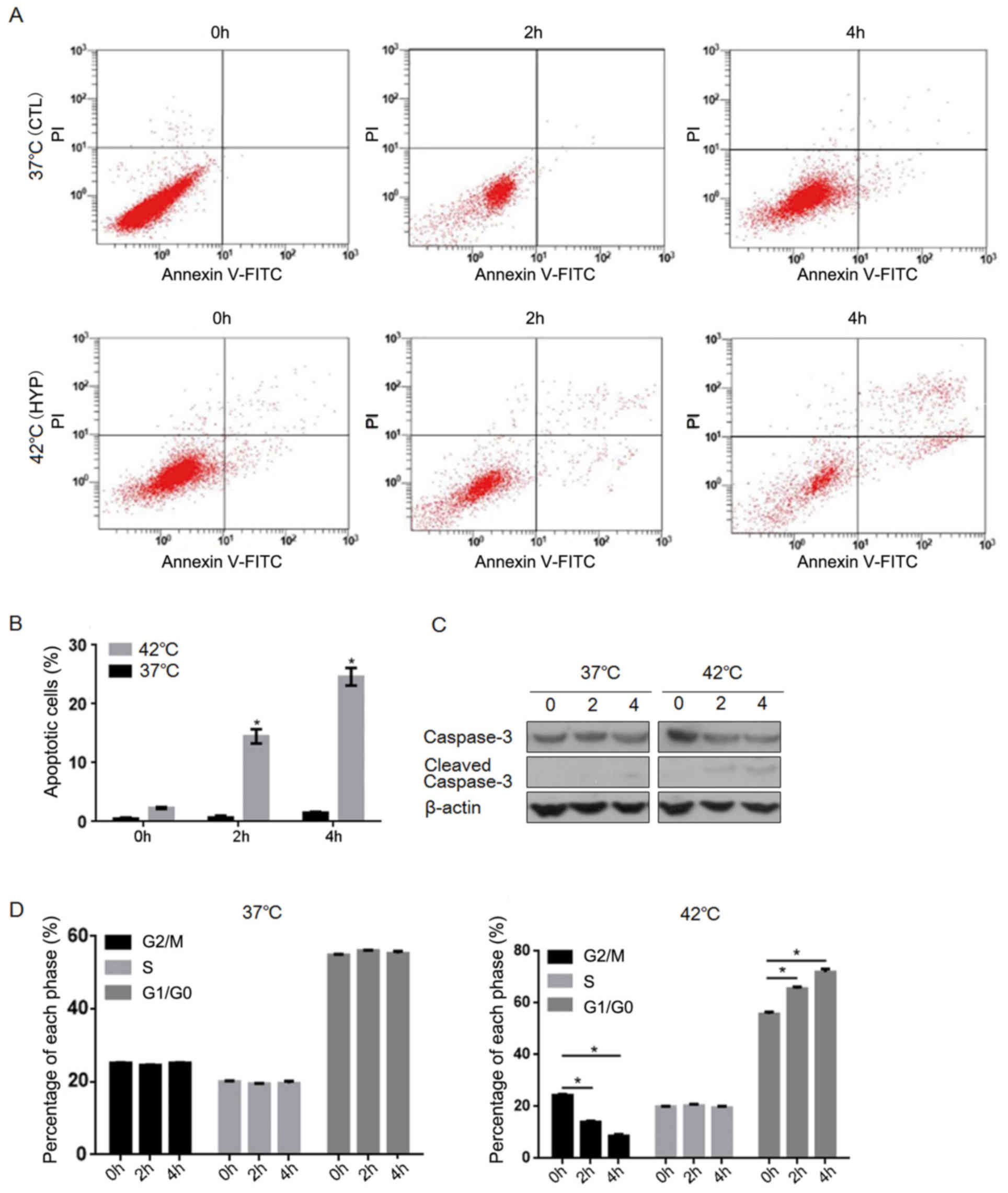
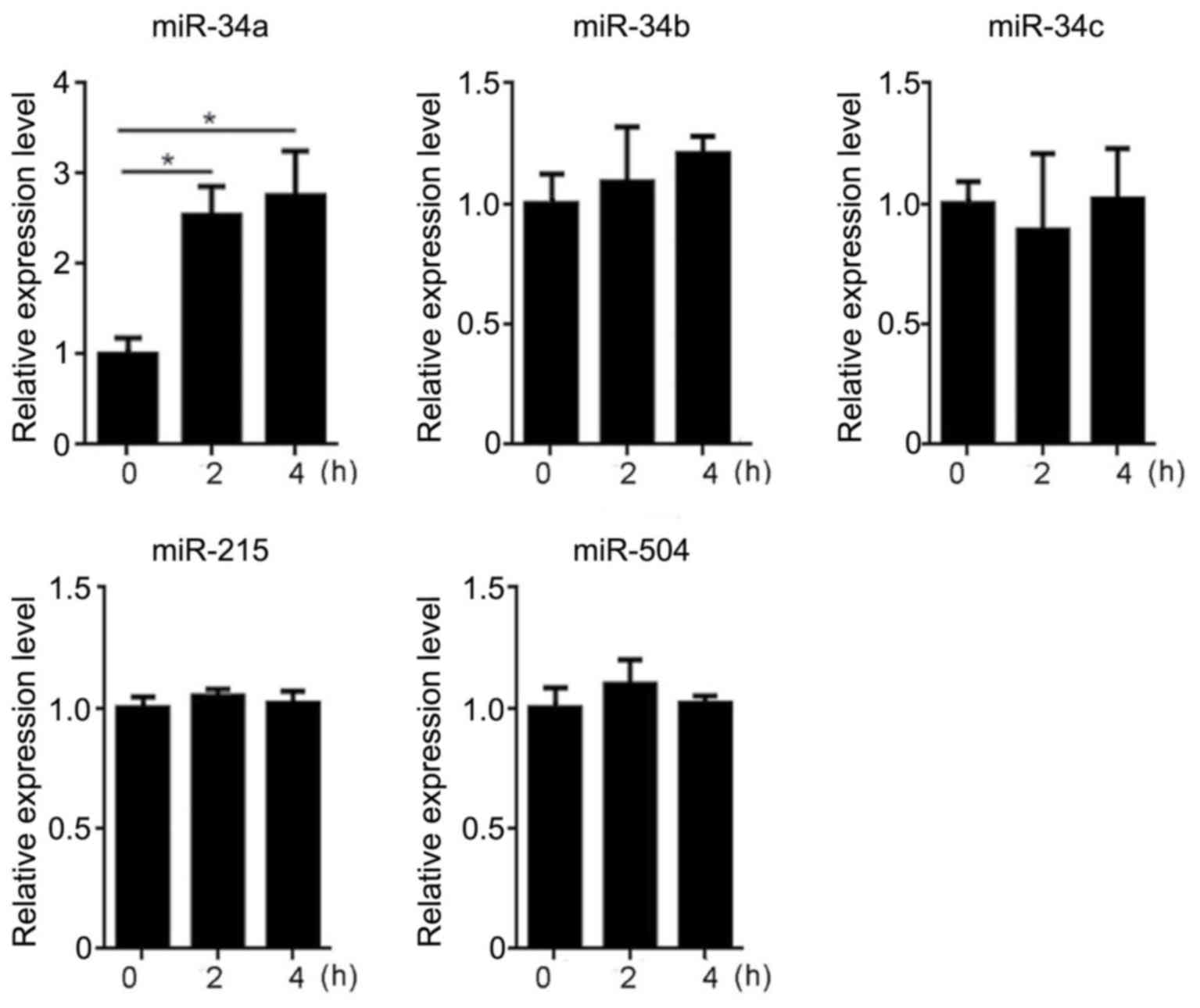
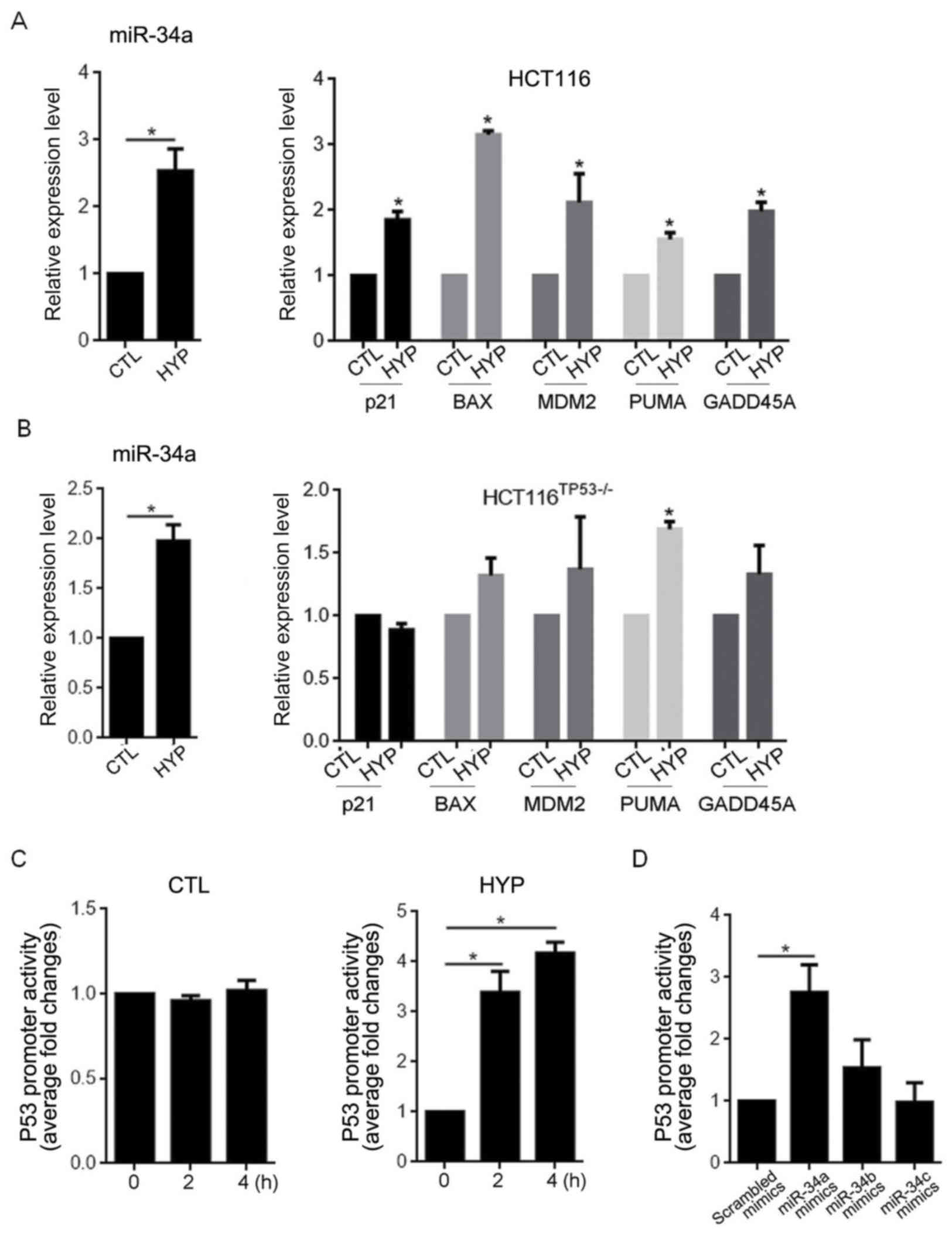
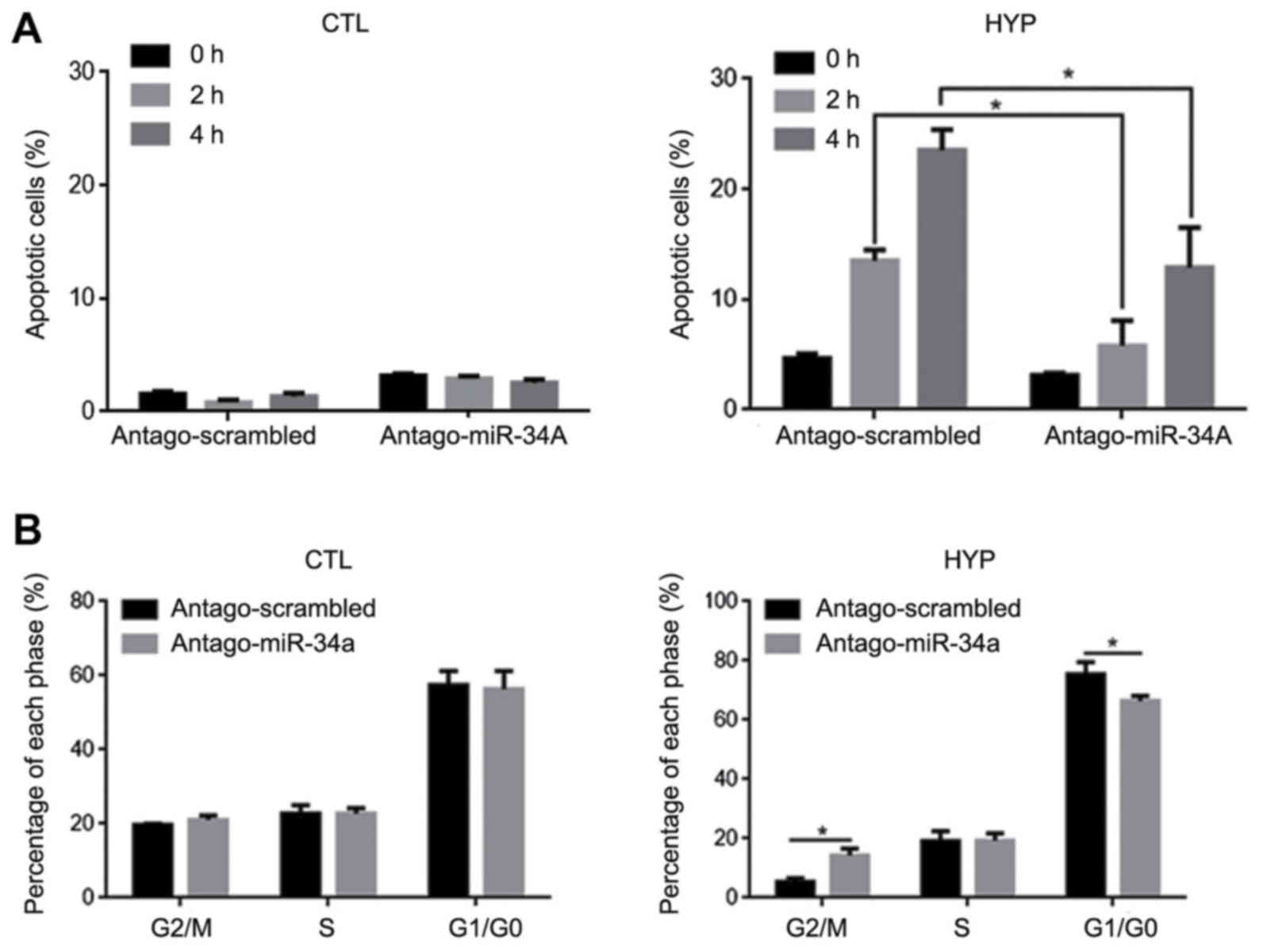
|
1
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Winawer SJ, Zauber AG, O'Brien MJ, Ho MN,
Gottlieb L, Sternberg SS, Waye JD, Bond J, Schapiro M, Stewart ET,
et al: Randomized comparison of surveillance intervals after
colonoscopic removal of newly diagnosed adenomatous polyps. The
National Polyp Study Workgroup. N Engl J Med. 328:901–906. 1993.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kornmann M, Formentini A, Ette C,
Henne-Bruns D, Kron M, Sander S, Baumann W, Kreuser ED, Staib L and
Link KH: Prognostic factors influencing the survival of patients
with colon cancer receiving adjuvant 5-FU treatment. Eur J Surg
Oncol. 34:1316–1221. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zlobec I and Lugli A: Prognostic and
predictive factors in colorectal cancer. J Clin Pathol. 61:561–569.
