropins, estrogens and
androgens promote OC progression, while gonadotropin-releasing
hormone (GnRH) and progesterone may serve as protective factors
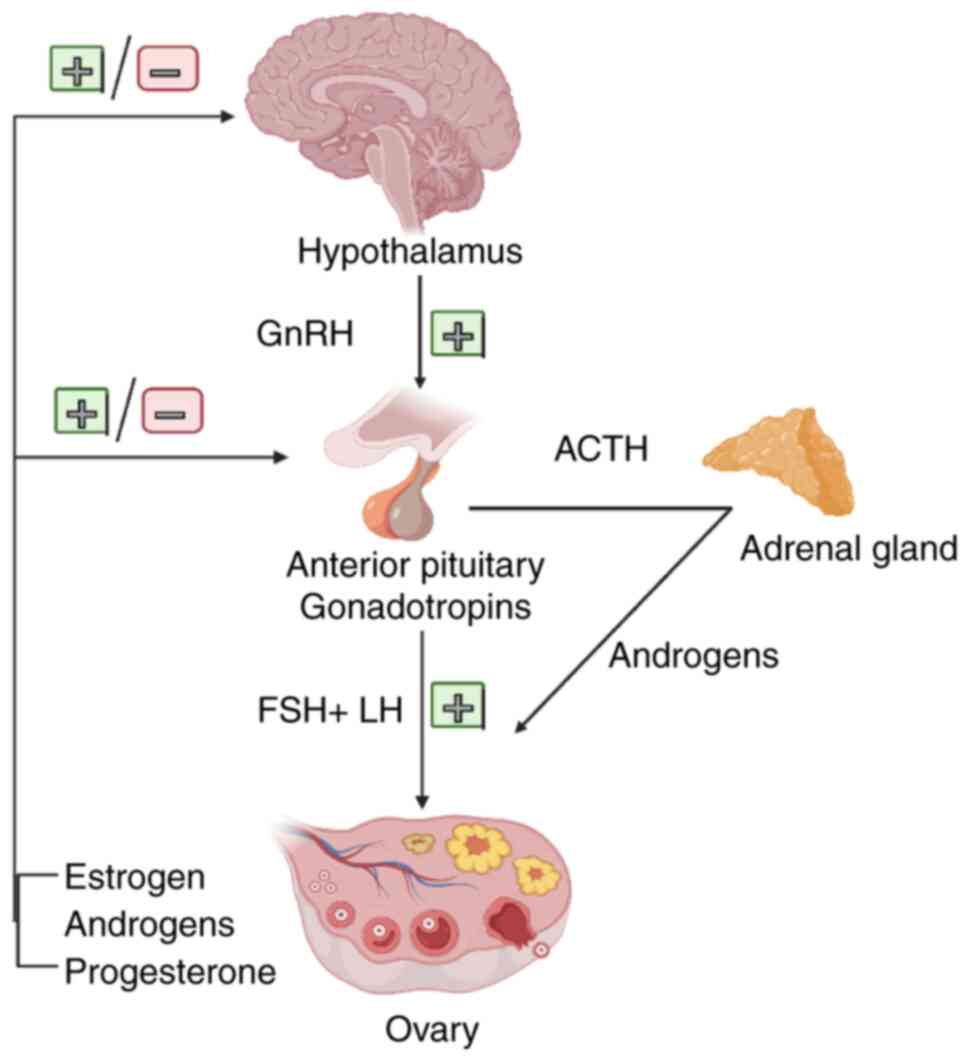
against OC (3,24). The endocrine regulation of the ovary
primarily relies on the neuroendocrine actions of the
hypothalamic-pituitary-ovary axis (Fig.
1). Experimental studies and clinicopathological findings have
demonstrated that hormone receptors are expressed in the normal
ovarian surface epithelium, as well as in ovarian cancer cells and
mediate the stimulatory or inhibitory effects of various hormones
on the development of these cells. Moreover, hormonal therapeutic
agents have been clinically evaluated in some patients with
recurrent or refractory ovarian tumors, mainly exhibiting average
efficiency and limited side-effects. For example, in the study by
Sieh et al (25), data from
almost 3,000 women with invasive EOC were analyzed u sing hormone
receptor assay and evidence of the prognostic role of ER and PR and
the potential hormonal sensitivity of EOC was provided. In
addition, Paleari and DeCensi (26)
conducted a meta-analysis of 53 clinical trials, including 2,490
patients and revealed an overall clinical benefit rate (CBR) of 41%
[95% confidence interval (CI), 0.34–0.48] for any endocrine
therapy. These results suggest that a greater understanding of the
mechanisms through which hormones affect OC cell development may
improve the effectiveness of hormone therapy for patients with this
type of cancer.
The present review conducted a literature search on
PubMed, Web of Science and Clinical Trials for relevant articles
published from inception to May, 2021 with no restrictions. The
search terms included ‘hormone therapy’ (or ‘hormone replacement
therapy’) and ‘ovarian cancer’ (or ‘ovarian carcinoma’ or ‘ovary
cancer’); ‘peptide hormones (gonadotropin-releasing hormone/GnRH,
gonadotropins, androgens, estrogens, and progestins) and ‘ovarian
cancer’ (or ‘ovarian carcinoma’ or ‘ovary cancer’). The reference
lists of the included studies were also reviewed for potential
available studies. The mechanisms involved in the hormonal
influences on the progression of OC (mainly EOC) are summarized,
mentioning peptide hormones (GnRH, gonadotropins, androgens,
estrogens and progestins), and the clinical efficacy and safety of
various hormonal therapies for OC are discussed.
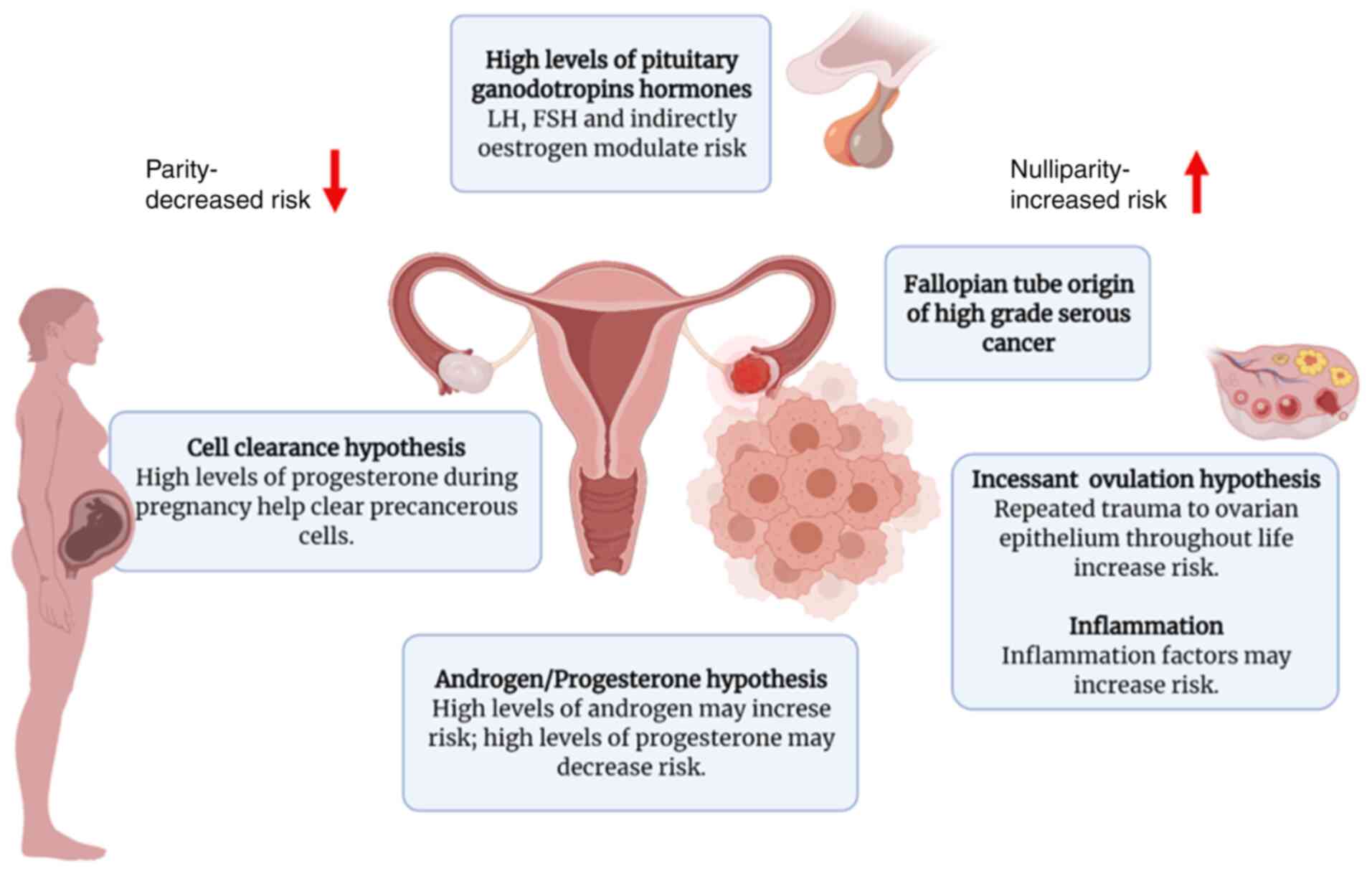
Epidemiological studies have implicated hormonal and
reproductive factors in the pathogenesis of OC. Several hormonal
hypotheses have been suggested thus far in an attempt to elucidate
the etiology of OC, including gonadotropin signaling, direct
influences exerted by progesterone and androgen, and incessant
ovulation (Fig. 2). The first
hypothesis of sex hormones as a potential mechanism underlying
ovarian carcinogenesis is the ‘gonadotropin hypothesis’ (27–29).
According to this hypothesis, OC advances due to excessive ovarian
tissue excitation by pituitary gonadotropins [follicle-stimulating
hormone (FSH) and luteinizing hormone (LH)] (27). Exposure to excess gonadotropins, which
is associated with menopause, ovulation or infertility treatment,
has been identified as an important risk factor for the development
of OC. Moreover, such a theory would also explain the decreased
risk of developing OC associated with the use of oral
contraceptives and pregnancy, which results in the decreased
exposure to gonadotropins owing to the negative feedback regulation
of steroid hormones onto the pituitary gland (30,31). In
contrast to the above, post-menopausal women with increased
gonadotropin levels and women suffering from polycystic ovary
syndrome (PCOS), who have upregulated circulating LH levels, are at
an increased risk of developing EOC (32). Evidence from several epidemiological
studies supports the gonadotropin theory. First, the rise in
circulating gonadotropin levels exhibits a strong temporal
association with the increased incidence of EOC (33,34).
Menopause occurs at the age of ~51 years and is accompanied by
variations in gonadotropin levels due to the cessation of the
menstrual cycle and the deterioration of ovarian function. When
ovarian function ceases completely, the negative feedback of
ovarian steroids on gonadotropins is lost. Within 2–3 years
following menopause, the gonadotropin levels can be particularly
high. Accordingly, the LH and FSH concentrations may peak 3–4-fold
(20–50 mIU/ml) and 10–20-fold (50–100 mIU/ml) compared with the
values during the proliferation stage of the menstrual cycle,
respectively. Subsequently, the levels of both gonadotropins
slightly decline in a gradual manner. The incidence of EOC markedly
increases at the age range in which the majority of women
experience menopause, a phenomenon consistent with the gonadotropin
theory. The mean age at onset of EOC is 57–59 years in the USA, and
half of the cases are aged >65 years at the time of diagnosis,
with 85–90% of the cases recorded in peri- or post-menopausal
women, whereas EOC occurs in only 10–15% of pre-menopausal women
(35,36). According to early information,
multiple pregnancies and the use of oral contraceptives are
established protective factors in terms of the incidence of EOC;
each additional pregnancy is associated with a 10–16% reduction in
the risk of developing EOC (37,38), and
the protective effect of oral contraceptives increases by 7% with
each year of use, reaching a 80% decrease among long-term users
(over a decade) (39). These data
regarding multiple pregnancies and the long-term use of oral
contraceptives support the gonadotropic theory, since these factors
are associated with low gonadotropin levels and the suppression of
incessant ovulation. Furthermore, late menopause and early menarche
are associated with a higher number of ovulations and exposure to
high gonadotropin levels, thereby increasing the risk of developing
EOC. The gonadotropin hypothesis is further supported by the
elevated gonadotropin levels found in the cysts and peritoneal
fluid of patients with EOC (40,41). In
particular, high concentrations of FSH in ascitic fluid have been
suggested to be inversely associated with survival (42), and EOC has been found to be associated
with significantly higher levels of FSH in serum and capsular fluid
compared with non-neoplastic ovarian lesions (43).
Another major hypothesis in the development of EOC,
namely incessant ovulation, was proposed in 1971 by Fathalla
(44). The causal mechanism through
which ovulation facilitates ovarian carcinogenesis remains unclear;
however, some possible theories have been suggested. During the
ovulatory process, the OSE is damaged and, subsequently, the repair
process occurs through extensive cell proliferation, increasing the
likelihood of spontaneous error and genome instability during DNA
replication (45). Later studies
under various disciplines proved the protective effect exerted by
oral contraceptives, provided further insight into the biological
mechanisms of ovulation, examined the possible pharmacological
production of luteinized unruptured follicles and presented
epidemiology-related evidence for this hypothesis (30,46). The
lifetime number of ovulatory cycles or years exhibits a positive
association with the risk of developing EOC, which supports the
hypothesis of incessant ovulation (47,48). The
levels of several inflammatory mediators (e.g., prostaglandins and
cytokines) are increased during ovulation and may enhance
mutagenesis (45). Moreover, the
‘androgen/progestin hypothesis’ is suggested to be a fundamental
mechanism underlying the development of EOC. According to this
hypothesis, the higher androgen levels observed in menopausal or
obese women, and in patients with PCOS, are associated with an
increased risk of developing EOC, whereas progesterone reduces the
risk of developing EOC (49,50). Although several in vitro and
animal studies have suggested a role for androgens in the
development of EOC (50–52), the epidemiological evidence is not
convincing.
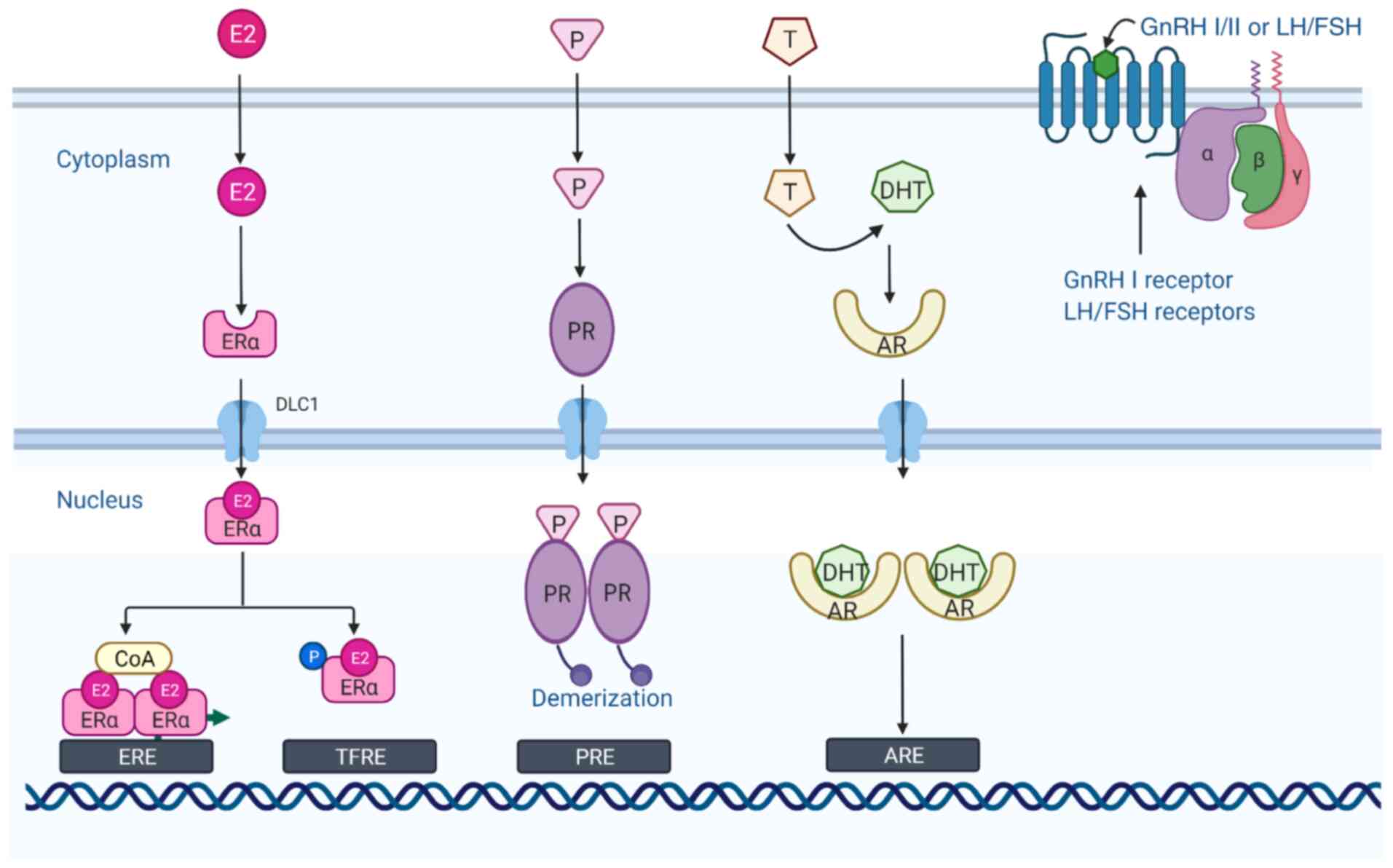
The two gonadotropins, FSH and LH, which share
similar chemical and structural characteristics, are synthesized in
the anterior pituitary, regulate gametogenesis and steroidogenesis
in the testes and the ovary in an endocrine manner, and they are
critical regulators of ovarian cell function (53). Specific receptors for gonadotropins
have been reported in EOC, as well as in normal OSE cells and in
fallopian tube cells (54,55). The LH receptor (LHR) and FSH receptor
(FSHR) are G-protein coupled seven-transmembrane domain receptors
(Fig. 3) that are highly expressed in
theca and granulosa cells, and play essential roles in reproductive
physiology when activated by their respective hormones (56). Over the past few years, FSHR
expression has been identified in the tumor vasculature of a number
of epithelial tumors, indicating a wider role for FSHR in
carcinogenesis (57,58). The expression status of various
gonadotropin receptors likely affects ovarian tumor progression
through various mechanisms. According to Lenhard et al
(59), patients with EOC expressing
higher levels of FSHR than LHR had a worse prognosis, whereas cases
expressing higher levels of LHR than FSHR exhibited an improved
overall survival. Two previous studies investigated the effects of
FSHR overexpression on immortalized OSE cells or benign ovarian
epithelial tumor cells and the advanced EOC cell line, OVCAR-3
(60,61). According to these studies, FSHR
overexpression may be associated with an increased oncogenic
potential and the increased proliferation of pre-neoplastic OSE
cells. In addition, EOC cells that expressed FSHR exhibited a
superior invasive ability (61).
