Introduction
Adoptive cellular immunotherapy (ACI), a developing
cancer therapeutic, is able to mobilize and strengthen the body's
immune system to kill cancer cells; however, clinical trials using
ACI have yielded low objective response rates (1). Inefficient T cell migration limits the
effect of cancer immunotherapy. Even large numbers of fully
activated tumor-specific cluster of differentiation
(CD)4+ T helper 1 and cytotoxic CD8+ T cells
may fail to reject established immunogenic murine or human tumors
due to their insufficient migration to tumor tissue (2,3). An
essential requirement for cancer immunotherapy is activation of
antigen-specific T cells and their migration to tumor sites
(4).
Radiotherapy has a synergistic effect on
immunotherapy. Low dose irradiation (LDI) of 2 Gy had been
demonstrated to induce the highest ratio of effector T cells to
FoxP3+ immunosuppressive regulatory T cells when
combined with adoptive T cell transfer (5). However, no previous reports have
investigated combining 2 Gy LDI with ACI to increase the migration
capacity of adoptive lymphocytes to tumor cells. The distribution
of immune cells injected into a tumor-bearing body after 2 Gy LDI
has not yet been described. Therefore, it would be worthwhile to
acquire visual evidence of the migration ability of immune cells
after 2 Gy LDI.
Chemokines and their receptors have a crucial role
in T cell recruitment to different tissues, and regulate both
homeostatic and inflammation-dependent homing of T cells (6–8).
However, little is understood about the chemokines involved in the
recruitment of effector T cells to tumors following irradiation.
Cancer cells produce several chemokines, largely to recruit
leukocytes that promote tolerance and immune escape, and to aid
tumor growth by enhanced angiogenesis. On the other hand, chemokine
(C-X-C motif) ligand 9 (CXCL9) and CXCL10 have been demonstrated to
recruit lymphocytes that reject the tumors (9,10).
Owing to the progression of fluorescence imaging
techniques, it is possible to trace the migration of cells in
living animal models (11). The
development of a near-infrared fluorescence imaging technique has
made it possible to trace living cells in deep tissue (12). Furthermore, fluorescent labeling of
immune cells with 1,1′-dioctadecyl-3,3,3′,3′-tetramethy
indotricarbocyanine iodide (DiR) does not adversely affect their
proliferation and cytotoxic function, and the labeling efficiency
is ~99.9% (13).
In the present study, a AGS-EBV preclinical model of
metastatic gastric adenocarcinoma was utilized to determine whether
2 Gy LDI is able to increase the migration capacity of cytotoxic T
lymphocytes (CTLs) to tumor cells. Whether LDI increased the
expression and release of chemokines CXCL9 and CXCL10 by AGS-EBV
cells and recruitment of tumor-specific activated CD8+
cells will be discussed. The present study monitored
antigen-specific CTLs dynamically labeled with the near-infrared
fluorescent dye, DiR. The present study highlights the therapeutic
potential of local LDI combined with ACI.
Materials and methods
Ethics statement
All animal protocols followed the experimental
procedures of the National Institutes of Health Guide for the Care
and Use of Laboratory Animals and were approved by the Ethics
Committee of Drum Tower Hospital. All donors provided their
informed written consent.
Cell line
Human gastric adenocarcinoma cell line AGS-EBV
(purchased from the Shanghai Institute of Biochemistry and Cell
Biology, Chinese Academy of Sciences, Shanghai, China) was cultured
in RPMI 1640 media (Sigma-Aldrich; Merck Millipore, Darmstadt,
Germany) supplemented with 10% fetal bovine serum (Invitrogen;
Thermo Fischer Scientific, Inc.) and 1 U/ml gentamicin (Gibco;
Thermo Fisher Scientific, Inc.). The culture was maintained in a
humidified atmosphere of 5% CO2 at 37°C.
Antigen peptides
Epstein-Barr virus (EBV)-latent membrane protein
(LMP)2A-derived peptide LMP2A356-364 (FLYALALLL) was synthesized
according to standard Fmoc solid phase synthesis methods
(GenScript, Nanjing, China). Peptide purity was determined using
analytical reverse-phase high performance liquid chromatography by
GenScript Corporation (Nanjing, China) and purity was routinely
>95%. Lyophilized peptides were diluted in dimethyl sulfoxide
and stored at −20°C.
Animals
A total of 30 female BALB/c nu/nu nude mice (6- to
8-weeks old; weight, 20±2 g) were obtained from the Animal Center
of the Nanjing University Medical School (Nanjing, China). All
animals were breed in a specific pathogen-free class barrier
system, had access to a 12-h light/dark cycle and were maintained
in a temperature controlled room (20–26°C) with a humidity of
50–80%. Mice had free access to water and food.
