Introduction
Danshen (root of Salviae miltiorrhizae) is
widely prescribed in traditional Chinese medicine (1,2), and
tanshinone IIA (C19H18O3) may be
obtained from it by extraction (3,4). It has
been demonstrated that tanshinone IIA exerts a variety of
biological activities in the cardiovascular system, serving as an
antioxidant and anticoagulant, and possesses anti-atherosclerotic,
anti-apoptotic and anti-hypertrophic properties (5–9).
Accumulating evidence suggests that tanshinone IIA reduces the area
of ischemic infarction and improves cardiac function (7,10).
Although the cardioprotective effects of tanshinone IIA have been
investigated for numerous years, the underlying molecular
mechanisms remain elusive.
MicroRNAs (miRNAs) are small endogenous ~22
nucleotide noncoding RNAs. They regulate post-transcriptional gene
expression by complementing the 3′-untranslated regions of their
target mRNAs, resulting in mRNA degradation or the inhibition of
translation (11,12). Due to their involvement in the
regulation of gene expression, miRNAs serve an important role in
cardiac function, including the conductance of electrical signals,
heart muscle contraction, development and morphogenesis; miRNAs are
also involved in the proliferation and apoptosis of cardiomyocytes,
cardiac hypertrophy and heart failure (13–15). In
particular, a number of studies suggest that miRNA-133 (miR-133)
serves an important role in the cardiovascular system (15–17). In
the present study, the role of miR-133 in the myocardial protective
effect of tanshinone IIA against hydrogen peroxide
(H2O2)-induced induce cell death was
investigated.
Akt is a vital regulator of cell survival that
antagonizes apoptosis; when activated, Akt phosphorylates its
downstream targets, contributing towards its inhibitory effect on
the apoptosis of cardiomyocytes induced by multiple stimuli,
including H2O2 (18,19).
Numerous putative downstream targets have been identified that may
contribute to the anti-apoptotic effects of Akt, including the
transcriptional nuclear factor (NF)-κB and B-cell lymphoma-2
(Bcl-2) (18). In the current study,
the role of the phosphatidylinositol 3-kinase
(PI3K)/Akt/NF-κB/Bcl-2 axis in the myocardial protection effect of
tanshinone IIA against oxidative stress-induced cell death was
investigated.
Materials and methods
Materials
Tanshinone IIA was purchased from Dasherb Corp.
(Shengyang, China). All the chemicals of reagent grade were
obtained from Sigma-Aldrich (St. Louis, MO, USA). Rabbit polyclonal
anti-Ser473 phospho-Akt (cat. no. 9271; 1:1,000) and rabbit
polyclonal anti-Akt antibodies (cat. no. 9272; 1:1,000) were
purchased from Cell Signaling Technology, Inc. (Danvers, MA, USA).
Rabbit polyclonal anti-Bcl-2 (cat. no. sc-492; 1:2,000) and rabbit
polyclonal anti-β-actin antibodies (cat. no. sc-7210; 1:2,000), and
goat anti-rabbit horseradish peroxidase (HRP)-conjugated secondary
antibodies (cat no. sc-2030; 1:10,000) were purchased from Santa
Cruz Biotechnology, Inc. (Dallas, TX, USA).
Cell culture and treatment
protocol
Cardiac H9c2 cells are cloned heart muscle cells
derived from embryonic rat hearts that retain a number of
cardiomyocyte phenotypes (20). The
cells used in the present study were derived from a CRL-1446 cell
culture obtained from the American Type Culture Collection
(Manassas, VA, USA). The cells were cultured in Dulbecco's modified
Eagle's medium (DMEM) supplemented with 10% fetal bovine serum
(both Gibco-BRL; Thermo Fisher Scientific, Inc.,Waltham, MA, USA),
100 U/ml penicillin and 100 µg/ml streptomycin (Sigma-Aldrich).
Cells were placed in a 95% humidified incubator containing 95% air
and 5% CO2 at 37°C with replenishment of the medium
every 3 days. Prior to the experiments, cells were starved of serum
for 24 h in DMEM supplemented with 1% fetal bovine serum. Next, the
starved cells were treated with H2O2 (50,
100, 200, 400 or 800 µM) and tanshinone IIA (0.01, 0.1, 0.3, 1, 3
or 10 µM) alone, or in combination for 24 h at 37°C. For
transfection experiments, cells were transfected with 50 nM miR-133
mimic or miR-133 inhibitor (Guangzhou RiboBio Co., Ltd., Guangzhou,
China); after 8 h, transfected cells were treated for 24 h at 37°C
with various combinations of H2O2 and
tanshinone IIA. For inhibitor experiments, cells were pre-incubated
with a selective PI3K inhibitor (100 nM wortmannin or 10 nM
LY294002; both Beyotime Institute of Biotechnology, Haimen, China)
for 30 min and then treated with H2O2 and/or
tanshinone IIA as described above.
Cell viability assay
Cell viability was determined using a Cell Counting
Kit-8 (CCK-8; Dojindo Molecular Technologies, Inc., Kumamoto,
Japan). Briefly, cells were cultured in 96-well plates and after 24
h, CCK-8 reagent was added to each well according to the
manufacturer's protocol. After a further 2 h of incubation, cell
viability was determined by measuring the absorbance at 450 nm
using a VICTOR X multi-label reader (PerkinElmer, Inc., Waltham,
MA, USA). Data were presented as a percentage of the control value.
