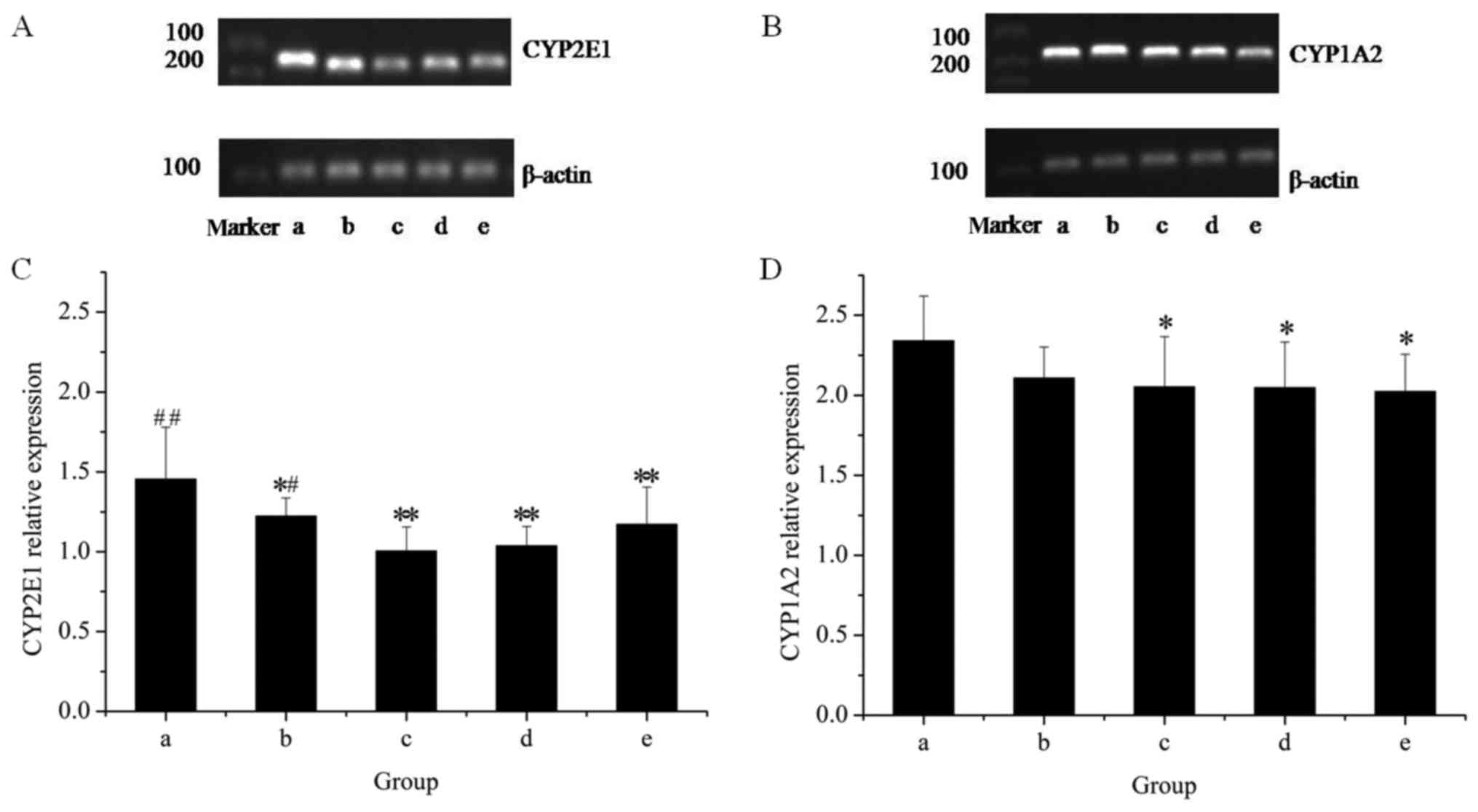
