Introduction
Porcine reproductive and respiratory syndrome
(PRRS), which is characterized by reproductive disorders in
pregnant sows and respiratory diseases in growing pigs, is a viral
disease caused by PRRS virus (PRRSV) (1,2). Since
its first outbreak in 1987 in USA (3), PRRS has spread to the major
pig-producing countries and regions (4), causing tremendous economic losses to
the world pig industry. The emergence and prevalence of highly
pathogenic (HP)-PRRSV with a 30-aa discontinuous deletion in its
nsp2-coding region devastated the Chinese swine industry in 2006
(5,6). PRRSV, a single positive-stranded RNA
virus, is classified into the genus Arterivirus of family
Arteriviridae in the order Nidovirales (7–9). This
virus can be divided into two genotypes, namely types 1 and 2
(10–12).
Studies have shown that PRRSV can utilize
post-transcriptional control to inhibit the production of type I
interferon (IFN) in its natural target cells (13,14).
PRRSV-infected target cells and host pig fail to induce the protein
production of type I IFN. IFN-α protein was undetectable in lungs
of PRRSV-infected pigs, and only a very small amount of IFN-α
protein could be detected or was undetectable in the culture
supernatant of PRRSV-infected peripheral blood mononuclear cells
and monocyte dendritic cells in vitro (15–17).
PRRSV cannot induce protein expression of IFN-α during viral
infection, furthermore it blocks the production of IFN-α induced by
porcine transmissible gastroenteritis virus (TGEV) or
double-stranded RNA polyinosinic-polycytidylic acid (poly I:C)
in vitro (17,18). However, the mRNA transcription of α
and IFN-α can be activated by PRRSV in host target cells in
vitro and in vivo (14,19,20).
These findings clearly indicate that PRRSV suppresses the
production of type I IFN in host by post-transcriptional and
translational control. Although several previous studies exploring
the molecular basis for PRRSV-mediated inhibition of type I IFN
have demonstrated that PRRSV-encoded nsp 1, nsp 2, and nsp 11 and N
protein play key roles in antagonizing the activation of IFN-β
promoter (21), these focused mainly
on the stage of IFN-β mRNA transcription, primarily in PRRSV
permissive MARC-145 cells or non-permissive human cell culture
systems. Therefore, these data have uncertain relevance to the host
cells naturally infected with PRRSV. To date, the
post-transcriptional mechanism concerning PRRSV-mediated innate
immune response escape, especially blocking the production of type
I IFN, during viral infection in host cells, remains unclear.
MicroRNAs (miRNAs) are small non-coding RNAs with
approximately 22 nucleotides in length, which can bind to the miRNA
seed sequence (2–8 nucleotides) complementarity motifs locating
usually within the 3′-untranslated region (3′UTR) of the target
mRNA to directly target gene silencing through mRNA cleavage,
transcription degradation or translational inhibition (22). Although miRNAs can be induced or
repressed directly by type I IFN, they play a crucial role in
regulating the innate immune response including suppression or
enhancement of type I IFN production (23). Moreover, during viral infection, not
only can host cellular miRNAs target directly viral RNAs or
regulate host antiviral genes to modify the cellular state for
performing antiviral functions, but also cellular miRNAs can be
manipulated by virus to facilitate viral replication (24,25).
Therefore, it is not surprising that PRRSV may interact with host
cellular miRNAs to evade the type I IFN response. Actually, several
previous studies have demonstrated that cellular miRNAs could be
modulated during PRRSV infection (26–28), and
some miRNAs could promote or inhibit PRRSV replication (29–32).
Simultaneously, the mRNA transcription of type I IFN could be
upregulated or downregulated by PRRSV-influenced cellular miRNAs
(33–35), however, no literature on how
PRRSV-modulating cellular miRNAs manipulate the protein expression
of type I IFN have been documented.
Different strains of PRRSV have different
sensitivity to IFN-α, and possess different abilities to inhibit
type I IFN induction (20), whilst
cellular miRNA expression profile in porcine alveolar macrophages
(PAMs) is also identified to be PRRSV strain-specific during viral
infection (26). The inhibition of
IFN-β by PRRSV is of significant difference among different type of
cells (36). PRRSV vaccine strain is
insensitive to type I IFN in MARC-145 cells, however, in primary
PAMs, PRRSV vaccine strain and virulent strain are sensitive to
type I IFN. PRRSV has been shown to activate IFN-β transcription in
porcine monocyte-derived dendritic cells (14), but in MARC-145 cells, PRRSV
inactivates and inhibits IFN-β transcription activated by poly I:C
(18). In addition to PRRSV strains
and cell types, different pig breeds may also exhibit distinct
characteristics during PRRSV infection. Compared with other pig
breeds, PRRSV replication and proliferation can be suppressed or
delayed in the PAMs of landrace pigs (37). Cellular miRNAs in lung tissues show
significantly differential expression between tongcheng and
landrace pigs during HP-PRRSV infection (27). Moreover, since the ultraviolet light-
and heat-inactivated PRRSV cannot suppress type I IFN production
induced by TGEV infection or poly I:C stimulation in PAMs, the
inhibition of type I IFN by PRRSV in natural host cell may be
pathogenicity-related (13).
Additionally, the pathogenicity between HP-PRRSV and low pathogenic
PRRSV strains are significantly different in vivo and in
vitro, therefore, the question of whether HP-PRRSV and low
pathogenic PRRSV show differences in the regulation and miRNA
modulation of type I IFN during viral infection, is of considerable
interest.
In the present study, we analyzed the inhibitory
effect of PRRSV infection on the post-transcriptionally protein
expression of porcine IFN-β, and further explored the relationship
between cellular miRNAs and IFN-β protein expression in primary
PAMs.
Materials and methods
Cells and virus
Primary PAMs were prepared as previously described
(38). PAM cell line 3D4/21
(CRL-2843) was obtained from the American Type Culture Collection
(ATCC) (Manassas, VA, USA). The cells were grown in RPMI-1640
medium (Thermo Fisher Scientific, Inc., Waltham, MA, USA)
supplemented with 10% fetal bovine serum (Hyclone Laboratories
Inc., Logan, UT, USA) at 37°C, with 5% CO2. The HP-PRRSV
JXwn06 (GenBank accession no. EF641008) and low pathogenic PRRSV
HB-1/3.9 (GenBank accession no. EU360130) were used in this study
(39).
miRNA target predictions
To analyze the interaction between cellular miRNAs
and porcine IFN-β 3′UTR, we selected a serial of miRNAs, especially
porcine let-7b, miR-26a, miR-34a and miR-145 obtained from miRBase
(http://www.mirbase.org), and predicted
miRNA-targeted binding sequences in porcine IFN-β 3′UTR, whose
sequences were from GenBank (http://www.ncbi.nlm.nih.gov/GenBank), by miRanda
(40), RNA hybrid (41) and PITA (42).
miRNA mimics and miRNA inhibitors
Based on the sequences of porcine cellular miRNAs,
including let-7b, miR-26a, miR-34a and miR-145, miRNA mimics and
miRNA inhibitors and negative control were designed (Table I), and were synthesized with
2′-O-methyl modification by Shanghai GenePharma Co., Ltd.
(Shanghai, China).
 | Table I.The sequences of the synthesized
porcine miRNA mimics and miRNA inhibitors. |
Table I.
The sequences of the synthesized
porcine miRNA mimics and miRNA inhibitors.
| miRNA | miRNA mimic
sequence (5′→3′) | miRNA inhibitor
sequence (5′→3′) |
|---|
| let-7b |
UGAGGUAGUAGGUUGUGUGGUU |
AACCACACAACCUACUACCUCA |
| miR-26a |
UUCAAGUAAUCCAGGAUAGGCU |
AGCCUAUCCUGGAUUACUUGAA |
| miR-34a |
UGGCAGUGUCUUAGCUGGUUGU |
ACAACCAGCUAAGACACUGCCA |
| miR-145 |
GUCCAGUUUUCCCAGGAAUCCCUU |
AAGGGAUUCCUGGGAAAACUGGAC |
| NC |
UUGUACUACACAAAAGUACUG |
CAGUACUUUUGUGUAGUACAA |
Plasmid construction
Three plasmids including pmirGLO-IFN-β 3′UTR,
pmirGLO-miRNA recognition elements (MRE)-wild type (wt) and
pmirGLO-MRE-mutant type (mut) were constructed based on the pmirGLO
dual-lucife-rase miRNA target expression vector (Promega Corp.,
Madison, WI, USA) following the manufacturer's instructions.
To construct pmirGLO-IFN-β 3′UTR, the PCR
amplification primers were designed by Primer Premier v5.0
according to the sequence of porcine IFN-β 3′UTR (GenBank accession
no. M86762). PmeI and XbaI restriction enzyme sites
(underlined letters) were introduced in the upstream forward primer
(5′-ATAGTTTAAACACATCTCCCCCCTGTGGCT-3′)
and downstream reverse primer (5′-TGCTCTAGAGGATCCTTAACCCACTGATCCAG-3′),
respectively. Taken PAMs genomic DNA as template, PCR was performed
to generate porcine IFN-β 3′UTR by high-fidelity
PrimeSTAR® HS DNA Polymerase (Takara Biotechnology Co.,
Ltd., Dalian, China) according to manufacturer's instructions.