The percentage cell viability was calculated as: Adrug /
Acontrol × 100.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
Total RNA samples were extracted using TRIzol
reagent (Thermo Fisher Scientific, Inc.) from cultured cells.
miR-133 expression levels were quantified using the Bulge-Loop
miRNA wRT-PCR Primer Set (Guangzhou RiboBio Co., Ltd.) in
conjunction with RT-qPCR using SYBR Green I dye (Beijing TransGen
Biotech Co., Ltd., Beijing, China). U6 (included in the Bulge-Loop
miRNA qRT-PCR Primer Set) was used as an internal control. The
relative expression of miR-133 was calculated and normalized to U6
using the comparative Cq method. Relative expression intensity
values were calculated as 2−ΔΔCq (16). The RT-qPCR was performed using the
SYBR green method with an Applied Biosystems 7500 Real-Time PCR
System (Thermo Fisher Scientific, Inc.).
Cell transfection with miR-133 mimic
or miR-133 inhibitor
Transfection was performed using Effectene
Transfection Reagent, according to the protocol recommended by the
manufacturer (Qiagen GmbH, Hilden, Germany). In brief, cells were
seeded into 96-well plates 1 day prior to transfection. Next, the
cells were transfected with an miR-133 mimic or an miR-133
inhibitor at a final concentration of 50 nM using Effectene
Transfection Reagent, in accordance with the manufacturer's
instructions.
Western blot analysis
Following treatment, H9c2 cells were harvested and
lysed in radioimmunoprecipitation assay buffer (Applygen
Technologies, Inc., Beijing, China). The whole cell lysates were
then resolved by 12% sodium dodecyl sulfate polyacrylamide gel
electrophoresis (SDS-PAGE) and transferred onto a 0.4
µm-polyvinylidene difluoride (PVDF) membrane (EMD Millipore,
Billerica, MA, USA). After blocking in 5% non-fat milk for 2 h at
room temperature, the PVDF membranes were probed with primary
antibody overnight. Following a 30-min wash with Tris-buffered
saline containing 0.1% Tween-20 (TBST), the membranes were
incubated with HRP-conjugated secondary antibody (1:10,000) for 1 h
at room temperature. The membranes were then washed with TBST for
30 min and visualized using an Enhanced Chemiluminescence detection
kit (Merck Millipore, Darmstadt, Germany). β-actin served used as
an internal control.
Statistical analysis
Data were analyzed using SPSS version 13.0 software
(SPSS, Inc., Chicago, IL, USA) and presented as the mean ± standard
deviation. Numeric variables were compared using one-way analysis
of variance. P<0.05 was considered to indicate a statistically
significant difference.
Results
Effect of tanshinone IIA on
H2O2 cytotoxity in H9c2 cells
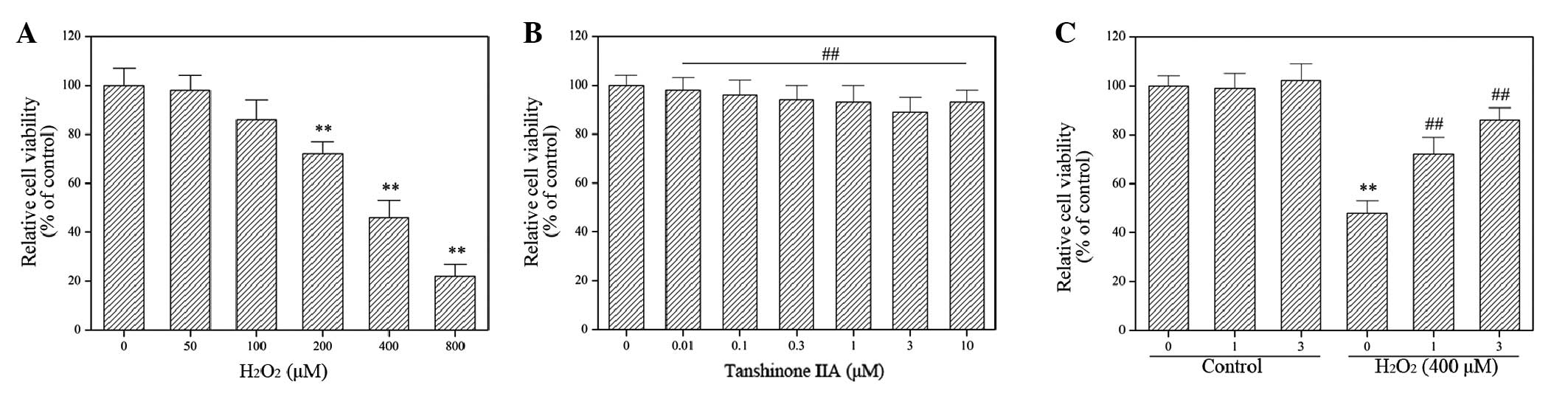
The molecular mechanisms underlying the effect of
tanshinone IIA on H2O2 cytotoxicity in H9c2
cells were investigated using an H9c2 cell line. The results
demonstrated that H2O2 induced H9c2 cell
death in a concentration-dependent manner (Fig. 1A), while tanshinone IIA displayed no
cytotoxic effect on H9c2 cells at any concentration studied
(Fig. 1B). In addition, the exposure
of H9c2 cells to tanshinone IIA significantly suppressed the
cytotoxic effect of H2O2 in dose-dependent
manner (P<0.05; Fig. 1C),
confirming the previously reported cardioprotective effects of
tanshinone IIA (21–24).
