Introduction
Mitochondria provides energy for almost all cellular
activities, thereby maintaining the cell's physiological functions
(1). In addition, mitochondria play
a vital role in cell physiological processes such as cell growth,
cell division, innate immunity, calcium homeostasis, stem cell
reprogramming, energy metabolism, and apoptosis (1,2).
Mitochondria can adjust their tubular network through morphological
changes, and fission and fusion affect the size and number of
mitochondria, which is termed mitochondrial dynamics (3).
Mitochondrial fission is primarily driven by
Dynein-related protein 1 (Drp1) (4), which is located in the cytoplasm under
physiological conditions. The activated Drp1 translocates to the
outer mitochondrial membrane, where it binds to its mitochondrial
adaptors (including FIS1 and MFF) to promote fission events
(5). In certain cases, cancer cells
exhibit high levels of fission mitochondrial fragmentation and
enhanced function, which leads to tumor growth and the development
of chemoresistance (6). The
activation and upregulation of Drp1 have been observed in various
types of cancer, such as lung cancer, breast cancer, glioblastoma,
colorectal cancer, pancreatic cancer, thyroid tumor, nasopharyngeal
carcinoma, and melanoma (7–13). Previous studies have found that the
loss of Drp1 leads to prolongation of the mitochondrial network,
inhibition of cell proliferation, and induction of spontaneous
apoptosis in a variety of cancer cells (6,14).
However, the role of Drp1 in bladder cancer remains unclear.
The ubiquitin-proteasome pathway is the primary
mechanism of protein catabolism in the cytoplasm and nucleus of
mammals and is involved in the degradation of >80% of the
proteins in the cell (15). The
deubiquitinating enzyme (DUB) catalyzes the process of removing
ubiquitin from ubiquitinated substrates, which is termed
deubiquitylation (16). Due to the
wide range of substrates, deubiquitylation plays an important role
in a variety of cellular functions. Previous studies have reported
that deubiquitylation dysregulation may the induction of various
diseases, including cancer (17,18).
As a subunit of the 19S regulatory particle in the 26S proteasome,
the 26S proteasome non-ATPase regulatory subunit 14 (PSMD14)
belongs to the JAB1/MPN+/MOV34 (JAMM) domain protease family of DUB
proteins and has been reported to be involved in the occurrence and
development of various types of cancer (19). Downregulation of PSMD14 can induce
G0/G1 phase arrest, and apoptosis, and reduce the proliferation and
epithelial-mesenchymal transition (EMT) of lung adenocarcinoma,
prostate cancer, and breast cancer cells (20–24). A
previous study found that PSMD14 exerts carcinogenic effects in
head and neck squamous cell carcinoma and glioma by inhibiting the
ubiquitination and degradation of cancer-related transcription
factor E2F1 (25,26). PSMD14 also promotes EMT in
esophageal squamous cell carcinoma by targeting SNAIL for
deubiquitylation and stabilization (27). Therefore, PSMD14 acts as an
oncogene, deubiquitinating a variety of protein substrates.
However, the function and mechanism of PSMD14 in bladder cancer
remain to be explored.
In the present study, it was found that PSMD14
functions upstream of Drp1 and is upregulated in patients with
bladder cancer, which indicated a potential mechanism of PSMD14 and
mitochondrial fission in cancer. The results of the present study
revealed the importance of PSMD14 in tumorigenesis and a novel
treatment strategy to attenuate mitochondrial fission by inhibiting
the PSMD14-Drp1 pathway.
Materials and methods
Clinical tissues
This study was approved by the Institutional Ethics
Committee of the Hunan Provincial People's Hospital and in
accordance with the Ethical Management Guidelines of Hunan
Provincial People's Hospital (approval no. 210139). A total of 32
bladder cancer tissue specimens and matched normal specimens were
collected from Hunan Provincial People's Hospital. Informed written
consent was obtained from all the participants.
Cell culture
T24 and 5637 cells (American Type Culture
Collection) were cultured in RPMI-1640 medium (Thermo Fisher
Scientific, Inc.) supplemented with 10% FBS, 100 mg/ml
streptomycin, and 100 units of penicillin. All the cells were
cultured in a humidified incubator at 37°C, supplied with 5%
CO2, 21% O2, and 74% N2.
siRNA transfections
Cells were first seeded in six-well plates and
cultured until they reached 30–50% confluence. siRNAs were
introduced to the cells through transfection. Cells were
transfected with Lipofectamine® 3000 Transfection
Reagent (Beyotime Institute of Biotechnology). For the present
study, six groups of cells were used: i) Lipofectamine®
3000 Transfection Reagent alone; ii) Lipofectamine® 3000
Transfection Reagent combined with 50 nM/µl NC siRNA; iii)
Lipofectamine® 3000 Transfection Reagent combined with
50 nM/µl si-PSMD14 (5′-AAGGTTGTTATTGATGCCTTCAGAT-3′); iv)
Lipofectamine® 3000 Transfection Reagent combined with
50 nM/µl si-NC for PSMD14 (5′-AAGTTGTGTTACGTACTTCATGGAT-3′); v)
Lipofectamine® 3000 Transfection Reagent combined with
50 nM/µl si-Drp1 (5′-AAGTGGTGACTTGTCTTCTTCGTAA-3′); vi)
Lipofectamine® 3000 Transfection Reagent combined with
or 50 nM/µl si-NC for DRP1 (5′-AAGAGTGGTTCTTCTCTTCGGTTAA-3′). Cells
were transfected for 24 h before subsequent experiments. Drp1
agonist Mdivi-1 (50 mg/ml; Beyotime Institute of Biotechnology) was
also used to manipulate Drp expression. The manufacturer's
instructions were followed during the transfection process, and the
efficiency of the transfection was confirmed using qPCR.
Immunohistochemical staining
The bladder tissue sections were treated first by
removing the paraffin using xylene and ethanol, and then performing
antigen retrieval. Following this, tissues were blocked using 3%
BSA and then stained using two different types of antibodies
anti-PSMD14 (cat. no. 4197S; Cell Signaling Technology, Inc.;
1:100) or anti-Drp1 (cat. no. 8570S; Cell Signaling Technology,
Inc.; 1:100) at 4°C overnight. To ensure the reliability of the
results, a control was also included by using mouse serum at the
same protein concentration as the antibody solution. Under a light
microscope at an ×100 magnification, the positively stained tissue
sections could be seen as appearing brown or tawny-brown in color.
Images of the tissue were taken using an optical microscope, and
the integrated optical density (IOD) values of the tissue sections
were then measured using Image-Pro Plus version 6.0 (Media
Cybernetics, Inc.). To ensure accuracy, five different fields of
view were selected from each tissue section, and three tissue
sections were used for each group to determine the positive IOD
values. The mean IOD values were then used to calculate the
relative expressions of PSMD14 and Drp1.