2008.PubMed/NCBI
|
|
5
|
Oei AL, Vriend LE, Crezee J, Franken NA
and Krawczyk PM: Effects of hyperthermia on DNA repair pathways:
One treatment to inhibit them all. Radiat Oncol. 10:1652015.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Fałkowska-Podstawka M and Wernicki A: Heat
shock proteins in health and disease. Pol J Vet Sci. 6:61–70.
2003.PubMed/NCBI
|
|
7
|
Kim SY, Lee DH, Song X, Bartlett DL, Kwon
YT and Lee YJ: Role of Bcl-xL/Beclin-1 in synergistic apoptotic
effects of secretory TRAIL-armed adenovirus in combination with
mitomycin C and hyperthermia on colon cancer cells. Apoptosis.
19:1603–1615. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kalamida D, Karagounis IV, Mitrakas A,
Kalamida S, Giatromanolaki A and Koukourakis MI: Fever-range
hyperthermia vs. Hypothermia effect on cancer cell viability,
proliferation and HSP90 expression. PLoS One. 10:e01160212015.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Qi D, Hu Y, Li J, Peng T, Su J, He Y and
Ji W: Hyperthermia induces apoptosis of 786-O cells through
suppressing Ku80 expression. PLoS One. 10:e01229772015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wilmink GJ, Roth CL, Ibey BL, Ketchum N,
Bernhard J, Cerna CZ and Roach WP: Identification of microRNAs
associated with hyperthermia-induced cellular stress response. Cell
Stress Chaperones. 15:1027–1038. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yu J, Liu F, Yin P, Zhu X, Cheng G, Wang
N, Lu A, Luan W, Zhang N, Li J, et al: Integrating miRNA and mRNA
expression profiles in response to heat stress-induced injury in
rat small intestine. Funct Integr Genomics. 11:203–213. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Oshlag JZ, Devasthanam AS and Tomasi TB:
Mild hyperthermia enhances the expression and induces oscillations
in the Dicer protein. Int J Hyperthermia. 29:51–61. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Olivier M, Eeles R, Hollstein M, Khan MA,
Harris CC and Hainaut P: The IARC TP53 database: New online
mutation analysis and recommendations to users. Hum Mutat.
19:607–614. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Efeyan A and Serrano M: p53: Guardian of
the genome and policeman of the oncogenes. Cell Cycle. 6:1006–1010.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Graeber TG, Osmanian C, Jacks T, Housman
DE, Koch CJ, Lowe SW and Giaccia AJ: Hypoxia-mediated selection of
cells with diminished apoptotic potential in solid tumours. Nature.
379:88–91. 1996. View
Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ren YL, Yu JH and Kinghorn AD: Development
of anticancer agents from Plant-derived Sesquiterpene lactones.
Curr Med Chem. 23:2397–2420. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Navarro F and Lieberman J: miR-34 and p53:
New insights into a complex functional relationship. PLoS One.