Involvement of miR-133 in the
mechanism of action of tanshinone IIA
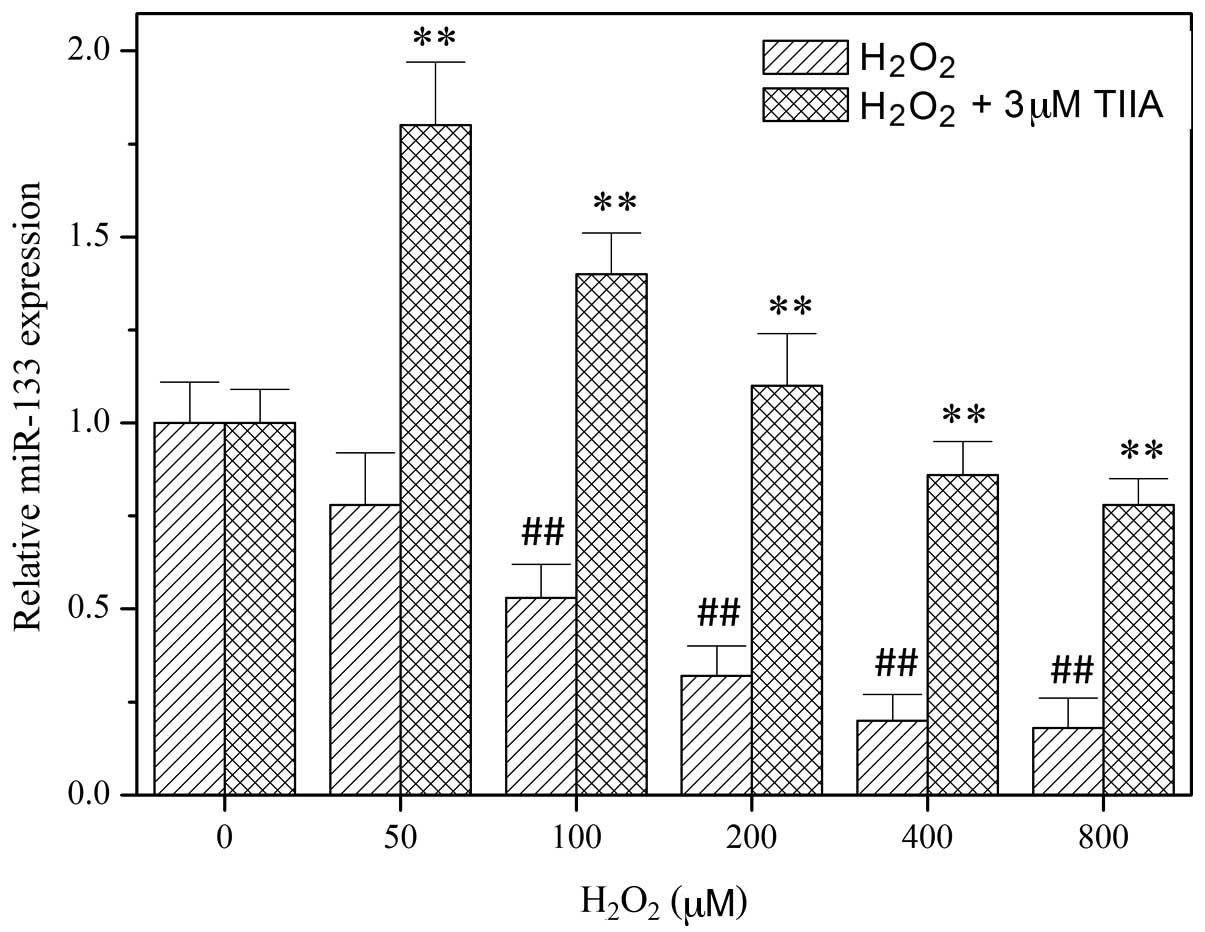
In order to study the role of miR-133 in the
function of tanshinone IIA, miR-133 expression levels in H9c2 cells
were determined using RT-qPCR following treatment with tanshinone
IIA and/or H2O2 (Fig. 2). The results demonstrated that
H2O2 decreases the expression of miR-133 in a
dose-dependent manner. However, the downregulation of miR-133 by
H2O2 was reversed by treatment with
tanshinone IIA (Fig. 2), indicating
that miR-133 is involved in the myocardial protective effect of
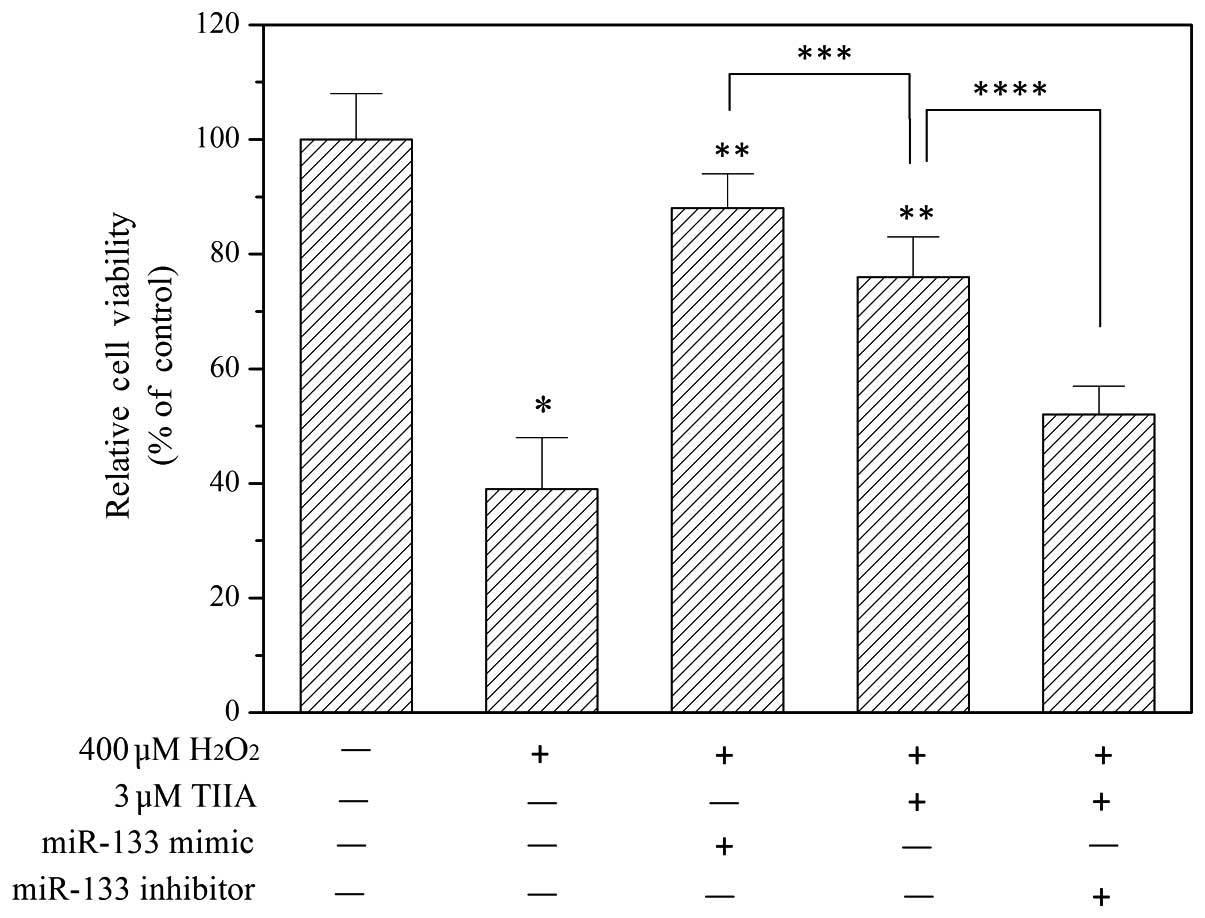
tanshinone IIA. To verify this, an miR-133 mimic and miR-133
inhibitor were transfected into H9c2 cells to potentiate and
suppress miR-133, respectively. As presented in Fig. 3, the miR-133 mimic alleviated