After digested with PmeI and XbaI, both porcine IFN-β
3′UTR gene fragments and pmirGLO dual firefly/Renilla
luciferase miRNA target expression vector were ligated together by
T4 DNA ligase (Promega Corp.) following the manufacturer's
protocol.
To generate pmirGLO-MRE-wt and pmirGLO-MRE-mut,
according to the predicted MRE in porcine IFN-β 3′UTR for let-7b,
miR-26a, miR-34a and miR-145, the DNA fragments of wild-type MRE
and mutant-type MRE (base mutations in the seed region) with
NotI restriction endonuclease sites were prepared by
annealing the primer pairs listed in Table II under the following conductions:
94°C 4 min, 70°C 10 sec, 65°C 10 sec, 60°C 10 sec, 58°C 10 sec,
55°C 10 sec, 55°C water bath to cool for 90 min, and then the
resulting product DNA fragments were cloned into the pmirGLO dual
firefly/Renilla luciferase miRNA target expression vector
previously digested with PmeI and XbaI, by T4 DNA
ligase according to manufacturer's instructions. All plasmids were
verified by digesting with NotI and sequencing.
 | Table II.Primers for wild-type and mutant-type
MRE in porcine IFN-β 3′UTR. |
Table II.
Primers for wild-type and mutant-type
MRE in porcine IFN-β 3′UTR.
| Primera | Primer sequence
(5′→3′)b |
|---|
| let-7b MRE-F | AAACTAGCGGCCGCTAGTATGTATTTAATTTTTTACCTTGT |
| let-7b MRE-R |
CTAGACAAGGTAAAAAATTAAATACATACTAGCGGCCGCTAGTTT |
| let-7b Mut-F | AAACTAGCGGCCGCTAGTATGTATTTAATTTTGATGGAGGT |
| let-7b Mut-R |
CTAGACCTCCATCAAAATTAAATACATACTAGCGGCCGCTAGTTT |
| miR-26a MRE-F | AAACTAGCGGCCGCTAGTCCCCTGTGGCTCTGGGAATTGACT |
| miR-26a MRE-R |
CTAGAGTCAATTCCCAGAGCCACAGGGGACTAGCGGCCGCTAGTTT |
| miR-26a Mut-F | AAACTAGCGGCCGCTAGTCCCCTGTGGCTCTGGGTGAACTCT |
| miR-26a Mut-R |
CTAGAGAGTTCACCCAGAGCCACAGGGGACTAGCGGCCGCTAGTTT |
| miR-34a MRE-F | AAACTAGCGGCCGCTAGTTTGACCATGTTGGCAATGATGT |
| miR-34a MRE-R |
CTAGACATCATTGCCAACATGGTCAAACTAGCGGCCGCTAGTTT |
| miR-34a Mut-F | AAACTAGCGGCCGCTAGTTTGACCATGTTGGGTGACGGGT |
| miR-34a Mut-R |
CTAGACCCGTCACCCAACATGGTCAAACTAGCGGCCGCTAGTTT |
| miR-145 MRE-F | AAACTAGCGGCCGCTAGTCTGTGGCTCTGGGAATTGACCT |
| miR-145 MRE-R |
CTAGAGGTCAATTCCCAGAGCCACAGACTAGCGGCCGCTAGTTT |
| miR-145 Mut-F | AAACTAGCGGCCGCTAGTCTGTGGCTCTGGGTTGACCTCT |
| miR-145 Mut-R |
CTAGAGAGGTCAACCCAGAGCCACAGACTAGCGGCCGCTAGTTT |
Transient transfection and PRRSV
infections
The 3D4/21 cells were seeded in 24-well cell culture
plates and incubated overnight until the cell density reached
70–80%, then transfected by FuGENE® HD Transfection
Reagent (Promega Corp.) according to manufacturer's instructions
using the modified method in our recent study (43). Briefly, 500 µl serum and
antibiotic-free opti-MEM were added in sterile 1.5 ml centrifuge
tube, then FuGENE® HD transfection reagent was added in
accordance with the proportion required, and mixed gently and
placed at room temperature for 15 min; 0.5 µg plasmid DNA and miRNA
mimics at a final concentration of 10 or 40 or 60 nM, were added
and mixed gently and placed at room temperature for 30 min. The
mixture was given to the culture plates, and then mixed gently,
treated at 37°C in a humidified incubator with 5% CO2
for 24 h.
Primary PAMs with a density of about
0.5–1×105 cells per ml were seeded in 24-well culture
plates, cultured at 37°C in a humidified incubator with 5%
CO2 overnight. After changing to fresh medium, the cells
were transfected transiently with miRNA mimics and inhibitors by
HiPerFect® transfection reagent (Qiagen China Co., Ltd.,
Shanghai, China) according to manufacturer's instructions as
previously described (44). Briefly,
miRNA mimics at a final concentration of 10 or 40 nM, or miRNA
inhibitors at 20 or 40 or 100 nM were added in 100 µl serum and
antibiotic-free opti-MEM in sterile 1.5 ml centrifuge tube, and
then the HiPerFect® transfection reagent in
corresponding volume ratio following the instructions was added,
and treated at room temperature for 5–10 min after being mixed. The
mixture was given to the culture plates after shaking gently,
cultured at 37°C in a humidified incubator with 5%
CO2.
For viral infections, prepared primary PAMs were
seeded in 6-well culture plates, and divided into three groups
including JXwn06-infected group, HB-1/3.9-infected group and
mock-infected group as control. In PRRSV-infected groups, PAMs were
inoculated with JXwn06 or HB-1/3.9 at MOI of 0.01 or 1, and then
cultured at 37°C in a humidified incubator with 5% CO2.
Cell samples and supernatant samples were collected at 0 h
post-infection (hpi), 12, 24, and 36 hpi to detect the RNA
expressions by real-time PCR and IFN-β protein expression by ELISA,
respectively.
Dual luciferase reporter assay
After 3D4/21 cells were co-transfected transiently
with miRNA mimics and plasmid DNA (pmirGLO-IFN-β 3′UTR
orpmirGLO-MRE-wt or pmirGLO-MRE-mut) for 24 h, dual luciferase
activity reporter assay was performed using
dual-luciferase® reporter assay system (Promega Corp.)
according to manufacturer's instructions. Briefly, the cell culture
medium in culture plates was removed, and washed twice with PBS. To
lyse cells fully, passive lysis buffer was added according to its
protocol, and treated at room temperature for 20 min. The lysate
was transferred into a 96-well plate, and then the plate was placed
at luciferase detector GloMAX™ 96 microplate luminometer (Promega
Corp.) to detect firefly luciferase activity and Renilla
luciferase activity. The data are shown as the ratio of firefly
luciferase activity value to Renilla luciferase activity
value for quantitative analysis of luciferase activity. Each
experiment was repeated three times.
RNA isolation and qRT-PCR
To extract total RNAs (including mRNA and miRNA)
from primary PAMs, the prepared cell samples were lysed by TRIzol
reagent (TransGen Biotech, Inc., Beijing, China) following the
manufacturer's instructions. Total RNAs extracted from PAMs were
digested by RQ1 RNase-Free DNase (Promega Corp.) to remove possible
residual DNA according to the manufacturer's protocol. The RNAs
concentration was determined, and total RNAs were subjected
immediately to reverse transcription or stored at −80°C for
use.
For detection of IFN-β mRNA, 1 µg purified total
RNAs and 0.5 µg random primer hexa-deoxyribonucleotide mixture
(Takara Biotechnology Co., Ltd.) was mixed together, and then
placed in water bath at 70°C for 5 min, immediately cooled on ice
for 5 min, the product was added to the following reaction
components for reverse transcription: 4 µl 5× M-MLV reaction
buffer, 1 µl 10 mM dNTPs, 0.5 µl 40 U/µl RNase inhibitor, 1 µl of
10 U/µl M-MLV reverse transcriptase (Promega Corp.), RNase-free
ddH2O was added to 20 µl. The mixture was incubated in
water bath at 37°C for 1 h, and then cDNA template was obtained.
The cDNA template was taken and diluted in the proportion of 1:20,
quantitative real-time PCR (qRT-PCR) was performed using 7500 Real
Time PCR system (Applied Biosystems; Thermo Fisher Scientific,
Inc.) and SYBR® Green real-time PCR master mix (Toyobo
Life Science, Osaka, Japan) according to manufacturer's
instructions with the primers for porcine IFN-β (45) and RPL4 (46) (Table
III). The reaction was in a 20 µl volume containing 10 µl 2×
SYBR® Green Real Time PCR Master Mix, 0.8 µl 10 pmol/µl
primer pair, 1 µl cDNA template, and 8.2 µl RNase-free
ddH2O. The PCR conditions comprised an initial
pre-denaturation at 95°C for 1 min, and 40 cycles of 95°C for 15
sec, 65°C for 15 sec, 72°C for 45 sec. To assess the specificity of
PCR products, melting curve analysis and size verification were
performed. Relative expression level of IFN-β gene was analyzed
using the ∆∆Ct method and the housekeeping gene RPL4 mRNA was used
as an internal control.
 | Table III.Primers for reverse transcription and
real-time PCR of porcine IFN-β and miRNAs. |
Table III.