These data indicate that FSHR may activate oncogenic pathways that
promote cell proliferation and invasive phenotypes, even in the
absence of FSH. Data from another study demonstrated that there was
a positive association between FSHR and aryl hydrocarbon receptor
levels, with their simultaneous expression observed in patients
with the least favorable EOC outcomes (62). Cheung et al (63) investigated the functional roles of
gonadotropin receptor expression in the progression of EOC and
demonstrated that the knockdown of FSHR and LHR expression was
associated with a more aggressive EOC phenotype and promoted
pro-metastatic behavior. The effects exerted by gonadotropins on
EOC have not yet been thoroughly investigated. Some studies have
demonstrated that gonadotropins (FSH and LH) promote cell migration
and invasion by inducing cyclooxygenase (COX)2 expression in EOC
cells, and that specific COX2 inhibitors significantly prevent
these effects (53,64). A recent study reported that
sphingosine kinase (SphK) served as a key mediator of FSH-induced
EOC cell proliferation, suggesting a novel strategy of using two
isozymes of SphK as drug targets for the treatment of EOC (65). By contrast, another study demonstrated
that in SKOV-3 cells, the overexpression of LHR upregulated the
expression of ERBB2, and the addition of LH further
increased ERBB2 expression, whereas it reduced cell
proliferation and motility, indicating that the upregulation of
ERBB2 independently failed to effectively decrease the
anti-proliferative effect exerted by LH on these cells (66). LH has also been implicated in the
development of EOC by inducing the secretion of VEGF via the
PI3K/AKT/mTOR pathway (67) and
upregulating survivin expression, leading to the inhibition of
apoptosis (68).
GnRH is a decapeptide hormone synthesized by the
hypothalamus and comprises 10 different amino acid residues. As a
key neuromodulator of the reproductive system, GnRH is secreted in
pulses and enters the anterior lobe of the pituitary gland via the
hypothalamic/pituitary portal circulation, acts on
gonadotropin-secreting cells in the anterior pituitary, and
regulates gamete formation and sex hormone production in the gonads
through the regulation of pituitary synthesis and the secretion of
FSH and LH. At present, there are three forms of GnRH found in most
vertebrates, namely GnRH-I, GnRH-II and GnRH-III, of which two
subtypes, GnRH-I and GnRH-II, are expressed in humans (69,70).
GnRH-I, also known as LH-releasing hormone, plays a role in
regulating ovarian proliferative activity. GnRH-I and its receptor
are expressed in ~80% of human ovarian epithelial tumors, EOC cell
lines and OSE cells (71), and their
activation by exogenous factors has been found to be associated
with the notable (time- and dose-dependent) and specific inhibition
of cell proliferation (72).
Currently, the protein expression of the GnRH-I receptor is
considered as a favorable prognostic factor in primary ovarian
tumors (73,74). GnRH-II has been identified in the
hypothalamus of chickens and has been reported to exhibit a
conserved structure in vertebrates. Similar to GnRH-I, GnRH-II is
expressed within a wide range of human ovarian cells, including OSE
cells, granulosa luteinizing cells, as well as in EOC cells
(75–77). It has been suggested that the
anti-proliferative effects of GnRH-II on tumor cells are mediated
via the GnRH-I receptor, as a functional GnRH-II receptor has not
yet been identified (78). Further
research on GnRH has demonstrated that, apart from the pituitary
gland and hypothalamus, GnRH and the relevant receptors are also
expressed in peripheral tissues (e.g., the ovaries, placenta,
endometrium and smooth muscle) and in certain malignant tumors
(e.g., OC, endometrial, breast and prostate cancers). GnRH must
bind to the high-affinity GnRH receptor (GnRH-R) to exert its
effects, i.e., the regulation of pituitary hormone release and
extra-pituitary products.
GnRH-as are produced by replacing or removing the
6th and 10th amino acids from naturally occurring GnRH, and their
biological properties are 50–100-fold stronger compared with those
of natural GnRH. The ultimate effects and efficacy of GnRH agonists
and antagonists are similar, differing in that antagonists have a
more rapid onset of action and do not have the initial surge in sex
hormone release. For example, the GnRH-a, degarelix, which is an
antagonist of gonadotropin receptors in the pituitary gland,
directly inhibits the synthesis and release of LH and FSH, and does
not have the initial surge characteristic of GnRH agonists
(79). In addition to protecting
ovarian function and improving the quality of life of patients,
GnRH-as may also act as antitumor agents through various
mechanisms. GnRH agonists and antagonists act by binding to GnRH-Rs
and are used in the treatmetn of steroid-dependent conditions,
including hormone-dependent tumors (80). The pharmacological application of GnRH
or the relevant synthetic analogs (agonists and antagonists) may
represent a valuable tool for stimulating or blocking gonadotropin
secretion, regulating the fertility of women suffering from
reproductive disorders and assisting with reproductive technologies
(81). GnRH-as can inhibit the
hypothalamic/pituitary axis and inhibit tumor growth by
downregulating FSH and LH secretion and by binding to GnRH-I and
GnRH-II receptors on the surface of cancer cells, thereby exerting
direct inhibitory effects on cancer cells through the regulation of
local autocrine and paracrine secretion (Table I). Following the administration of
GnRH agonists, FSH and LH levels are increased for a short period
of time; following long-term administration, the number of GnRH-Rs
decreases, inhibiting FSH and LH release and thus, the secretion of
estrogen and progesterone by the ovaries. GnRH antagonists exert
opposite effects. By directly binding to the GnRH-R, they inhibit
the release of FSH and LH without stimulating the pituitary gland,
thereby reducing the level of progesterone and estrogen in the
blood, and inhibiting sex hormone-dependent tumor development
(82). GnRH does not only play a key
role in mammalian reproductive regulation; however, it has also
been widely reported to exert significant antitumor
(anti-proliferative and anti-metastatic) effects by regulating the
activation of local GnRH-R (83,84). The
antitumor mechanisms of action of GnRH-I in OC are considered to
involve the desensitization or downregulation of GnRH-I receptors
within the pituitary gland, thereby reducing gonadotropin
secretion, which in turn leads to a decrease in gonadotropin
steroids acting as tumor growth-promoting factors (85,86). In
addition to regulating gonadotropin and relevant receptors in
vitro, GnRH-I and the relevant analogs have also been found to
exert a direct inhibitory effect on normal epithelial cells and EOC
cell lines in vitro and in vivo, leading to cell
cycle arrest based on the increase in the protein levels of p53 and
p21 (87).
After binding to G-protein α(i), activated GnRH-R
induces a phosphotyrosine phosphatase (PTP) that is subject to
EGF-triggered tyrosine autophosphorylation of the EGF receptor,
which leads to a reduction in cell proliferation and mitogenic
signal transduction (88). This
process prevents growth factor-driven mitogenic signaling, leading
to the EGFR-induced expression of MAPK (72) and c-fos (89), as well as in the inhibition of cell
proliferation (90). In addition,
GnRH-I has been reported to have the ability to trigger JunD-DNA
binding, leading to an increased cell number and a decreased DNA
synthesis at the G0/G1 phase of the cell
cycle, thereby reducing cell proliferation (91). The GnRH-I analog, leuprorelin, may
also induce the apoptosis of tumor cells through the Fas-ligand/Fas
mechanism. Data from two research teams suggested that elevated
levels of Fas ligand in tumors expressing GnRH-R were likely to
promote apoptotic cell death by targeting intratumoral Fas-positive
cells, thereby exerting growth inhibitory effects on GnRH-sensitive
tumors (92,93). However, the GnRH-I agonist,
triptorelin (D-Trp-6-LH-RH), has been reported to reduce apoptosis
induced by NF-κB activation triggered by the cytotoxic agent,
doxorubicin (94–96). GnRH-II has been reported to exert a
more prominent anti-proliferative effect on EOC cells compared with
equimolar concentrations of GnRH-I agonists. The inhibitory effect
exerted by GnRH-II on the proliferation of EOC cells in humans
notably exceeds that exerted by the highly active GnRH-I agonist,
triptorelin (97). GnRH-I and GnRH-II
agonists inhibit the mitogenic signaling of growth factor receptors
based on the activation of PTP, thus leading to decreased
proliferation of cancer cells (88).
Unlike GnRH-I and GnRH-II agonists, GnRH-II antagonists mainly
promote the apoptosis of EOC cells (97). Apoptosis triggered by GnRH-II
antagonists occurs through intrinsic apoptotic pathways: This is
mediated via the activity of the pro-apoptotic protein, Bax,
induced by MAPKs p38 and JNK, followed by caspase-3 activation, the
release cytoplasmic cytochrome c, and the probable loss of
mitochondrial membrane potential (97,98). The
aforementioned antitumor effects were confirmed in nude mice, as a
previous study demonstrated that antagonistic analogs of GnRH-II
significantly reduced the growth of mouse EOC xenograft tumors
without notable side-effects (97).
Since the existence of a GnRH-II receptor in humans is
controversial, there is a tendency to infer that the
anti-proliferative effects of both GnRH-I and GnRH-II are mediated
through the GnRH-I receptor (99,100). Of
note, the GnRH antagonist, AEZ-115, has exhibited substantial
antitumor activity in endometrial and EOC cells; however, this
antitumor effect is not mediated by tumor GnRH-Rs (90). Based on the GnRH-R tumor-specific
signaling in gynecological cancers, such as OC, and the particular
distribution of GnRH-R, gene therapy by employing GnRH-a as
inducers of therapeutically related gene transcription has been
successfully developed and tested in vitro, as well as in
athymic mice with EOC cell xenografts (101).
The inhibitory effect of gonadotropins and the
anti-proliferative effect exerted by GnRH-a binding to the receptor
form the rationale for their use in various hormone-dependent
tumors. In a limited number of clinical studies, GnRH-I agonists
have been assessed for their potential as third-line therapy in
women with recurrent (mostly platinum-resistant) and refractory OC,
and with the failure of at least one regimen of chemotherapy. A
summary of 18 clinical trials (102–119),
dating back to 1988, that have employed a wide range of GnRH-as
(triptorelin, goserelin and leuprolide) for the treatment of
patients with relapsed/platinum-resistant OC is presented in
Table II. Initially, Parmar et
al (102,120) reported on patients with advanced EOC
who relapsed following conventional treatment and were treated with
slow-release triptorelin microcapsules once per month. During
treatment, they observed stable disease (SD) in 5 patients and
evidence of clinical and/or radiological partial remission (PR) in
6 patients, where the tumor size decreased by >50%. This result
suggests that the clinical benefit of therapy with GnRH agonists
for recurrent OC is highly encouraging. Subsequently, based on the
inhibition of LH/FSH secretion by GnRH agonists, several clinical
trials using triptorelin and leuprolide acetate in advanced
recurrent EOC demonstrated associated objective remission and/or
disease stabilization in 10–50% of patients (103,112,113,117).
In addition, in 2014, 42 women suffering from platinum-refractory
or -resistant GnRH-R-positive EOC were evaluated for zoptarelin
efficacy and toxicity; of these 42 patients, 6 (14.3%) achieved a
partial response and 16 (38%) had SD (121). However, regardless of the
aforementioned encouraging results, the benefits of employing
GnRH-a as a treatment for advanced OC remain controversial.
A prospective, double-blind, randomized clinical
research was carried out to assess whether the addition of the GnRH
agonist, triptorelin, to the course of common platinum-based
chemotherapy prolongs the survival of patients with stage III or IV
EOC who have previously undergone surgery; however, that study
reported that the use of triptorelin to inhibit the release of
gonadotropins did not inhibit disease progression or prolong the
overall survival of the patients (107). In 2001, the European Organization
for Research and Treatment of Cancer completed the largest trial of
a GnRH agonist to date. In that study, 74 women with progressive OC
who were treated with platinum-based chemotherapy received
intramuscular injections of the GnRH agonist triptorelin. However,
according to the results presented, triptorelin treatment was only
moderately effective in patients already treated with
platinum-based chemotherapy (106).
In a recent phase II clinical trial, tamoxifen and goserelin were
combined to treat patients with advanced EOC who had developed
recurrence following chemotherapy. Although ‘endocrine responses’
were observed in ~50% of the patients, including SD (38.5%),
partial response (7.7%) and complete response (3.8%), there was no
consistent association observe4d between LH/FSH suppression and
tumor progression (111). Similarly,
in two other pilot studies in which patients with advanced OC were
treated independently with platinum-containing chemotherapy or
chemotherapy plus triptorelin, no significant differences were
observed between the two groups in terms of drug response, survival
and time to progression (122,123).
The ineffectiveness of GnRH agonists when combined with
chemotherapeutic agents may be due to their direct
anti-proliferative effect being neutralized by chemotherapy-related
anti-apoptotic activity, as confirmed by in vitro cell
experiments. By contrast, Rzepka-Górska et al (124) reported that the combination of
chemotherapy and the GnRH-a, goserelin, yielded favorable results
in advanced OC and, in that study, patients in the combination
group exhibited higher overall and 5-year survival rates compared
with the chemotherapy group; in addition, serum LH levels were
significantly lower in the combination group. Based on this result,
it was concluded that combination therapy for advanced OC was
effective and that GnRH-a may be an effective adjuvant therapy for
OC (124).
As GnRH agonists have failed to meet the
expectations in the clinical setting, the application of high doses
of GnRH antagonists warrants further in-depth investigations. Since
GnRH-I antagonists do not possess intrinsic gonadotropin activity,
the initial ‘burst’ phenomenon observed with agonist therapy can be
avoided, which renders antagonists more tolerable and blocks
gonadotropin secretion within a short period of time. High-dose
cetrorelix, a GnRH-I antagonist, was used in a phase II trial for
the treatment of OC or Müllerian tumors refractory to platinum
chemotherapy: All cases who relapsed following standard
chemotherapy received 10 mg cetrorelix daily. A total of 3 patients
(18%) experienced partial remission following cetrorelix treatment,
lasting for 9, 16 and 17 weeks, respectively, and 6 patients (35%)
had SD for 1–12 months (119). These
results suggest that GnRH antagonists may be considered for the
palliative treatment of patients with platinum-resistant OC.
The biological effects of androgens [testosterone
and dihydrotestosterone (DHT), as well as certain androgens
produced by the adrenal glands and ovaries] are typically mediated
via the androgen receptor (AR), a steroid hormone receptor that is
a member of the nuclear receptor superfamily (125–127).
Under basal conditions, AR exhibits inactivity and can bind towards
heat shock proteins and certain cellular partners. When activated
by androgens, it induces a cascade of events (e.g., ligand binding,
dissociation from heat shock protein, phosphorylation and
dimerization) and is involved in nuclear translocation. Specific to
the nucleus, AR binds to specific DNA sequences known as androgen
response elements in the nucleus and binds to various AR cofactors
to form complexes. The AR complex does not only alter the
expression of genes involved in multiple physiological and
pathological functions; however, under certain pathological
conditions, AR may also become activated in the absence of
androgens. For example, the activation of AR by IL-6 in human
prostate carcinoma cells does not require the presence of androgens
(128). Androgen/AR signals have
been reported to promote metastasis and tumorigenesis in various
malignancies, including prostate carcinoma, for which androgen
deprivation therapy remains the primary treatment option (129), but also in other types of cancer,
such as breast (130) and bladder
cancer (131).
In addition, there is accumulating evidence to
indicate that AR and related signaling pathways are involved in the
development and progression of OC. The expression of AR in OC was
first demonstrated by Hamilton et al (132) with the use of ligand binding tests.