oxidative injury in H9c2 cells. Furthermore, the myocardial
protective effect of tanshinone IIA was inhibited by the miR-133
inhibitor (Fig. 3).
Role of Akt activation in the
mechanism of action of tanshinone IIA
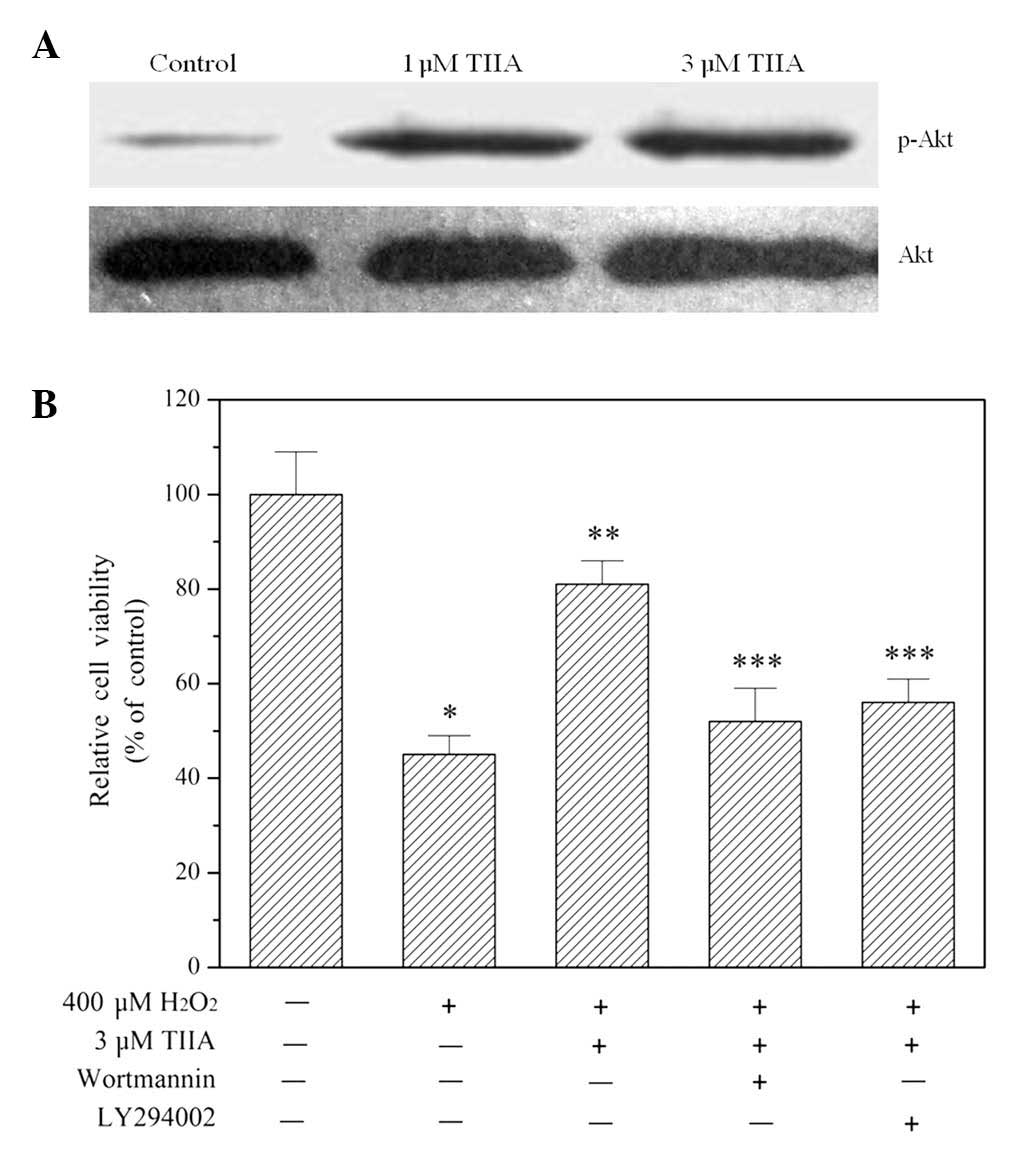
The results of western blot analysis demonstrated
that treatment with tanshinone IIA promoted Akt phosphorylation at
the amino acid serine 473 in a concentration-dependent manner
(Fig. 4A), indicating that the
PI3K/Akt signaling pathway was activated. Blocking the signaling
pathway using the PI3K-specific inhibitors wortmannin and LY294002
eliminated the ability of tanshinone IIA to protect H9c2 cells from
oxidative injury (Fig. 4B).
Role of Bcl-2 expression in the
mechanism of action of tanshinone IIA
Expression of Bcl-2 is regulated by the