Induction of LMP2A-specific CTLs by
LMP2A peptide pulsed myeloid dendritic cells (mDCs) in vitro
Human CTLs of healthy donors were generated as
described previously (14). In
brief, mDCs were harvested after 5 days of culture in the presence
of recombinant human (rh) granulocyte macrophage colony-stimulating
factor (200 ng/ml) and rh interleukin (IL)-4 (100 ng/ml; both
PeproTech, Inc., Rocky Hill, NJ, USA), adjusted to 5×105
cells/ml in 24-well plates and incubated at 37°C for 48 h with
EBV-LMP2A peptide (25 µg/ml). Tumor necrosis factor (TNF)-α (200
ng/ml; PeproTech, Inc.) was added to the culture media, which was
then incubated at 37°C for 24 h. T cells were co-cultured with
LMP2A-DCs at a ratio of 10:1 in 24-well plates in 1 ml AIM-V medium
(Life Technologies; Thermo Fisher Scientific, Inc.) supplemented
with 10% human AB serum (Gibco; Thermo Fisher Scientific, Inc.) at
37°C in 5% CO2 for 5–7 days. Cells were stimulated with
fresh peptide-pulsed autologous DCs twice daily for 7 days. After 3
days, rhIL-2 was added at a final concentration of 20 U/ml to all
wells and this was repeated every 2–3 days thereafter. On day seven
after the final stimulation, cells were harvested and LMP2A356-364
epitope-specific cytolytic activity of T lymphocytes was measured
using tetramer technology. Briefly, the single cell suspension was
adjusted to 5×106 cells/ml and 1 ml was inserted into a
1.5-ml Eppendorf tube. Following washing two times with phosphate
buffered saline (PBS), the cells were suspended in 5 µl
phycoerythrin-labeled LMP2A-356 tetramer (Kuangbo Biotechnology
Corp, Beijing, China), incubated in the dark at 4°C for 20 min and
washed two times with PBS again. Subsequently, 2.5 µl
PerCP-Cy5.5-labeled human CD3 antibodies (BD Biosciences, Franklin
Lakes, NJ, USA; cat. no. 6203-25; 1:40 dilution) and 2.5 µl
allophycocyanin-labeled human CD8 antibodies (BD Biosciences; cat.
no. 641400; 1:40 dilution) were added and the mixture was incubated
in the dark at 4°C for 20 min. Subsequently, cells were washed with
PBS two times again and suspended to 300 µl PBS prior to being
examined using a flow cytometer. Data was analyzed using CellQuest
software (Version 5.1, BD Biosciences).
Detection of secretion of chemokines
in tumor cells
Gastric carcinoma AGS-EBV cells were incubated in
RPMI 1640 medium with no serum for 12 h to starve the cells.
Subsequently, the cells were plated at 1×104 cells/well in
duplicate wells of a 96-well plate. Chemokine concentrations were
measured 24 h after 2 or 5 Gy radiation or mock treatment. CXCL9
and CXCL10 release in culture supernatant was determined using
ELISA kits (RayBiotech, Inc., Norcross, GA, USA; cat. nos. QC123
and QC23).
Transwell migration assay
Transwell migration assays were performed using 3-µm
Transwell chambers (Corning, Inc., Corning, NY, USA). Briefly,
5×105 T cells were added to the upper chamber in 100 µl
of AIM V supplemented with 1% bovine serum albumin (chemotaxis
buffer) (Life Corporation). The lower chamber contained 600 µl of
chemotaxis buffer and 1×105 AGS-EBV cells 24 h after
they had been irradiated with 2 or 5 Gy or not treated. After
incubation for 4 h at 37°C, cells that migrated inzto the lower
chamber were collected and counted.
Fluorescent labeling of immune
cells
DiR is a lipophilic, near-infrared fluorescent
cyanine dye used for labeling the cytoplasmic membrane. CTLs were
suspended at a concentration of 1×106 cells/ml. DiR
working solution (1 µg/ul) was added into the cell suspension and
incubated for 30 min at 37°C with 10 ul of dye used per
106 cells. Following this, the dye was cleared away with
two washes using AIM V medium.
Establishment of a gastric carcinoma
model
A total of 20 female nude mice were injected
subcutaneously with 1×107 AGS-EBV cells. After tumor
formation (2–3 week later), nude mice bearing gastric carcinomas
were randomly divided into two groups: Intravenous injection of
CTLs (CTL-i.v.) group and intravenous injection of CTLs plus local
2 Gy LDI at tumor sites (CTL-i.v. + LDI) group.
Adoptive transfer and LDI
Tumors were either not irradiated or locally
irradiated with a 2-Gy dose of radiation using an X-RAD 320
Biological Irradiator (Precision X-Ray, North Branford, CT, USA) 1
day prior to transfer of CTLs. Saline suspensions of CTLs labeled
with DiR were constructed at a concentration of 1×108
cells/ml and each tumor-bearing mouse received an infusion of 0.1
ml (1×107) labeled cells.
Fluorescence live imaging (FLI)
After infusion of CTLs, each mouse was anesthetized
with 2% isoflurane (Yuyan Instruments, Shanghai, China) and FLI was
performed using the Xenogen IVIS-Spectrum Imaging System (Xenogen;
Caliper Life Sciences, Inc., Hopkinton, MA, USA). Imaging
examination times were set as follows: Day 0, 1 h after infusion;
2, 4, 8 and 24 h after infusion (day 1); and 2, 6, 9 and 12 days
after infusion. Living Image v.4.1 software (PerkinElmer, Waltham,
MA, USA) was used to draw and calculate the region of interest.
Flow cytometry to detect the content
of CTLs in tumor tissue
Recipient tumor-bearing mice were sacrificed 8 h
after injection of DiR-labeled CTLs. The tumors of these mice were
isolated. Single cell suspensions of tumor tissues were prepared
and the CTL content was detected using flow cytometry. The single
cell suspension was adjusted to 1–2×107 cells/ml and 1
ml was inserted into a 1.5-ml Eppendorf tube. Following washing two
times with PBS, the cells were suspended in 5 µl CD3 antibodies (BD
Biosciences; cat. no. 6203-25, 1:20 dilution) respectively, and
incubated in the dark at 4°C for 30 min. Subsequently, cells were
washed with PBS two times again and suspended to 1 ml PBS prior to
being examined using a flow cytometer. Analysis was performed using
CellQuest software (Version 5.1, BD Biosciences).