According to in-depth research, AR expression has been detected in
~90% of EOCs using biochemical receptor tests (133) and in 43.5–86% of EOCs using
immunohistochemistry (134,135). Some investigators have attempted to
determine whether AR is differentially expressed in various
histological subtypes of OC. According to Cardillo et al
(136) the expression of AR varies
widely across various histological subtypes of OC. The
aforementioned result was verified by Elattar et al
(137), who reported that AR was
expressed in 43.7% of EOC samples, with the highest expression
levels observed in serous carcinomas (47.5%). Similarly, according
to de Toledo et al (138),
AR-positive expression tended to exhibit a higher prevalence in
serous tumors compared with that in non-serous tumors. The
association between AR expression and other clinicopathological
characteristics of OC, such as tumor stage and grade, was also
assessed. According to Jönsson et al (139), a negative AR expression was
associated with high-grade cancer. By contrast, four other studies
on malignant ovarian tumors reported that AR expression exhibited
no association with tumor FIGO stage (135,136,138,140).
Furthermore, according to a previous study, matched primary and
metastatic serous EOC samples exhibited non-significant differences
in the levels of nuclear AR (140).
In summary, AR expression is more frequently detected in serous
compared with non-serous ovarian tumors; however, whether AR
expression levels are crucial for OC progression requires further
in-depth investigation.
Currently, a number of factors are known to be
involved in regulating the transcriptional activity of AR,
including not only various androgenic and androgen-derived
compounds, but also the expression levels and variants of AR per
se. Edmondson et al (141)
first demonstrated that OSE cells were androgen-responsive, and the
addition of androgen to eight primary human OSE cell lines cultured
in vitro was able to promote the proliferation of these
cells and reduce cell apoptosis. In other in vitro
experiments, DHT and testosterone were shown to notably stimulate
the proliferation of ovarian tumor and normal ovarian cell
lines/cultures (142). Co-treating
these cells with the anti-androgen 4-hydroxyflutamide also
demonstrated that this androgen-stimulating effect was reversible
(142). In another study, DHT
stimulation was reported to increase cell division in six of 11
primary cultured EOC cell lines. The proportion of cells in the S
phase also increased from 4.4% in serum-free medium to 8.3% in 100
nM DHT-stimulated cells (137). In
that study, it was also found that AR nuclear expression was
positively associated with an increased fraction in the S phase in
response to androgen stimulation, while the expression of AR in the
immunohistochemical nuclear and cytoplasm was significantly
decreased following chemotherapy (137). This may explain the low response
rates observed in clinical trials among patients who have received
aggressive prior chemotherapy. Other androgens, such as
androstenedione (143) and
methylosome (144), have also been
shown to increase the proliferation and induce cell motility and
invasion in EOC lines. Animal models have also been used to
evaluate the effect of androgens on EOC progression. According to
Silva et al (145),
testosterone therapy in guinea pigs promoted ovarian epithelial
cell proliferation, leading to the formation of papillomas on the
ovarian surface, small adenomas in the ovarian parenchyma and
benign cysts. Similarly, in a mouse xenotransplantation model, DHT
treatment significantly induced tumor growth (146), and the AR inhibitor enzalutamide was
shown to antagonize the effects of DHT (147). These observations in animal models
and cell lines suggest that androgen/AR signaling can critically
stimulate EOC growth and promote EOC progression. The activation of
AR signaling is likely to be associated with the sensitivity of
malignant tumors to conventional chemotherapeutic agents, as
suggested by research on prostate cancer (148) and bladder urothelial carcinoma
(125). AR expression has also been
found to be considerably upregulated in the paclitaxel-resistant
SKOV3 subline (149).
Correspondingly, as previously demonstrated, in
paclitaxel-resistant SKOV3 cells, the silencing of AR via RNA
interference increased cell sensitivity to chemotherapy and
enhanced paclitaxel-mediated apoptosis (149,150).
Conversely, the activation of AR with the agonist DHT has been
shown to upregulate the expression of paclitaxel resistance target
genes (150).
As aforementioned, preclinical evidence suggests
that androgen/AR signaling is associated with the progression of
OC, and targeting AR is a promising treatment strategy. However, to
date, only a small number of clinical trials have been conducted to
evaluate the efficacy of anti-androgen therapy in OC, with or
without GnRH agonists (Table III)
(151–155). Notably, androgen deprivation therapy
(e.g., bicalutamide and flutamide), with or without GnRH agonists,
is extensively applied in patients with prostate cancer without
severe side-effects, and functions by inhibiting pituitary LH
release and ultimately reducing androgen production (49,156). In
a previous study, Tumolo et al (151) assessed the use of flutamide, a
non-steroidal AR antagonist, in patients with EOC who relapsed and
progressed following platinum-based chemotherapy. First, 68
eligible patients were included, 32 of whom had completed oral
flutamide therapy (750 mg/day) for at least 2 months. Of the 32
patients in the study, only 2 responded to the treatment and the
disease was stable over a mean period of 24 weeks in 9 patients.
Based on the results of that trial, it was concluded that flutamide
was ineffective as a treatment for patients who had previously
received extensive chemotherapy, and that it is associated with
certain side-effects, such as nausea and vomiting. In another phase
II study, Vassilomanolakis et al (152) assessed the response of patients with
stage III or IV EOC to flutamide (300 mg/day) following
chemotherapy. The outcome of that clinical trial was not
satisfactory, with only 1 (4.3%) of the 23 evaluated patients
exhibiting a partial response and 2 patients (8.7%) having SD, with
the remaining 20 patients experiencing disease progression within 3
months. In addition, bicalutamide, a non-steroidal drug exhibiting
anti-androgen characteristics, was tested in patients with grade II
or higher EOC in remission (154).
However, the results of that study demonstrated that the oral
bicalutamide administration (30 mg/day) combined with subcutaneous
goserelin injection (3.6 mg/4 weeks) did not prolong
progression-free survival (PFS) in 35 patients. In another later
study, Gruessner et al (153)
investigated the effects of preoperative oral flutamide therapy
(125 mg/day for 6 weeks) on biomarker levels in blood and tissue
samples from 12 patients at a high risk of developing OC and 47
controls, 47% of whom had BRCA mutations. The results
demonstrated that flutamide significantly reduced the expression of
colony stimulating factor (CSF)-1 and Erb-B2 receptor tyrosine
kinase 4 (ERBB4) in the ovarian stroma and was well-tolerated by
the patients. Enzalutamide is an oral AR signaling inhibitor
capable of blocking the binding of androgens to AR and preventing
AR nuclear translocation, DNA binding and coactivator recruitment
(157). In 2012, enzalutamide was
approved by the US Food and Drug Administration for the treatment
of castration-resistant prostate carcinoma. A phase II study is
currently underway to evaluate enzalutamide in women with
AR-positive OC (NCT 01974765) (155).
Evidence from limited clinical trials indicates that
only a small proportion of patients with EOC benefit from androgen
deprivation therapy consisting of non-steroidal anti-androgen drugs
with generally tolerable side-effects. Therefore, further research
is required to determine the actual benefit of androgen deprivation
therapy and the optimal regimen, and to select appropriate drug
candidates by evaluating AR expression, AR polymorphism/fragment
variation and downstream targets of AR activity.
Estrogens belong to the family of steroidal organic
compounds and are mainly synthesized from androgens through the
aromatase activity of granulosa cells in the ovaries. In fact, the
term ‘estrogen’ refers to a group of female hormones, the most
common of which are estrone, estradiol and estriol. Serum estrogen
concentrations increase from the metaphase, consistent with the
formation of dominant follicles. In the follicular fluid chambers,
estrogen levels are 1,000-fold higher compared with those in
peripheral blood. All these estrogens can bind to nuclear and
membrane ERs with varying affinities and response strengths
(158). Estrogen signal transduction
is mediated by several estrogen isomers, the most important of
which are the classical ERs, namely ERα and ERβ. Estrogen, as a
steroid hormone, can cross the plasma membrane and interact with
ERα and ERβ in the cells, acting directly by binding to DNA
sequences. ER complexes bind directly or indirectly to DNA; thus,
estrogen-mediated signaling events can be genomic as well as
non-genomic. Genomic effects involve the migration of ER complexes
to the nucleus and direct interaction with chromatin on specific
DNA sequences, known as estrogen response elements (158,159).
On the other hand, non-genomic effects involve the indirect
regulation of gene expression through a variety of intracellular
signaling events. The G-protein coupled receptor (GPR)30, also
known as G-protein coupled ER1, has been extensively investigated
as a membrane ER that activates non-genomic estrogen signaling
pathways in a variety of tissues (160).
ERα is closely associated with endocrine sensitivity
in breast cancer and is a significant mediator of the estrogen
response in OC (161,162). Multiple studies have investigated
the expression of ERα in EOC (163–166);
however, the largest study was conducted by the Ovarian Tumor
Tissue Analysis consortium in 2013 (25). That study investigated 2,933 women and
found that HGSOC, LGSOC and endometrioid OC exhibited the highest
levels of ER positivity at 81, 88 and 77%, respectively;
furthermore, these tumor types have exhibited responses to
endocrine therapy (tamoxifen and aromatase inhibitors) in multiple
clinical studies. By contrast, ERα expression was detected in only
20 and 21% of clear cells in mucinous and clear cell carcinomas,
respectively. ERβ is known to be expressed as five different
isoforms, namely ERβ1-5; however, only the full-length form of ERβ1
is able to bind to agonist or antagonist ligands and has a more
complex function (167). Rutherford
et al (168) found higher
levels of ERβ expression in normal OSE cells, lower levels of ERβ
expression in primary EOC, and the absence of ERβ expression in
metastatic tumors. These results indicate that there may be
fundamental differences in ERβ expression from the normal ovarian
tissue to primary OC to metastatic tumor (168).
ER is highly expressed in several EOCs and is a
potential target for endocrine therapy. Studies using in
vitro experiments and in vivo animal models of EOC
support the hypothesis that ER expression levels are a crucial
determinant of the response to treatment with selective ER
modulators. In addition, epidemiological studies have demonstrated
that the long-term use of estrogen-only therapy increases the risk
of developing OC in women, supporting the hypothesis that estrogen
signaling contributes to the etiology of the disease.
The previous evaluation of the role of ERβ
suggested that this receptor isoform functions as a tumor
suppressor in EOC by reducing tumor growth and deterring
metastasis. These actions rely on the fact that ERβ inhibits ERα
expression and decreases estradiol-induced cell proliferation
(174,175). In addition, the restoration of ERβ
expression in EOC cells has been shown to lead to a decrease in
proliferation and invasion, while apoptosis is enhanced (176). Similarly, the overexpression of ERβ1
induced by transfection with an overexpression vector in the SKOV3
EOC cell line (177) or the ES-2
cell line (178) has been shown to
result in reduced proliferation and motility, and increased
apoptosis. The gene ERβ, is localized on chromosome 14q, and this
region is frequently deleted in OC (176). Consistent with its tumor-suppressive
effect, the high expression of ERβ1 in the cytoplasm of EOC cells
has been found to be strongly associated with a longer disease-free
and overall survival of patients (179). In contrast to the inhibitory role of
ERβ1, both ERβ2 and ERβ5 have been associated with pro-migratory
and invasive activities. ERβ2 overexpression has been shown to
increase cell migration and invasion, but not the proliferation of
EOC cells (179). The mitochondrial
activity of cytoplasmic ERβ2 signaling in serous carcinomas has
been found to be associated with BAD binding, leading to reduced
apoptosis, thereby serving a pro-survival role (174). ERβ5 also plays an important role in
tumor progression and has been shown to promote cell proliferation,
and enhance cell migration and invasion. High levels of ERβ5
expression have been found in the nuclei of tumor cells from
patients with clinically advanced EOC (particularly serous and
clear cell carcinomas), which has also been associated with poor
patient survival, although some researchers have reported that
cytoplasmic ERβ5 expression may be associated with more favorable
patient outcomes (179). The cell
cycle is affected by the presence of ERβ by reducing the proportion
of cells in the S phase, increasing the number of cells in the
G2/M phase, and inducing apoptosis (167,175). A
suppressive effect of ERβ on cyclin D1 expression has been
reported, and its expression appears to be enhanced in response to
estrogen antagonists (180).
Indirect evidence that ERβ is implicated in ovarian carcinogenesis
is the fact that its expression has been found to be significantly
associated with survival in human and animal models (181,182). A
recent study revealed that ERβ-positive nuclear staining was
associated with a decreased PFS (183). On the other hand, patients with
hormone receptor-negative OC have been shown to have a favorable
prognosis (184).
Several clinical trials have been conducted using
selective ER modulators (mainly tamoxifen) and aromatase inhibitors
(letrozole and anastrozole), which have exhibited activity against
OC (low response rate, yet apparently SD) and these are discussed
below (Tables IV and V) (108,185–213).
Tamoxifen was the first selective ER modulator to
be evaluated in clinical trials for OC and is still in use today;
it is considered to function as a selective ER modulator that
competes with estrogen for binding to the ERα and, thus, functions
as an ER antagonist. The majority of studies were designed as
single-arm studies to assess whether tamoxifen can induce a
response in EOC; however, they did not evaluate the effects of
tamoxifen on symptom control, the quality of life or the survival
time of patients. It has been reported that the overall mean
response rate of this treatment is 10–15%, and the disease
stability rate is 30–40% (214–216).
In clinical trials in which tamoxifen was applied, the majority of
patients had undergone major pre-treatment procedures, such as
chemotherapy, and several studies did not differentiate between
ER-positive patients. Perez-Gracia and Carrasco (215) reported an overall response rate of
26% and a complete response rate of 9% in the analysis of trials
using tamoxifen in at least 50% of cases not having received
multiple prior treatments, compared with a clinical study treating
patients with severe disease with an effectiveness rate of only 4%
(215). For 19 years, non-steroidal
aromatase inhibitors have been studied for their antitumor
activity, which appears to be comparable to that of tamoxifen, by
inhibiting the conversion of androgens to estrogens, thereby
reducing circulating estrogen levels. Paleari et al
(216) reviewed 53 endocrine therapy
trials including a total of 2,490 patients in their meta-analysis.
The clinical benefit rate (CBR; percentage of total patients
exhibiting complete response, partial response or SD in all
endocrine therapy evaluations) reached 41%; the CBR for tamoxifen
was 43% (23 trials) and the CBR for aromatase inhibitors was 39%
(10 trials), demonstrating that the effectiveness of the two types
of drugs was comparable for patients with advanced EOC (216). According to a recently conducted
trial (PARAGON), the application of anastrozole in a phase II study
on asymptomatic cases suffering from ER- and PR-positive recurrent
EOC with CA125 progression was assessed (217). The response rate reached 4%, and the
CBR reached 35%, which was disappointing, given that these cases
only had limited disease and had previously received single
chemotherapy treatment.
Two recent reports described studies evaluating the
use of endocrine therapy at their respective centers and provided
insight into settings outside of HGSOC trials. An analysis of 97
patients treated at the Royal Marsden Hospital (London, UK)
investigated the use of tamoxifen and letrozole for advanced EOC
(91% HGSOC) (198). More than a
quarter of the patients had previously received five or more types
of chemotherapy, and half of these had an unknown ER status,
whereas the CBR reached 60% (tamoxifen, 65%; and letrozole, 56%).
Cases responding to letrozole had a significantly longer response
time (198). A 25-year analysis of
269 cases with HGSOC in Edinburgh revealed a comparable overall
response rate for letrozole and tamoxifen (8 and 11%, respectively)
and CBR (41 and 33%, respectively). Cases with a high ER expression
and a longer treatment-free interval were most likely to benefit
from these treatments (218). The
conclusions of these two analyses are consistent with the findings
of clinical trials, demonstrating that tamoxifen or letrozole
constitute reasonable treatment options for patients with
ER-positive HGSOC, with a comparable overall response rate, CBR and
disease stability. Endocrine therapy may be a promising alternative
therapy for LGSOC, which is less sensitive to chemotherapy.
Gershenson et al (219)
identified a 9% response rate and 61% disease stabilization rate in
a retrospective analysis of 64 LGSOC cases who had received a total
of 89 hormonal regimens. The PFS for cases receiving hormonal
maintenance therapy (primarily letrozole or tamoxifen) was 65
months, compared with 26 months for cases under observation only
(P<0.001) (219). That study was
followed-up by Fader et al (220), who also retrospectively explored the
use of adjuvant hormonal therapy following surgery without
chemotherapy, with promising results. A stage III trial initiated
in 2019 (NRG-GY019) is also currently ongoing. Specifically, the
comparison is between paclitaxel/carboplatin + letrozole vs.
letrozole independently for stage II–IV LGSOC (221). To date, there is limited information
available on the sensitivity of endometrioid OC to hormone therapy.
In the Royal Marsden High Grade Ovarian Cancer Study, a total of 5
patients with high-grade endometrioid OC were treated with
endocrine therapy and, encouragingly, 3 patients exhibited partial
remission, while the remaining 2 patients had SD (198). In a study on letrozole reported by
Bowman et al (204), 4/11
endometrioid OC cases had SD compared with 4/43 serous carcinoma
cases. Moreover, estrogen-targeting therapies have exhibited
considerable promise in the treatment of GCTs. In a review article
summarizing aromatase inhibitors as single agents, 25 cases with
known outcomes were described. The response rate to aromatase
inhibitors in these patients was 48% (12/25) and the clinical
benefit rate was 76% (19/25) (222).