10:e01327672015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yamakuchi M, Ferlito M and Lowenstein CJ:
miR-34a repression of SIRT1 regulates apoptosis. Proc Natl Acad Sci
USA. 105:pp. 13421–13426. 2008, View Article : Google Scholar : PubMed/NCBI
|
|
19
|
el-Deiry WS, Tokino T, Velculescu VE, Levy
DB, Parsons R, Trent JM, Lin D, Mercer WE, Kinzler KW and
Vogelstein B: WAF1, a potential mediator of p53 tumor suppression.
Cell. 75:817–825. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Sullivan KD, Gallant-Behm CL, Henry RE,
Fraikin JL and Espinosa JM: The p53 circuit board. Biochim Biophys
Acta. 1825:229–244. 2012.PubMed/NCBI
|
|
22
|
Campbell SC, Novick AC, Belldegrun A,
Blute ML, Chow GK, Derweesh IH, Faraday MM, Kaouk JH, Leveillee RJ,
Matin SF, et al: Guide lines for management of the clinical T1
renal mass. J Urol. 182:1271–1279. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Bordonaro M, Shirasawa S and Lazarova DL:
In Hyperthermia Increased ERK and WNT signaling suppress colorectal
cancer cell growth. Cancer (Basel). 8:pii:E492016. View Article : Google Scholar
|
|
24
|
Hobohm U, Stanford JL and Grange JM:
Pathogen-associated molecular pattern in cancer immunotherapy. Crit
Rev Immunol. 28:95–107. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Trieb K, Sztankay A, Amberger A, Lechner H
and Grubeck-Loebenstein B: Hyperthermia inhibits proliferation and
stimulates the expression of differentiation markers in cultured
thyroid carcinoma cells. Cancer Lett. 87:65–71. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Guan J, Stavridi E, Leeper DB and Iliakis
G: Effects of hyperthermia on p53 protein expression and activity.
J Cell Physiol. 190:365–374. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Okamoto K, Shinoura N, Egawa N, Asai A,
Kirino T, Shibasaki F and Shitara N: Adenovirus-mediated transfer
of p53 augments hyperthermia-induced apoptosis in U251 glioma
cells. Int J Radiat Oncol Biol Phys. 50:525–531. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Li T, Kon N, Jiang L, Tan M, Ludwig T,
Zhao Y, Baer R and Gu W: Tumor suppression in the absence of
p53-mediated cell cycle arrest, apoptosis, and senescence. Cell.
149:1269–1283. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Gu ZT, Wang H, Li L, Liu YS, Deng XB, Huo
SF, Yuan FF, Liu ZF, Tong HS and Su L: Heat stress induces
apoptosis through transcription-independent p53-mediated
mitochondrial pathways in human umbilical vein endothelial cell.
Sci Rep. 4:44692014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Juan LJ, Shia WJ, Chen MH, Yang WM, Seto
E, Lin YS and Wu CW: Histone deacetylases specifically
down-regulate p53-dependent gene activation. J Biol Chem.
275:20436–20443. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Luo J, Nikolaev AY, Imai S, Chen D, Su F,
Shiloh A, Guarente L and Gu W: Negative control of p53 by Sir2alpha
promotes cell survival under stress. Cell. 107:137–148. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Luo J, Su F, Chen D, Shiloh A and Gu W:
Deacetylation of p53 modulates its effect on cell growth and
apoptosis. Nature. 408:377–381. 2000. View
Article : Google Scholar : PubMed/NCBI
|
|
33
|
Vaziri H, Dessain SK, Eaton Ng E, Imai SI,
Frye RA, Pandita TK, Guarente L and Weinberg RA: hSIR2(SIRT1)
functions as an NAD-dependent p53 deacetylase. Cell. 107:149–159.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Finch RA, Donoviel DB, Potter D, Shi M,
Fan A, Freed DD, Wang CY, Zambrowicz BP, Ramirez-Solis R, Sands AT
and Zhang N: Mdmx is a negative regulator of p53 activity in vivo.
Cancer Res. 62:3221–3225. 2002.PubMed/NCBI
|