Primers for reverse transcription and
real-time PCR of porcine IFN-β and miRNAs.
| Primera | Primer sequence
(5′→3′)b |
|---|
| IFN-β-F |
TGCAACCACCACAATTCC |
| IFN-β-R |
CTGAGAATGCCGAAGATCTG |
| RPL4-F |
CAAGAGTAACTACAACCTTC |
| RPL4-R |
GAACTCTACGATGAATCTTC |
| miRNA RT
primer |
GCGAGCACAGAATTAATACGACTCACTATAGGT |
| let-7b-F |
CGGTGAGGTAGTAGGTTGTGTGGTT |
| miR-26a-F |
CGGTTCAAGTAATCCAGGATAGGCT |
| miR-34a-F |
GGTGGCAGTGTCTTAGCTGGTTGT |
| miR-145-F |
GTCCAGTTTTCCCAGGAATCCCT |
| Uni-miRNA-R |
GCGAGCACAGAATTAATACGACTCAC |
| U6-F |
CTCGCTTCGGCAGCACA |
| U6-R |
AACGCTTCACGAATTTGCGT |
For detection of miRNAs, 1 µg of the purified total
cellular RNAs was used for polyadenylation using E. coli
Poly (A) polymerase (New England BioLabs Inc., Ipswich, MA, USA)
according to manufacturer's instructions. To obtain cDNA template
of miRNAs, the polyadenylated total cellular RNAs were
reverse-transcribed with a universal adapter primer (miRNA RT
Primer) or U6 downstream primer (U6-R) (Table III) using M-MLV reverse
transcriptase in accordance with manufacturer's instructions. Based
on miRNAs sequences in miRBase, the specific primers for porcine
let-7b, miR-26a, miR-34a, miR-145 and internal reference U6, were
designed and synthesized (Table
III). Quantitative real-time PCR were performed with the
primers for miRNAs using 7500 Real Time PCR system (Applied
Biosystems; Thermo Fisher Scientific, Inc.) and SYBR®
Green Real Time PCR Master Mix (Toyobo Life Science) in accordance
with manufacturer's instructions, the specificity of PCR products
were assessed as described above. The relative expression levels of
miRNAs were normalized internally utilizing U6 as a reference using
the ∆∆Ct method as previously described (44).
ELISA for porcine IFN-β
To determine the protein induction of IFN-β in
culture supernatants of primary PAMs, porcine IFN-β detection was
performed using a commercial ELISA kit (DZE40056; DongGe
Biotechnology, Beijing, China) according to manufacturer's
instructions. After the cell concentration was adjusted, the
prepared primary PAMs were seeded in six-well cell culture plates
incubated in 10% RPMI-1640 medium at 37°C in a humidified cell
incubator with 5% CO2. Non-adherent cells were removed
at about 2.5 h after the incubation, and then complete growth
medium was added to continue incubating overnight. Primary PAMs
were infected with PRRSV JXwn06 or HB-1/3.9, or transfected
transiently using miRNA mimics or inhibitors, or cultured in fresh
10% RPMI-1640 growth medium containing a final concentration of 50
µg/ml poly I:C (Sigma-Aldrich, Inc., St. Louis, MO, USA), meanwhile
mock-infected or negative control-transfected or mock-treated cells
acted as control group, respectively. The cell culture supernatant
samples were harvested at different time points and used for
detection of porcine IFN-β protein by ELISA. The corresponding
IFN-β concentration in samples was obtained by their OD values
according to the standard curve following manufacturer's
instructions. The data are shown as relative expression levels of
IFN-β protein normalized to control group.
Statistical analysis
Experimental data are presented as means ± standard
error of the mean. Significant differences of the variability among
different groups were analyzed by two-way ANOVA test or t-test
using GraphPad Prism (version 5.0; GraphPad Software, Inc., La
Jolla, CA, USA) software. A P<0.05 was considered to indicate a
statistically significant difference.
Results
PRRSV inhibits post-transcriptionally
the protein production of IFN-β in primary PAMs
To analyze the differences in mRNA transcription and
protein expression of IFN-β in PAMs following PRRSV infection,
primary PAMs were infected with JXwn06 or HB-1/3.9 at MOI of 0.01
or 1, while mock-infected cells served as negative control. Cell
samples and supernatant samples were collected at 0, 12, 24 and 36
hpi. Cell samples were used to analyze the mRNA transcription level
of IFN-β by real-time PCR. Supernatant samples were subjected to
detection of the protein concentration of IFN-β by ELISA kit. The
results showed that compared with mock group, IFN-β mRNA
transcription levels increased from 12 to 36 hpi both in JXwn06-
and HB-1/3.9-infected PAMs (Fig.
1A), indicating that mRNA transcription of porcine IFN-β was
upregulated in primary PAMs early infected with PRRSV.
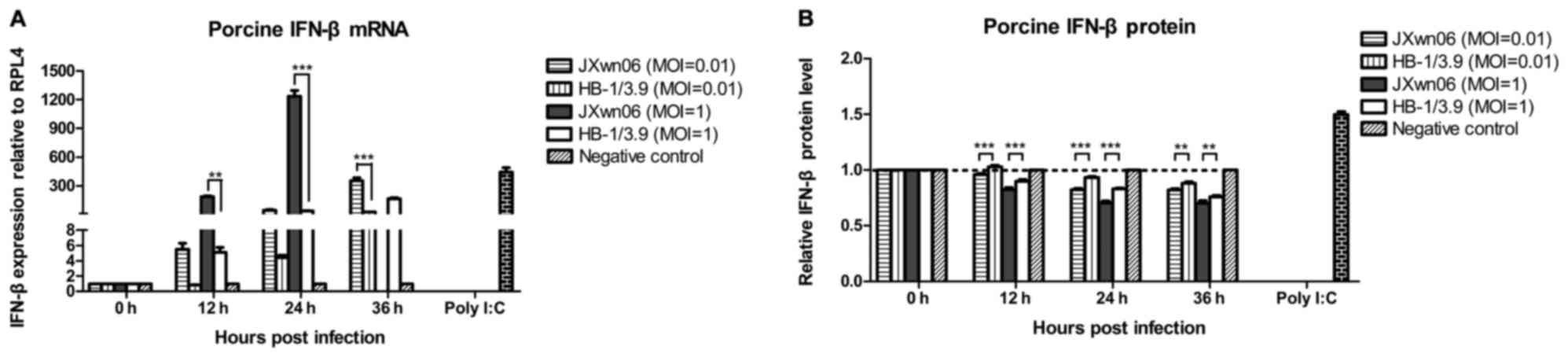 | Figure 1.Inhibition of the IFN-β protein
expression in PRRSV-infected PAMs by post-transcriptional control.
(A) The mRNA levels of porcine IFN-β in PRRSV-infected PAMs and (B)
its protein expression levels in the culture supernatants of
PRRSV-infected PAMs were detected by real-time PCR and ELISA,
respectively. Primary PAMs were infected with JXwn06 or HB-1/3.9 at
MOI of 0.01 or 1, and then the cell and supernatant samples
harvested at 0, 12, 24 and 36 hpi, were measured by real-time PCR
and ELISA, respectively, while mock-infected primary PAMs served as
negative control and primary PAMs stimulated by 50 µg/ml poly I:C
for 24 h were used as positive control. Relative expression level
of IFN-β gene was analyzed using the ∆∆Ct method and the
housekeeping gene RPL4 mRNA was used as an internal control. The
IFN-β protein data are shown as relative expression levels
normalized to negative control group at each time point,
respectively. The data represent means ± standard error of the mean
of three independent experiments (**P<0.01; ***P<0.001).
Since the primary PAMs infected with JXwn06 at MOI of 1 were
disrupted at 36 hpi, the data in relation to IFN-β mRNA
transcription could not be analyzed. IFN-β, interferon-β. |
The transcription levels of porcine IFN-β at MOI of
1 were higher than that at MOI of 0.01 in both JXwn06- and
HB-1/3.9-infected PAMs, showing that the regulation effect of PRRSV
on porcine IFN-β mRNA transcription is dose-dependent in primary
PAMs infected with PRRSV. IFN-β mRNA transcription levels in
JXwn06-infected PAMs were higher compared with HB-1/3.9-infected
PAMs. At MOI of 0.01, the transcription levels of porcine IFN-β
mRNA were significantly different at 36 hpi between JXwn06- and
HB-1/3.9-infected PAMs (P<0.001). At MOI of 1, the transcription
levels of porcine IFN-β mRNA were significantly different between
the two strains of PRRSV at 12 (P<0.05) and 24 hpi (P<0.001),
respectively (Fig. 1A). These data
indicated that porcine IFN-β mRNA transcription exhibited stronger
upregulation effect in HP-PRRSV JXwn06-infected PAMs in
vitro than that in low pathogenic PRRSV HB-1/3.9-infected PMAs
at the same initial viral infectious dose during early
infection.