Statistical analysis
All statistical analyses were performed using SPSS
v.17.0 software (SPSS, Inc., Chicago, IL, USA). One way analysis of
variance and Newman-Keuls multiple comparison tests were used to
compare the differences between multiple groups. Unpaired
two-tailed Student's t-tests were used to analyze differences
between two groups. Data were presented as the mean ± standard
deviation. P<0.05 was considered to indicate a statistically
significant difference.
Results
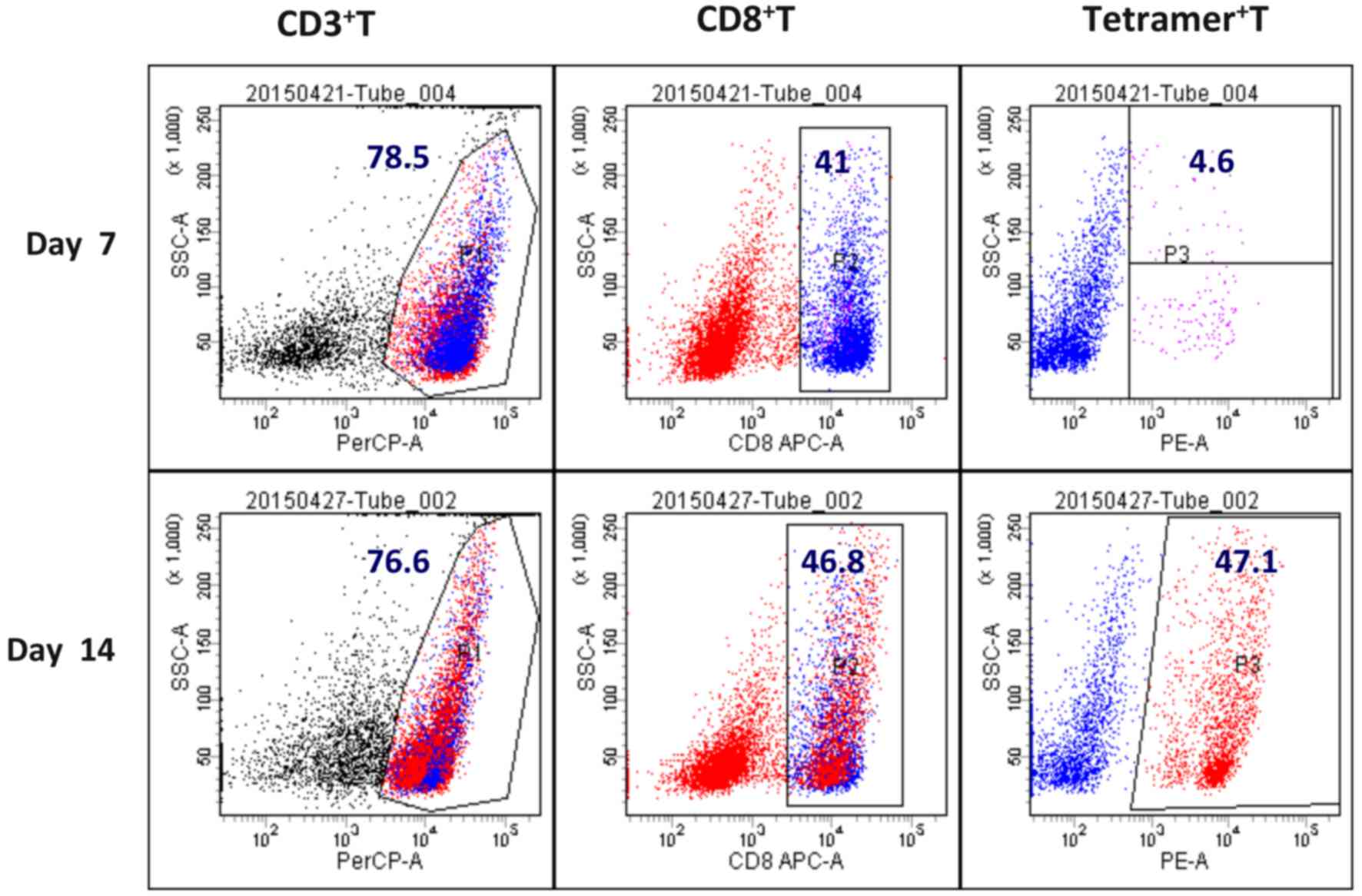
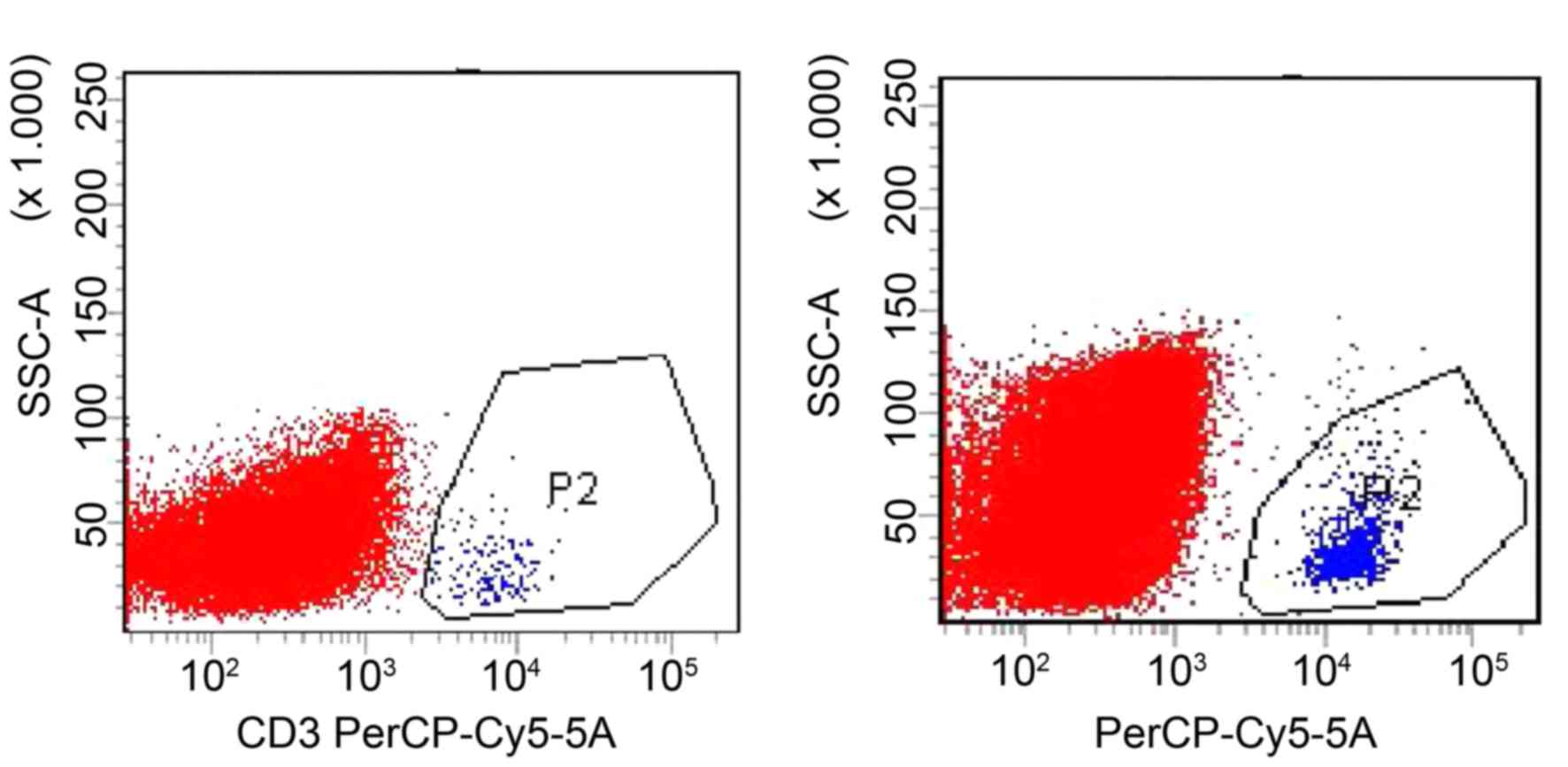
Content of LMP2A-specific CTLs
The content of LMP2A-specific T cells in the CTL
population was detected by EBV-LMP2A-356 tetramer technology. The
EBV-LMP2A-356 antigen peptide-loaded DC induced peripheral blood
mononuclear cells into CTLs in vitro. At 14 days after the
cells were cultured, a small amount (5×106) of CTLs were
incubated with fluorescein-labelled EBV-LMP2A-356 tetramers for 30
min, and CD3 and CD8 were subsequently labelled with a flow