Immunofluorescent staining
Cells that had been grown on slides were fixed with
4% paraformaldehyde solution for 15 min at room temperature, and
then permeabilized with 0.1% Triton X-100 at room temperature for
15 min. The samples were washed three times with PBS and then
sealed with a 5% BSA solution at room temperature for 45 min. Next,
the primary antibody was incubated with the sample overnight at
4°C. The primary antibodies used were: Anti-PSMD14 (cat. no. 4197S;
1:200; Cell Signaling Technology, Inc.), anti-Drp1 (cat. no. 8570S;
1:200; Cell Signaling Technology, Inc.), or anti-TOM20 (cat. no.
42406S; 1:200; Cell Signaling Technology, Inc.). DAPI (cat. no.
4083S; Cell Signaling Technology, Inc. 1:200) was used to
counterstain the nuclei. The secondary antibodies included Alexa
FluoR 555 goat anti-rabbit (cat. no. A27017; 1:400; Invitrogen;
Thermo Fisher Scientific, Inc.). A total of five different fields
of view were selected and randomly scored using a fluorescence
microscope (magnification, ×40, Olympus Corporation).
Western blotting
Cells were collected and lysed using RIPA buffer
(Beyotime Institute of Biotechnology) for 30 min at 4°C. The
nuclear protein was collected using cytoplasmic and nuclear
extraction kit (cat. no. A37487; Invitrogen; Thermo Fisher
Scientific, Inc.). The protein lysates were quantified by BCA
Protein Assay Kit (Beyotime Institute of Biotechnology). SDS gels
(8–12%) were used for SDS-PAGE. The resolved proteins were then
transferred to PVDF. To block the membranes, a 10% BSA solution was
dissolved in TBS buffer containing 0.05% Tween 20 and added to the
membranes at room temperature for 1 h. The primary antibody was
then added to TBS containing 0.05% Tween 20 and incubated for at
least 10 h at 4°C. The primary antibodies used were anti-PSMD14
(cat. no. 4197S; Cell Signaling Technology, Inc.; 1:1,000) and
anti-Drp1 (cat. no. 8570S; Cell Signaling Technology, Inc.;
1:1,000). To ensure accuracy, the protein content was normalized
using GAPDH (cat. no. 5174S; Cell Signaling Technology, Inc.;
1:8,000) as the internal control. After washing the membrane three
times, the anti-rabbit second antibody (cat. no. 7074S; Cell
Signaling Technology, Inc.; 1:8,000) and anti-mouse mice secondary
antibody (cat. no. 7076S; Cell Signaling Technology, Inc.; 1:8,000)
in TBS containing 0.05% Tween 20 were used at room temperature for
1 h. After three additional washes, the bands were detected by
enhanced chemiluminescence and autoradiography. The experiments
were repeated three times, and the results were evaluated using
Image Lab 6.0 (Bio-Rad Laboratories, Inc.). Drp1 agonist Mdivi-1
(50 mg/ml; Beyotime Institute of Biotechnology) was also used to
manipulate the expression of Drp1. The blots were grouped and
cropped from the same gel.
RNA extraction and qPCR
Total RNA was extracted using TRIzol®
reagent (Invitrogen; Thermo Fisher Scientific, Inc.) and RNA purity
was assessed using a spectrophotometer. First-strand cDNA was
synthesized using a cDNA synthesis kit (Promega Corporation) at
65°C for 5 min according to the manufacturer's protocol.
Subsequently, cDNA was amplified by qPCR using an Applied
Biosystems SYBR Green mix kit and the ABI 7900 Real-Time PCR system
(both Applied Biosystems; Thermo Fisher Scientific, Inc.) following
the manufacturer's instructions. qPCR was performed as follows:
Denaturation at 94°C for 4 min; followed by 40 cycles of
amplification at 94°C for 30 sec, hybridization at 56°C for 30 sec,
and extension at 72°C for 30 sec. Quantification of mRNA was
automatically conducted using SDS version 1.3 software (Applied
Biosystems; Thermo Fisher Scientific, Inc.), and the
2−∆∆Cq method was employed to calculate mRNA expression
(28,29). The sequences of the primers used
were: PSMD14 forward, 5′-GTCAGTGTGGAGGCAGTTGATC-3′, and reverse,
5′-CCACACCAGAAAGCCAACAACC-3′; DRP1 forward,
5′-TCACCCGGAGACCTCTCATTC-3′ and reverse,
5′-GGTTCAGGGCTTACTCCCTTAT-3′, and β-actin forward,
5′-CGAGCGCGGCTACAGCTT-3′ and reverse,
5′-TCCTTAATGTCACGCACGATTT-3′.
Cell viability assay
T24 and 5637 cells in the cell culture flask were
digested with 0.25% trypsin. Then these cells were seeded in a
96-well culture plate at a density of 1×104 cells/ml for
24 h. After 24 h of incubation, 10 µl CCK-8 reagent was added to
each well for 2 h. The optical density was measured at a wavelength
of 450 nm using a Bio-Rad 680 microplate reader. Each experiment
was performed in triplicates.
In vitro ubiquitination assay
Cells were transfected with si-PSMD14. After
transfection for 48 h, the cells were treated with MG132 (15 µm;
Beyotime Institute of Biotechnology) overnight and then lysed with
RIPA lysis buffer, follow by passing 5 times through a 21-gauge
needle. Whole-cell extract (1 mg) was used for immunoprecipitation
with 1 µg anti-PSMD14 (cat. no. 4197S; Cell Signaling Technology,
Inc.) or anti-Drp1 (cat. no. 8570S; Cell Signaling Technology,
Inc.). The proteins that had been pulled down were subjected to
SDS/PAGE for immunoblot analysis.
Statistical analysis
Each experiment was repeated at least three times.
The categorical data were assessed using a Fisher's exact test, and
the quantitative data are presented as the mean ± SEM. All
statistical analyses were performed using SPSS version 19.0 (IBM
Corp.). The differences between groups were compared using a
Student's t-test or ANOVA followed by a post hoc Dunnett's test or
Tukey's test. P<0.05 was considered to indicate a statistically
significant difference.