On the contrary, normalized to negative control
group at each time point, the protein relative expression levels of
IFN-β in PRRSV-infected PAMs were downregulated (Fig. 1B), suggesting that IFN-β protein
expression was inhibited by PRRSV in primary PAMs during early
infection. The concentration of IFN-β protein in the culture
supernatants of negative control PAM cells increased higher than
that in the culture supernatants of PRRSV-infected PAM cells at
each time point, although the absolute concentration of IFN-β
protein in the culture supernatants of PRRSV-infected PAM cells
augmented very little as time went on during early viral infection,
the relative concentration of IFN-β protein normalized to negative
control decreased. To better analyze the relationship between PRRSV
infection and IFN-β protein expression and better observe the
variation in the IFN-β protein expression levels of PRRSV-infected
PAMs, the IFN-β protein data were shown as relative expression
levels normalized to negative control group at each time point
(Fig. 1B). The relative protein
expression levels of porcine IFN-β at MOI of 1 were lower than that
at MOI of 0.01 in both JXwn06- and HB-1/3.9-infected PAMs,
indicating that the PRRSV-mediated inhibition of porcine IFN-β
protein relative expression in primary PAMs is in dose-dependent
manner during early infection. The downregulation of IFN-β protein
relative expression in JXwn06-infected PAMs was more significant
than that in HB-1/3.9-infected PAMs (Fig. 1B) (P<0.05), suggesting that the
inhibition effect of porcine IFN-β protein expression by HP-PRRSV
was stronger. Although it seemed to be small variation in porcine
IFN-β protein production in PAMs among different treated groups, it
still made biological sense, because the succedent signaling
cascades would amplify the IFN-β effect thousands of times
(47).
As a whole, the above results demonstrated that mRNA
transcription levels of IFN-β in primary PAMs early infected with
PRRSV were upregulated, whereas its protein relative expression
levels were downregulated, suggesting that PRRSV infection
suppresses the protein expression of IFN-β in PAMs by
post-transcriptional control, and HP-PRRSV has stronger effect on
the downregulation of IFN-β protein relative expression than low
pathogenic PRRSV at the same initial viral infectious dose during
early infection in PAMs in vitro.
Porcine let-7b, miR-26a, miR-34a and miR-145
could inhibit protein expression of IFN-β in primary PAMs by
directly targeting sequences within porcine IFN-β 3′UTR.
To understand the post-transcriptional molecular
basis of the porcine IFN-β protein expression in PAMs, the roles of
porcine miRNAs were investigated. Since some miRNAs had been
demonstrated to regulate IFN-β protein expression in primary
primate macrophages (47), to
predict the interaction between cellular miRNAs and porcine IFN-β
3′UTR in primary PAMs, the binding sequences targeted by porcine
miRNAs including let-7b, miR-26a, miR-34a and miR-145 within IFN-β
3′UTR were analyzed using miRanda, RNA hybrid and PITA. The results
showed that the cellular miRNAs let-7b, miR-26a, miR-34a and
miR-145, could target porcine IFN-β 3′UTR, and the binding
sequences were located at 160–181, 9–31, 27–47 and 12–32 bp in
porcine IFN-β 3′UTR, respectively. The specific interacting keys
between these miRNAs and IFN-β 3′UTR are shown in Fig. 2A.
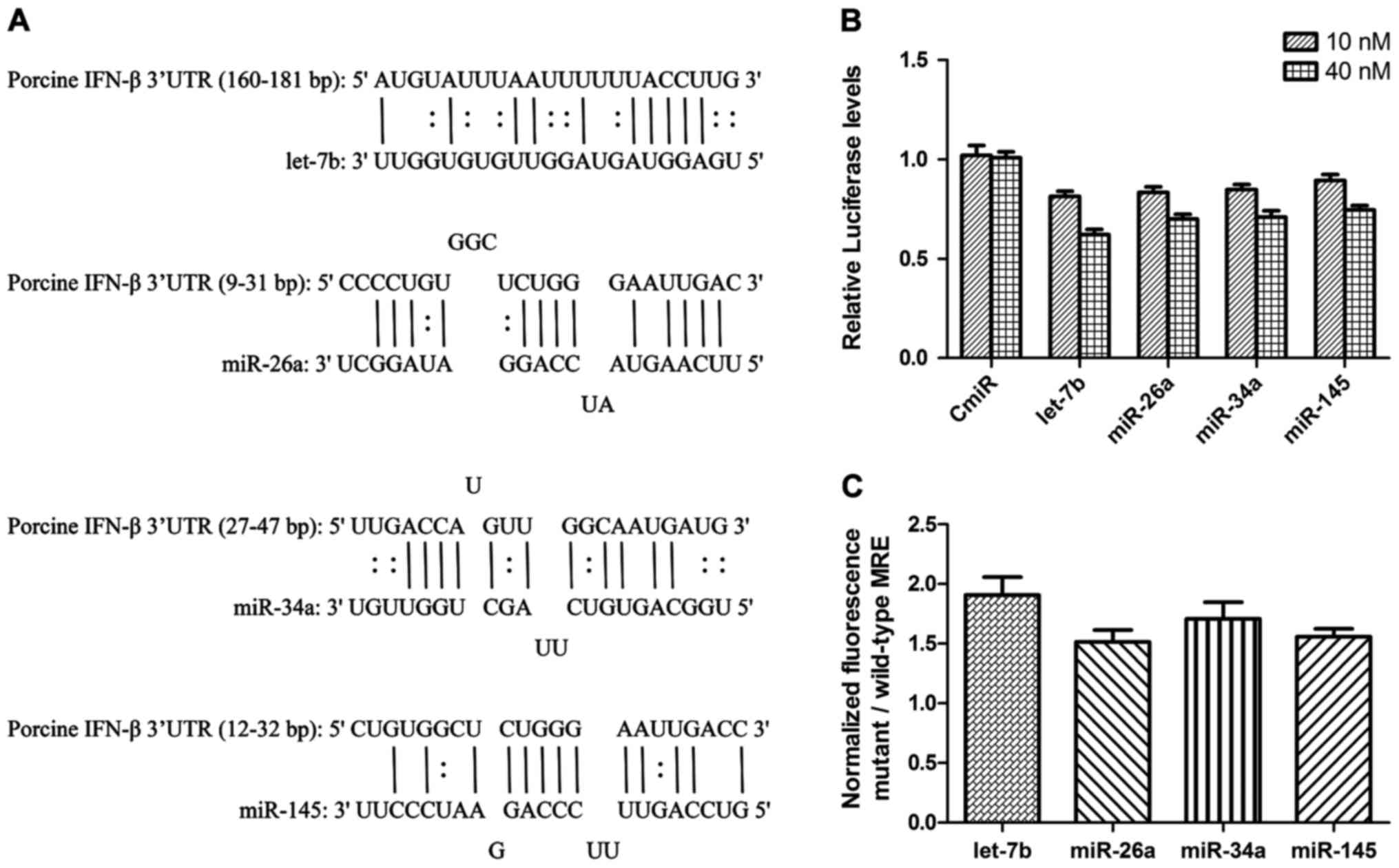 | Figure 2.miRNAs target sequences located in
the porcine IFN-β 3′UTR. miRNA recognition elements (MRE) in the
porcine IFN-β 3′UTR for porcine let-7b, miR-26a, miR-34a and
miR-145, were predicted using miRanda, RNA hybrid and PITA. (A)
Porcine IFN-β 3′UTR gene was cloned into the downstream of firefly
luciferase in pmirGLO dual firefly/Renilla luciferase miRNA
target expression vector, and the expression of pmirGLO-IFN-β 3′UTR
luciferase was inhibited in 3D4/21 cells transfected with miRNA
mimics at a final concentration of 10 or 40 nM, (B) 3D4/21 cells
were co-transfected with pmirGLO-MRE-wtor pmirGLO-MRE-mut and miRNA
mimics at 60 nM, the fluorescence results are shown as the ratio of
mt to wt MREs (C) the data represent means ± standard error of the
mean of three independent experiments. IFN-β, interferon-β; 3′UTR,
3′ untranslated region; MRE, miRNA recognition elements; mt, mutant
type; wt, wild-type. |
By using pmirGLO-IFN-β 3′UTR, pmirGLO-MRE-wt and
pmirGLO-MRE-mut, the interactions between miRNAs and porcine IFN-β
3′UTR were verified. The 3D4/21 cells were co-transfected
transiently with pmirGLO-IFN-β 3′UTR and a final concentration of
10 or 40 nM miRNA mimics, and dual luciferase activity reporter
assay was performed by dual-luciferase® reporter assay
system. As shown in Fig. 2B, miRNA
mimics could inhibit the expression of firefly luciferase in
pmirGLO-IFN-β 3′UTR. Compared with the control miRNA (CmiR),
relative luciferase activity was suppressed by miRNA mimics,
including let-7b, miR-26a, miR-34a and miR-145 at 10 nM, while at
40 nM the inhibition of four miRNAs was stronger. The 3D4/21 cells
were co-transfected with pmirGLO-MRE-wt or pmirGLO-MRE-mut and
miRNA mimics at 60 nM, dual luciferase activity reporter assay was
then performed. The results showed that miRNA mimics inhibited the
expression of firefly luciferase of pmirGLO-MRE-wt, whereas the
inhibition of firefly luciferase expression by miRNA was weakened
for pmirGLO-MRE-mut. The relative luciferase activity ratio between
pmirGLO-MREs-mut and pmirGLO-MREs-wt was greater than 1 (Fig. 2C). These results suggested that
let-7b, miR-26a, miR-34a and miR-145 were capable of interacting
with the predicted miRNA binding sequence in porcine IFN-β 3′UTR,
resulting in the protein expression inhibition of porcine
IFN-β.