According to a previous analysis, 9 out of 9 patients responded to
aromatase inhibitors. Although the number of patients in those
studies was limited, they supported aromatase inhibitors as a
potential alternative to chemotherapy (223).
Progesterone (P4) is a steroid hormone mainly
generated from the corpus luteum in the ovaries during the luteal
phase or the second half of the menstrual cycle. The adrenal glands
and the placenta during pregnancy also produce small amounts of
progesterone (224,225). Thus, from the beginning of
menstruation to the end of menopause, there is a monthly cycle of
hormone exposure, and the regulation of the growth and
differentiation of the female reproductive tract system and breast
tissue (225). Pregnancy interrupts
this cyclic process, as high progesterone levels are continuously
required for fetal growth, the maintenance of uterine/placental
integrity and breast development during lactation (226). Progesterone-dependent effects and
related biological actions in different tissues and tumors are
mediated by two PR subtypes, namely PR-A and PR-B. PR-B is the
major subtype required for mammary gland development and expansion,
while PR-A is required for normal uterine development and
reproductive activity (227). PR is
a polypeptide expressed in the cytoplasm, and once bound to
progesterone, it translates into the nucleus and regulates the
expression of a specific set of genes. Limited studies have
reported information on the relative expression of PR in tissue
samples from human OC subtypes. Diep et al (228) assessed the percentage of PR-positive
tumors among primary histological subtypes of OSE-derived OC in 504
tissue samples. According to their results, 35% of ovarian tumors
were PR-positive, with the maximal total PR expression found in the
endometrioid (67%) and serous (35%; LGSOC, 64%) subtypes (228). Subsequently, the International
Consortium for Ovarian Tissue Analysis analyzed the proportion of
tumors positive for PR staining in ~3,000 invasive epithelial
ovarian tumors and reported the highest expression in endometrioid
(67.4%) and LGSOC (57.4%); intermediate expression in HGSOC
(31.1%); and the lowest expression in the mucinous (16.4%) and
clear cell subtypes (8.0%) (25).
Additionally, that study investigated the prognostic implications
of PR expression in ovarian tumors highly expressing PR (staining
of 50% tumor cell nuclei). PR expression was associated with a
significantly favorable survival for HGSOC, and significantly
improved disease-specific survival independent of patient age,
tumor grade, site and stage for endometrioid OC (25). To the best of our knowledge, only
three studies to date have reported the differential expression of
PR isoforms in ovarian tumors (229–231).
These studies reported the dominance of PR-B expression in all
subtypes of ovarian tumors; furthermore, PR-B is usually expressed
in serous subtypes, whereas PR-A is weakly expressed in mucinous
and serous OC, and its expression is low or absent in tumors
compared with that of PR-B in comparison with normal and malignant
ovarian tissues.
The first indication that progesterone may be
involved in the regulation of OC is derived from the observation
that the use of progestin-containing oral contraceptives appears to
prevent the occurrence of OC (232).
Moreover, the incidence of OC is increased among women with
progesterone deficiency, while the high levels of serum
progesterone during pregnancy are associated with a lower risk of
developing OC (233,234). The protective effect of progesterone
against OC development is lost in post-menopausal women, as the
serum levels of progesterone are negligible following menopause
(235). In addition, women with a
history of twin pregnancies exhibit a lower risk of developing OC,
which may be associated with the higher serum levels of
progesterone in the maternal circulation in twin compared with
singleton pregnancies (236,237). The molecular mechanisms through
which progesterone exerts its protective effects against OC are not
yet well understood. Both proliferative and inhibitory effects of
progesterone on OC cells have been reported in cell line assays,
which may be attributed, at least in part, to the differential
effects of progesterone on the two receptors, PR-A and PR-B, and
their relative expression in target cells. Several independent
in vitro studies have demonstrated the anti-proliferative
effects of progesterone at higher concentrations in EOC cells,
primarily through the induction of apoptosis (238–240).
The activation of progesterone signaling can inhibit ovulation,
antagonize the growth-promoting effects of estrogen, and regulate
EOC cell proliferation and apoptosis (241). High concentrations of estrogen
combined with progesterone may induce the apoptosis of EOC cells by
promoting the expression of let-7a and microRNA-34b, and decreasing
the expression of Bcl-2 (241). In
another study, however, it was demonstrated that the combination of
tamoxifen and progesterone treatment induced apoptosis similar to
that induced by treatment with progesterone independently, with no
additional anticancer effect on EOC cells (242).
The precursor form of the steroid hormone
pregnenolone reduces ovarian cell proliferation and viability by
downregulating the expression of PR (243). Recently, Pedernera et al
(244) observed that progesterone
treatment significantly reduced cell survival in endometrioid OC.
That study demonstrated the protective effects of progesterone
against OC and indicated that the presence of PRs may suppress the
progression of endometrioid OC. Of note, as demonstrated in a
previous study, the progesterone metabolite, allopregnanolone,
increased the proliferation of and Ki67 expression in EOC IGROV-1
cells, whereas the expression of cleaved caspase-3 was unaltered
(239). Furthermore, progesterone
and allopregnanolone increased the migratory capacity of IGROV-1
cells in a concentration-dependent manner (239). Moreover, it has been demonstrated
that PR membrane component-1 (PGRMC1) plays an important role in
promoting EOC cell viability by binding to progesterone, and that
the attenuation of PGRMC1 function by small interfering RNA
increases the sensitivity of EOC cells to cisplatin (237,245).
The prolonged presence of the anti-progestin, mifepristone, has
also been shown to block the repopulation of EOC cells that escaped
platinum or platinum/paclitaxel treatment, providing evidence of
the long-term use of anti-progestins as anti-repopulation therapy
for cells that escape other effective chemotherapies (246–248).
However, as progesterone/PR signaling is complex, further research
on the intricate details of its role in tumor progression is
required before it can be considered as a potential clinical target
in OC treatment.
Despite the high expression levels of endocrine
response receptors, hormonal therapy plays merely a secondary role
in EOC treatment. Epidemiological evidence coupled with the
findings of in vivo and in vitro studies suggests the
modulation of PR levels or activity as a form of endocrine therapy
for EOC (Table VI) (189,249–262).
In a phase II clinical trial conducted in 2000, 34 patients with
recurrent EOC who no longer responded to cisplatin/paclitaxel
chemotherapy were administered 200 mg oral mifepristone daily for
28 days. Of these patients, 9 responded to mifepristone, exhibiting
a decrease in tumor size by at least 50% or a 50% decrease in the
levels of CA125, which is used to assess disease recurrence
(261). However, the results from
another phase II clinical trial including 24 patients with advanced
EOC who received standard chemotherapy and relapsed within 6
months, demonstrated that only 1 patient had an objective response
to a 28-day regimen of 200 mg mifepristone administered daily
(262). This clinical evidence
appears to be less convincing in terms of the number of patients
included, the lack of biomarkers to predict the response, and the
fact that the studies did not report hormone receptor expression
levels in OC tissue. Niwa et al (263) investigated the effects of the
combination of medroxyprogesterone acetate (MPA) with primary
adjuvant chemotherapy for advanced EOC in 2008. Both PFS and
overall survival were significantly longer in the cases treated
with the combination of MPA and platinum-based chemotherapy
compared with those in the control group. Zheng et al
(264) reviewed the value of PR
ligands in OC treatment by examining 13 clinically related trials
that included 432 cases with recurrent or refractory OC treated
with megestrol acetate or MPA. A total of 10 cases (2.3%) had a
complete response, 21 (4.9%) had a partial response and 47 (10.9%)
had SD. The authors of that study concluded that the effectiveness
achieved by progesterone preparations in recurrent EOC was not
confirmed by existing research. In addition, the anti-progestin,
mifepristone, is employed in the treatment of platinum-resistant
EOC. PR can be induced by estrogen (and by tamoxifen, if serving as
an agonist), and several trials have investigated the effectiveness
achieved by combination hormonal therapy that targets the mentioned
crosstalk (206,265,266).
One noticeable clinically related response was observed in a trial
of 65 patients with refractory EOC who were administered
medroxyprogesterone and sequential ethinyl estradiol, achieving a
response rate of 14% and SD in 20% of the patients (267). Given the recent preclinical data and
basic research findings, it is expected that drugs targeting
different PR subtypes (PR-A vs. PR-B), or drugs targeting other
progesterone-binding receptors, will be developed and used in the
near future.
Findings by epidemiology researches on menopausal
hormone replacement therapy (HRT) and the risk of developing OC are
inconsistent. HRT exhibits an association with an increased risk of
developing OC. Nevertheless, some researchers have reported that
they found no such association, whereas other researchers have
reported a positive association within the single histology
subdivided type. As concluded from previous a review and
meta-analysis of information (1966 and 2006), examining the
application of post-menopausal hormone therapy (HT), an upregulated
risk of developing OC by 30% was observed in contrast to no HT; it
was thus indicated that the risk of developing OC with estrogen
therapy (ET) was independently higher than that related to estrogen
plus progestin therapy (EPT) (268).
Nevertheless, the Million Women Study examined 2,273 incident cases
of OC and with 948,576 females, reported an increased risk of
developing OC with hormone replacement therapy, whereas an
insignificant differential influence exerted by ET vs. EPT was
achieved (269); similarly, another
nationwide study found no evidence of risk associated with vaginal
ET or transdermal vs. oral EPT (270). Furthermore, in other studies, cyclic
treating process were reported to increase the risk of OC, and no
statistics-related significant differences were found from the
cyclic combined regimen (28,271). A national prospective cohort study
covering overall Danish females aged 50–79 years from 1995–2005
suggested an increased risk of developing OC with EPT and estrogen
therapy, exhibiting an insignificant effect exerted by various
doses, the length of use, routes of administration, progestin types
and regimens (22). Based on the
investigation of the identical cohort of cases recruited, as was
reported, the risk of developing OC was altered in accordance with
the histology of the tumor: In contrast to never users, females
undergoing unopposed oral estrogen therapy exhibited an increased
risk of developing ovarian serous and endometrial-like tumors,
whereas they exhibited a decreased risk of developing mucinous
tumors (272). According to a
previous study analyzing the risk of developing OC in
post-menopausal females treated with estradiol-progestin, the
elevated risk of developing OC associated with EPT use over a
period of 5 years was only observed in serous and mixed cancers,
while the risk of mucinous cancers was reduced (270). A recently conducted meta-analysis
reported by Liu et al (273)
concluded that EPT during menopause may increase the risk of
developing OC, particularly in terms of endometrioid and serous
tumors. In a collaborative re-analysis of 52 epidemiological
studies in the OC Epidemiology Research Collaborative (274), the relative risk (RR) of HRT for a
duration of ≥5 years was more notable than that observed with past
users who had terminated the therapy <5 years earlier. It was
thus concluded that this increased risk was likely to be primarily
or entirely involving a cause; if causal, female users who had been
on hormone therapy for 5 years from the age of ~50 years would have
one additional OC for every 1,000 users and, if their prognosis was
characteristic, one additional OC-related death for every 1,700
users.
The effects of post-operative HRT on
non-progression and total survival within cases of EOC is also
controversial. The present review article can support the
assumption that, i.e., HRT following surgery cannot adversely
affect the non-progression and total survival of EOC cases. A
retrospective study that included 77 cases supported the assumption
that HRT following surgery failed to not significantly influence
non-progression and total survival in cases suffering from EOC.
Likewise, various HRTs (an estrogen-tibolone integration, tibolone
independently, or estrogen independently) failed to notably affect
the prognosis of patients with EOC (275). This finding was validated in another
meta-analysis that covered 419 EOC survivors using HRT and 1,029
non-users (276). In a recent
meta-analysis that included 350 cases suffering from EOC, the
authors concluded that HRT may slightly improve the total survival
of cases who had undergone surgical treatment, and there may be a
minimal or no effect of HRT use on non-progression survival
(24).
In summary, OC is a malignancy depending on
hormones in which steroid hormones and the relevant receptor
critically affect its advancement. Although hormone therapy is
effective in cases with advanced or recurrent OC, and has a low
profile of toxicity, studies evaluating the therapeutic value of
hormone therapy in OC have not been conclusive due to small sample
sizes, different pathological types of OC, different hormone
receptor-expressing states within OC cells, and the lack of
molecular markers. In addition, the lack of corresponding NCT
numbers for a number of clinical trials also does not facilitate
the readers' understanding of the trials and may affect the
authenticity of the trial results. Therefore, multicenter,
prospective, randomized trial studies are required to confirm its
efficacy. In addition, further studies are warranted to determine
whether the combination of hormones and chemotherapeutic drugs can
improve the effectiveness of chemotherapy, and whether this can be
used as therapy for OC, as well as to identify the molecular
markers of hormone therapy. In cases suffering from ER-positive
breast carcinoma, hormone therapy has been used as part of systemic
cancer treatment, and in endometrial cancer, hormone therapy
functions as a second-line treatment option in terms of cancer
metastasis. The ovary as an endocrine organ and the application of
hormone therapy in OC patients, is worthy of further study. In
future clinical treatment, hormone receptor expression should be
routinely tested, and the expression should be standardized and
classified into treatment considerations. In addition, different
treatment options should be adopted according to the various
pathological types of OC.
Not applicable.
The present study was supported by the National
Major Scientific and Technological Special Project for the
‘Significant New Drugs Development’ (grant no. 2018ZX09201018-013)
and the National Natural Science Foundation of China (grant no.
81821002).
Not applicable.
HL and YL wrote the initial manuscript and confirm
the authenticity of all the raw data. YW created the figures and
contributed to writing the material and providing new ideas. XZ and
XQ revised the manuscript and approved the final version. All
authors (HL, YL, YW, XZ, XQ) read and approved the final
manuscript.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2019. CA Cancer J Clin. 69:7–34. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality Worldwide for 36 Cancers in
185 Countries. CA Cancer J Clin. 68:394–424. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Reid BM, Permuth JB and Sellers TA:
Epidemiology of ovarian cancer: A review. Cancer Biol Med. 14:9–32.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Chen VW, Ruiz B, Killeen JL, Coté TR, Wu
XC and Correa CN: Pathology and classification of ovarian tumors.
Cancer. 97 (Suppl 10):S2631–S2642. 2003. View Article : Google Scholar
|
|
5
|
Prat J: New insights into ovarian cancer
pathology. Ann Oncol. 23 (Suppl 10):x111–x117. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Peres LC, Cushing-Haugen KL, Anglesio M,
Wicklund K, Bentley R, Berchuck A, Kelemen LE, Nazeran TM, Gilks
CB, Harris HR, et al: Histotype classification of ovarian
carcinoma: A comparison of approaches. Gynecol Oncol. 151:53–60.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Duska LR and Kohn EC: The new
classifications of ovarian, fallopian tube, and primary peritoneal
cancer and their clinical implications. Ann Oncol. 28 (Suppl
8):viii8–viii12. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Meinhold-Heerlein I, Fotopoulou C, Harter
P, Kurzeder C, Mustea A, Wimberger P, Hauptmann S and Sehouli J:
The new WHO classification of ovarian, fallopian tube, and primary
peritoneal cancer and its clinical implications. Arch Gynecol
Obstet. 293:695–700. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Bowtell DD, Böhm S, Ahmed AA, Aspuria PJ,
Bast RC Jr, Beral V, Berek JS, Birrer MJ, Blagden S, Bookman MA, et
al: Rethinking ovarian cancer II: Reducing mortality from
high-grade serous ovarian cancer. Nat Rev Cancer. 15:668–679. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Patch AM, Christie EL, Etemadmoghadam D,
Garsed DW, George J, Fereday S, Nones K, Cowin P, Alsop K, Bailey
PJ, et al: Whole-genome characterization of chemoresistant ovarian
cancer. Nature. 521:489–494. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Hunter SM, Anglesio MS, Ryland GL, Sharma
R, Chiew YE, Rowley SM, Doyle MA, Li J, Gilks CB, Moss P, et al:
Molecular profiling of low grade serous ovarian tumours identifies
novel candidate driver genes. Oncotarget. 6:37663–37677. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Wilbur MA, Shih IM, Segars JH and Fader
AN: Cancer implications for patients with endometriosis. Semin
Reprod Med. 35:110–116. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Munksgaard PS and Blaakaer J: The
association between endometriosis and gynecological cancers and
breast cancer: A review of epidemiological data. Gynecol Oncol.
123:157–163. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wiegand KC, Shah SP, Al-Agha OM, Zhao Y,
Tse K, Zeng T, Senz J, McConechy MK, Anglesio MS, Kalloger SE, et
al: ARID1A mutations in endometriosis-associated ovarian
carcinomas. N Engl J Med. 363:1532–1543. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ayhan A, Mao TL, Seckin T, Wu CH, Guan B,
Ogawa H, Futagami M, Mizukami H, Yokoyama Y, Kurman RJ and Shih
IeM: Loss of ARID1A expression is an early molecular event in tumor
progression from ovarian endometriotic cyst to clear cell and
endometrioid carcinoma. Int J Gynecol Cancer. 22:1310–1315. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Hollis RL and Gourley C: Genetic and
molecular changes in ovarian cancer. Cancer Biol Med. 13:236–247.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yarmolinsky J, Relton CL, Lophatananon A,
Muir K, Menon U, Gentry-Maharaj A, Walther A, Zheng J, Fasching P,
Zheng W, et al: Appraising the role of previously reported risk
factors in epithelial ovarian cancer risk: A Mendelian
randomization analysis. PLoS Med. 16:e10028932019. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
La Vecchia C: Ovarian cancer: Epidemiology
and risk factors. Eur J Cancer Prev. 26:55–62. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Liao Y, Tu C, Song X and Cai L: Case
report: Analysis of BRCA1 and BRCA2 gene mutations in a hereditary
ovarian cancer family. J Assist Reprod Genet. 37:1489–1495. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Daly MB, Pal T, Berry MP, Buys SS, Dickson
P, Domchek SM, Elkhanany A, Friedman S, Goggins M, Hutton ML, et
al: Genetic/Familial high-risk assessment: Breast, ovarian, and
pancreatic, version 2.2021, NCCN clinical practice guidelines in
oncology. J Natl Compr Canc Netw. 19:77–102. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Onwude J: Hormone therapy and ovarian
cancer. Lancet. 386:1037–1038. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Mørch LS, Løkkegaard E, Andreasen AH,
Krüger-Kjaer S and Lidegaard O: Hormone therapy and ovarian cancer.