cytometry antibody. It was demonstrated that the
EBV-LMP2A-356-specific CTLs accounted for 47.1% of CD8+
T cells (Fig. 1).
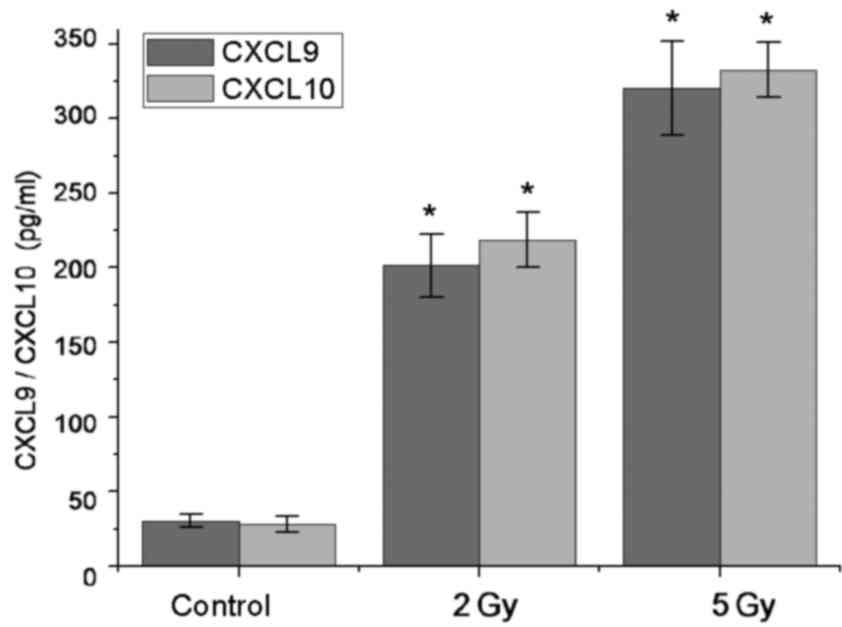
Radiation induces upregulation of
CXCL9 and CXCL10 release in tumor cells
Chemotaxis assays were performed to detect the
secretion of chemokines CXCL9 and CXCL10 by AGS-EBV cells that were
able to recruit antitumor effector T cells. ELISA test results
demonstrated that there was not significant CXCL9 and CXCL10
release in AGS-EBV cells at the basal condition (CXCL9, 30.23±4.31
pg/ml; CXCL10, 28.21±5.34 pg/ml). However, in 2 Gy-irradiated tumor
cells, soluble CXCL9 and CXCL10 significantly increased by ~7-fold
(CXCL9, 201.32±21.37 pg/ml; CXCL10, 218.54±18.37 pg/ml) compared
with non-irradiated tumor cells (P<0.05). In 5 Gy-irradiated
tumor cells, soluble CXCL9 and CXCL10 was significantly increased
by ~10-fold (CXCL9, 320.47±31.47 pg/ml; CXCL10, 332.56±18.37 pg/ml)
compared with non-irradiated tumor cells (P<0.05; Fig. 2).