There is a mutual regulation between
miRNAs and IFN-β protein expression in PAMs
The miRNA-regulating protein expression of IFN-β in
primary PAMs was analyzed. By using miRNA HiPerFect®
transfection reagent, the primary PAMs were transfected transiently
with a final concentration of 10 or 40 nM miRNAs mimics, including
let-7b, miR-26a, miR-34a, miR-145 and CmiR, or a final
concentration of 20 or 100 nM miRNA inhibitors including four miRNA
inhibitors and negative control (CmiRi). At about 3.5 h
post-transfection, a final concentration of 50 µg/ml poly I:C was
used to stimulate the PAMs for 24 h, cell culture supernatant
samples were then subjected to detect porcine IFN-β protein
expression by ELISA kit. As shown in Fig. 3A and B, compared with the negative
control group, miRNA mimics including let-7b, miR-26a, miR-34a and
miR-145, were capable of inhibiting the protein production of
porcine IFN-β in primary PAMs at 10 nM, while at 40 nM the
inhibition effect of four miRNAs were significantly stronger. In
contrast, compared with the negative control group, except for the
miR-145, three of the four miRNA inhibitors, were able to weaken
the suppression effect of porcine cellular endogenous miRNAs in
primary PAMs on IFN-β protein expression at 20 nM, while at 100 nM
the relieving effect of four miRNA inhibitors were significantly
stronger.
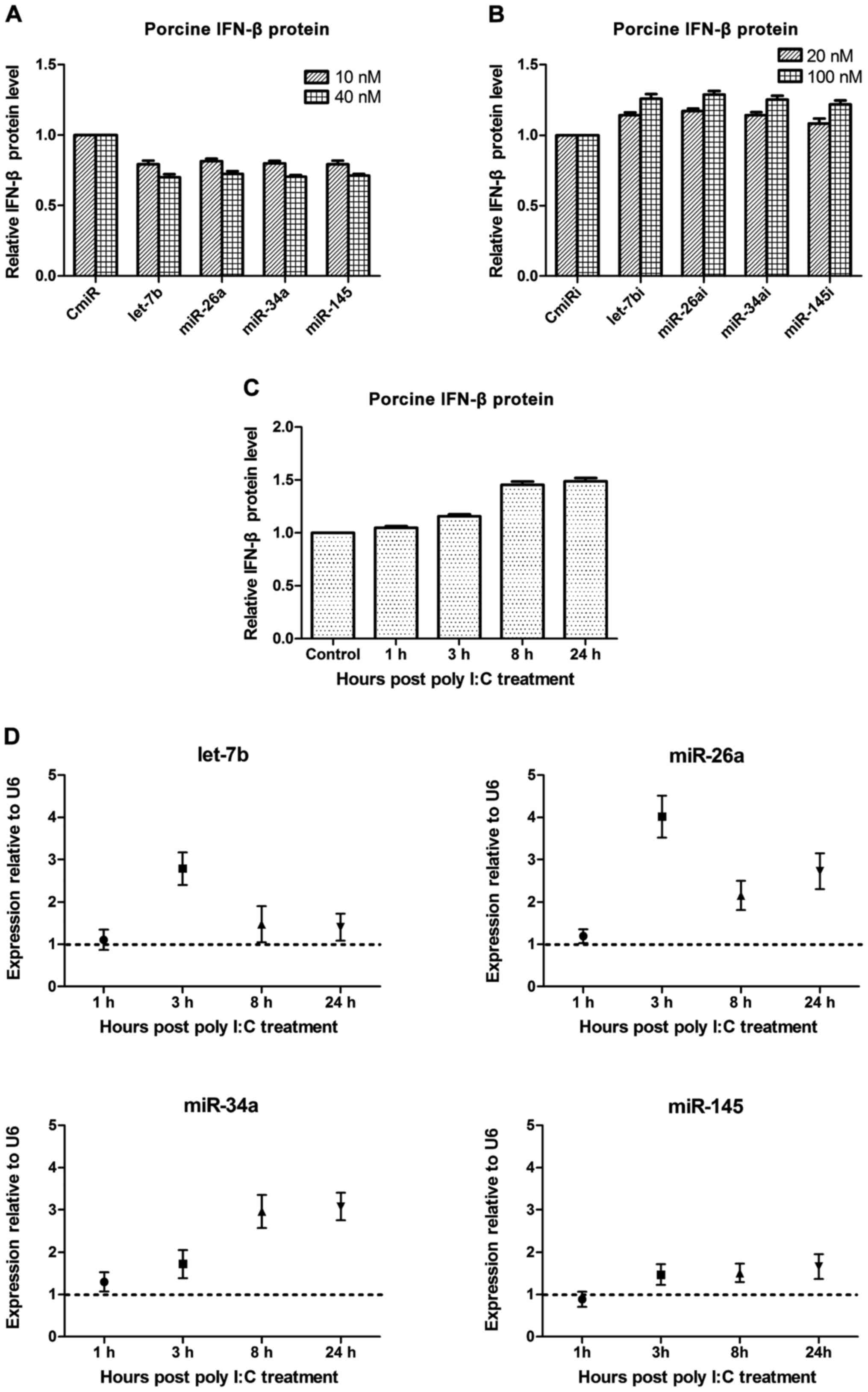 | Figure 3.The mutual regulation between miRNAs
and IFN-β protein in PAMs. (A) 10 or 40 nM four miRNA mimics and
negative control miRNA mimic (CmiR), and (B) 20 or 100 nM four
miRNA inhibitors and negative control miRNA inhibitor (CmiRi), were
transfected transiently into primary PAMs, which were then
stimulated by 50 µg/ml poly I:C for 24 h. The protein expression
levels of porcine IFN-β in the supernatant of PAMs at 24 hpi were
measured by ELISA. The IFN-β protein expression could be suppressed
by miRNA mimics (A) and the native endogenous miRNAs inhibition
effect on IFN-β protein expression could be relieved by miRNA
inhibitors (B) in primary PAMs. (C) Primary PAMs were treated by 50
µg/ml poly I:C to stimulate the production of porcine IFN-β, whose
protein levels were monitored by a commercial porcine IFN-β ELISA
kit at 1, 3 8 and 24 h post-treatment. Mock-treated PAMs were used
as control. (D) qRT-PCR analysis of miRNAs expression including
let-7b, miR-26a, miR-34a and miR-145, in primary PAMs stimulated by
50 µg/ml poly I:C at 1, 3, 8 and 24 h after treatment. The native
IFN-β protein induction stimulated by poly I:C could upregulate the
expression of endogenous miRNAs in primary PAMs. The results were
normalized to U6 expression and mock-treated controls according to
∆∆Ct method. The data represent means ± standard error of the mean
of three independent experiments. |
The native IFN-β protein-regulating expression of
cellular endogenous miRNAs in primary PAMs was further analyzed.
Cell culture supernatant samples and cell samples were collected at
1, 3, 8 and 24 h after poly I:C treatment, respectively. The
protein expression levels of IFN-β in the supernatants were
measured by ELISA kit, miRNAs including let-7b, miR-26a, miR-34a
and miR-145 in the cell samples were detected by real-time PCR
using ∆∆Ct method. As shown in Fig.
3C, the expression levels of porcine IFN-β protein were
increased subsequently at 3 h after primary PAMs were stimulated by
poly I:C. As shown in Fig. 3D,
compared with untreated cells, the levels of native endogenous
miRNAs in primary PAMs were increased with varying degrees
(normalized to U6 expression and mock-treated controls). Taken
together, native IFN-β protein expression and cellular endogenous
miRNAs could be induced by poly I:C together, and cellular
endogenous miRNAs were increased only at or after the first
augmentation of native IFN-β protein induction in primary PAMs,
indicating that the increasing porcine native IFN-β protein could
lead to the upregulation of the endogenous miRNA levels.
Furthermore, the miR-26a and miR-34a had a significant increase,
and let-7b was initially increased, and then decreased; the miR-145
showed an increase at a low level; the let-7b and miR-26a were
upregulated significantly in the early stage of IFN-β protein
expression at a low level, demonstrating that it was exquisite for
native IFN-β protein to modulate the expression of cellular
endogenous miRNAs in primary PAMs.
In short, the four cellular miRNAs could modulate
the protein induction of IFN-β in PAMs, meanwhile, the native IFN-β
induced in PAMs could also regulate the expression of four
endogenous miRNAs, suggesting that a negative feedback loop might
exist in the regulation of porcine IFN-β protein on the expression
of cellular miRNAs including let-7b, miR-26a, miR-34a and
miR-145.