JAMA. 302:298–305. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Colombo N, Sessa C, du Bois A, Ledermann
J, McCluggage WG, McNeish I, Morice P, Pignata S, Ray-Coquard I,
Vergote I, et al: ESMO-ESGO consensus conference recommendations on
ovarian cancer: Pathology and molecular biology, early and advanced
stages, borderline tumours and recurrent disease†. Ann Oncol.
30:672–705. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Saeaib N, Peeyananjarassri K,
Liabsuetrakul T, Buhachat R and Myriokefalitaki E: Hormone
replacement therapy after surgery for epithelial ovarian cancer.
Cochrane Database Syst Rev. 1:CD0125592020.PubMed/NCBI
|
|
25
|
Sieh W, Köbel M, Longacre TA, Bowtell DD,
deFazio A, Goodman MT, Høgdall E, Deen S, Wentzensen N, Moysich KB,
et al: Hormone-receptor expression and ovarian cancer survival: An
ovarian tumor tissue analysis consortium study. Lancet Oncol.
14:853–862. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Paleari L and DeCensi A: Endocrine therapy
in ovarian cancer: Where do we stand? Curr Opin Obstet Gynecol.
30:17–22. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Cramer DW and Welch WR: Determinants of
ovarian cancer risk. II. Inferences regarding pathogenesis. J Natl
Cancer Inst. 71:717–721. 1983.PubMed/NCBI
|
|
28
|
Riman T, Dickman PW, Nilsson S, Correia N,
Nordlinder H, Magnusson CM, Weiderpass E and Persson IR: Hormone
replacement therapy and the risk of invasive epithelial ovarian
cancer in Swedish women. J Natl Cancer Inst. 94:497–504. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Rodriguez C, Patel AV, Calle EE, Jacob EJ
and Thun MJ: Estrogen replacement therapy and ovarian cancer
mortality in a large prospective study of US women. JAMA.
285:1460–1465. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Risch HA: Hormonal etiology of epithelial
ovarian cancer, with a hypothesis concerning the role of androgens
and progesterone. J Natl Cancer Inst. 90:1774–1786. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Konishi I, Kuroda H and Mandai M: Review:
Gonadotropins and development of ovarian cancer. Oncology. 57
(Suppl 2):S45–S48. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Schildkraut JM, Schwingl PJ, Bastos E,
Evanoff A and Hughes C: Epithelial ovarian cancer risk among women
with polycystic ovary syndrome. Obstet Gynecol. 88:554–559. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Goulis DG and Mantzoros CS: Reproductive
endocrinology: Novel insights into pathophysiology and clinical
management. Metabolism. 86:1–2. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Cardenas C, Alvero AB, Yun BS and Mor G:
Redefining the origin and evolution of ovarian cancer: A hormonal
connection. Endocr Relat Cancer. 23:R411–R422. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Trabert B, Coburn SB, Falk RT, Manson JE,
Brinton LA, Gass ML, Kuller LH, Rohan TE, Pfeiffer RM, Qi L, et al:
Circulating estrogens and postmenopausal ovarian and endometrial
cancer risk among current hormone users in the Women's Health
Initiative Observational Study. Cancer Causes Control.
30:1201–1211. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Webb PM and Jordan SJ: Epidemiology of
epithelial ovarian cancer. Best Pract Res Clin Obstet Gynaecol.
41:3–14. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Risch HA, Marrett LD, Jain M and Howe GR:
Differences in risk factors for epithelial ovarian cancer by
histologic type. Results of a case-control study. Am J Epidemiol.
144:363–372. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Hankinson SE, Colditz GA, Hunter DJ,
Willett WC, Stampfer MJ, Rosner B, Hennekens CH and Speizer FE: A
prospective study of reproductive factors and risk of epithelial
ovarian cancer. Cancer. 76:284–290. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Moorman PG, Havrilesky LJ, Gierisch JM,
Coeytaux RR, Lowery WJ, Peragallo Urrutia R, Dinan M, McBroom AJ,
Hasselblad V, Sanders GD and Myers ER: Oral contraceptives and risk
of ovarian cancer and breast cancer among high-risk women: A
systematic review and meta-analysis. J Clin Oncol. 31:4188–4198.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Rzepka-Górska I, Chudecka-Głaz A and
Kosmowska B: FSH and LH serum/tumor fluid ratios and malignant
tumors of the ovary. Endocr Relat Cancer. 11:315–321. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Halperin R, Pansky M, Vaknin Z, Zehavi S,
Bukovsky I and Schneider D; Sackler Faculty of Medicine, Tel Aviv
University, Israel, : Luteinizing hormone in peritoneal and ovarian
cyst fluids: A predictor of ovarian carcinoma. Eur J Obstet Gynecol
Reprod Biol. 110:207–210. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Chen FC, Oskay-Ozcelik G, Bühling KJ,
Köpstein U, Mentze M, Lichtenegger W and Sehouli J: Prognostic
value of serum and ascites levels of estradiol, FSH, LH and
prolactin in ovarian cancer. Anticancer Res. 29:1575–1578.
2009.PubMed/NCBI
|
|
43
|
Chudecka-Głaz A and Rzepka-Górska I:
Concentrations of follicle stimulating hormone are increased in
ovarian tumor fluid: Implications for the management of ovarian
cancer. Eur J Gynaecol Oncol. 29:37–42. 2008.PubMed/NCBI
|
|
44
|
Fathalla MF: Incessant ovulation-a factor
in ovarian neoplasia? Lancet. 2:1631971. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Fleming JS, Beaugié CR, Haviv I,
Chenevix-Trench G and Tan OL: Incessant ovulation, inflammation and
epithelial ovarian carcinogenesis: Revisiting old hypotheses. Mol
Cell Endocrinol. 247:4–21. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Fathalla MF: Incessant ovulation and
ovarian cancer-a hypothesis re-visited. Facts Views Vis Obgyn.
5:292–297. 2013.PubMed/NCBI
|
|
47
|
Peres LC, Moorman PG, Alberg AJ, Bandera
EV, Barnholtz-Sloan J, Bondy M, Cote ML, Funkhouser E, Peters ES,
Schwartz AG, et al: Lifetime number of ovulatory cycles and
epithelial ovarian cancer risk in African American women. Cancer
Causes Control. 28:405–414. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Yang HP, Murphy KR, Pfeiffer RM, George N,
Garcia-Closas M, Lissowska J, Brinton LA and Wentzensen N: Lifetime
number of ovulatory cycles and risks of ovarian and endometrial
cancer among postmenopausal women. Am J Epidemiol. 183:800–814.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Mizushima T and Miyamoto H: The role of
androgen receptor signaling in ovarian cancer. Cells. 8:1762019.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Gibson DA, Simitsidellis I, Collins F and
Saunders PT: Evidence of androgen action in endometrial and ovarian
cancers. Endocr Relat Cancer. 21:T203–T218. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Kollara A, Shathasivam P, Park S,
Ringuette MJ and Brown TJ: Increased androgen receptor levels and
signaling in ovarian cancer cells by VEPH1 associated with
suppression of SMAD3 and AKT activation. J Steroid Biochem Mol
Biol. 196:1054982020. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Huang SL, Chang TC, Chao CCK and Sun NK:
Role of the TLR4-androgen receptor axis and genistein in
taxol-resistant ovarian cancer cells. Biochem Pharmacol.
177:1139652020. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Feng D, Zhao T, Yan K, Liang H, Liang J,
Zhou Y, Zhao W and Ling B: Gonadotropins promote human ovarian
cancer cell migration and invasion via a cyclooxygenase 2-dependent
pathway. Oncol Rep. 38:1091–1098. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Parrott JA, Doraiswamy V, Kim G, Mosher R
and Skinner MK: Expression and actions of both the follicle
stimulating hormone receptor and the luteinizing hormone receptor
in normal ovarian surface epithelium and ovarian cancer. Mol Cell
Endocrinol. 172:213–222. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Zheng W, Magid MS, Kramer EE and Chen YT:
Follicle-stimulating hormone receptor is expressed in human ovarian
surface epithelium and fallopian tube. Am J Pathol. 148:47–53.
1996.PubMed/NCBI
|
|
56
|
Chambers GM, Sullivan EA, Ishihara O,
Chapman MG and Adamson GD: The economic impact of assisted
reproductive technology: A review of selected developed countries.
Fertil Steril. 91:2281–2294. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Pawlikowski Łódź M: Expression of follicle
stimulating hormone receptors in intra-tumoral vasculature and in
tumoral cells-the involvement in tumour progression and the
perspectives of application in cancer diagnosis and therapy.
Endokrynol Pol. 69:192–198. 2018.PubMed/NCBI
|
|
58
|
Robin B, Planeix F, Sastre-Garau X, Pichon
C, Olesen TK, Gogusev J and Ghinea N: Follicle-stimulating hormone
receptor expression in endometriotic lesions and the associated
vasculature: An immunohistochemical study. Reprod Sci. 23:885–891.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Lenhard M, Lennerová T, Ditsch N, Kahlert
S, Friese K, Mayr D and Jeschke U: Opposed roles of
follicle-stimulating hormone and luteinizing hormone receptors in
ovarian cancer survival. Histopathology. 58:990–994. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Choi JH, Choi KC, Auersperg N and Leung
PC: Overexpression of follicle-stimulating hormone receptor
activates oncogenic pathways in preneoplastic ovarian surface
epithelial cells. J Clin Endocrinol Metab. 89:5508–5516. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Zhang Z, Jia L, Feng Y and Zheng W:
Overexpression of follicle-stimulating hormone receptor facilitates
the development of ovarian epithelial cancer. Cancer Lett.
278:56–64. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Deuster E, Mayr D, Hester A, Kolben T,
Zeder-Göß C, Burges A, Mahner S, Jeschke U, Trillsch F and Czogalla
B: Correlation of the aryl hydrocarbon receptor with FSHR in
ovarian cancer patients. Int J Mol Sci. 20:28622019. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Cheung J, Lokman NA, Abraham RD,
Macpherson AM, Lee E, Grutzner F, Ghinea N, Oehler MK and
Ricciardelli C: Reduced gonadotrophin receptor expression is
associated with a more aggressive ovarian cancer phenotype. Int J
Mol Sci. 22:712020. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Lau MT, Wong AS and Leung PC:
Gonadotropins induce tumor cell migration and invasion by
increasing cyclooxygenases expression and prostaglandin E(2)
production in human ovarian cancer cells. Endocrinology.
151:2985–2993. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Song K, Dai L, Long X, Wang W and Di W:
Follicle-stimulating hormone promotes the proliferation of
epithelial ovarian cancer cells by activating sphingosine kinase.
Sci Rep. 10:138342020. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Warrenfeltz SW, Lott SA, Palmer TM, Gray
JC and Puett D: Luteinizing hormone-induced up-regulation of ErbB-2
is insufficient stimulant of growth and invasion in ovarian cancer
cells. Mol Cancer Res. 6:1775–1785. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Liao H, Zhou Q, Gu Y, Duan T and Feng Y:
Luteinizing hormone facilitates angiogenesis in ovarian epithelial
tumor cells and metformin inhibits the effect through the mTOR
signaling pathway. Oncol Rep. 27:1873–1878. 2012.PubMed/NCBI
|
|
68
|
Zhang Z, Liao H, Chen X, Zheng Y, Liu Y,
Tao X, Gu C, Dong L, Duan T, Yang Y, et al: Luteinizing hormone
upregulates survivin and inhibits apoptosis in ovarian epithelial
tumors. Eur J Obstet Gynecol Reprod Biol. 155:69–74. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Sakai T, Shiraishi A, Kawada T, Matsubara
S, Aoyama M and Satake H: Invertebrate Gonadotropin-releasing
hormone-related peptides and their receptors: An update. Front
Endocrinol (Lausanne). 8:2172017. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Jankowska AG, Andrusiewicz M, Fischer N
and Warchol PJ: Expression of hCG and GnRHs and their receptors in
endometrial carcinoma and hyperplasia. Int J Gynecol Cancer.
20:92–101. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Gründker C, Schlotawa L, Viereck V, Eicke
N, Horst A, Kairies B and Emons G: Antiproliferative effects of the
GnRH antagonist cetrorelix and of GnRH-II on human endometrial and
ovarian cancer cells are not mediated through the GnRH type I
receptor. Eur J Endocrinol. 151:141–149. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Kang SK, Tai CJ, Cheng KW and Leung PC:
Gonadotropin- releasing hormone activates mitogen-activated protein
kinase in human ovarian and placental cells. Mol Cell Endocrinol.
170:143–151. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Gründker C, Günthert AR, Westphalen S and
Emons G: Biology of the gonadotropin-releasing hormone system in
gynecological cancers. Eur J Endocrinol. 146:1–14. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Wilkinson SJ, Kucukmetin A, Cross P, Darby
S, Gnanapragasam VJ, Calvert AH, Robson CN and Edmondson RJ:
Expression of gonadotrophin releasing hormone receptor I is a
favorable prognostic factor in epithelial ovarian cancer. Hum
Pathol. 39:1197–1204. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Peng C, Fan NC, Ligier M, Väänänen J and
Leung PC: Expression and regulation of gonadotropin-releasing
hormone (GnRH) and GnRH receptor messenger ribonucleic acids in
human granulosa-luteal cells. Endocrinology. 135:1740–1746. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Choi KC, Auersperg N and Leung PC:
Expression and antiproliferative effect of a second form of
gonadotropin-releasing hormone in normal and neoplastic ovarian
surface epithelial cells. J Clin Endocrinol Metab. 86:5075–5078.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Choi JH, Choi KC, Auersperg N and Leung
PC: Differential regulation of two forms of gonadotropin-releasing
hormone messenger ribonucleic acid by gonadotropins in human
immortalized ovarian surface epithelium and ovarian cancer cells.
Endocr Relat Cancer. 13:641–651. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Kim KY, Choi KC, Auersperg N and Leung PC:
Mechanism of gonadotropin-releasing hormone (GnRH)-I and
-II-induced cell growth inhibition in ovarian cancer cells: Role of
the GnRH-I receptor and protein kinase C pathway. Endocr Relat
Cancer. 13:211–220. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Padula AM: GnRH analogues-agonists and
antagonists. Anim Reprod Sci. 88:115–126. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Limonta P, Marelli MM, Moretti R,
Marzagalli M, Fontana F and Maggi R: GnRH in the human female
reproductive axis. Vitam Horm. 107:27–66. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Newton CL, Riekert C and Millar RP:
Gonadotropin-releasing hormone analog therapeutics. Minerva
Ginecol. 70:497–515. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Newton CL, Anderson RC and Millar RP:
Therapeutic neuroendocrine agonist and antagonist analogs of
hypothalamic neuropeptides as modulators of the
hypothalamic-pituitary- gonadal axis. Endocr Dev. 30:106–129. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Wuntakal R, Seshadri S, Montes A and Lane
G: Luteinising hormone releasing hormone (LHRH) agonists for the
treatment of relapsed epithelial ovarian cancer. Cochrane Database
Syst Rev. 2016:CD0113222016.PubMed/NCBI
|
|
84
|
Limonta P, Montagnani Marelli M, Mai S,
Motta M, Martini L and Moretti RM: GnRH receptors in cancer: From
cell biology to novel targeted therapeutic strategies. Endocr Rev.
33:784–811. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Gründker C and Emons G: The role of
gonadotropin-releasing hormone in cancer cell proliferation and
metastasis. Front Endocrinol (Lausanne). 8:1872017. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Zhang Y, Lu ZY, Yan XH, Wang JJ, Feng WW,
Ding JX and Hua KQ: Study on the effects of gonadotropin-releasing
hormone analogues in the inhibition of ovarian cancer transplanted
tumors and in the protection of ovarian function after chemotherapy
on nude mice. Zhonghua Fu Chan Ke Za Zhi. 46:892–897. 2011.(In
Chinese). PubMed/NCBI
|
|
87
|
Völker P, Gründker C, Schmidt O, Schulz KD
and Emons G: Expression of receptors for luteinizing
hormone-releasing hormone in human ovarian and endometrial cancers:
Frequency, autoregulation, and correlation with direct
antiproliferative activity of luteinizing hormone-releasing hormone
analogues. Am J Obstet Gynecol. 186:171–179. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Gründker C, Völker P and Emons G:
Antiproliferative signaling of luteinizing hormone-releasing
hormone in human endometrial and ovarian cancer cells through G
protein alpha(I)-mediated activation of phosphotyrosine
phosphatase. Endocrinology. 142:2369–2380. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Gründker C, Völker P, Schulz KD and Emons
G: Luteinizing hormone-releasing hormone agonist triptorelin and
antagonist cetrorelix inhibit EGF-induced c-fos expression in human
gynecological cancers. Gynecol Oncol. 78:194–202. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Engel JB, Hahne JC, Häusler SF, Meyer S,
Segerer SE, Diessner J, Dietl J and Honig A: Peptidomimetic GnRH
antagonist AEZS-115 inhibits the growth of ovarian and endometrial
cancer cells. Anticancer Res. 32:2063–2068. 2012.PubMed/NCBI
|
|
91
|
Günthert AR, Gründker C, Hollmann K and
Emons G: Luteinizing hormone-releasing hormone induces JunD-DNA
binding and extends cell cycle in human ovarian cancer cells.