PI3K/Akt/NF-κB signaling pathway (25). Since tanshinone IIA was observed to
activate PI3K/Akt signaling in H9c2 cells (Fig. 4A) and protect H9c2 cells from
oxidative stress-induced death (Fig.
4B), activation of the PI3K/Akt signaling pathway by tanshinone
IIA appears to be instrumental to the survival of H9c2 cells. This
was further investigated by evaluating the expression of Bcl-2.
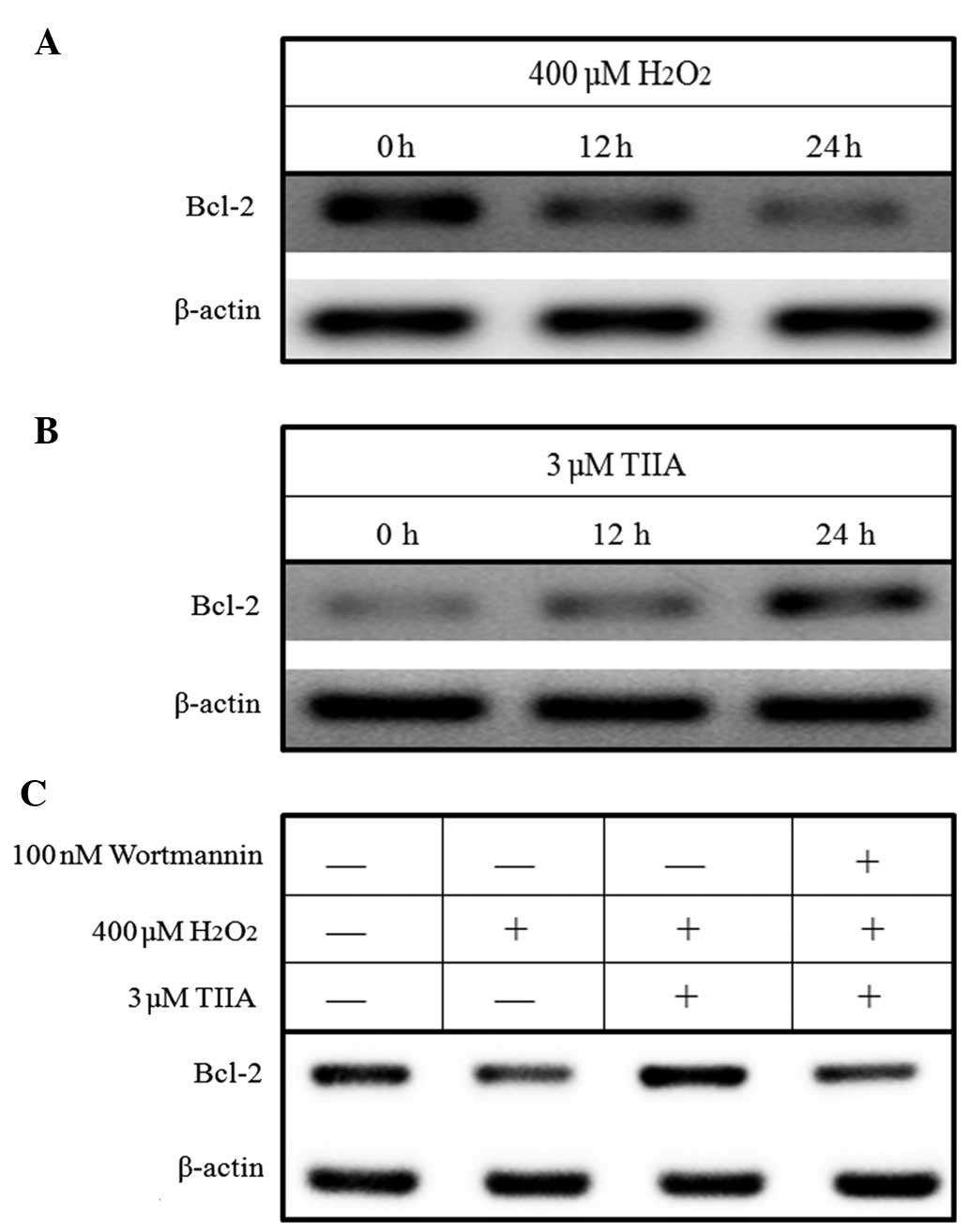
Western blotting results demonstrated that
H2O2 decreased the expression of Bcl-2 in
H9c2 cells in a time-dependent manner, whereas tanshinone IIA
increased Bcl-2 expression in H9c2 cells (Fig. 5A and B). Furthermore, the results
demonstrated that the downregulation of Bcl-2 expression by
H2O2 in H9c2 cells is reversed by treatment
with tanshinone IIA (Fig. 5C).