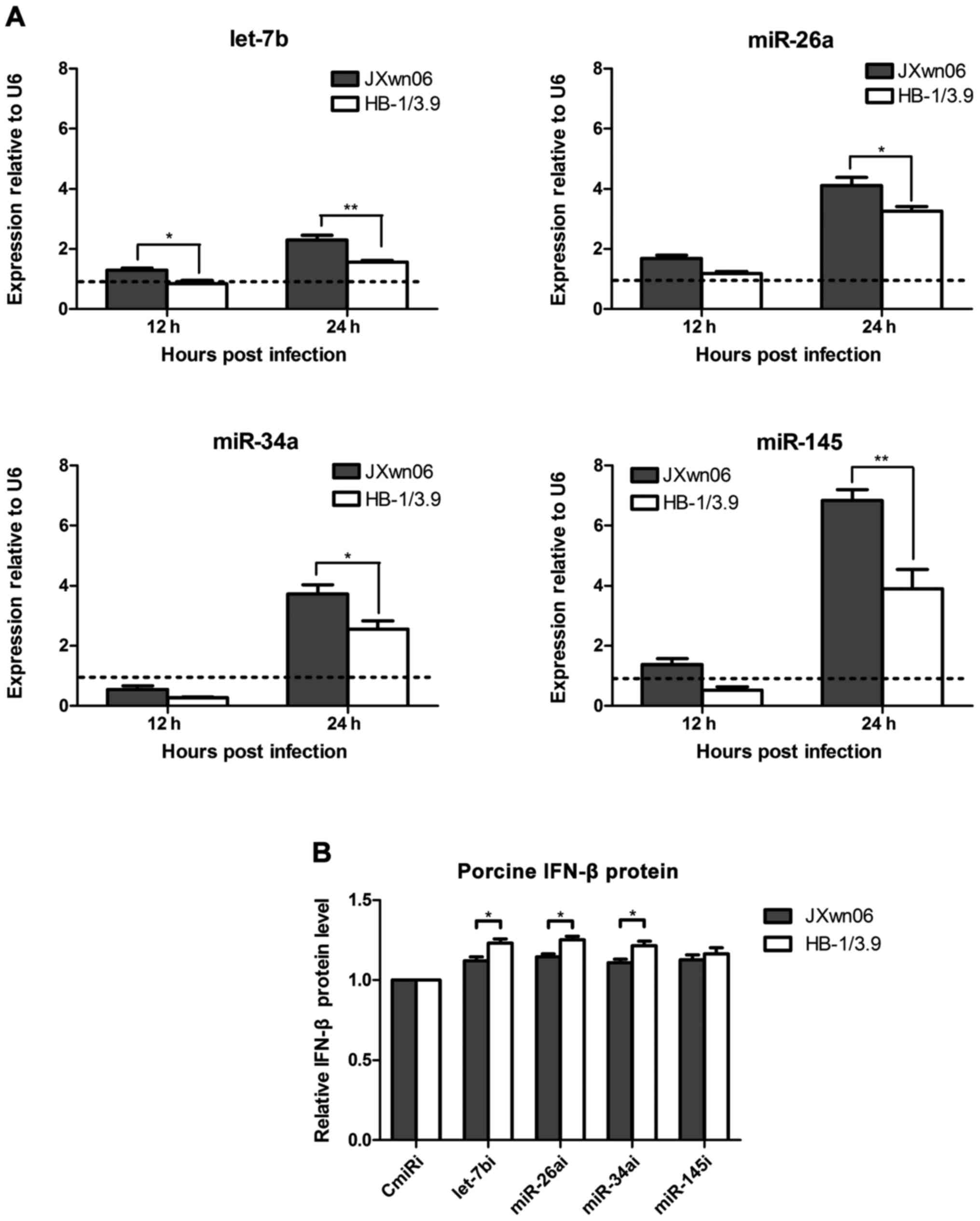
PRRSV infection suppresses IFN-β
protein expression in PAMs by upregulating cellular miRNAs
In order to investigate the modulation effect of the
four cellular miRNAs during PRRSV infection, the levels of miRNAs
in PRRSV JXwn06-infected and HB-1/3.9-infected primary PAMs were
detected by real-time PCR. The results showed that the cellular
miRNAs including let-7b, miR-26a, miR-34a and miR-145 in primary
PAMs were increased along with time proceeding following PRRSV
infection (Fig. 4A). The levels of
four miRNAs could be upregulated approximately by 2–7 fold at 24
hpi, indicating that PRRSV infection is capable of inducing the
generation of cellular let-7b, miR-26a, miR-34a and miR-145 during
the early infection. The levels of let-7b, miR-26a, miR-34a and
miR-145 in JXwn06-infected PAMs were significantly higher than
those in HB-1/3.9-infected PAMs at 24 hpi (P<0.05 or P<0.01),
suggesting that the HP-PRRSV JXwn06 exhibits a higher ability of
inducing let-7b, miR-26a, miR-34a and miR-145 levels than low
pathogenic PRRSV HB-1/3.9 during early infection.
The roles of miRNAs in PRRSV-mediated inhibition of
porcine IFN-β protein expression were further analyzed. Primary
PAMs were transfected transiently with miRNA inhibitors including
the antagonists of let-7b, miR-26a, miR-34a and miR-145 at a final
concentration of 40 nM, and then infected with JXwn06 or HB-1/3.9
at MOI of 1. The porcine IFN-β protein expression was measured by
ELISA kit. As shown in Fig. 4B, the
protein expression of IFN-β was upregulated in the miRNA
inhibitor-transfected primary PAMs during PRRSV infection at 24
hpi, suggesting that the miRNA inhibitors including the antagonists
of let-7b, miR-26a, miR-34a and miR-145, were able to relieve
PRRSV-mediating inhibition of porcine IFN-β protein expression to a
certain extent. Compared with PRRSV-infected PAMs transfected with
all miRNA inhibitors, but miRNA-145 inhibitor, the protein
expression of IFN-β in HB-1/3.9-infected PAMs were higher than that
in JXwn06-infected PAMs (P<0.05), indicating that an equal
amount of three miRNA inhibitors more effectively relieve low
pathogenic PRRSV-mediated inhibition of porcine IFN-β protein
expression. This provided further evidence that HP-PRRSV-mediating
inhibition of porcine IFN-β protein expression was stronger than
low pathogenic PRRSV.
Discussion
Type I IFN-(α/β), most efficiently induced by
leukocytes such as macrophages and dendritic cells, play multiple
important functions in the innate and adaptive immune responses of
host against various virus infections (48,49).
PAMs are important host target cells for PRRSV infection and
replication. It is helpful for understanding the pathogenesis of
PRRSV to investigate the interaction between the virus and its host
cells. Previous studies have demonstrated that PRRSV is a poor
inducer of type I IFN in vitro and in vivo (1,50). In
the present study, cellular miRNA-mediated regulation of IFN-β
protein expression was investigated in primary PAMs infected with
PRRSV in vitro. Our results showed that the transcription
level of IFN-β mRNA was increased, but its protein expression was
decreased in PRRSV-infected PAMs in the early stage, suggesting
that there was a post-transcriptional inhibition mechanism of IFN-β
protein expression. Cellular miRNAs, including let-7b, miR-26a,
miR-34a and miR-145, were capable of downregulating the porcine
IFN-β protein expression via targeting the binding sequences of
IFN-β 3′UTR, while porcine native IFN-β protein could modulate
cellular endogenous miRNAs expression using a negative feedback
mechanism. Moreover, the four miRNAs were upregulated in
PRRSV-infected PAMs, and their inhibitors could relieve
PRRSV-mediated suppression of porcine IFN-β protein expression.
Previous study has shown that post-transcriptional
mechanism was involved in type I IFN induction in host cells during
PRRSV infection (13). Here, we
found that IFN-β mRNA transcription could be activated, but its
protein production was inhibited in PRRSV-infected PAMs during
early infection, suggesting that PRRSV-mediated inhibition of IFN-β
protein production occurred in a post-transcriptional manner. Our
results are consistent with previous studies using PAMs and porcine
monocyte-derived dendritic cells (14,20).
Studies have demonstrated that some nsps and N protein of PRRSV
were involved in inhibiting the IFN-β promoter in MARC-145 cells
and human cells (21), it is
debatable whether these results are similar in natural host cells
of PRRSV, as the findings reported on mouse hepatitis virus
(51).
Different pathogenic PRRSV isolates might be of
distinct features in post-transcriptional control of IFN-β. Our
results presented that HP-PRRSV JXwn06 and low pathogenic PRRSV
HB-1/3.9 induced differential levels of IFN-β protein expression in
PAMs in vitro during early infection, and the
post-transcriptional inhibition of IFN-β induced by JXwn06 was
stronger than that by HB-1/3.9 at the same initial viral infectious
dose, indicating that different pathogenic PRRSV strains have
different ability in the regulation of IFN-β protein expression
in vitro. Our data are consistent with the study that the
IFN phenotypes induced by PRRSV field isolates were distinct in
vitro (20), and is similar to
the study that the Chinese HP-PRRSV isolate displayed more
inhibitory effect than PRRSV strain VR-2332 in the suppression of
CpG-ODN-induced IFN-α responses by enriched plasmacytoid dendritic
cells (pDC) (50). The differential
IFN-β response of PAMs to different pathogenic PRRSV strains may be
due to the viral distinct characteristics or variation in
efficiency of viral infection and replication. Chinese HP-PRRSV
JXwn06 possess higher replication efficiency and infection capacity
than low pathogenic PRRSV HB-1/3.9 in PAMs in vitro at the
same initial infectious dose [data not shown, as our similar recent
study described by Li et al (52)], which may contribute to the stronger
post-transcriptional inhibition effect of porcine IFN-β protein
expression by HP-PRRSV. Since IFN-β is the first batch of host
genes expression after PRRSV infection and a few fold changes in
its protein level leads to almost amplifying the IFN-β effect
thousands of times by the signaling cascades (47), the differences between HP-PRRSV and
low pathogenic PRRSV in the inhibition of IFN-β induction in PAMs
during early infection might correlate with their different
virulence and replication efficiency in vivo. Compared with
wild-type viruses, UV- and heat-inactivated PRRSV fail to suppress
type I IFN production in vitro induced by TGEV or poly I:C,
indicating that virus replication and cytopathogenicity are
essential for PRRSV to inhibit the expression of type I IFN
(1,53). Previous studies showed that type I
IFN induction correlated with the virulence of some viruses
(54). Additionally, our recent
study showed that HP-PRRSV and low pathogenic PRRSV elicited
differential TNF-α subtype production in vitro, suggesting
that different pathogenic isolates of PRRSV might possess different
capacity to induce other cytokines in vitro (43).