Biochem Biophys Res Commun. 294:11–15. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Imai A, Takagi A, Horibe S, Takagi H and
Tamaya T: Fas and Fas ligand system may mediate antiproliferative
activity of gonadotropin-releasing hormone receptor in endometrial
cancer cells. Int J Oncol. 13:97–100. 1998.PubMed/NCBI
|
|
93
|
Imai A, Takagi A, Horibe S, Takagi H and
Tamaya T: Evidence for tight coupling of gonadotropin-releasing
hormone receptor to stimulated Fas ligand expression in
reproductive tract tumors: Possible mechanism for hormonal control
of apoptotic cell death. J Clin Endocrinol Metab. 83:427–431. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Meyer C, Sims AH, Morgan K, Harrison B,
Muir M, Bai J, Faratian D, Millar RP and Langdon SP: Transcript and
protein profiling identifies signaling, growth arrest, apoptosis,
and NF-κB survival signatures following GNRH receptor activation.
Endocr Relat Cancer. 20:123–136. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Mo Y, Peng P, Zhou R, He Z, Huang L and
Yang D: Regulation of gonadotropin-releasing hormone (GnRH)
receptor-I expression in the pituitary and ovary by a GnRH agonist
and antagonist. Reprod Sci. 17:68–77. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Gründker C, Schulz K, Günthert AR and
Emons G: Luteinizing hormone-releasing hormone induces nuclear
factor kappaB-activation and inhibits apoptosis in ovarian cancer
cells. J Clin Endocrinol Metab. 85:3815–3820. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Gründker C, Günthert AR, Millar RP and
Emons G: Expression of gonadotropin-releasing hormone II (GnRH-II)
receptor in human endometrial and ovarian cancer cells and effects
of GnRH-II on tumor cell proliferation. J Clin Endocrinol Metab.
87:1427–1430. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Fister S, Günthert AR, Aicher B, Paulini
KW, Emons G and Gründker C: GnRH-II antagonists induce apoptosis in
human endometrial, ovarian, and breast cancer cells via activation
of stress-induced MAPKs p38 and JNK and proapoptotic protein Bax.
Cancer Res. 69:6473–6481. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Morgan K, Conklin D, Pawson AJ, Sellar R,
Ott TR and Millar RP: A transcriptionally active human type II
gonadotropin-releasing hormone receptor gene homolog overlaps two
genes in the antisense orientation on chromosome 1q.12.
Endocrinology. 144:423–436. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Neill JD, Musgrove LC and Duck LW: Newly
recognized GnRH receptors: Function and relative role. Trends
Endocrinol Metab. 15:383–392. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Gründker C, Huschmand Nia A and Emons G:
Gonadotropin- releasing hormone receptor-targeted gene therapy of
gynecologic cancers. Mol Cancer Ther. 4:225–231. 2005.PubMed/NCBI
|
|
102
|
Parmar H, Phillips RH, Rustin G, Lightman
SL and Schally AV: Therapy of advanced ovarian cancer with
D-Trp-6-LH-RH (decapeptyl) microcapsules. Biomed Pharmacother.
42:531–538. 1988.PubMed/NCBI
|
|
103
|
Jäger W, Wildt L and Lang N: Some
observations on the effect of a GnRH analog in ovarian cancer. Eur
J Obstet Gynecol Reprod Biol. 32:137–148. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Carnino F, Iskra L, Fuda G, Foglia G,
Odicino F, Bruzzone M, Chiara S, Gadducci A and Ragni N: The
treatment of progressive ovarian carcinoma with D-Trp-LHRH
(Decapeptyl). Gruppo Oncologico Nord ovest (GONO). Eur J Cancer.
30A:1903–1904. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Ron IG, Wigler N, Merimsky O, Inbar MJ and
Chaitchik S: A phase II trial of D-Trp-6-LHRH (decapeptyl) in
pretreated patients with advanced epithelial ovarian cancer. Cancer
Invest. 13:272–275. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Duffaud F, van der Burg ME, Namer M,
Vergote I, Willemse PH, ten Bokkel Huinink W, Guastalla JP, Nooij,
Kerbrat P, Piccart M, et al: D-TRP-6-LHRH (Triptorelin) is not
effective in ovarian carcinoma: An EORTC gynaecological cancer
Co-operative group study. Anticancer Drugs. 12:159–162. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Emons G, Ortmann O, Teichert HM, Fassl H,
Löhrs U, Kullander S, Kauppila A, Ayalon D, Schally A and
Oberheuser F: Luteinizing hormone-releasing hormone agonist
triptorelin in combination with cytotoxic chemotherapy in patients
with advanced ovarian carcinoma. A prospective double blind
randomized trial. Decapeptyl Ovarian Cancer Study Group. Cancer.
78:1452–1460. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Jager W, Sauerbrei W, Beck E, Maassen V,
Stumpfe M, Meier W, Kuhn W and Janicke F: A randomized comparison
of triptorelin and tamoxifen as treatment of progressive ovarian
cancer. Anticancer Res. 15:2639–2642. 1995.PubMed/NCBI
|
|
109
|
Sevelda P, Vavra N, Fitz R, Barrada M,
Salzer H, Baur M and Dittrich C: Goserelin a GnRH-analogue as
third-line therapy of refractory epithelial ovarian cancer. Int J
Gynecol Cancer. 2:160–162. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Lind MJ, Cantwell BM, Millward MJ,
Robinson A, Proctor M, Simmons D, Carmichael J and Harris AL: A
phase II trial of goserelin (Zoladex) in relapsed epithelial
ovarian cancer. Br J Cancer. 65:621–623. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Hasan J, Ton N, Mullamitha S, Clamp A,
McNeilly A, Marshall E and Jayson GC: Phase II trial of tamoxifen
and goserelin in recurrent epithelial ovarian cancer. Br J Cancer.
93:647–651. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Marinaccio M, D'Addario V, Serratì A,
Pinto V and Cagnazzo G: Leuprolide acetate as a salvage-therapy in
relapsed epithelial ovarian cancer. Eur J Gynaecol Oncol.
17:286–288. 1996.PubMed/NCBI
|
|
113
|
Paskeviciute L, Roed H and Engelholm S: No
rules without exception: Long-term complete remission observed in a
study using a LH-RH agonist in platinum-refractory ovarian cancer.
Gynecol Oncol. 86:297–301. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
du BA, Meier W, Lück HJ, Emon G, Moebus V,
Schroeder W, Costa S, Bauknecht T, Olbricht S, Jackisch C, et al:
Chemotherapy versus hormonal treatment in platinum- and
Paclitaxel-refractory ovarian cancer: A randomised trial of the
german arbeitsgemeinschaft gynaekologische onkologie (AGO) study
group ovarian cancer. Ann Oncol. 13:251–257. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Balbi G, Piano LD, Cardone A and Cirelli
G: Second-line therapy of advanced ovarian cancer with GnRH
analogs. Int J Gynecol Cancer. 14:799–803. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Kavanagh JJ, Roberts W, Townsend P and
Hewitt S: Leuprolide acetate in the treatment of refractory or
persistent epithelial ovarian cancer. J Clin Oncol. 7:115–118.
1989. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Bruckner HW and Motwani BT: Treatment of
advanced refractory ovarian carcinoma with a gonadotropin-releasing
hormone analogue. Am J Obstet Gynecol. 161:1216–1218. 1989.
View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Miller DS, Brady MF and Barrett RJ: A
phase II trial of leuprolide acetate in patients with advanced
epithelial ovarian carcinoma. A Gynecologic Oncology Group study.
Am J Clin Oncol. 15:125–128. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Verschraegen CF, Westphalen S, Hu W, Loyer
E, Kudelka A, Völker P, Kavanagh J, Steger M, Schulz KD and Emons
G: Phase II study of cetrorelix, a luteinizing hormone-releasing
hormone antagonist in patients with platinum-resistant ovarian
cancer. Gynecol Oncol. 90:552–559. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Parmar H, Rustin G, Lightman SL, Phillips
RH, Hanham IW and Schally AV: Response to D-Trp-6-luteinising
hormone releasing hormone (Decapeptyl) microcapsules in advanced
ovarian cancer. Br Med J (Clin Res Ed). 296:12291988. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Emons G, Gorchev G, Sehouli J, Wimberger
P, Stähle A, Hanker L, Hilpert F, Sindermann H, Gründker C and
Harter P: Efficacy and safety of AEZS-108 (INN: Zoptarelin
doxorubicin acetate) an LHRH agonist linked to doxorubicin in women
with platinum refractory or resistant ovarian cancer expressing
LHRH receptors: A multicenter phase II trial of the ago-study group
(AGO GYN 5). Gynecol Oncol. 133:427–432. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Medl M, Peters-Engel C, Fuchs G and
Leodolter S: Triptorelin (D-Trp-6-LHRH) in combination with
carboplatin-containing polychemotherapy for advanced ovarian
cancer: A pilot study. Anticancer Res. 13:2373–2376.
1993.PubMed/NCBI
|
|
123
|
Falkson CI, Falkson HC and Falkson G:
Cisplatin versus cisplatin plus D-Trp-6-LHRH in the treatment of
ovarian cancer: A pilot trial to investigate the effect of the
addition of a GnRH analogue to cisplatin. Oncology. 53:313–317.
1996. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Rzepka-Górska I, Chudecka-Glaz A, Kosmider
M and Malecha J: GnRH analogues as an adjuvant therapy for ovarian
cancer patients. Int J Gynaecol Obstet. 81:199–205. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Mizushima T, Tirador KA and Miyamoto H:
Androgen receptor activation: A prospective therapeutic target for
bladder cancer? Expert Opin Ther Targets. 21:249–257. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Fujii S and Kagechika H: Androgen receptor
modulators: A review of recent patents and reports (2012–2018).
Expert Opin Ther Pat. 29:439–453. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Gucalp A and Traina TA: The androgen
receptor: Is it a promising target? Ann Surg Oncol. 24:2876–2880.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Ueda T, Mawji NR, Bruchovsky N and Sadar
MD: Ligand-independent activation of the androgen receptor by
interleukin-6 and the role of steroid receptor coactivator-1 in
prostate cancer cells. J Biol Chem. 277:38087–38094. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Pagliarulo V: Androgen deprivation therapy
for prostate cancer. Adv Exp Med Biol. 1096:1–30. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Rahim B and O'Regan R: AR Signaling in
breast cancer. Cancers (Basel). 9:212017. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Antonarakis ES: AR Signaling in human
malignancies: Prostate cancer and beyond. Cancers (Basel).
10:222018. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Hamilton TC, Davies P and Griffiths K:
Androgen and oestrogen binding in cytosols of human ovarian
tumours. J Endocrinol. 90:421–431. 1981. View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Kühnel R, de Graaff J, Rao BR and Stolk
JG: Androgen receptor predominance in human ovarian carcinoma. J
Steroid Biochem. 26:393–397. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Chadha S, Rao BR, Slotman BJ, van
Vroonhoven CC and van der Kwast TH: An immunohistochemical
evaluation of androgen and progesterone receptors in ovarian
tumors. Hum Pathol. 24:90–95. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Sheach LA, Adeney EM, Kucukmetin A,
Wilkinson SJ, Fisher AD, Elattar A, Robson CN and Edmondson RJ:
Androgen-related expression of G-proteins in ovarian cancer. Br J
Cancer. 101:498–503. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Cardillo MR, Petrangeli E, Aliotta N,
Salvatori L, Ravenna L, Chang C and Castagna G: Androgen receptors
in ovarian tumors: Correlation with oestrogen and progesterone
receptors in an immunohistochemical and semiquantitative image
analysis study. J Exp Clin Cancer Res. 17:231–237. 1998.PubMed/NCBI
|
|
137
|
Elattar A, Warburton KG, Mukhopadhyay A,
Freer RM, Shaheen F, Cross P, Plummer ER, Robson CN and Edmondson
RJ: Androgen receptor expression is a biological marker for
androgen sensitivity in high grade serous epithelial ovarian
cancer. Gynecol Oncol. 124:142–147. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
de Toledo MC, Sarian LO, Sallum LF,
Andrade LL, Vassallo J, de Paiva Silva GR, Pinto GA, Soares FA,
Fonseca CD and Derchain SF: Analysis of the contribution of
immunologically-detectable HER2, steroid receptors and of the
‘triple-negative’ tumor status to disease-free and overall survival
of women with epithelial ovarian cancer. Acta Histochem.
116:440–447. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Jönsson JM, Arildsen NS, Malander S,
Måsbäck A, Hartman L, Nilbert M and Hedenfalk I: Sex Steroid
hormone receptor expression affects ovarian cancer survival. Transl
Oncol. 8:424–433. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Butler MS, Ricciardelli C, Tilley WD and
Hickey TE: Androgen receptor protein levels are significantly
reduced in serous ovarian carcinomas compared with benign or
borderline disease but are not altered by cancer stage or
metastatic progression. Horm Cancer. 4:154–164. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Edmondson RJ, Monaghan JM and Davies BR:
The human ovarian surface epithelium is an androgen responsive
tissue. Br J Cancer. 86:879–885. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Syed V, Ulinski G, Mok SC, Yiu GK and Ho
SM: Expression of gonadotropin receptor and growth responses to key
reproductive hormones in normal and malignant human ovarian surface
epithelial cells. Cancer Res. 61:6768–6776. 2001.PubMed/NCBI
|
|
143
|
Nourbakhsh M, Golestani A, Zahrai M,
Modarressi MH, Malekpour Z and Karami-Tehrani F: Androgens
stimulate telomerase expression, activity and phosphorylation in
ovarian adenocarcinoma cells. Mol Cell Endocrinol. 330:10–16. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Ligr M, Patwa RR, Daniels G, Pan L, Wu X,
Li Y, Tian L, Wang Z, Xu R, Wu J, et al: Expression and function of
androgen receptor coactivator p44/Mep50/WDR77 in ovarian cancer.
PLoS One. 6:e262502011. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Silva EG, Tornos C, Fritsche HA Jr,
el-Naggar A, Gray K, Ordonez NG, Luna M and Gershenson D: The
induction of benign epithelial neoplasms of the ovaries of guinea
pigs by testosterone stimulation: A potential animal model. Mod
Pathol. 10:879–883. 1997.PubMed/NCBI
|
|
146
|
Sawada M, Terada N, Wada A, Mori Y,
Yamasaki M, Saga T and Endo K: Estrogen- and androgen-responsive
growth of human ovarian adenocarcinoma heterotransplanted into nude
mice. Int J Cancer. 45:359–363. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Park BY, Grisham RN, den Hollander B,
Thapi D, Berman T, de Stanchina E, Zhou Q, Iyer G, Aghajanian C and
Spriggs DR: Tumor Inhibition by Enzalutamide in a xenograft model
of ovarian cancer. Cancer Invest. 34:517–520. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Proverbs-Singh T, Feldman JL, Morris MJ,
Autio KA and Traina TA: Targeting the androgen receptor in prostate
and breast cancer: Several new agents in development. Endocr Relat
Cancer. 22:R87–R106. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Sun NK, Huang SL, Chang PY, Lu HP and Chao
CC: Transcriptomic profiling of taxol-resistant ovarian cancer
cells identifies FKBP5 and the androgen receptor as critical
markers of chemotherapeutic response. Oncotarget. 5:11939–11956.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Sun NK, Huang SL, Lu HP, Chang TC and Chao
CC: Integrative transcriptomics-based identification of cryptic
drivers of taxol-resistance genes in ovarian carcinoma cells:
Analysis of the androgen receptor. Oncotarget. 6:27065–27082. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Tumolo S, Rao BR, van der Burg ME,
Guastalla JP, Renard J and Vermorken JB: Phase II trial of
flutamide in advanced ovarian cancer: An EORTC Gynaecological
Cancer Cooperative Group study. Eur J Cancer. 30A:911–914. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
152
|
Vassilomanolakis M, Koumakis G, Barbounis
V, Hajichristou H, Tsousis S and Efremidis A: A phase II study of
flutamide in ovarian cancer. Oncology. 54:199–202. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Gruessner C, Gruessner A, Glaser K,
AbuShahin N, Zhou Y, Laughren C, Wright H, Pinkerton S, Yi X,
Stoffer J, et al: Flutamide and biomarkers in women at high risk
for ovarian cancer: Preclinical and clinical evidence. Cancer Prev
Res (Phila). 7:896–905. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
154
|
Levine D, Park K, Juretzka M, Esch J,
Hensley M, Aghajanian C, Lewin S, Konner J, Derosa F, Spriggs D, et
al: A phase II evaluation of goserelin and bicalutamide in patients
with ovarian cancer in second or higher complete clinical disease
remission. Cancer. 110:2448–2456. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Grisham RN, Giri DD, Iasonos A, Zhou Q,
Girshman J, McGrath SP, O'Cearbhaill RE, Sabbatini P, Tew WP,
Michael Hyman D, et al: A phase II trial of enzalutamide in
patients with androgen receptor positive (AR+) ovarian, primary
peritoneal or fallopian tube cancer and one, two, or three prior
therapies. J Clin Oncol. 35 (Suppl 15):TPS56102017. View Article : Google Scholar
|
|
156
|
Miyamoto H, Messing EM and Chang C:
Androgen deprivation therapy for prostate cancer: Current status
and future prospects. Prostate. 61:332–353. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
157
|
Tran C, Ouk S, Clegg NJ, Chen Y, Watson
PA, Arora V, Wongvipat J, Smith-Jones PM, Yoo D, Kwon A, et al:
Development of a second-generation antiandrogen for treatment of
advanced prostate cancer. Science. 324:787–790. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
158
|
Fuentes N and Silveyra P: Estrogen
receptor signaling mechanisms. Adv Protein Chem Struct Biol.