However, this reversion process is inhibited by the PI3K-specific
inhibitor wortmannin (Fig. 5C),
indicating that tanshinone IIA upregulates Bcl-2 expression by
activating the PI3K/Akt signaling pathway.
Discussion
Tanshinone IIA, one of the most abundant
tanshinones, exhibits a variety of cardiovascular activities,
including vasorelaxation, and protection against
ischemia-reperfusion injury and the effects of antiarrhythmia
(4,7,26). A
number of studies have demonstrated that tanshinone IIA increases
coronary blood flow and protects the heart against cardiac injury
(7,10,22,23,27–31). The
molecular mechanisms underlying the cardioprotective effects of
tanshinone IIA were investigated in the present study. The results
demonstrated that tanshinone IIA protects H9c2 cells from
H2O2-induced cell death, which confirms the
cardioprotective effect of tanshinone IIA in the oxidative stress
injury model in vitro. In addition, the current study
provides evidence that miR-133, Akt activation and Bcl-2 are
involved in the cardioprotective effects of tanshinone IIA against
oxidative stress-induced cell death.
miRNAs serve an important role in the proliferation
and apoptosis of cardiomyocytes, cardiac hypertrophy and heart
failure (13–15). Previous studies have demonstrated
that muscle-specific miRNAs (miR-1 and miR-133) are important
regulators of myogenesis (32). In
addition, their upregulation or downregulation may influence the
pathogenesis of cardiac diseases (33–35). He
et al reported that miR-133 provides protection in
myocardial ischemic post-conditioning via the regulation of the
initiator caspase, caspase-9 (36).
In the present study, changes in the expression of
miR-133 that were associated with the cardioprotectve effects of
tanshinone IIA against H2O2-induced cell
death were investigated. The results demonstrated that tanshinone
IIA reversed the reduction in miR-133 expression levels induced by
H2O2 in H9c2 cells. In addition, transfection
with an miR-133 inhibitor attenuated the cardioprotective effects
of tanshinone IIA against H2O2-induced cell
death in H9c2 cells, indicating that miR-133 serves a vital role in
the cardioprotective action of tanshinone IIA.
The PI3K signaling pathway controls cardiomyocytes
survival and function (37). The
downstream effects of the PI3K signaling pathway are primarily
mediated by Akt, a serine/threonine kinase, to coordinate a variety
of intracellular signals, and thereby regulate cell proliferation
and survival (38). Activation of
the PI3K/Akt signaling pathway has been shown to protect the
myocardium from myocardial injury and prevent the apoptosis of
cardiomyocytes (38). In order to
explore whether the protective effects of tanshinone IIA against
H2O2-induced cell death are associated with
the PI3K/Akt signaling pathway, the current study investigated the
effects of tanshinone IIA on the phosphorylation of Akt and the
effects of PI3K inhibition on the cardioprotective effects of
tanshinone IIA against H2O2-induced cell
death. The results demonstrated that tanshinone IIA increased the
phosphorylation of Akt at serine 473 in a dose-dependent manner. In
addition, the blockade of Akt phosphorylation with a PI3K inhibitor
(wortmannin or LY294002) eliminated the cardioprotective effects of
tanshinone IIA against H2O2-induced cell
death, suggesting that tanshinone IIA exerts cardioprotective
effects against oxidative stress-induced cell death via the
activation of the PI3K/Akt signaling pathway.
The PI3K/Akt signaling pathway can upregulate the
expression of anti-apoptotic genes (18,19). For
example, Akt activates inhibitor of κB (IκB) kinases (IKKs),
resulting in the activation of NF-κB (18,19), its
translocation to the nucleus and the transcription of
anti-apoptotic genes, such as BCL-2 (39,40). In
the present study it was observed that H2O2
decreased the expression of Bcl-2 in a time-dependent manner, while
tanshinone IIA increased Bcl-2 expression in a time-dependent
manner. In addition, the reduction of Bcl-2 expression induced by
H2O2 was attenuated by tanshinone IIA, and
this effect was suppressed by pre-treatment with the PI3K
inhibitor, wortmannin.