Post-transcriptional control of the IFN system is
involved in multiple levels of regulation including mRNA stability,
alternative splicing, translation, and post-translational effects
(55). miRNAs have been shown to
play important roles in regulating the IFN-α/β response at the
post-transcriptional level (23,56).
Given that there is a subset of IFN-α and a single IFN-β subtype,
and neither a single IFN-α subtype nor all IFN-α can be activated
by reported stimuli (57), we chose
porcine IFN-β to investigate the interaction between miRNAs and
type I IFN. In the present study, our results indicated that
porcine IFN-β protein expression in primary PAMs could be
downregulated by cellular let-7b, miR-26a, miR-34a and miR-145
which are capable of targeting the binding sequences of porcine
IFN-β 3′UTR. Similar phenomenon has been described by an early
study using primary primate macrophages (47). It is possible that porcine IFN-β
response is modulated at different levels in primary PAMs, even
more miRNAs may be involved in this regulation. Therefore, further
studies are required to discover additional mechanisms or miRNAs in
primary PAMs.
PRRSV has evolved several strategies to evade and
antagonize anti-viral immune responses of host including using
miRNAs (58). Data presented in this
study showed that cellular endogenous miRNAs including let-7b,
miR-26a, miR-34a and miR-145, were upregulated in PAMs during early
infection of PRRSV, and miRNA antagonists enhanced the production
of porcine IFN-β protein, implying that PRRSV may utilize the
cellular miRNAs including let-7b, miR-26a, miR-34a and miR-145, to
negatively regulate porcine IFN-β response post-transcriptionally,
and further to facilitate virus replication, since porcine IFN-β
has been shown to protect PAMs from PRRSV infection (59). Interestingly, similar to Li et
al (35), a recent study has
demonstrated that miR-26a overexpressed in MARC-145 cells triggered
IFN-β signaling pathway during PRRSV infection, but the protein
expression of IFN-β was not increased, indicating that miR-26a may
modulate the IFN-β expression post-transcriptionally (34). Similarly, since miR-373 could
downregulate the transcription of IFN-β via targeting cellular
factors such as NFIA, NFIB, IRAK1, IRAK4 and IRF1, PRRSV could
upregulate the expression of miR-373 by elevating Sp1 expression in
MARC-145 cells during viral infection (33), which suggests that PRRSV is capable
of altering cellular miRNA expression to modulate immune response
and to facilitate the viral immune escape. Consistent with this
possibility, PRRSV infection can not only suppress host miR-125b
and poly I:C-induced miR-23, but also elevate miR-146a, suggesting
that these miRNA-mediated regulations might be strategies for PRRSV
to evade the type I IFN response (30,33,60,61).
Supporting this, many viruses could induce miRNAs to escape from
immune responses such as enterovirus 71-induced miR-146a in the
mouse model, HCV-induced miR-21 in hepatocytes and respiratory
syncytial virus-induced let-7b in DCs (62–64). Our
present data suggested that porcine IFN-β production could be
augmented by suppressing the expression of endogenous miRNAs
including let-7b, miR-26a, miR-34a and miR-145, which could be a
new approach in future exploration to prevent PRRSV infection.
In conclusion, our findings for the first time
demonstrated that the protein expression of IFN-β could be
suppressed by porcine cellular miRNAs, including let-7b, miR-26a,
miR-34a and miR-145, in PAMs by directly targeting the sequences
within the porcine IFN-β 3′UTR. PRRSV could inhibit the IFN-β
protein expression in PAMs by means of upregulating the levels of
the four porcine miRNAs during early viral infection, which may
contribute to the post-transcriptional control of IFN-β in
PRRSV-infected host cells and to the establishment of viral
persistent infection. The molecular mechanism by which PRRSV
upregulates cellular miRNAs in host cells will be explored in the
future.
Acknowledgements
The present study was supported by The Major Program
of National Natural Science Funds from The National Natural Science
Foundation of China (31490603), The National Key Basic Research
PlanGrant (2014CB542700) from the Chinese Ministry of Science and
Technology, and the Earmarked Fund for Modern Agro-industry
Technology Research System of China (CARS-35) from the Chinese
Ministry of Agriculture of People's Republic of China.
References
|
1
|
Albina E: Epidemiology of porcine
reproductive and respiratory syndrome (PRRS): An overview. Vet
Microbiol. 55:309–316. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Rossow KD: Porcine reproductive and
respiratory syndrome. Vet Pathol. 35:1–20. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Keffaber K: Reproductive failure of
unknown etiology. Am Assoc Swine Prac News. 1:1–9. 1989.
|
|
4
|
Cho JG and Dee SA: Porcine reproductive
and respiratory syndrome virus. Theriogenology. 66:655–662. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Tian K, Yu X, Zhao T, Feng Y, Cao Z, Wang
C, Hu Y, Chen X, Hu D, Tian X, et al: Emergence of fatal PRRSV
variants: Unparalleled outbreaks of atypical PRRS in China and
molecular dissection of the unique hallmark. PLoS One. 2:e5262007.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Zhou L and Yang H: Porcine reproductive
and respiratory syndrome in China. Virus Res. 154:31–37. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Cavanagh D: Nidovirales: A new order
comprising Coronaviridae and Arteriviridae. Arch Virol.
142:629–633. 1997.PubMed/NCBI
|
|
8
|
Conzelmann KK, Visser N, Van Woensel P and
Thiel HJ: Molecular characterization of porcine reproductive and
respiratory syndrome virus, a member of the arterivirus group.
Virology. 193:329–339. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Meulenberg JJ, Hulst MM, de Meijer EJ,
Moonen PL, den Besten A, de Kluyver EP, Wensvoort G and Moormann
RJ: Lelystad virus, the causative agent of porcine epidemic
abortion and respiratory syndrome (PEARS), is related to LDV and
EAV. Virology. 192:62–72. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Mardassi H, Mounir S and Dea S:
Identification of major differences in the nucleocapsid protein
genes of a Québec strain and European strains of porcine
reproductive and respiratory syndrome virus. J Gen Virol.
75:681–685. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Meng XJ, Paul PS, Halbur PG and Lum MA:
Phylogenetic analyses of the putative M (ORF 6) and N (ORF 7) genes
of porcine reproductive and respiratory syndrome virus (PRRSV):
Implication for the existence of two genotypes of PRRSV in the USA
and Europe. Arch Virol. 140:745–755. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Nelson EA, Christopher-Hennings J, Drew T,
Wensvoort G, Collins JE and Benfield DA: Differentiation of U.S.
and European isolates of porcine reproductive and respiratory
syndrome virus by monoclonal antibodies. J Clin Microbiol.
31:3184–3189. 1993.PubMed/NCBI
|
|
13
|
Wang X and Christopher-Hennings J:
Post-transcriptional control of type I interferon induction by
porcine reproductive and respiratory syndrome virus in its natural
host cells. Viruses. 4:725–733. 2012. View
Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhang H, Guo X, Nelson E,
Christopher-Hennings J and Wang X: Porcine reproductive and
respiratory syndrome virus activates the transcription of
interferon alpha/beta (IFN-α/β) in monocyte-derived dendritic cells
(Mo-DC). Vet Microbiol. 159:494–498. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Albina E, Carrat C and Charley B:
Interferon-alpha response to swine arterivirus (PoAV), the porcine
reproductive and respiratory syndrome virus. J Interferon Cytokine
Res. 18:485–490. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Buddaert W, Van Reeth K and Pensaert M: In
vivo and in vitro interferon (IFN) studies with the porcine
reproductive and respiratory syndrome virus (PRRSV). Adv Exp Med
Biol. 440:461–467. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Calzada-Nova G, Schnitzlein WM, Husmann RJ
and Zuckermann FA: North American porcine reproductive and
respiratory syndrome viruses inhibit type I interferon production
by plasmacytoid dendritic cells. J Virol. 85:2703–2713. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Miller LC, Laegreid WW, Bono JL,
Chitko-McKown CG and Fox JM: Interferon type I response in porcine
reproductive and respiratory syndrome virus-infected MARC-145
cells. Arch Virol. 149:2453–2463. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Chung HK, Lee JH, Kim SH and Chae C:
Expression of interferon-alpha and Mx1 protein in pigs acutely
infected with porcine reproductive and respiratory syndrome virus
(PRRSV). J Comp Pathol. 130:299–305. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Lee SM, Schommer SK and Kleiboeker SB:
Porcine reproductive and respiratory syndrome virus field isolates
differ in in vitro interferon phenotypes. Vet Immunol Immunopathol.
102:217–231. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Sun Y, Han M, Kim C, Calvert JG and Yoo D:
Interplay between interferon-mediated innate immunity and porcine
reproductive and respiratory syndrome virus. Viruses. 4:424–446.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Lin S and Gregory RI: MicroRNA biogenesis
pathways in cancer. Nat Rev Cancer. 15:321–333. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Forster SC, Tate MD and Hertzog PJ:
MicroRNA as type I interferon-regulated transcripts and modulators
of the innate immune response. Front Immunol. 6:3342015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Cullen BR: MicroRNAs as mediators of viral
evasion of the immune system. Nat Immunol. 14:205–210. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Skalsky RL and Cullen BR: Viruses,
microRNAs, and host interactions. Annu Rev Microbiol. 64:123–141.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Cong P, Xiao S, Chen Y, Wang L, Gao J, Li
M, He Z, Guo Y, Zhao G, Zhang X, et al: Integrated miRNA and mRNA
transcriptomes of porcine alveolar macrophages (PAM cells)
identifies strain-specific miRNA molecular signatures associated
with H-PRRSV and N-PRRSV infection. Mol Biol Rep. 41:5863–5875.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Li J, Chen Z, Zhao J, Fang L, Fang R, Xiao
J, Chen X, Zhou A, Zhang Y, Ren L, et al: Difference in microRNA
expression and editing profile of lung tissues from different pig
breeds related to immune responses to HP-PRRSV. Sci Rep.