116:135–170. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Björnström L and Sjöberg M: Estrogen
receptor-dependent activation of AP-1 via non-genomic signalling.
Nucl Recept. 2:32004. View Article : Google Scholar : PubMed/NCBI
|
|
160
|
Kumar A and Foster TC: G Protein-Coupled
estrogen receptor: Rapid effects on hippocampal-dependent spatial
memory and synaptic plasticity. Front Endocrinol (Lausanne).
11:3852020. View Article : Google Scholar : PubMed/NCBI
|
|
161
|
O'Donnell AJ, Macleod KG, Burns DJ, Smyth
JF and Langdon SP: Estrogen receptor-alpha mediates gene expression
changes and growth response in ovarian cancer cells exposed to
estrogen. Endocr Relat Cancer. 12:851–866. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
162
|
Arnal JF, Lenfant F, Metivier R, Flouriot
G, Henrion D, Adlanmerini M, Fontaine C, Gourdy P, Chambon P,
Katzenellenbogen B and Katzenellenbogen J: Membrane and nuclear
estrogen receptor alpha actions: From tissue specificity to medical
implications. Physiol Rev. 97:1045–1087. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
163
|
Ghasemi A, Saeidi J, Mohtashami M and
Hashemy SI: Estrogen-independent role of ERα in ovarian cancer
progression induced by leptin/Ob-Rb axis. Mol Cell Biochem.
458:207–217. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
164
|
Bogush TA, Basharina AA, Bogush EA,
Ryabinina OM, Tjulandina AS and Tjulandin SA: Estrogen receptors
alpha and beta in ovarian cancer: Expression level and prognosis.
Dokl Biochem Biophys. 482:249–251. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
165
|
Andersen CL, Sikora MJ, Boisen MM, Ma T,
Christie A, Tseng G, Park Y, Luthra S, Chandran U, Haluska P, et
al: Active estrogen receptor-alpha signaling in ovarian cancer
models and clinical specimens. Clin Cancer Res. 23:3802–3812. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
166
|
Chan KK, Wei N, Liu SS, Xiao-Yun L, Cheung
AN and Ngan HY: Estrogen receptor subtypes in ovarian cancer: A
clinical correlation. Obstet Gynecol. 111:144–151. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
167
|
Bossard C, Busson M, Vindrieux D, Gaudin
F, Machelon V, Brigitte M, Jacquard C, Pillon A, Balaguer P,
Balabanian K and Lazennec G: Potential role of estrogen receptor
beta as a tumor suppressor of epithelial ovarian cancer. PLoS One.
7:e447872012. View Article : Google Scholar : PubMed/NCBI
|
|
168
|
Rutherford T, Brown WD, Sapi E, Aschkenazi
S, Muñoz A and Mor G: Absence of estrogen receptor-beta expression
in metastatic ovarian cancer. Obstet Gynecol. 96:417–421. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
169
|
Hoffmann M, Gogola J and Ptak A: Apelin
abrogates the stimulatory effects of 17β-estradiol and insulin-like
growth factor-1 on proliferation of epithelial and granulosa
ovarian cancer cell lines via crosstalk between APLNR and
ERα/IGF1R. Mol Biol Rep. 46:6325–6338. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
170
|
Liu H, Yan Y, Wen H, Jiang X, Cao X, Zhang
G and Liu G: A novel estrogen receptor GPER mediates proliferation
induced by 17β-estradiol and selective GPER agonist G-1 in estrogen
receptor α (ERα)-negative ovarian cancer cells. Cell Biol Int.
38:631–638. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
171
|
Chan KK, Leung TH, Chan DW, Wei N, Lau GT,
Liu SS, Siu MK and Ngan HY: Targeting estrogen receptor subtypes
(ERalpha and ERbeta) with selective ER modulators in ovarian
cancer. J Endocrinol. 221:325–336. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
172
|
Park SH, Cheung LW, Wong AS and Leung PC:
Estrogen regulates Snail and Slug in the down-regulation of
E-cadherin and induces metastatic potential of ovarian cancer cells
through estrogen receptor alpha. Mol Endocrinol. 22:2085–2098.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
173
|
Moll F, Katsaros D, Lazennec G, Hellio N,
Roger P, Giacalone PL, Chalbos D, Maudelonde T, Rochefort H and
Pujol P: Estrogen induction and overexpression of fibulin-1C mRNA
in ovarian cancer cells. Oncogene. 21:1097–1107. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
174
|
Ciucci A, Zannoni GF, Travaglia D, Scambia
G and Gallo D: Mitochondrial estrogen receptor β2 drives
antiapoptotic pathways in advanced serous ovarian cancer. Hum
Pathol. 46:1138–1146. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
175
|
Kyriakidis I and Papaioannidou P: Estrogen
receptor beta and ovarian cancer: A key to pathogenesis and
response to therapy. Arch Gynecol Obstet. 293:1161–1168. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
176
|
Lazennec G: Estrogen receptor beta, a
possible tumor suppressor involved in ovarian carcinogenesis.
Cancer Lett. 231:151–157. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
177
|
Treeck O, Pfeiler G, Mitter D, Lattrich C,
Piendl G and Ortmann O: Estrogen receptor {beta}1 exerts
antitumoral effects on SK-OV-3 ovarian cancer cells. J Endocrinol.
193:421–433. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
178
|
Liu J, Viswanadhapalli S, Garcia L, Zhou
M, Nair BC, Kost E, Rao Tekmal R, Li R, Rao MK, Curiel T, et al:
Therapeutic utility of natural estrogen receptor beta agonists on
ovarian cancer. Oncotarget. 8:50002–50014. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
179
|
Chan KKL, Siu MKY, Jiang YX, Wang JJ, Wang
Y, Leung THY, Liu SS, Cheung ANY and Ngan HYS: Differential
expression of estrogen receptor subtypes and variants in ovarian
cancer: Effects on cell invasion, proliferation and prognosis. BMC
Cancer. 17:6062017. View Article : Google Scholar : PubMed/NCBI
|
|
180
|
Chan KKL, Siu MKY, Jiang YX, Wang JJ,
Leung THY and Ngan HYS: Estrogen receptor modulators genistein,
daidzein and ERB-041 inhibit cell migration, invasion,
proliferation and sphere formation via modulation of FAK and
PI3K/AKT signaling in ovarian cancer. Cancer Cell Int. 18:652018.
View Article : Google Scholar : PubMed/NCBI
|
|
181
|
Fekete T, Rásó E, Pete I, Tegze B, Liko I,
Munkácsy G, Sipos N, Rigó J Jr and Györffy B: Meta-analysis of gene
expression profiles associated with histological classification and
survival in 829 ovarian cancer samples. Int J Cancer. 131:95–105.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
182
|
Schüler-Toprak S, Weber F, Skrzypczak M,
Ortmann O and Treeck O: Estrogen receptor β is associated with
expression of cancer associated genes and survival in ovarian
cancer. BMC Cancer. 18:9812018. View Article : Google Scholar : PubMed/NCBI
|
|
183
|
Fujimoto J, Hirose R, Sakaguchi H and
Tamaya T: Clinical significance of expression of estrogen receptor
alpha and beta mRNAs in ovarian cancers. Oncology. 58:334–341.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
184
|
van Kruchten M, van der Marel P, de Munck
L, Hollema H, Arts H, Timmer-Bosscha H, de Vries E, Hospers G and
Reyners A: Hormone receptors as a marker of poor survival in
epithelial ovarian cancer. Gynecol Oncol. 138:634–639. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
185
|
van der Vange N, Greggi S, Burger CW,
Kenemans P and Vermorken JB: Experience with hormonal therapy in
advanced epithelial ovarian cancer. Acta Oncol. 34:813–820. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
186
|
Rolski J and Pawlicki M: Evaluation of
efficacy and toxicity of tamoxifen in patients with advanced
chemotherapy resistant ovarian cancer. Ginekol Pol. 69:586–589.
1998.(In Polish). PubMed/NCBI
|
|
187
|
Tropé C, Marth C and Kaern J: Tamoxifen in
the treatment of recurrent ovarian carcinoma. Eur J Cancer. 36
(Suppl 4):S59–S61. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
188
|
Van Der Velden J, Gitsch G, Wain GV,
Friedlander ML and Hacker NF: Tamoxifen in patients with advanced
epithelial ovarian cancer. Int J Gynecol Cancer. 5:301–305. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
189
|
Ahlgren JD, Ellison NM, Gottlieb RJ,
Laluna F, Lokich JJ, Sinclair PR, Ueno W, Wampler GL, Yeung KY, Alt
D, et al: Hormonal palliation of chemoresistant ovarian cancer:
Three consecutive phase II trials of the Mid-atlantic oncology
program. J Clin Oncol. 11:1957–1968. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
190
|
Jakobsen A, Bertelsen K and Sell A: Cyclic
hormonal treatment in ovarian cancer. A phase-II trial. Eur J
Cancer Clin Oncol. 23:915–916. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
191
|
Pagel J RC and Thorpe S: Treatment of
advanced ovarian carcinoma with tamoxifen: A phase II trial. Proc
2nd Eur Conf Clin Oncol. May 29–1983.(Epub ahead of print).
|
|
192
|
Osborne RJ, Malik ST, Slevin ML, Harvey
VJ, Spona J, Salzer H and Williams CJ: Tamoxifen in refractory
ovarian cancer: The use of a loading dose schedule. Br J Cancer.
57:115–116. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
193
|
Slevin ML, Harvey VJ, Osborne RJ, Shepherd
JH, Williams CJ and Mead GM: A phase II study of tamoxifen in
ovarian cancer. Eur J Cancer Clin Oncol. 22:309–312. 1986.
View Article : Google Scholar : PubMed/NCBI
|
|
194
|
Shirey DR, Kavanagh JJ Jr, Gershenson DM,
Freedman RS, Copeland LJ and Jones LA: Tamoxifen therapy of
epithelial ovarian cancer. Obstet Gynecol. 66:575–578.
1985.PubMed/NCBI
|
|
195
|
Hatch KD, Beecham JB, Blessing JA and
Creasman WT: Responsiveness of patients with advanced ovarian
carcinoma to tamoxifen. A Gynecologic Oncology Group study of
second-line therapy in 105 patients. Cancer. 68:269–271. 1991.
View Article : Google Scholar : PubMed/NCBI
|
|
196
|
Weiner SA, Alberts DS, Surwit EA, Davis J
and Grosso D: Tamoxifen therapy in recurrent epithelial ovarian
carcinoma. Gynecol Oncol. 27:208–213. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
197
|
Schwartz PE, Keating G, MacLusky N,
Naftolin F and Eisenfeld A: Tamoxifen therapy for advanced ovarian
cancer. Obstet Gynecol. 59:583–588. 1982.PubMed/NCBI
|
|
198
|
George A, McLachlan J, Tunariu N, Della
Pepa C, Migali C, Gore M, Kaye S and Banerjee S: The role of
hormonal therapy in patients with relapsed high-grade ovarian
carcinoma: A retrospective series of tamoxifen and letrozole. BMC
Cancer. 17:4562017. View Article : Google Scholar : PubMed/NCBI
|
|
199
|
Hurteau JA, Brady MF, Darcy KM, McGuire
WP, Edmonds P, Pearl ML, Ivanov I, Tewari KS, Mannel RS, Zanotti K
and Benbrook DM: Randomized phase III trial of tamoxifen versus
thalidomide in women with biochemical-recurrent-only epithelial
ovarian, fallopian tube or primary peritoneal carcinoma after a
complete response to first-line platinum/taxane chemotherapy with
an evaluation of serum vascular endothelial growth factor (VEGF): A
Gynecologic Oncology Group Study. Gynecol Oncol. 119:444–450. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
200
|
Schwartz PE, Chambers JT, Kohorn EI,
Chambers SK, Weitzman H, Voynick IM, MacLusky N and Naftolin F:
Tamoxifen in combination with cytotoxic chemotherapy in advanced
epithelial ovarian cancer. A prospective randomized trial. Cancer.
63:1074–1078. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
201
|
Panici PB, Greggi S, Amoroso M, Scambia G,
Battaglia FA, Gebbia V, Salerno G, Paratore MP and Mancuso S: A
combination of platinum and tamoxifen in advanced ovarian cancer
failing platinum-based chemotherapy: Results of a Phase II study.
Int J Gynecol Cancer. 11:438–444. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
202
|
Markman M, Webster K, Zanotti K, Peterson
G, Kulp B and Belinson J: Phase 2 trial of carboplatin plus
tamoxifen in platinum-resistant ovarian cancer and primary
carcinoma of the peritoneum. Gynecol Oncol. 94:404–408. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
203
|
Wagner U, du Bois A, Pfisterer J, Huober
J, Loibl S, Lück HJ, Sehouli J, Gropp M, Stähle A, Schmalfeldt B,
et al: Gefitinib in combination with tamoxifen in patients with
ovarian cancer refractory or resistant to platinum-taxane based
therapy-a phase II trial of the AGO Ovarian Cancer Study Group
(AGO-OVAR 2.6). Gynecol Oncol. 105:132–137. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
204
|
Bowman A, Gabra H, Langdon SP, Lessells A,
Stewart M, Young A and Smyth JF: CA125 response is associated with
estrogen receptor expression in a phase II trial of letrozole in
ovarian cancer: Identification of an endocrine-sensitive subgroup.
Clin Cancer Res. 8:2233–2239. 2002.PubMed/NCBI
|
|
205
|
Colon-Otero G, Weroha SJ, Foster NR,
Haluska P, Hou X, Wahner-Hendrickson AE, Jatoi A, Block MS, Dinh
TA, Robertson MW and Copland JA: Phase 2 trial of everolimus and
letrozole in relapsed estrogen receptor-positive high-grade ovarian
cancers. Gynecol Oncol. 146:64–68. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
206
|
del Carmen MG, Fuller AF, Matulonis U,
Horick NK, Goodman A, Duska LR, Penson R, Campos S, Roche M and
Seiden MV: Phase II trial of anastrozole in women with asymptomatic
müllerian cancer. Gynecol Oncol. 91:596–602. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
207
|
Kavanagh JJ, Hu W, Fu S, Deavers M, Moore
C, Coleman RL, Levenback CF, Shen D, Zheng HG, YF Y, et al:
Anti-tumor activity of letrozole in patients with recurrent
advanced low malignant potential or low-grade serous ovarian
tumors. J Clin Oncol. 25 (Suppl 18):S55822007. View Article : Google Scholar : PubMed/NCBI
|
|
208
|
Krasner CN, Debernardo RL, Findley M,
Penson R, Matulonis U, Atkinson T, Roche M and Seiden MV: Phase II
trial of anastrazole in combination with gefitinib in women with
asymptomatic mullerian cancer. J Clin Oncol. 23 (Suppl
16):S50632005. View Article : Google Scholar
|
|
209
|
Papadimitriou CA, Markaki S, Siapkaras J,
Vlachos G, Efstathiou E, Grimani I, Hamilos G, Zorzou M and
Dimopoulos MA: Hormonal therapy with letrozole for relapsed
epithelial ovarian cancer. Long-term results of a phase II study.
Oncology. 66:112–117. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
210
|
Ramirez PT, Schmeler KM, Milam MR,
Slomovitz BM, Smith JA, Kavanagh JJ, Deavers M, Levenback C,
Coleman RL and Gershenson DM: Efficacy of letrozole in the
treatment of recurrent platinum- and taxane-resistant high-grade
cancer of the ovary or peritoneum. Gynecol Oncol. 110:56–59. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
211
|
Smyth JF, Gourley C, Walker G, MacKean MJ,
Stevenson A, Williams AR, Nafussi AA, Rye T, Rye R, Stewart M, et
al: Antiestrogen therapy is active in selected ovarian cancer
cases: The use of letrozole in estrogen receptor-positive patients.
Clin Cancer Res. 13:3617–3622. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
212
|
Tchekmedyian NS, Liem AK, Quan ET, Burtzo
DM and Ucar K: Aromatase inhibitor therapy for estrogen receptor
positive ovarian cancer. J Clin Oncol. 24 (Suppl 18):S150382006.