Results
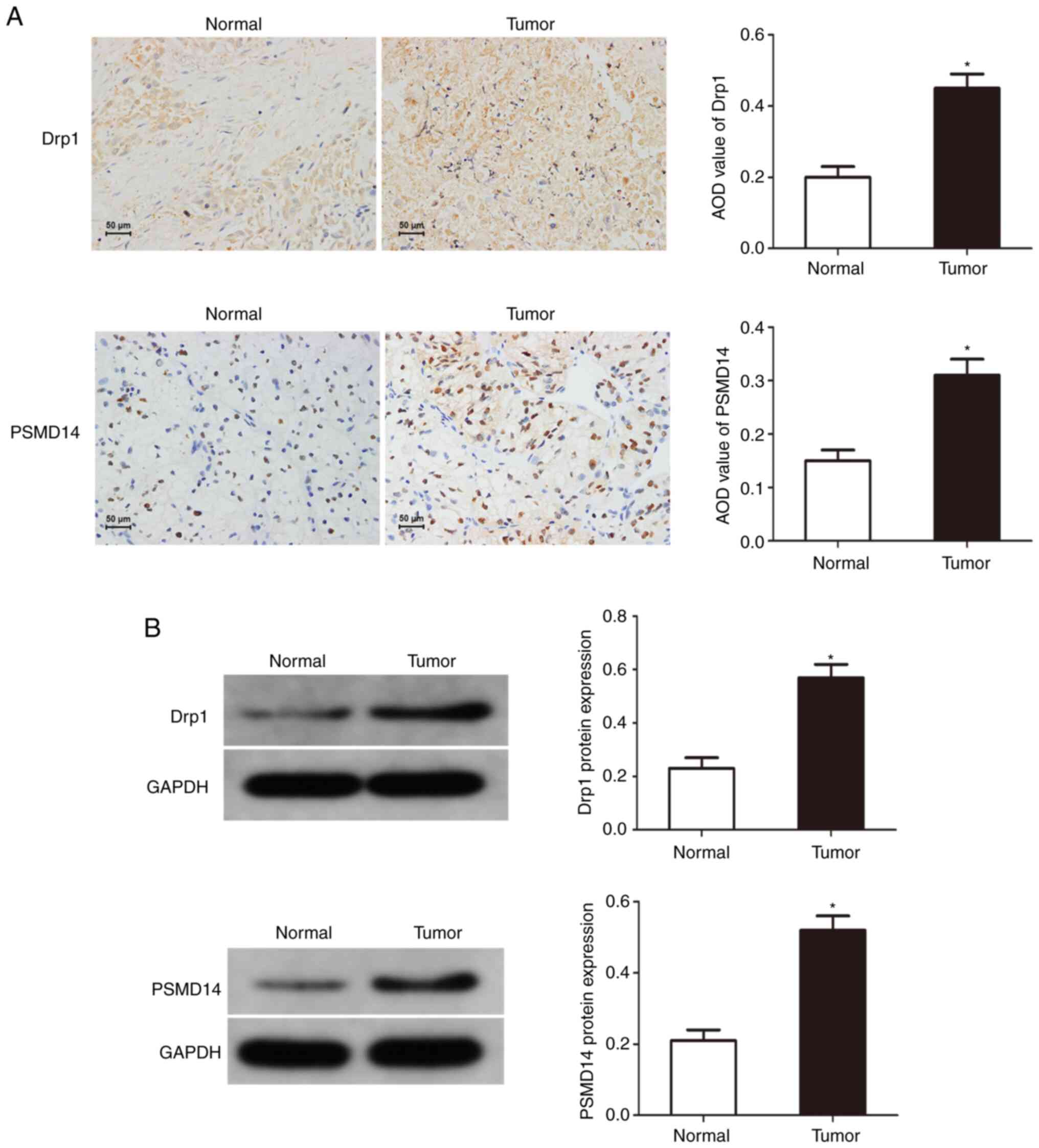
Drp1 and PSMD14 expression are
upregulated in bladder cancer
To reveal the potential function of PSMD14 in
cancer, immunohistochemical staining and western blotting were used
to determine the expression of Drp1 and PSMD14 in bladder cancer
tissues, and it was found that compared with normal tissues, Drp1
and PSMD14 expression were abnormally upregulated in bladder cancer
tissues (Fig. 1). These results
suggest that Drp1 and PSMD14 may play important roles in the
occurrence and development of bladder cancer.
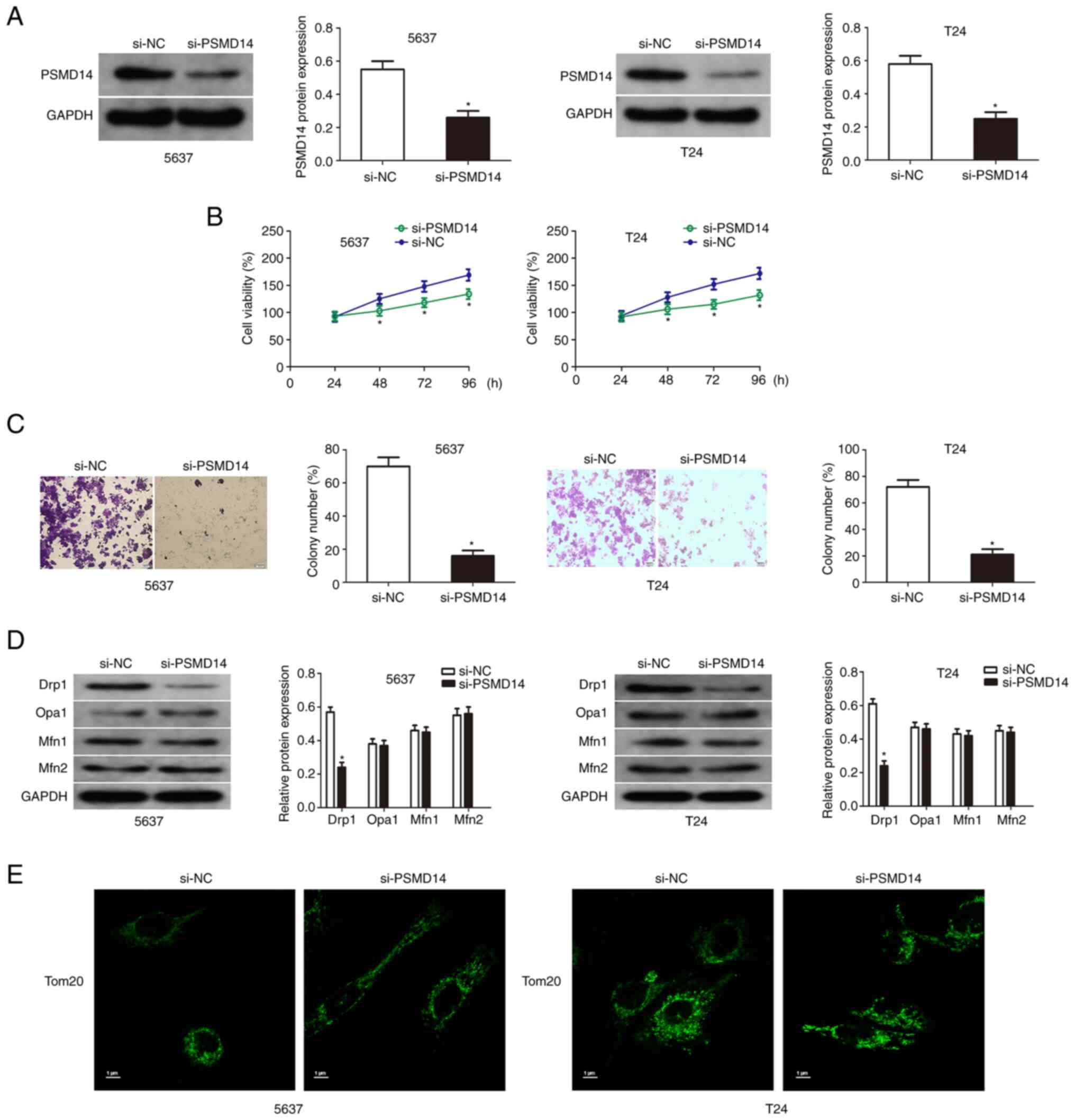
Knockdown of PSMD14 inhibits the
growth of bladder cancer cells
The expression of PSMD14 of T24 and 5637 cells
transfected with PSMD14 siRNA was detected by western blotting
(Fig. 2A). Then cell viability
assay and colony formation assays were used to detect the
proliferation and colony formation of the transfected bladder
cancer cells compared with the respective control cells. The
results showed that the knockdown of PSMD14 significantly reduced
the growth and proliferation capacity of T24 and 5637 cells
(Fig. 2B and C). Mitochondrial
fusion in the bladder cancer cells was assessed to explore the
relationship between PSMD14 and mitochondrial dynamics. PSMD14
knockdown did not affect the expression of mitochondrial fusion
factors (OPA1, Mfn1, and Mfn2) by immunofluorescent staining, and
the levels of mitochondrial factor-Drp1 were downregulated by
PSMD14 knockdown (Fig. 2D). Drp1
induced mitochondria to produce several fragments, and PSMD14
knockdown blocked this process (Fig.