5:95492015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wang C, Zhang Y, Luo J, Ding H, Liu S,
Amer S, Xie L, Lyv W, Su W, Li M, et al: Identification of miRNomes
reveals ssc-miR-30d-R_1 as a potential therapeutic target for PRRS
viral infection. Sci Rep. 6:248542016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Li L, Gao F, Jiang Y, Yu L, Zhou Y, Zheng
H, Tong W, Yang S, Xia T, Qu Z, et al: Cellular miR-130b inhibits
replication of porcine reproductive and respiratory syndrome virus
in vitro and in vivo. Sci Rep. 5:170102015. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wang D, Cao L, Xu Z, Fang L, Zhong Y, Chen
Q, Luo R, Chen H, Li K and Xiao S: MiR-125b reduces porcine
reproductive and respiratory syndrome virus replication by
negatively regulating the NF-κB pathway. PLoS One. 8:e558382013.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Xiao S, Wang X, Ni H, Li N, Zhang A, Liu
H, Pu F, Xu L, Gao J, Zhao Q, et al: MicroRNA miR-24-3p promotes
porcine reproductive and respiratory syndrome virus replication
through suppression of heme oxygenase-1 expression. J Virol.
89:4494–4503. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zhang Q, Huang C, Yang Q, Gao L, Liu HC,
Tang J and Feng WH: MicroRNA-30c modulates type I IFN responses to
facilitate porcine reproductive and respiratory syndrome virus
infection by targeting JAK1. J Immunol. 196:2272–2282. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Chen J, Shi X, Zhang X, Wang A, Wang L,
Yang Y, Deng R and Zhang GP: MicroRNA-373 facilitated the
replication of porcine reproductive and respiratory syndrome virus
by its negative regulation of type I interferon Induction. J Virol.
91:e01311–e01316. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Jia X, Bi Y, Li J, Xie Q, Yang H and Liu
W: Cellular microRNA miR-26a suppresses replication of porcine
reproductive and respiratory syndrome virus by activating innate
antiviral immunity. Sci Rep. 5:106512015. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Li L, Wei Z, Zhou Y, Gao F, Jiang Y, Yu L,
Zheng H, Tong W, Yang S, Zheng H, et al: Host miR-26a suppresses
replication of porcine reproductive and respiratory syndrome virus
by upregulating type I interferons. Virus Res. 195:86–94. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
He D, Overend C, Ambrogio J, Maganti RJ,
Grubman MJ and Garmendia AE: Marked differences between MARC-145
cells and swine alveolar macrophages in IFNβ-induced activation of
antiviral state against PRRSV. Vet Immunol Immunopathol. 139:57–60.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Ait-Ali T, Wilson AD, Westcott DG,
Clapperton M, Waterfall M, Mellencamp MA, Drew TW, Bishop SC and
Archibald AL: Innate immune responses to replication of porcine
reproductive and respiratory syndrome virus in isolated Swine
alveolar macrophages. Viral Immunol. 20:105–118. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Zhang H, Guo X, Ge X, Chen Y, Sun Q and
Yang H: Changes in the cellular proteins of pulmonary alveolar
macrophage infected with porcine reproductive and respiratory
syndrome virus by proteomics analysis. J Proteome Res. 8:3091–3097.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Zhou L, Zhang J, Zeng J, Yin S, Li Y,
Zheng L, Guo X, Ge X and Yang H: The 30-amino-acid deletion in the
Nsp2 of highly pathogenic porcine reproductive and respiratory
syndrome virus emerging in China is not related to its virulence. J
Virol. 83:5156–5167. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
John B, Enright AJ, Aravin A, Tuschl T,
Sander C and Marks DS: Human MicroRNA targets. PLoS Biol.
2:e3632004. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Rehmsmeier M, Steffen P, Hochsmann M and
Giegerich R: Fast and effective prediction of microRNA/target
duplexes. RNA. 10:1507–1517. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Kertesz M, Iovino N, Unnerstall U, Gaul U
and Segal E: The role of site accessibility in microRNA target
recognition. Nat Genet. 39:1278–1284. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
He Q, Li Y, Zhou L, Ge X, Guo X and Yang
H: Both Nsp1β and Nsp11 are responsible for differential TNF-α
production induced by porcine reproductive and respiratory syndrome
virus strains with different pathogenicity in vitro. Virus Res.
201:32–40. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Guo XK, Zhang Q, Gao L, Li N, Chen XX and
Feng WH: Increasing expression of microRNA 181 inhibits porcine
reproductive and respiratory syndrome virus replication and has
implications for controlling virus infection. J Virol.
87:1159–1171. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Loving CL, Brockmeier SL and Sacco RE:
Differential type I interferon activation and susceptibility of
dendritic cell populations to porcine arterivirus. Immunology.
120:217–229. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Nygard AB, Jørgensen CB, Cirera S and
Fredholm M: Selection of reference genes for gene expression
studies in pig tissues using SYBR green qPCR. BMC Mol Biol.
8:672007. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Witwer KW, Sisk JM, Gama L and Clements
JE: MicroRNA regulation of IFN-beta protein expression: Rapid and
sensitive modulation of the innate immune response. J Immunol.
184:2369–2376. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Biron CA: Role of early cytokines,
including alpha and beta interferons (IFN-alpha/beta), in innate
and adaptive immune responses to viral infections. Semin Immunol.
10:383–390. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Theofilopoulos AN, Baccala R, Beutler B
and Kono DH: Type I interferons (alpha/beta) in immunity and
autoimmunity. Annu Rev Immunol. 23:307–336. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Baumann A, Mateu E, Murtaugh MP and
Summerfield A: Impact of genotype 1 and 2 of porcine reproductive
and respiratory syndrome viruses on interferon-α responses by
plasmacytoid dendritic cells. Vet Res (Faisalabad). 44:332013.
View Article : Google Scholar
|
|
51
|
Rose KM and Weiss SR: Murine coronavirus
cell type dependent interaction with the type I interferon
response. Viruses. 1:689–712. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Li Y, Zhou L, Zhang J, Ge X, Zhou R, Zheng
H, Geng G, Guo X and Yang H: Nsp9 and Nsp10 contribute to the fatal
virulence of highly pathogenic porcine reproductive and respiratory
syndrome virus emerging in China. PLoS Pathog. 10:e10042162014.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Carrigan DR and Knox KK: Identification of
interferon-resistant subpopulations in several strains of measles
virus: Positive selection by growth of the virus in brain tissue. J
Virol. 64:1606–1615. 1990.PubMed/NCBI
|
|
54
|
Marcus PI, Rojek JM and Sekellick MJ:
Interferon induction and/or production and its suppression by
influenza A viruses. J Virol. 79:2880–2890. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Khabar KS and Young HA:
Post-transcriptional control of the interferon system. Biochimie.
89:761–769. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Sedger LM: microRNA control of interferons
and interferon induced anti-viral activity. Mol Immunol.
56:781–793. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Hertzog PJ and Williams BR: Fine tuning
type I interferon responses. Cytokine Growth Factor Rev.
24:217–225. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Huang C, Zhang Q and Feng WH: Regulation
and evasion of antiviral immune responses by porcine reproductive
and respiratory syndrome virus. Virus Res. 202:101–111. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Overend C, Mitchell R, He D, Rompato G,
Grubman MJ and Garmendia AE: Recombinant swine beta interferon
protects swine alveolar macrophages and MARC-145 cells from
infection with Porcine reproductive and respiratory syndrome virus.
J Gen Virol. 88:925–931. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
A Hicks J, Yoo D and Liu HC:
Characterization of the microRNAome in porcine reproductive and
respiratory syndrome virus infected macrophages. PLoS One.
8:e820542013. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Zhang Q, Guo XK, Gao L, Huang C, Li N, Jia
X, Liu W and Feng WH: MicroRNA-23 inhibits PRRSV replication by
directly targeting PRRSV RNA and possibly by upregulating type I
interferons. Virology 450–451. 1–195. 2014.
|
|
62
|
Chen Y, Chen J, Wang H, Shi J, Wu K, Liu
S, Liu Y and Wu J: HCV-induced miR-21 contributes to evasion of
host immune system by targeting MyD88 and IRAK1. PLoS Pathog.
9:e10032482013. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Ho BC, Yu IS, Lu LF, Rudensky A, Chen HY,
Tsai CW, Chang YL, Wu CT, Chang LY, Shih SR, et al: Inhibition of
miR-146a prevents enterovirus-induced death by restoring the
production of type I interferon. Nat Commun. 5:33442014. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Thornburg NJ, Hayward SL and Crowe JE Jr:
Respiratory syncytial virus regulates human microRNAs by using
mechanisms involving beta interferon and NF-κB. MBio. 3:e0022012.
View Article : Google Scholar
|