View Article : Google Scholar : PubMed/NCBI
|
|
213
|
Verma S, Alhayki M, Le T, Baines K,
Rambout L, Hopkins L and Fung Kee Fung M: Phase II study of
exemestane (E) in refractory ovarian cancer (ROC). J Clin Oncol. 24
(Suppl 18):S50262006. View Article : Google Scholar
|
|
214
|
Williams C, Simera I and Bryant A:
Tamoxifen for relapse of ovarian cancer. Cochrane Database Syst
Rev. 2010:CD0010342010.PubMed/NCBI
|
|
215
|
Perez-Gracia JL and Carrasco EM: Tamoxifen
therapy for ovarian cancer in the adjuvant and advanced settings:
Systematic review of the literature and implications for future
research. Gynecol Oncol. 84:201–209. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
216
|
Paleari L, Gandini S, Provinciali N,
Puntoni M, Colombo N and DeCensi A: Clinical benefit and risk of
death with endocrine therapy in ovarian cancer: A comprehensive
review and meta-analysis. Gynecol Oncol. 146:504–513. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
217
|
Kok PS, Beale P, O'Connell RL, Grant P,
Bonaventura T, Scurry J, Antill Y, Goh J, Sjoquist K, DeFazio A, et
al: PARAGON (ANZGOG-0903): A phase 2 study of anastrozole in
asymptomatic patients with estrogen and progesterone
receptor-positive recurrent ovarian cancer and CA125 progression. J
Gynecol Oncol. 30:e862019. View Article : Google Scholar : PubMed/NCBI
|
|
218
|
Stanley B, Hollis RL, Nunes H, Towler JD,
Yan X, Rye T, Dawson C, Mackean MJ, Nussey F, Churchman M, et al:
Endocrine treatment of high grade serous ovarian carcinoma;
quantification of efficacy and identification of response
predictors. Gynecol Oncol. 152:278–285. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
219
|
Gershenson DM, Bodurka DC, Coleman RL, Lu
KH, Malpica A and Sun CC: Hormonal maintenance therapy for women
with low-grade serous cancer of the ovary or peritoneum. J Clin
Oncol. 35:1103–1111. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
220
|
Fader AN, Bergstrom J, Jernigan A, Tanner
EJ III, Roche KL, Stone RL, Levinson KL, Ricci S, Wethingon S, Wang
TL, et al: Primary cytoreductive surgery and adjuvant hormonal
monotherapy in women with advanced low-grade serous ovarian
carcinoma: Reducing overtreatment without compromising survival?
Gynecol Oncol. 147:85–91. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
221
|
Slomovitz B, Gourley C, Carey MS, Malpica
A, Shih IM, Huntsman D, Fader AN, Grisham RN, Schlumbrecht M, Sun
CC, et al: Low-grade serous ovarian cancer: State of the science.
Gynecol Oncol. 156:715–725. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
222
|
Yang A, Curtin J and Muggia F: Ovarian
adult-type granulosa cell tumor: Focusing on endocrine-based
therapies. Int J Endocrine Oncol. 52018.doi: 10.2217/ije-2017-0021.
PubMed/NCBI
|
|
223
|
van Meurs HS, van Lonkhuijzen LR, Limpens
J, van der Velden J and Buist MR: Hormone therapy in ovarian
granulosa cell tumors: A systematic review. Gynecol Oncol.
134:196–205. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
224
|
Graham JD and Clarke CL: Physiological
action of progesterone in target tissues. Endocr Rev. 18:502–519.
1997. View Article : Google Scholar : PubMed/NCBI
|
|
225
|
Diep CH, Daniel AR, Mauro LJ, Knutson TP
and Lange CA: Progesterone action in breast, uterine, and ovarian
cancers. J Mol Endocrinol. 54:R31–R53. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
226
|
Mesiano S, Wang Y and Norwitz ER:
Progesterone receptors in the human pregnancy uterus: Do they hold
the key to birth timing? Reprod Sci. 18:6–19. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
227
|
Conneely OM, Mulac-Jericevic B and Lydon
JP: Progesterone- dependent regulation of female reproductive
activity by two distinct progesterone receptor isoforms. Steroids.
68:771–778. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
228
|
Diep CH, Charles NJ, Gilks CB, Kalloger
SE, Argenta PA and Lange CA: Progesterone receptors induce
FOXO1-dependent senescence in ovarian cancer cells. Cell Cycle.
12:1433–1449. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
229
|
Akahira J, Inoue T, Suzuki T, Ito K, Konno
R, Sato S, Moriya T, Okamura K, Yajima A and Sasano H: Progesterone
receptor isoforms A and B in human epithelial ovarian carcinoma:
Immunohistochemical and RT-PCR studies. Br J Cancer. 83:1488–1494.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
230
|
Akahira J, Suzuki T, Ito K, Kaneko C,
Darnel AD, Moriya T, Okamura K, Yaegashi N and Sasano H:
Differential expression of progesterone receptor isoforms A and B
in the normal ovary, and in benign, borderline, and malignant
ovarian tumors. Jpn J Cancer Res. 93:807–815. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
231
|
Lenhard M, Tereza L, Heublein S, Ditsch N,
Himsl I, Mayr D, Friese K and Jeschke U: Steroid hormone receptor
expression in ovarian cancer: Progesterone receptor B as prognostic
marker for patient survival. BMC Cancer. 12:5532012. View Article : Google Scholar : PubMed/NCBI
|
|
232
|
Havrilesky LJ, Moorman PG, Lowery WJ,
Gierisch JM, Coeytaux RR, Urrutia RP, Dinan M, McBroom AJ,
Hasselblad V, Sanders GD and Myers ER: Oral contraceptive pills as
primary prevention for ovarian cancer: A systematic review and
meta-analysis. Obstet Gynecol. 122:139–147. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
233
|
Troisi R, Bjørge T, Gissler M, Grotmol T,
Kitahara CM, Myrtveit Saether SM, Sköld C, Sørensen HT, Trabert B
and Glimelius I: The role of pregnancy, perinatal factors and
hormones in maternal cancer risk: A review of the evidence. J
Intern Med. 283:430–445. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
234
|
Han KH, Kim MK, Kim HS, Chung HH and Song
YS: Protective effect of progesterone during pregnancy against
ovarian cancer. J Cancer Prev. 18:113–122. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
235
|
Eisenhofer G, Peitzsch M, Kaden D, Langton
K, Pamporaki C, Masjkur J, Tsatsaronis G, Mangelis A, Williams TA,
Reincke M, et al: Reference intervals for plasma concentrations of
adrenal steroids measured by LC-MS/MS: Impact of gender, age, oral
contraceptives, body mass index and blood pressure status. Clin
Chim Acta. 470:115–124. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
236
|
Ji J, Försti A, Sundquist J and Hemminki
K: Risks of breast, endometrial, and ovarian cancers after twin
births. Endocr Relat Cancer. 14:703–711. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
237
|
Peluso JJ: Progesterone signaling mediated
through progesterone receptor membrane component-1 in ovarian cells
with special emphasis on ovarian cancer. Steroids. 76:903–909.
2011.PubMed/NCBI
|
|
238
|
Wu NY, Huang HS, Chao TH, Chou HM, Fang C,
Qin CZ, Lin CY, Chu TY and Zhou HH: Progesterone prevents
high-grade serous ovarian cancer by inducing necroptosis of
p53-defective fallopian tube epithelial cells. Cell Rep.
18:2557–2565. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
239
|
Pelegrina LT, de Los Ángeles Sanhueza M,
Ramona Cáceres AR, Cuello-Carrión D, Rodriguez CE and Laconi MR:
Effect of progesterone and first evidence about allopregnanolone
action on the progression of epithelial human ovarian cancer cell
lines. J Steroid Biochem Mol Biol. 196:1054922020. View Article : Google Scholar : PubMed/NCBI
|
|
240
|
McGlorthan L, Paucarmayta A, Casablanca Y,
Maxwell GL and Syed V: Progesterone induces apoptosis by activation
of caspase-8 and calcitriol via activation of caspase-9 pathways in
ovarian and endometrial cancer cells in vitro. Apoptosis.
26:184–194. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
241
|
Xie YL, Yang YJ, Tang C, Sheng HJ, Jiang
Y, Han K and Ding LJ: Estrogen combined with progesterone decreases
cell proliferation and inhibits the expression of Bcl-2 via
microRNA let-7a and miR-34b in ovarian cancer cells. Clin Transl
Oncol. 16:898–905. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
242
|
Lee JY, Shin JY, Kim HS, Heo JI, Kho YJ,
Kang HJ, Park SH and Lee JY: Effect of combined treatment with
progesterone and tamoxifen on the growth and apoptosis of human
ovarian cancer cells. Oncol Rep. 27:87–93. 2012.PubMed/NCBI
|
|
243
|
Shin YY, Kang EJ, Jeong JS, Kim MJ, Jung
EM, Jeung EB and An BS: Pregnenolone as a potential candidate for
hormone therapy for female reproductive disorders targeting ERβ.
Mol Reprod Dev. 86:109–117. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
244
|
Pedernera E, Gómora MJ, Morales-Vásquez F,
Pérez-Montiel D and Mendez C: Progesterone reduces cell survival in
primary cultures of endometrioid ovarian cancer. J Ovarian Res.
12:152019. View Article : Google Scholar : PubMed/NCBI
|
|
245
|
Peluso JJ, Liu X, Saunders MM, Claffey KP
and Phoenix K: Regulation of ovarian cancer cell viability and
sensitivity to cisplatin by progesterone receptor membrane
component-1. J Clin Endocrinol Metab. 93:1592–1599. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
246
|
Gamarra-Luques CD, Goyeneche AA, Hapon MB
and Telleria CM: Mifepristone prevents repopulation of ovarian
cancer cells escaping cisplatin-paclitaxel therapy. BMC Cancer.
12:2002012. View Article : Google Scholar : PubMed/NCBI
|
|
247
|
Gamarra-Luques CD, Hapon MB, Goyeneche AA
and Telleria CM: Resistance to cisplatin and paclitaxel does not
affect the sensitivity of human ovarian cancer cells to
antiprogestin-induced cytotoxicity. J Ovarian Res. 7:452014.
View Article : Google Scholar : PubMed/NCBI
|
|
248
|
Telleria CM: Repopulation of ovarian
cancer cells after chemotherapy. Cancer Growth Metastasis. 6:15–21.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
249
|
Mangioni C, Franceschi S, Vecchia CL and
D'Incalci M: High-dose medroxyprogesterone acetate (MPA) in
advanced epithelial ovarian cancer resistant to first- or
second-line chemotherapy. Gynecol Oncol. 12:314–318. 1981.
View Article : Google Scholar : PubMed/NCBI
|
|
250
|
Slayton RE, Pagano M and Creech RH:
Progestin therapy for advanced ovarian cancer: A phase II Eastern
Cooperative Oncology Group trial. Cancer Treat Rep. 65:895–896.
1981.PubMed/NCBI
|
|
251
|
Aabo K, Pedersen AG, Haid I and
Dombernowsky P: High-dose medroxyprogesterone acetate (MPA) in
advanced chemotherapy-resistant ovarian carcinoma: A phase II
study. Cancer Treat Rep. 66:407–408. 1982.PubMed/NCBI
|
|
252
|
Tropé C, Johnsson JE, Sigurdsson K and
Simonsen E: High-dose medroxyprogesterone acetate for the treatment
of advanced ovarian carcinoma. Cancer Treat Rep. 66:1441–1443.
1982.PubMed/NCBI
|
|
253
|
Hamerlynck JV, Maskens AP, Mangioni C, van
der Burg ME, Wils JA, Vermorken JB and Rotmensz N: Phase II trial
of medroxyprogesterone acetate in advanced ovarian cancer: An EORTC
Gynecological Cancer Cooperative Group Study. Gynecol Oncol.
22:313–316. 1985. View Article : Google Scholar : PubMed/NCBI
|
|
254
|
Malfetano J, Beecham JB, Bundy BN and
Hatch KD: A phase II trial of medroxyprogesterone acetate in
epithelial ovarian cancers. A Gynecologic Oncology Group study. Am
J Clin Oncol. 16:149–151. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
255
|
Geisler HE: The use of high-dose megestrol
acetate in the treatment of ovarian adenocarcinoma. Semin Oncol. 12
(Suppl 1):S20–S22. 1985.
|
|
256
|
Sikic BI, Scudder SA, Ballon SC, Soriero
OM, Christman JE, Suey L, Ehsan MN, Brandt AE and Evans TL:
High-dose megestrol acetate therapy of ovarian carcinoma: A phase
II study by the Northern California Oncology Group. Semin Oncol. 13
(Suppl 4):S26–S32. 1986.PubMed/NCBI
|
|
257
|
Belinson JL, McClure M and Badger G:
Randomized trial of megestrol acetate vs. megestrol
acetate/tamoxifen for the management of progressive or recurrent
epithelial ovarian carcinoma. Gynecol Oncol. 28:151–155. 1987.
View Article : Google Scholar : PubMed/NCBI
|
|
258
|
Veenhof CH, van der Burg ME, Nooy M,
Aalders JG, Pecorelli S, Oliveira CF, Rotmensz N and Vermorken JB:
Phase II study of high-dose megestrol acetate in patients with
advanced ovarian carcinoma. Eur J Cancer. 30A:697–698. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
259
|
Wiernik PH, Greenwald ES, Ball H, Young JA
and Vogl S: High-dose megestrol acetate in the treatment of
patients with ovarian cancer who have undergone previous treatment:
Eastern Cooperative Oncology Group Study PD884. Am J Clin Oncol.
21:565–567. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
260
|
Wilailak S, Linasmita V and Srisupundit S:
Phase II study of high-dose megestrol acetate in
platinum-refractory epithelial ovarian cancer. Anticancer Drugs.
12:719–724. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
261
|
Rocereto TF, Saul HM, Aikins JA Jr and
Paulson J: Phase II study of mifepristone (RU486) in refractory
ovarian cancer. Gynecol Oncol. 77:429–432. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
262
|
Rocereto TF, Brady WE, Shahin MS, Hoffman
JS, Small L, Rotmensch J and Mannel RS: A phase II evaluation of
mifepristone in the treatment of recurrent or persistent epithelial
ovarian, fallopian or primary peritoneal cancer: A gynecologic
oncology group study. Gynecol Oncol. 116:332–334. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
263
|
Niwa K, Onogi K, Wu Y, Mori H, Harrigan RC
and Tamaya T: Clinical implication of medroxyprogesterone acetate
against advanced ovarian carcinoma: A pilot study. Eur J Gynaecol
Oncol. 29:252–255. 2008.PubMed/NCBI
|
|
264
|
Zheng H, Kavanagh JJ, Hu W, Liao Q and Fu
S: Hormonal therapy in ovarian cancer. Int J Gynecol Cancer.
17:325–338. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
265
|
Laurelli G, Di Vagno G, Scaffa C, Losito
S, Del Giudice M and Greggi S: Conservative treatment of early
endometrial cancer: Preliminary results of a pilot study. Gynecol
Oncol. 120:43–46. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
266
|
Bonaventura A, O'Connell RL, Mapagu C,
Beale PJ, McNally OM, Mileshkin LR, Grant PT, Hadley AM, Goh JCH,
Sjoquist KM, et al: Paragon (ANZGOG-0903): Phase 2 study of
anastrozole in women with estrogen or progesterone
receptor-positive platinum-resistant or -refractory recurrent
ovarian cancer. Int J Gynecol Cancer. 27:900–906. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
267
|
Freedman RS, Saul PB, Edwards CL, Jolles
CJ, Gershenson DM, Jones LA, Atkinson EN and Dana WJ: Ethinyl
estradiol and medroxyprogesterone acetate in patients with
epithelial ovarian carcinoma: A phase II study. Cancer Treat Rep.
70:369–373. 1986.PubMed/NCBI
|
|
268
|
Greiser CM, Greiser EM and Dören M:
Menopausal hormone therapy and risk of ovarian cancer: Systematic
review and meta-analysis. Hum Reprod Update. 13:453–463. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
269
|
Beral V, Bull D, Green J and Reeves G:
Ovarian cancer and hormone replacement therapy in the Million Women
Study. Lancet. 369:1703–1710. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
270
|
Koskela-Niska V, Lyytinen H, Riska A,
Pukkala E and Ylikorkala O: Ovarian cancer risk in postmenopausal
women using estradiol-progestin therapy-a nationwide study.
Climacteric. 16:48–53. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
271
|
Trabert B, Wentzensen N, Yang HP, Sherman
ME, Hollenbeck A, Danforth KN, Park Y and Brinton LA: Ovarian
cancer and menopausal hormone therapy in the NIH-AARP diet and
health study. Br J Cancer. 107:1181–1187. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
272
|
Morch LS, Lokkegaard E, Andreasen AH,
Kjaer SK and Lidegaard O: Hormone therapy and different ovarian
cancers: A national cohort study. Am J Epidemiol. 175:1234–1242.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
273
|
Liu Y, Ma L, Yang X, Bie J, Li D, Sun C,
Zhang J, Meng Y and Lin J: Menopausal hormone replacement therapy
and the risk of ovarian cancer: A meta-analysis. Front Endocrinol
(Lausanne). 10:8012019. View Article : Google Scholar : PubMed/NCBI
|
|
274
|
Beral V, Gaitskell K, Hermon C, Moser K,
Reeves G and Peto R: Menopausal hormone use and ovarian cancer
risk: Individual participant meta-analysis of 52 epidemiological
studies. Lancet. 385:1835–1842. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
275
|
Wen Y, Huang H, Huang H, Wu M, Shen K and
Pan L: The safety of postoperative hormone replacement therapy in
epithelial ovarian cancer patients in China. Climacteric.
16:673–681. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
276
|
Li D, Ding CY and Qiu LH: Postoperative
hormone replacement therapy for epithelial ovarian cancer patients:
A systematic review and meta-analysis. Gynecol Oncol. 139:355–362.
2015. View Article : Google Scholar : PubMed/NCBI
|