2E).
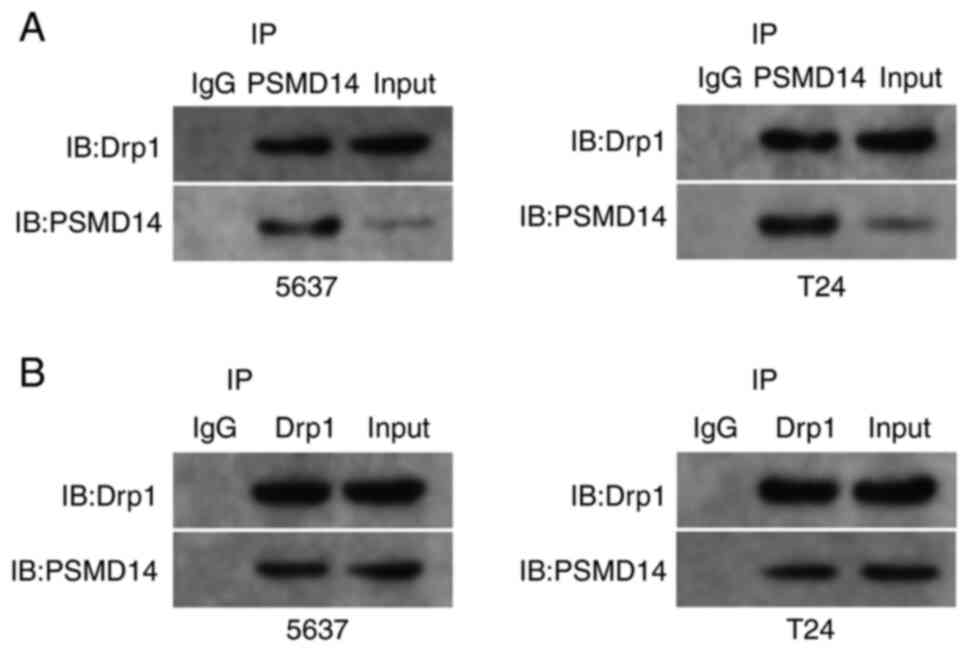
The interaction mode between PSMD14
and Drp1
Next, whether endogenous Drp1 interacted with
endogenous PSMD14 was assessed. Endogenous Drp1 and PSMD14 proteins
were co-immunoprecipitated (Fig.
3A), and this finding was confirmed through reciprocal co-IP
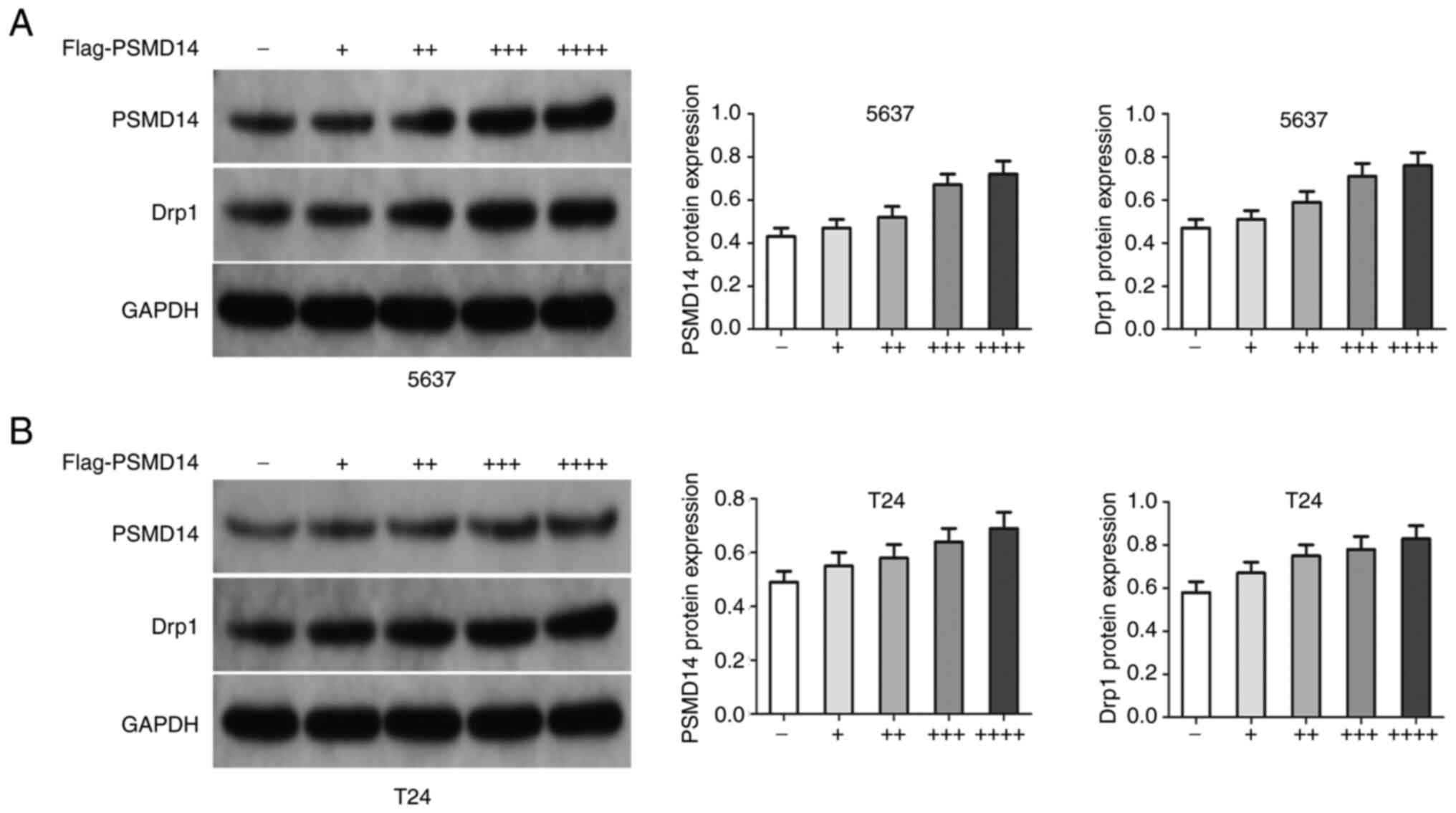
test (Fig. 3B). Then, si-PSMD14
transfected T24 and 5637 cells were treated with exogenous PSMD14.