In conclusion, the results from the present study
suggest that tanshinone IIA exerts its cardioprotective effects
against H2O2-induced cell death by
upregulating the expression of Bcl-2 via the activation of the
PI3K/Akt signaling pathway. In addition, tanshinone IIA is able to
protect H9c2 cells from oxidative stress-induced cell death.
Tanshinone IIA may potentially be used to treat heart diseases
involving oxidative stress; however, further studies are required
in order to define and clarify the rationale for its clinical
use.
References
|
1
|
Fish JM, Welchons DR, Kim YS, Lee SH, Ho
WK and Antzelevitch C: Dimethyl lithospermate B, an extract of
Danshen, suppresses arrhythmogenesis associated with the Brugada
syndrome. Circulation. 113:1393–1400. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Chang PN, Mao JC, Huang SH, Ning L, Wang
ZJ, On T, Duan W and Zhu YZ: Analysis of cardioprotective effects
using purified Salvia miltiorrhiza extract on isolated rat
hearts. J Pharmacol Sci. 101:245–249. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Che AJ, Zhang JY, Li CH, Chen XF, Hu ZD
and Chen XG: Separation and determination of active components in
Radix Salviae miltiorrhizae and its medicinal preparations by
nonaqueous capillary electrophoresis. J Sep Sci. 27:569–575. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zhou L, Zuo Z and Chow MS: Danshen: An
overview of its chemistry, pharmacology, pharmacokinetics and
clinical use. J Clin Pharmacol. 45:1345–1359. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Adams JD, Wang R, Yang J and Lien EJ:
Preclinical and clinical examinations of Salvia miltiorrhiza
and its tanshinones in ischemic conditions. Chin Med. 1:32006.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Cheng TO: Cardiovascular effects of
Danshen. Int J Cardiol. 121:9–22. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Gao J, Yang G, Pi R, Li R, Wang P, Zhang
H, Le K, Chen S and Liu P: Tanshinone IIA protects neonatal rat
cardiomyocytes from adriamycin-induced apoptosis. Transl Res.
151:79–87. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Yang L, Zou X, Liang Q, Chen H, Feng J,
Yan L, Wang Z, Zhou D, Li S, Yao S and Zheng Z: Sodium tanshinone
IIA sulfonate depresses angiotensin II-induced cardiomyocyte
hypertrophy through MEK/ERK pathway. Exp Mol Med. 39:65–73. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yang R, Liu A, Ma X, Li L, Su D and Liu J:
Sodium tanshinone IIA sulfonate protects cardiomyocytes against
oxidative stress-mediated apoptosis through inhibiting JNK
activation. J Cardiovasc Pharmacol. 51:396–401. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Shanghai Cooperative Group for the Study
of Tanshinone IIA: Therapeutic effect of sodium tanshinone IIA
sulfonate in patients with coronary heart disease. A double blind
study. J Tradit Chin Med. 4:20–24. 1984.PubMed/NCBI
|
|
11
|
Bartel DP: Micrornas: Genomics,
biogenesis, mechanism and function. Cell. 116:281–297. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Jackson RJ and Standart N: How do
microRNAs regulate gene expression? Sci STKE. 2007:re12007.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Roy S, Khanna S, Hussain SR, Biswas S,
Azad A, Rink C, Gnyawali S, Shilo S, Nuovo GJ and Sen CK: MicroRNA
expression in response to murine myocardial infarction: Mir-21
regulates fibroblast metalloprotease-2 via phosphatase and tensin
homologue. Cardiovasc Res. 82:21–29. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
van Rooij E, Sutherland LB, Thatcher JE,
DiMaio JM, Naseem RH, Marshall WS, Hill JA and Olson EN:
Dysregulation of microRNAs after myocardial infarction reveals a
role of miR-29 in cardiac fibrosis. Proc Natl Acad Sci USA.
105:13027–13032. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Shan H, Zhang Y, Lu Y, Zhang Y, Pan Z, Cai
B, Wang N, Li X, Feng T, Hong Y and Yang B: Downregulation of
miR-133 and miR-590 contributes to nicotine-induced atrial
remodelling in canines. Cardiovasc Res. 83:465–472. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhang L, Wu Y, Li Y, Xu C, Li X, Zhu D,
Zhang Y, Xing S, Wang H, Zhang Z and Shan H: Tanshinone IIa
improves miR-133 expression through MAPK ERK1/2 pathway in hypoxic
cardiac myocytes. Cell Physiol Biochem. 30:843–852. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Takaya T, Ono K, Kawamura T, Takanabe R,
Kaichi S, Morimoto T, Wada H, Kita T, Shimatsu A and Hasegawa K:
MicroRNA-1 and microRNA-133 in spontaneous myocardial
differentiation of mouse embryonic stem cells. Circ J.
73:1492–1497. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Matsui T and Rosenzweig A: Convergent
signal transduction pathways controlling cardiomyocyte survival and
function: The role of PI 3-kinase and Akt. J Mol Cell Cardiol.
38:63–71. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Amaravadi R and Thompson CB: The survival
kinases Akt and Pim as potential pharmacological targets. J Clin
Invest. 115:2618–2624. 2005. View
Article : Google Scholar : PubMed/NCBI
|
|
20
|
Hescheler J, Meyer R, Plant S, Krautwurst
D, Rosenthal W and Schultz G: Morphological, biochemical and
electrophysiological characterization of a clonal cell (H9c2) line
from rat heart. Circ Res. 69:1476–1486. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Hong HJ, Liu JC, Cheng TH and Chan P:
Tanshinone IIA attenuates angiotensin II-induced apoptosis via Akt
pathway in neonatal rat cardiomyocytes. Acta Pharmacol Sin.