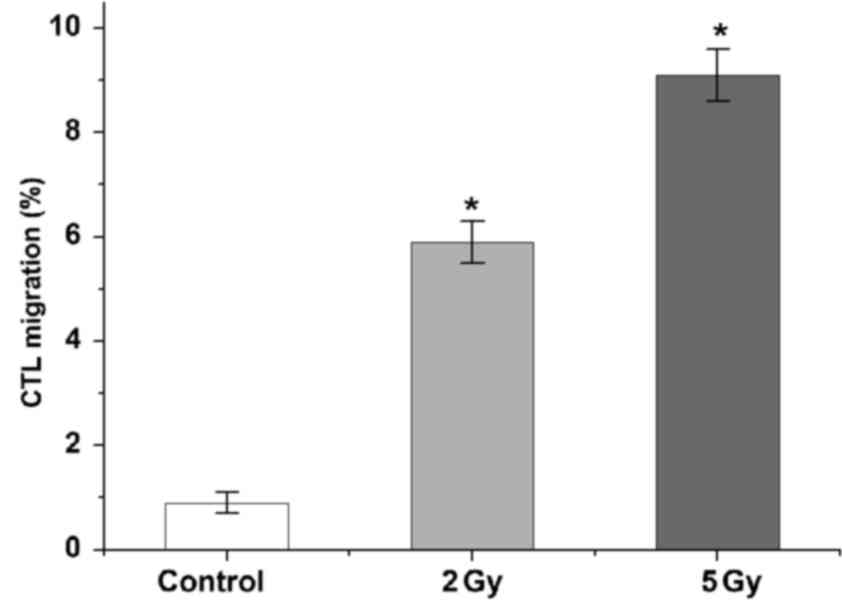
LDI attracts CTLs to tumor cells in
vitro
Transwell migration assays demonstrated that both 2
and 5 Gy LDI promoted immune cell migration to tumor sites. There
was no evident immune cell migration towards non-irradiated gastric
cancer cells (0.9±0.2%). As expected, migration of CTLs towards
irradiated AGS-EBV cells was significantly increased ~6-fold after
2 Gy LDI (5.9±0.4%) and ~9-fold after 5 Gy LDI (9.1±0.5%;
P<0.05; Fig. 3).
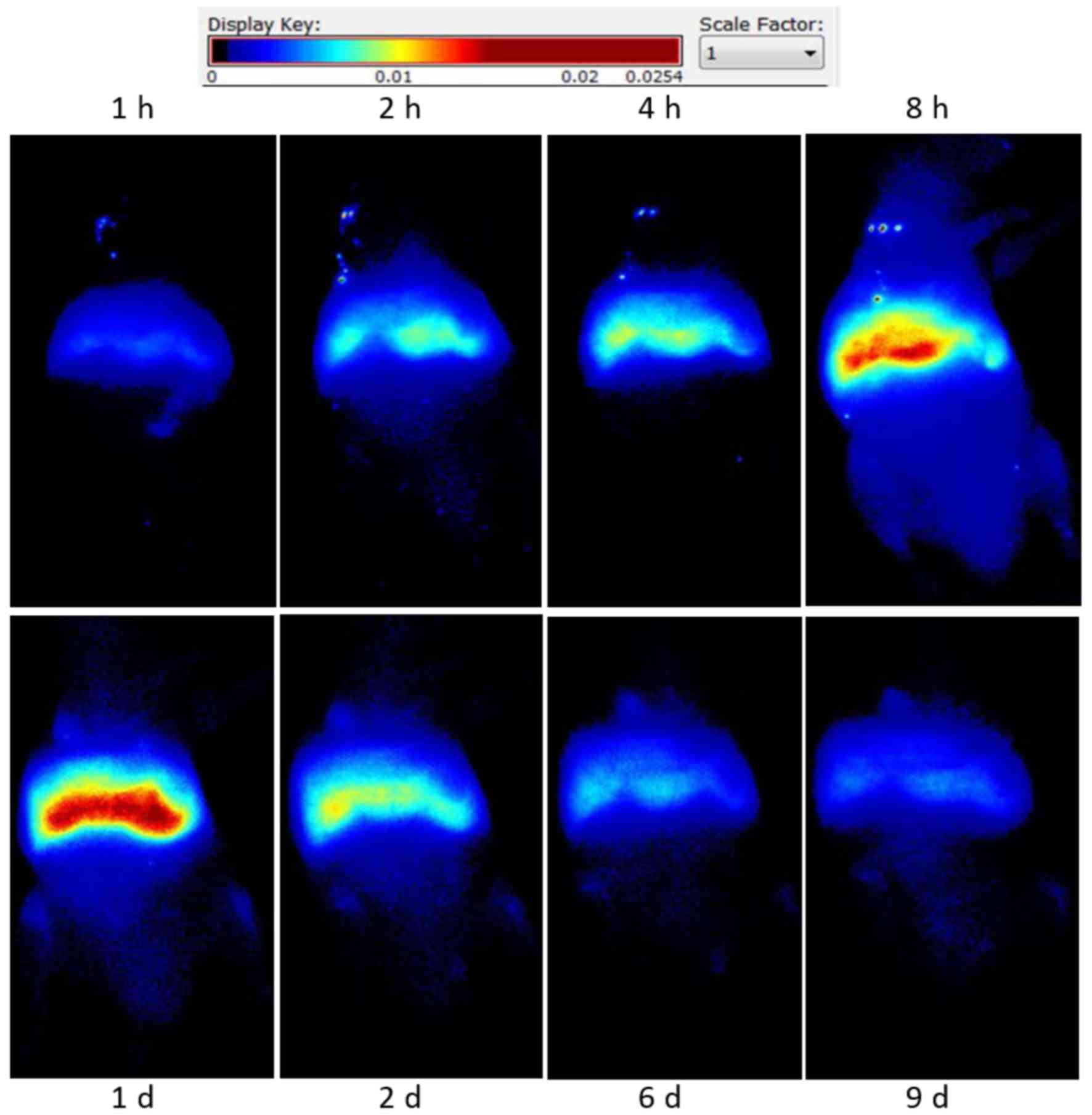
Distribution of CTLs in tumor-bearing
mice
DiR-labeled CTLs were injected into tumor-bearing
nude mice, intravenously. Fluorescent signal intensity was observed
at different times using fluorescence imaging. CTLs were
predominantly accumulated in the liver and spleen region, their
levels peaked at 24 h and gradually decreased and cleared at 9 days
(Fig. 4).