The results showed that the PSMD14 resulted in an increase in Drp1
in T24 and 5637 cells (Fig. 4A and
B).
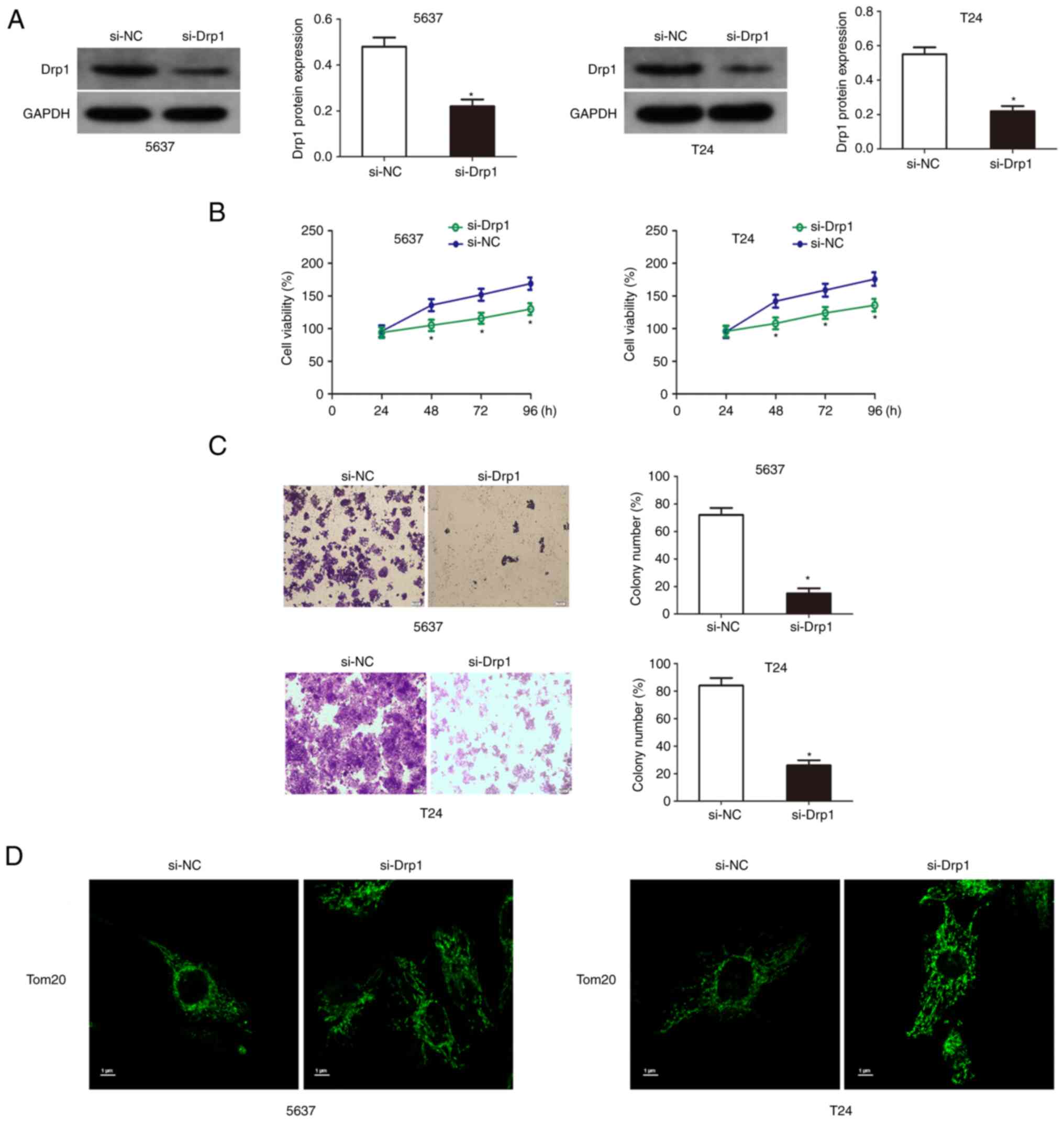
Knockdown of Drp1 inhibits the growth
of bladder cancer cells
The results of western blotting results showed that
Drp1 was significantly downregulated in T24 and 5637 cells
transfected with si-Drp1 (Fig. 5A).
Then cell viability and colony formation assays were used to detect
the proliferation and colony formation ability of Drp1 knockdown
bladder cancer cells. The results showed that the knockdown of Drp1
significantly reduced the growth and proliferation capacity of T24
and 5637 cells (Fig. 5B and C).
Additionally, Drp1 knockdown significantly increased mitochondrial
length and inhibited mitochondrial fission (Fig. 5D).
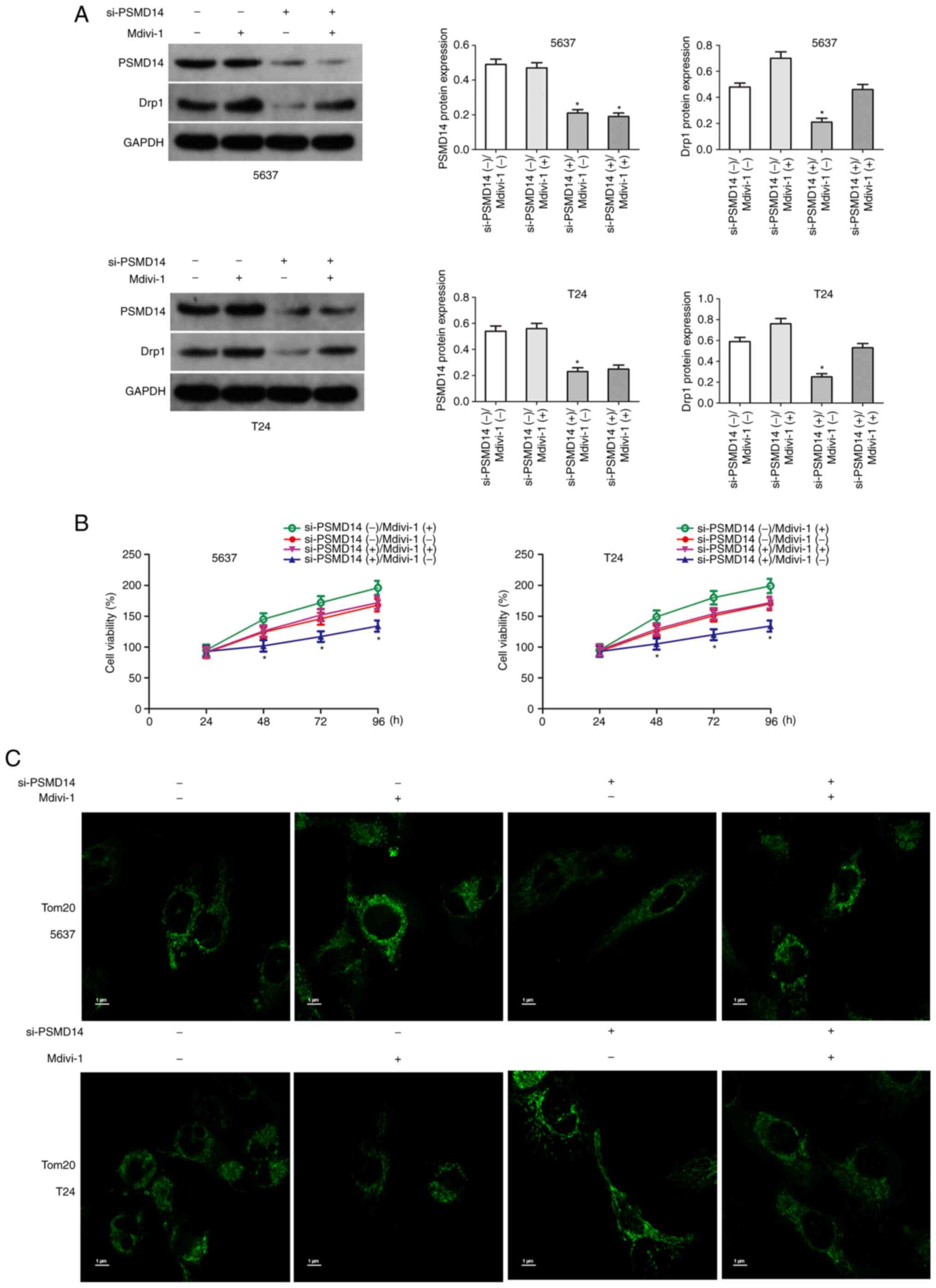
Upregulation of Drp1 partially
restores the carcinogenic effects of PSMD14
The above results confirmed that Drp1 plays an
important role in PSMD14-mediated bladder cancer. Next, whether the
upregulation of Drp1 reversed the anticancer effects mediated by
PSMD14 knockdown was assessed. Drp1 agonist Mdivi-1 (50 mg/ml) was
used to upregulate Drp1 in bladder cancer cells (Fig. 6A). Then the proliferation of
si-PSMD14 transfected bladder cancer cells treated with Mdivi-1 was
determined. The results of the cell viability assays showed that
Mdivi-1 significantly increased the growth of T24 and 5637 cells
transfected with si-PSMD14 (Fig.