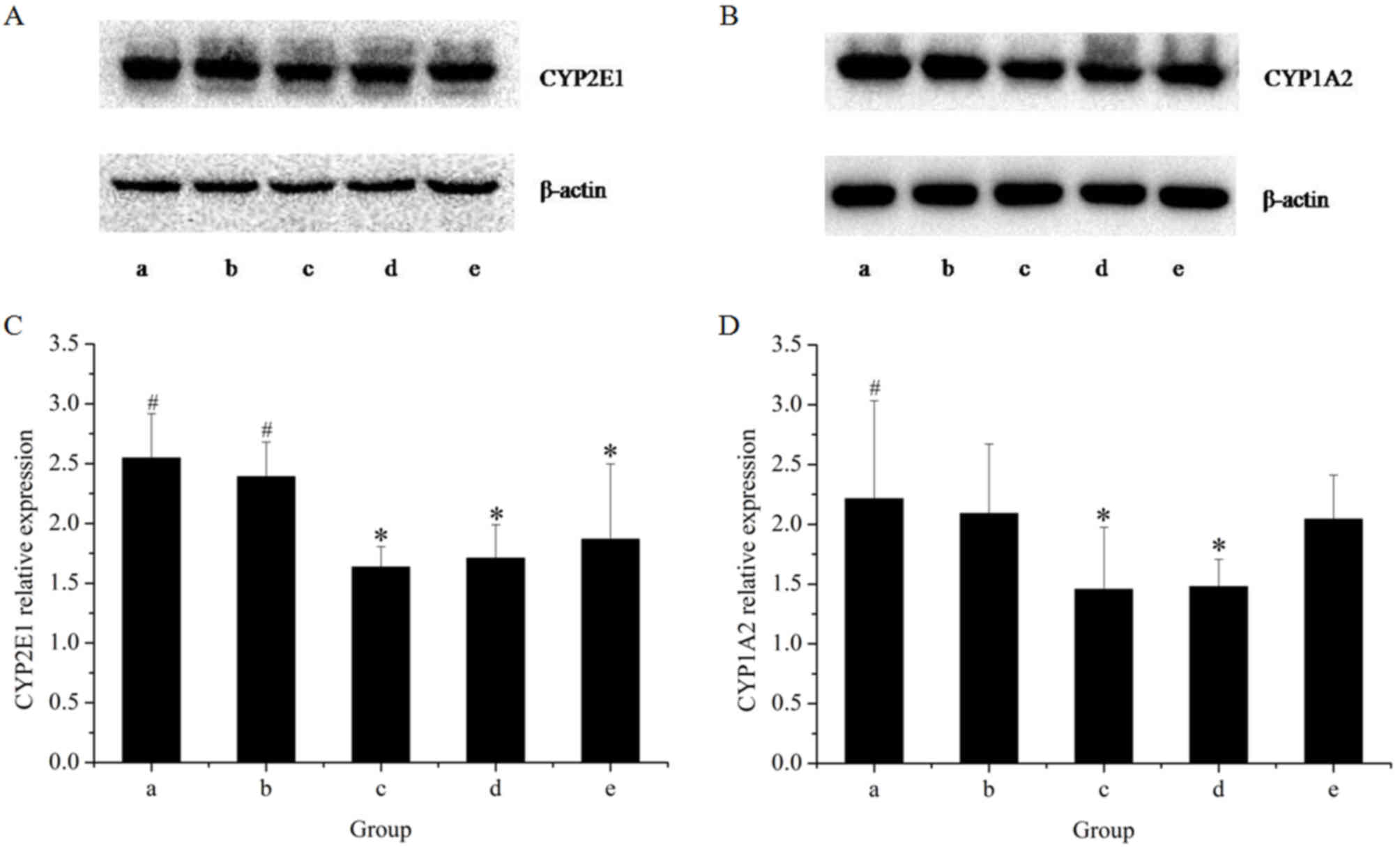