Effect of LDI on the distribution of
CTLs in tumor-bearing mice
Intravenous-infused CTLs displayed enhanced
accumulation in tumor sites after 2 Gy LDI, with CTL levels peaking
at 8 h. These levels then gradually decreased and cleared at ~24 h
(Fig. 4). Enhanced accumulation of
CTLs at un-irradiated tumor sites was not observed in the control
group. Living Image v.4.1 software was used to draw and calculate
the region of interest.
Effect of LDI on the migration of CTLs
to tumor sites in vivo
Flow cytometry results demonstrated that only a
small proportion of lymphocytes were detected in the tumor tissue
of the CTL-i.v. group, with a ratio of T lymphocytes in the single
cell suspension of tumor tissue of 0.15±0.03%. A significantly
increased number of lymphocytes (~9-fold) was detected in the
CTL-i.v. + LDI group compared with the CTL-i.v. group, with a ratio
of T lymphocytes in the single cell suspension of tumor tissue of
1.45±0.14% (P<0.05; Fig. 5).
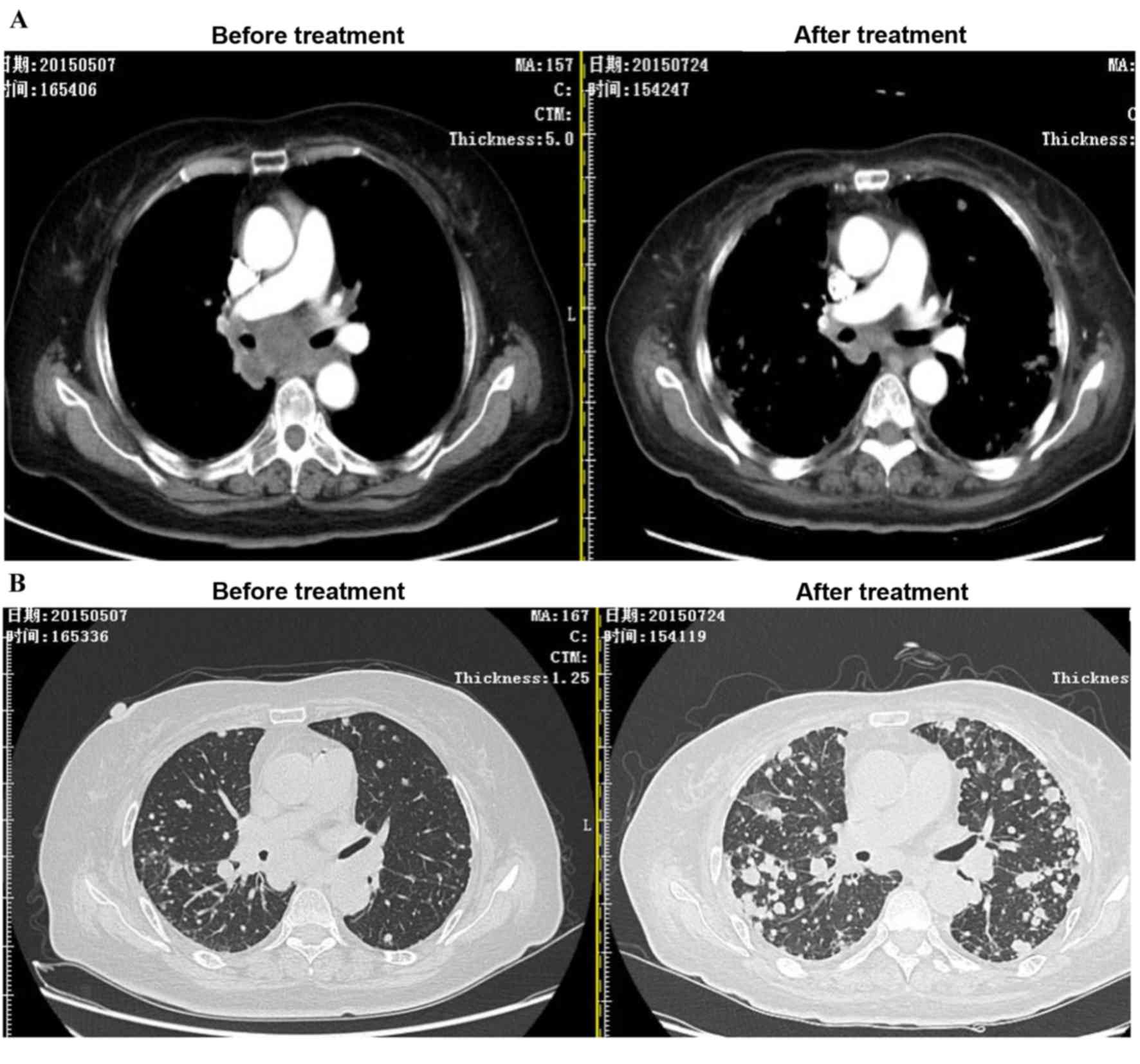
Effect of LDI in a patient with
metastatic gastric cancer
In a case study, ACI combined with 2 Gy LDI was
performed on a stage IV cancer patient with a metastatic
mediastinum tumor. As expected, after two courses of LDI + ACI
treatment, the size of the mediastinum tumor was markedly reduced
compared to the tumor size before treatment; however, the lesions
as a result of radiotherapy were increased (Fig. 6).