6B). Mdivi-1 treatment significantly reduced mitochondrial
length and promoted mitochondrial fission (Fig. 6C).
Discussion
The ubiquitin-proteasome system is the primary
mechanism of protein catabolism in the mammalian cytoplasm and
nucleus, and it participates in the degradation of most proteins in
the cell (30). The
ubiquitin-proteasome system is modified by ubiquitination and
deubiquitylation to maintain the dynamic balance of intracellular
proteins and is one of the important balance systems for
maintaining the stability of the intracellular environment.
Additional evidence has shown that the changes in expression levels
of deubiquitinating enzymes are closely related to human diseases,
such as autoimmune diseases and tumors (31–34).
PSMD14 plays an important role under normal physiological
conditions, including the repair process of DNA damage, embryonic
cell development, cell differentiation, and cell apoptosis
(33–35). The abnormal expression of PSMD14
disrupts the dynamic balance of proteins in cells, leading to a
variety of diseases including tumors (36,37).
The results confirmed that the progression of bladder cancer is
significantly associated with upregulated expression of PSMD14.
PSMD14 can affect the malignant biological behavior of tumor cells,
such as apoptosis resistance, proliferation, chemotherapy
resistance, invasion, and metastasis by inhibiting the
ubiquitination and degradation of specific proteins. A previous
study showed that high expression of PSMD14 increased the
resistance of colon cancer cells to chemotherapeutic drugs
(38). Zhu et al (36) found that PSMD14 induced the invasion
and metastasis of esophageal cancer by regulating the
ubiquitination and degradation of the EMT transcription factor
Snail. PSMD14 reversed the ubiquitination of GRB2 and promoted cell
growth and metastasis of hepatocellular carcinoma (20). PSMD14 can also regulate the
stability of ALK2 to make colorectal cancer cells more resistant to
chemotherapy (37). PSMD14 can also
determine the fate of its substrate protein. PSMD14 promotes
substrate transfer and proteolysis by removing the ubiquitin label
and induces the release of substrate proteins from the proteasome
to avoid degradation (36,39). Therefore, an increasing number of
studies are suggesting that PSMD14 may be used as a marker in the
ubiquitin-proteasome system to determine the fate of substrates.
Here, it was found that PSMD14 knockdown can inhibit cell growth of
bladder cancer, suggesting that PSMD14 plays an important role in
the progression of bladder cancer.
Mitochondria are the central hub of cellular energy
metabolism, biosynthesis, and signal transduction, and can help
cells sense stress and adapt to the environment; thus, they play a
special role in the occurrence and development of tumors (40). Disturbances in mitochondrial
dynamics have been observed in different tumors. Mitochondrial
fusion and fission proteins of tumor cells are often abnormally
expressed, and mitochondrial morphology has also been shown to be
altered (41–43). The expression levels of the
mitochondrial division protein Drp1 were upregulated in liver
cancer, breast cancer, and lung cancer (44–46).
The increased mitochondrial fission in tumor cells suggests the
changes in mitochondrial dynamics in tumors, and the intervention
of disordered mitochondrial fusion or fission may be a potential
tumor therapeutic target for tumors (47). The present study found that Drp1
expression is upregulated, and mitochondrial fission increased
significantly in bladder cancer cells. Furthermore, PSMD14 could
deubiquitinate and stabilize Drp1.
Mitochondrial dynamics also play a vital role in the
regulation of the cell cycle. Mitra et al (48) revealed that the cycle of
mitochondrial division and fusion is highly coupled with cell cycle
progression. In the process of cell division, the mitochondria
assigned to each daughter cell are divided from those in the parent
cells. In the G1 and G2 phases, a network is formed between the
mitochondria. However, in the S phase and mitotic phase,
mitochondria are present in a highly fragmented state, which may be
due to the increased expression of Drp1 during the cell cycle,
which promotes the division of mitochondria (6). Mitra et al (48) also found that inhibiting the
expression of Drp1 in cells can induce cell cycle arrest in the G1
phase. Previous studies have confirmed that overexpression of Drp1
promotes the proliferation of cancer cells by accelerating the
transition of cells in the G1/S phase (6,49).
Kitamura et al (50) found
that Drp1 knockdown can arrest cell cycle progression of skin
squamous cell carcinoma at the G2/M phase. Studies have found that
overexpression of Drp1 can promote the proliferation of breast
cancer, lung cancer, and glioma cells; however, the underlying
mechanism has not been fully elucidated (51–53).
The present study confirmed that the upregulation of Drp1 could
reverse the anticancer effects mediated by PSMD14 knockdown. These
results have shown that the overexpression of Drp1 may be related
to tumor cell proliferation. The primary limitation of the present
study is the lack of in vivo experiments, and thus, this
will be taken into consideration in our future studies.
In conclusion, at present, the role of PSMD14 in the
occurrence and development of cancer has not been fully elucidated.
The results indicated that PSMD14 is closely related to the
occurrence and development of bladder cancer. In addition, the
present study proposes for the first time that PSMD14 mediates the
mitochondrial fission of bladder cancer cells by deubiquitinating
and stabilizing Drp1. PSMD14 as an important oncogenic protein of
bladder cancer, may thus serve as a novel anti-cancer target.