31:1569–1575. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhang MQ, Zheng YL, Chen H, Tu JF, Shen Y,
Guo JP, Yang XH, Yuan SR, Chen LZ, Chai JJ, et al: Sodium
tanshinone IIA sulfonate protects rat myocardium against
ischemia-reperfusion injury via activation of PI3K/Akt/FOXO3A/Bim
pathway. Acta Pharmacol Sin. 34:1386–1396. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wei B, Li WW, Ji J, Hu QH and Ji H: The
cardioprotective effect of sodium tanshinone IIA sulfonate and the
optimizing of therapeutic time window in myocardial
ischemia/reperfusion injury in rats. Atherosclerosis. 235:318–327.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Wu WY, Wang WY, Ma YL, Yan H, Wang XB, Qin
YL, Su M, Chen T and Wang YP: Sodium tanshinone IIA silate inhibits
oxygen-glucose deprivation/recovery-induced cardiomyocyte apoptosis
via suppression of the NF-κb/TNF-α pathway. Br J Pharmacol.
169:1058–1071. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wang B, Shravah J, Luo H, Raedschelders K,
Chen DD and Ansley DM: Propofol protects against hydrogen
peroxide-induced injury in cardiac H9c2 cells via Akt activation
and Bcl-2 up-regulation. Biochem Biophys Res Commun. 389:105–111.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Sun DD, Wang HC, Wang XB, Luo Y, Jin ZX,
Li ZC, Li GR and Dong MQ: Tanshinone IIA: A new activator of human
cardiac KCNQ1/KCNE1 (I(Ks)) potassium channels. Eur J Pharmacol.
590:317–321. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Shang Q, Xu H and Huang L: Tanshinone IIA:
A promising natural cardioprotective agent. Evid Based Complement
Alternat Med. 2012:7164592012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Gao S, Liu Z, Li H, Little PJ, Liu P and
Xu S: Cardiovascular actions and therapeutic potential of
tanshinone IIA. Atherosclerosis. 220:3–10. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Tong Y, Xu W, Han H, Chen Y, Yang J, Qiao
H, Hong D, Wu Y and Zhou C: Tanshinone IIA increases recruitment of
bone marrow mesenchymal stem cells to infarct region via
up-regulating stromal cell-derived factor-1/CXC chemokine receptor
4 axis in a myocardial ischemia model. Phytomedicine. 18:443–450.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Yuan X, Jing S, Wu L, Chen L and Fang J:
Pharmacological postconditioning with tanshinone IIA attenuates
myocardial ischemia-reperfusion injury in rats by activating the
phosphatidylinositol 3-kinase pathway. Exp Ther Med. 8:973–977.
2014.PubMed/NCBI
|
|
31
|
Song W, Pu J and He B: Tanshinol protects
human umbilical vein endothelial cells against hydrogen
peroxide-induced apoptosis. Mol Med Rep. 10:2764–2770.
2014.PubMed/NCBI
|
|
32
|
Zhao Y, Samal E and Srivastava D: Serum
response factor regulates a muscle-specific microRNA that targets
Hand2 during cardiogenesis. Nature. 436:214–220. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Yang B, Lin H, Xiao J, Lu Y, Luo X, Li B,
Zhang Y, Xu C, Bai Y, Wang H, et al: The muscle-specific microRNA
miR-1 regulates cardiac arrhythmogenic potential by targeting GJA1
and KCNJ2. Nat Med. 13:486–491. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
34
|
Carè A, Catalucci D, Felicetti F, Bonci D,
Addario A, Gallo P, Bang ML, Segnalini P, Gu Y, Dalton ND, et al:
MicroRNA-133 controls cardiac hypertrophy. Nat Med. 13:613–618.
2007. View
Article : Google Scholar : PubMed/NCBI
|
|
35
|
Dong DL, Chen C, Huo R, Wang N, Li Z, Tu
YJ, Hu JT, Chu X, Huang W and Yang BF: Reciprocal repression
between microRNA-133 and calcineurin regulates cardiac hypertrophy:
A novel mechanism for progressive cardiac hypertrophy.
Hypertension. 55:946–952. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
He B, Xiao J, Ren AJ, Zhang YF, Zhang H,
Chen M, Xie B, Gao XG and Wang YW: Role of miR-1 and miR-133a in
myocardial ischemic postconditioning. J Biomed Sci. 18:222011.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Hausenloy DJ and Yellon DM: Reperfusion
injury salvage kinase signalling: Taking a risk for
cardioprotection. Heart Fail Rev. 12:217–234. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Matsui T, Tao J, del Monte F, Lee KH, Li
L, Picard M, Force TL, Franke TF, Hajjar RJ and Rosenzweig A: Akt
activation preserves cardiac function and prevents injury after
transient cardiac ischemia in vivo. Circulation. 104:330–335. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Gustin JA, Korgaonkar CK, Pincheira R, Li
Q and Donner DB: Akt regulates basal and induced processing of
NF-kappaB2 (p100) to p52. J Biol Chem. 281:16473–16481. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Luo JL, Kamata H and Karin M: The
anti-death machinery in IKK/NF-kappaB signaling. J Clin Immunol.
25:541–550. 2005. View Article : Google Scholar : PubMed/NCBI
|