Discussion
In the present study it was demonstrated, for the
first time, that 2 Gy LDI is able to markedly upregulate the
release of chemokines CXCL9 and CXCL10 from human gastric cancer
cells. It was also demonstrated that local tumor LDI increases the
migration of activated CTLs to tumor sites, both in vitro
and in vivo.
An essential requirement for cancer immunotherapy is
activation of antigen-specific T cells and their homing to tumors.
Although clinical studies on antitumoral immunization have reported
the presence of immune effector cells in the periphery or at the
injection site, the therapeutic antitumor responses remain limited
(2,15). The most obvious reason for the lack
of clinical response is insufficient tumor infiltration of immune
effector cells (4).
Radiotherapy is a standard treatment used in
oncology and may be very precisely targeted to tumors to minimize
immunosuppressive side effects, which is an advantage over systemic
chemotherapy (16). Recently, the
possibility of combining local radiotherapy with immunotherapy has
been explored (17). The ability of
irradiation to convert tumors into inflamed peripheral tissues may
be exploited to overcome obstacles at the effector phase of the
antitumor immune response and improve the therapeutic efficacy of
immunotherapy (17). However,
irradiation alone has been demonstrated to increase the number of
intratumoral myeloid-derived suppressor cells (MDSCs) (18). The number of MDSCs has not been
demonstrated to increase when irradiation is combined with
immunization (19). Furthermore,
Klug et al (5) demonstrated
that LDI of 2 Gy resulted in the highest ratio of effector T cells
to FoxP3+ immunosuppressive regulatory T cells when
combined with adoptive T cell transfer. The ratio of
antigen-specific CD8+ T cells and MDSCs in tumors
increased by ~85-fold compared with the control group (5). Based on these findings, the present
study used 2 Gy LDI. Additionally, LDI induces minimal damage to
surrounding normal tissue, can be widely applied and is
particularly suitable for recurrent tumors that have received
radiotherapy previously (20). LDI
may be more suitable as a clinical treatment when combined with
immunotherapy.
Effective T cell migration into peripheral tissues
occurs at sites of infection-induced inflammation, through
interaction with endothelial cells and is guided by chemokines
secreted by activated cells (21,22). A
study by Matsumura et al (23) demonstrated that ionizing radiation
markedly enhanced the secretion of CXCL16 by murine and human
breast cancer cells, resulting in the recruitment of antitumor
effector T cells to sites of inflammation. Soluble CXCL9 and CXCL10
has been demonstrated to induce strong chemotaxis of activated T
cells and natural killer cells, which express high levels of
CXCR3+ (2,24,25).
Similarly, the results of the present study indicate that
LDI-treated EBV-AGS tumor cells induce strong
CXCL9/CXCL10-dependent chemotaxis of activated CD8+
cells in vitro (Fig. 3), and
that local tumor LDI in vivo may enhance the recruitment of
tumor-specific CD8+ cells (Fig. 4).
The mechanisms by which LDI regulates trafficking of
T cells to solid tumors remain largely undefined. Klug et al
(5) also demonstrated that local
neoadjuvant LDI resulted in normalization of aberrant vasculature
and efficient recruitment of tumor-specific T cells in human
pancreatic carcinomas, which led to T cell-mediated tumor rejection
and prolonged survival in xenotransplant murine tumor models. From
the present study, it is evident that 2 Gy LDI is able to markedly
enhance the secretion of chemokines CXCL9 and CXCL10 by human
gastric cancer cells, which have an important role in the
recruitment of tumor-specific CTLs to tumor sites. Localized 2 Gy
LDI induces enhanced intratumoral levels of immune cell populations
that are crucial for stimulating the antitumor responses in the
effector phase of the antitumor immune response. The underlying
molecular mechanisms are, at least partly, due to
irradiation-induced changes in chemokine levels.
In conclusion, the present study demonstrates that
local 2 Gy LDI of tumors induces upregulation of chemokine
secretion, resulting in the recruitment of activated T cells to the
tumor. These results suggest that LDI may be an easily translatable
strategy to overcome immune barriers at the effector phase of
ACI.
References
|
1
|
Smyth MJ, Ngiow SF, Ribas A and Teng MW:
Combination cancer immunotherapies tailored to the tumour
microenvironment. Nat Rev Clin Oncol. 13:143–158. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Abastado JP: The next challenge in cancer
immunotherapy: Controlling T-cell traffic to the tumor. Cancer Res.