Acknowledgements
Not available.
Funding
This project is supported by the Hunan Provincial Health
Commission (grant no. 20200053), Changsha Municipal Natural Science
Foundation (grant no. kq2014096), and the Natural Science
Foundation of Hunan Province (grant no. 2022JJ70099).
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
WX contributed to the conception and design of the
study. WS performed the experiments. ZL and MX analyzed data and
wrote this manuscript. WX revised and reviewed the manuscript. WS,
MX, ZL and WX confirm the authenticity of all the raw data.
Ethics approval and consent to
participate
This study was approved by the Institutional Ethics
Committee of the Hunan Provincial People's Hospital and in
accordance with the Ethical Management Guidelines of Hunan
Provincial People's Hospital (approval no. 210139). Written
informed consent was obtained from all participants.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Zong WX, Rabinowitz JD and White E:
Mitochondria and cancer. Mol Cell. 61:667–676. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Rangaraju V, Lewis TJ Jr, Hirabayashi Y,
Bergami M, Motori E, Cartoni R, Kwon SK and Courchet J: Pleiotropic
mitochondria: The influence of mitochondria on neuronal development
and disease. J Neurosci. 39:8200–8208. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Tilokani L, Nagashima S, Paupe V and
Prudent J: Mitochondrial dynamics: Overview of molecular
mechanisms. Essays Biochem. 62:341–360. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Schmitt K, Grimm A, Dallmann R,
Oettinghaus B, Restelli LM, Witzig M, Ishihara N, Mihara K,
Ripperger JA, Albrecht U, et al: Circadian control of DRP1 activity
regulates mitochondrial dynamics and bioenergetics. Cell Metab.
27:657–666.e5. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zhou BH, Wei SS, Jia LS, Zhang Y, Miao CY
and Wang HW: Drp1/Mff signaling pathway is involved in
fluoride-induced abnormal fission of hepatocyte mitochondria in
mice. Sci Total Environ. 725:1381922020. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ma Y, Wang L and Jia R: The role of
mitochondrial dynamics in human cancers. Am J Cancer Res.
10:1278–1293. 2020.PubMed/NCBI
|
|
7
|
Chauhan SS, Toth RK, Jensen CC, Casillas
AL, Kashatus DF and Warfel NA: PIM kinases alter mitochondrial
dynamics and chemosensitivity in lung cancer. Oncogene.
39:2597–2611. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhou D, Ren K, Wang M, Wang J, Li E, Hou
C, Su Y, Jin Y, Zou Q, Zhou P and Liu X: Long non-coding RNA
RACGAP1P promotes breast cancer invasion and metastasis via
miR-345-5p/RACGAP1-mediated mitochondrial fission. Mol Oncol.
15:543–559. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kamradt ML, Jung JU, Pflug KM, Lee DW,
Fanniel V and Sitcheran R: NIK promotes metabolic adaptation of
glioblastoma cells to bioenergetic stress. Cell Death Dis.
12:2712021. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zhang BY, Zhang L, Chen YM, Qiao X, Zhao
SL, Li P, Liu JF, Wen X and Yang J: Corosolic acid inhibits
colorectal cancer cells growth as a novel HER2/HER3
heterodimerization inhibitor. Br J Pharmacol. 178:1475–1491. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Courtois S, de Luxán-Delgado B,
Penin-Peyta L, Royo-García A, Parejo-Alonso B, Jagust P, Alcalá S,
Rubiolo JA, Sánchez L, Sainz B Jr, et al: Inhibition of
mitochondrial dynamics preferentially targets pancreatic cancer
cells with enhanced tumorigenic and invasive potential. Cancers
(Basel). 13:6982021. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zhou TJ, Zhang SL, He CY, Zhuang QY, Han
PY, Jiang SW, Yao H, Huang YJ, Ling WH, Lin YC and Lin ZN:
Downregulation of mitochondrial cyclooxygenase-2 inhibits the
stemness of nasopharyngeal carcinoma by decreasing the activity of
dynamin-related protein 1. Theranostics. 7:1389–1406. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ferraz LS, Costa RTD, Costa CAD, Ribeiro
CAJ, Arruda DC, Maria-Engler SS and Rodrigues T: Targeting
mitochondria in melanoma: Interplay between MAPK signaling pathway
and mitochondrial dynamics. Biochem Pharmacol. 178:1141042020.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Qian W, Wang J and Van Houten B: The role
of dynamin-related protein 1 in cancer growth: A promising
therapeutic target? Expert Opin Ther Targets. 17:997–1001. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Luza S, Opazo CM, Bousman CA, Pantelis C,
Bush AI and Everall IP: The ubiquitin proteasome system and
schizophrenia. Lancet Psychiatry. 7:528–537. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhu D, Xu R, Huang X, Tang Z, Tian Y,
Zhang J and Zheng X: Deubiquitinating enzyme OTUB1 promotes cancer
cell immunosuppression via preventing ER-associated degradation of
immune checkpoint protein PD-L1. Cell Death Differ. 28:1773–1789.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Park HB, Kim JW and Baek KH: Regulation of
Wnt signaling through ubiquitination and deubiquitination in
cancers. Int J Mol Sci. 21:39042020. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Sun T, Liu Z and Yang Q: The role of
ubiquitination and deubiquitination in cancer metabolism. Mol
Cancer. 19:1462020. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Sukenik S, Braunstein I and Stanhill A: An
arsenite relay between PSMD14 and AIRAP enables revival of
proteasomal DUB activity. Biomolecules. 11:13172021. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Lv J, Zhang S, Wu H, Lu J, Lu Y, Wang F,
Zhao W, Zhan P, Lu J, Fang Q, et al: Deubiquitinase PSMD14 enhances
hepatocellular carcinoma growth and metastasis by stabilizing GRB2.
Cancer Lett. 469:22–34. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Luo G, Hu N, Xia X, Zhou J and Ye C: RPN11
deubiquitinase promotes proliferation and migration of breast
cancer cells. Mol Med Rep. 16:331–338. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Yu W, Li J, Wang Q, Wang B, Zhang L, Liu
Y, Tang M, Xu G, Yang Z, Wang X, et al: Targeting POH1 inhibits
prostate cancer cell growth and enhances the suppressive efficacy
of androgen deprivation and docetaxel. Prostate. 79:1304–1315.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhang L, Xu H, Ma C, Zhang J, Zhao Y, Yang
X, Wang S and Li D: Upregulation of deubiquitinase PSMD14 in lung
adenocarcinoma (LUAD) and its prognostic significance. J Cancer.
11:2962–2971. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Byrne A, McLaren RP, Mason P, Chai L,
Dufault MR, Huang Y, Liang B, Gans JD, Zhang M, Carter K, et al:
Knockdown of human deubiquitinase PSMD14 induces cell cycle arrest
and senescence. Exp Cell Res. 316:258–271. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Jing C, Duan Y, Zhou M, Yue K, Zhuo S, Li
X, Liu D, Ye B, Lai Q, Li L, et al: Blockade of deubiquitinating
enzyme PSMD14 overcomes chemoresistance in head and neck squamous
cell carcinoma by antagonizing E2F1/Akt/SOX2-mediated stemness.
Theranostics. 11:2655–2669. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zhi T, Jiang K, Xu X, Yu T, Zhou F, Wang
Y, Liu N and Zhang J: ECT2/PSMD14/PTTG1 axis promotes the
proliferation of glioma through stabilizing E2F1. Neuro Oncol.
21:462–473. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Jing C, Li X, Zhou M, Zhang S, Lai Q, Liu
D, Ye B, Li L, Wu Y, Li H, et al: The PSMD14 inhibitor Thiolutin as
a novel therapeutic approach for esophageal squamous cell carcinoma
through facilitating SNAIL degradation. Theranostics. 11:5847–5862.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Rao X, Huang X, Zhou Z and Lin X: An
improvement of the 2ˆ(−delta delta CT) method for quantitative
real-time polymerase chain reaction data analysis. Biostat
Bioinforma Biomath. 3:71–85. 2013.PubMed/NCBI
|
|
29
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Asmamaw MD, Liu Y, Zheng YC, Shi XJ and
Liu HM: Skp2 in the ubiquitin-proteasome system: A comprehensive
review. Med Res Rev. 40:1920–1949. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Wang J and Maldonado MA: The
ubiquitin-proteasome system and its role in inflammatory and
autoimmune diseases. Cell Mol Immunol. 3:255–261. 2006.PubMed/NCBI
|
|
32
|
Singh SR, Meyer-Jens M, Alizoti E, Bacon
WC, Davis G, Osinska H, Gulick J, Reischmann-Düsener S, Orthey E,
McLendon PM, et al: A high-throughput screening identifies ZNF418
as a novel regulator of the ubiquitin-proteasome system and
autophagy-lysosomal pathway. Autophagy. 17:3124–3139. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Narayanan S, Cai CY, Assaraf YG, Guo HQ,
Cui Q, Wei L, Huang JJ, Ashby CR Jr and Chen ZS: Targeting the
ubiquitin-proteasome pathway to overcome anti-cancer drug
resistance. Drug Resist Updat. 48:1006632020. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Yerlikaya A, Kanbur E, Stanley BA and
Tümer E: The ubiquitin-proteasome pathway and epigenetic
modifications in cancer. Anticancer Agents Med Chem. 21:20–32.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Sakai W, Yuasa-Sunagawa M, Kusakabe M,
Kishimoto A, Matsui T, Kaneko Y, Akagi JI, Huyghe N, Ikura M, Ikura
T, et al: Functional impacts of the ubiquitin-proteasome system on
DNA damage recognition in global genome nucleotide excision repair.
Sci Rep. 10:197042020. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Zhu R, Liu Y, Zhou H, Li L, Li Y, Ding F,
Cao X and Liu Z: Deubiquitinating enzyme PSMD14 promotes tumor
metastasis through stabilizing SNAIL in human esophageal squamous
cell carcinoma. Cancer Lett. 418:125–134. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Seo D, Jung SM, Park JS, Lee J, Ha J, Kim
M and Park SH: The deubiquitinating enzyme PSMD14 facilitates tumor
growth and chemoresistance through stabilizing the ALK2 receptor in
the initiation of BMP6 signaling pathway. EbioMedicine. 49:55–71.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Fire A, Xu S, Montgomery MK, Kostas SA,
Driver SE and Mello CC: Potent and specific genetic interference by
double-stranded RNA in Caenorhabditis elegans. Nature. 391:806–811.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Yao T and Cohen RE: A cryptic protease
couples deubiquitination and degradation by the proteasome. Nature.
419:403–407. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Vyas S, Zaganjor E and Haigis MC:
Mitochondria and cancer. Cell. 166:555–566. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Dai W and Jiang L: Dysregulated
mitochondrial dynamics and metabolism in obesity, diabetes, and
cancer. Front Endocrinol (Lausanne). 10:5702019. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Chan DC: Mitochondrial dynamics and its
involvement in disease. Annu Rev Pathol. 15:235–259. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Rodrigues T and Ferraz LS: Therapeutic
potential of targeting mitochondrial dynamics in cancer. Biochem
Pharmacol. 182:1142822020. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Deng X, Liu J, Liu L, Sun X, Huang J and
Dong J: Drp1-mediated mitochondrial fission contributes to
baicalein-induced apoptosis and autophagy in lung cancer via
activation of AMPK signaling pathway. Int J Biol Sci. 16:1403–1416.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Moghaddam FD, Mortazavi P, Hamedi S,
Nabiuni M and Roodbari NH: Apoptotic effects of melittin on 4T1
breast cancer cell line is associated with up regulation of Mfn1
and Drp1 mRNA expression. Anticancer Agents Med Chem. 20:790–799.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Zhan L, Cao H, Wang G, Lyu Y, Sun X, An J,
Wu Z, Huang Q, Liu B and Xing J: Drp1-mediated mitochondrial
fission promotes cell proliferation through crosstalk of p53 and
NF-κB pathways in hepatocellular carcinoma. Oncotarget.
7:65001–65011. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Hu J, Zhang Y, Jiang X, Zhang H, Gao Z, Li
Y, Fu R, Li L, Li J, Cui H and Gao N: ROS-mediated activation and
mitochondrial translocation of CaMKII contributes to Drp1-dependent
mitochondrial fission and apoptosis in triple-negative breast
cancer cells by isorhamnetin and chloroquine. J Exp Clin Cancer
Res. 38:2252019. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Mitra K, Wunder C, Roysam B, Lin G and
Lippincott-Schwartz J: A hyperfused mitochondrial state achieved at
G1-S regulates cyclin E buildup and entry into S phase. Proc Natl
Acad Sci USA. 106:11960–11965. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Liang J, Yang Y, Bai L, Li F and Li E:
DRP1 upregulation promotes pancreatic cancer growth and metastasis
through increased aerobic glycolysis. J Gastroenterol Hepatol.
35:885–895. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Kitamura S, Yanagi T, Imafuku K, Hata H,
Abe R and Shimizu H: Drp1 regulates mitochondrial morphology and
cell proliferation in cutaneous squamous cell carcinoma. J Dermatol
Sci. 88:298–307. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Xie Q, Wu Q, Horbinski CM, Flavahan WA,
Yang K, Zhou W, Dombrowski SM, Huang Z, Fang X, Shi Y, et al:
Mitochondrial control by DRP1 in brain tumor initiating cells. Nat
Neurosci. 18:501–510. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Zhao J, Zhang J, Yu M, Xie Y, Huang Y,
Wolff DW, Abel PW and Tu Y: Mitochondrial dynamics regulates
migration and invasion of breast cancer cells. Oncogene.
32:4814–4824. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Rehman J, Zhang HJ, Toth PT, Zhang Y,
Marsboom G, Hong Z, Salgia R, Husain AN, Wietholt C and Archer SL:
Inhibition of mitochondrial fission prevents cell cycle progression
in lung cancer. FASEB J. 26:2175–2186. 2012. View Article : Google Scholar : PubMed/NCBI
|