72:2159–2161. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Fridman WH, Galon J, Pagès F, Tartour E,
Sautès-Fridman C and Kroemer G: Prognostic and predictive impact of
intra- and peritumoral immune infiltrates. Cancer Res.
71:5601–5605. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Draghiciu O, Nijman HW and Daemen T: From
tumor immunosuppression to eradication: Targeting homing and
activity of immune effector cells to tumors. Clin Dev Immunol.
2011:4390532011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Klug F, Prakash H, Huber PE, Seibel T,
Bender N, Halama N, Pfirschke C, Voss RH, Timke C, Umansky L, et
al: Low-dose irradiation programs macrophage differentiation to an
iNOS+/M1 phenotype that orchestrates effective T cell
immunotherapy. Cancer Cell. 24:589–602. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Domanska UM, Kruizinga RC, Nagengast WB,
Timmer-Bosscha H, Huls G, de Vries EG and Walenkamp AM: A review on
CXCR4/CXCL12 axis in oncology: No place to hide. Eur J Cancer.
49:219–230. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kang TH, Mao CP, Lee SY, Chen A, Lee JH,
Kim TW, Alvarez RD, Roden RB, Pardoll D, Hung CF and Wu TC:
Chemotherapy acts as an adjuvant to convert the tumor
microenvironment into a highly permissive state for
vaccination-induced antitumor immunity. Cancer Res. 73:2493–2504.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Marchesi F, Grizzi F, Laghi L, Mantovani A
and Allavena P: Molecular mechanisms of pancreatic cancer
dissemination: The role of the chemokine system. Curr Pharm Des.
18:2432–2438. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Franciszkiewicz K, Boissonnas A, Boutet M,
Combadière C and Mami-Chouaib F: Role of chemokines and chemokine
receptors in shaping the effector phase of the antitumor immune
response. Cancer Res. 72:6325–6332. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ohtani H, Jin Z, Takegawa S, Nakayama T
and Yoshie O: Abundant expression of CXCL9 (MIG) by stromal cells
that include dendritic cells and accumulation of CXCR3+ T cells in
lymphocyte-rich gastric carcinoma. J Pathol. 217:21–31. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Frangioni JV: In vivo near-infrared
fluorescence imaging. Curr Opin Chem Biol. 7:626–634. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
He X, Gao J, Gambhir SS and Cheng Z:
Near-infrared fluorescent nanoprobes for cancer molecular imaging:
Status and challenges. Trends Mol Med. 16:574–583. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
DU X, Wang X, Ning N, Xia S, Liu J, Liang
W, Sun H and Xu Y: Dynamic tracing of immune cells in an orthotopic
gastric carcinoma mouse model using near-infrared fluorescence live
imaging. Exp Ther Med. 4:221–225. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Liu G, Yao K, Wang B, Chen Y, Zhou F, Guo
Y, Xu J and Shi H: Immunotherapy of Epstein-Barr virus associated
malignancies using mycobacterial HSP70 and LMP2A356-364 epitope
fusion protein. Cell Mol Immunol. 6:423–431. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Draghiciu O, Nijman HW and Daemen T: From
tumor immunosuppression to eradication: Targeting homing and
activity of immune effector cells to tumors. Clin Dev Immunol.
2011:4390532011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Demaria S, Pilones KA, Vanpouille-Box C,
Golden EB and Formenti SC: The optimal partnership of radiation and
immunotherapy: From preclinical studies to clinical translation.
Radiat Res. 182:170–181. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Reynders K and De Ruysscher D:
Radiotherapy and immunotherapy: Improving cancer treatment through
synergy. Prog Tumor Res. 42:67–78. 2015.PubMed/NCBI
|
|
18
|
Kalbasi A, June CH, Haas N and Vapiwala N:
Radiation and immunotherapy: A synergistic combination. J Clin
Invest. 123:2756–2763. 2013. View
Article : Google Scholar : PubMed/NCBI
|
|
19
|
Soukup K and Wang X: Radiation meets
immunotherapy - a perfect match in the era of combination therapy?
Int J Radiat Biol. 91:299–305. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Patel NR, Lanciano R, Sura K, Yang J,
Lamond J, Feng J, Good M, Gracely EJ, Komarnicky L and Brady L:
Stereotactic body radiotherapy for re-irradiation of lung cancer
recurrence with lower biological effective doses. J Radiat Oncol.
4:65–70. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Bryant J, Ahern DJ and Brennan FM: CXCR4
and vascular cell adhesion molecule 1 are key chemokine/adhesion
receptors in the migration of cytokine-activated T cells. Arthritis
Rheum. 64:2137–2146. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Lim JY, Gerber SA, Murphy SP and Lord EM:
Type I interferons induced by radiation therapy mediate recruitment
and effector function of CD8(+) T cells. Cancer Immunol Immunother.
63:259–271. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Matsumura S, Wang B, Kawashima N,
Braunstein S, Badura M, Cameron TO, Babb JS, Schneider RJ, Formenti
SC, Dustin ML and Demaria S: Radiation-induced CXCL16 release by
breast cancer cells attracts effector T cells. J Immunol.
181:3099–3107. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zhu Q, Han X, Peng J, Qin H and Wang Y:
The role of CXC chemokines and their receptors in the progression
and treatment of tumors. J Mol Histol. 43:699–713. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Balkwill FR: The chemokine system and
cancer. J Pathol. 226:148–157. 2012. View Article : Google Scholar : PubMed/NCBI
|