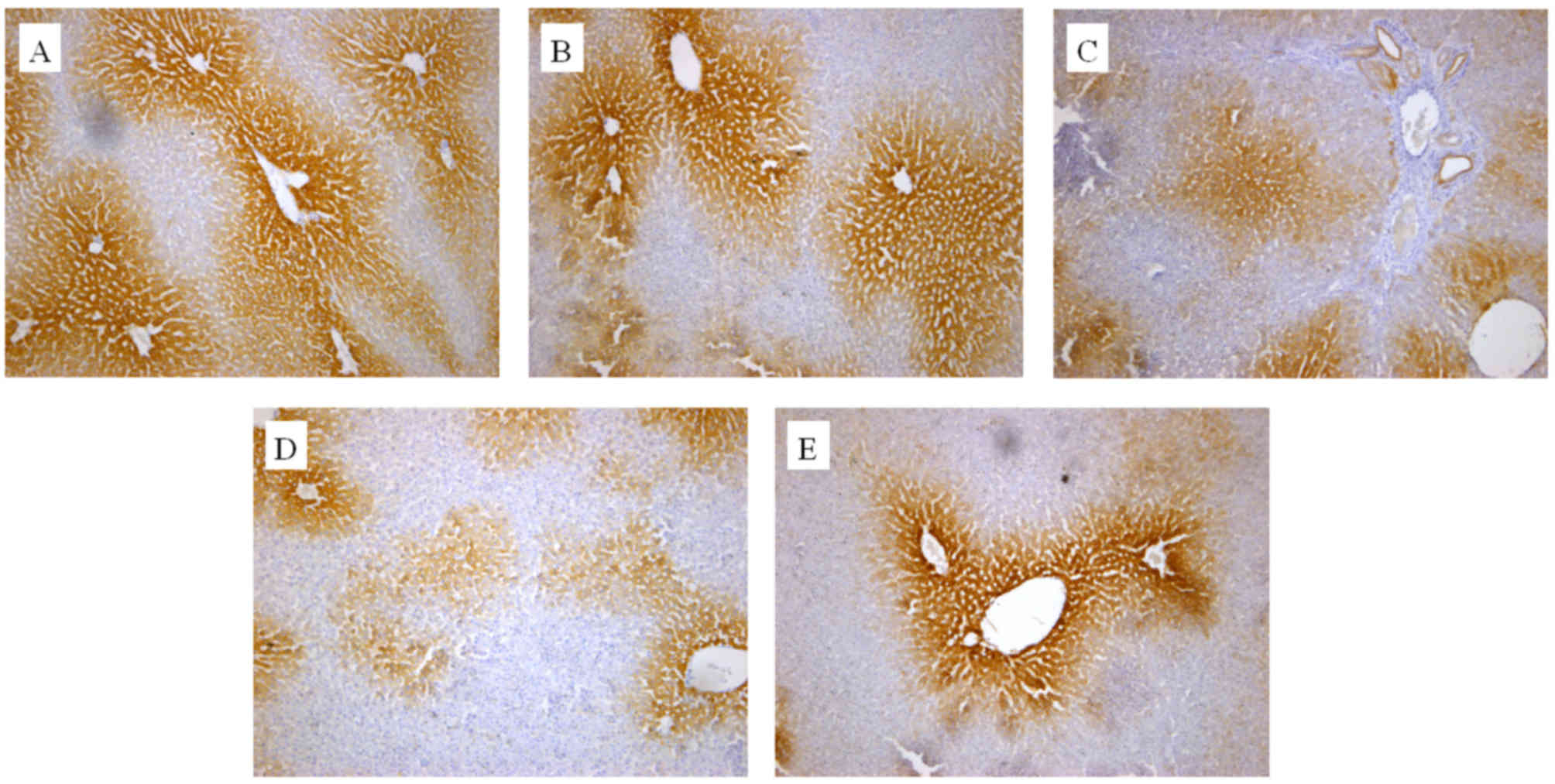
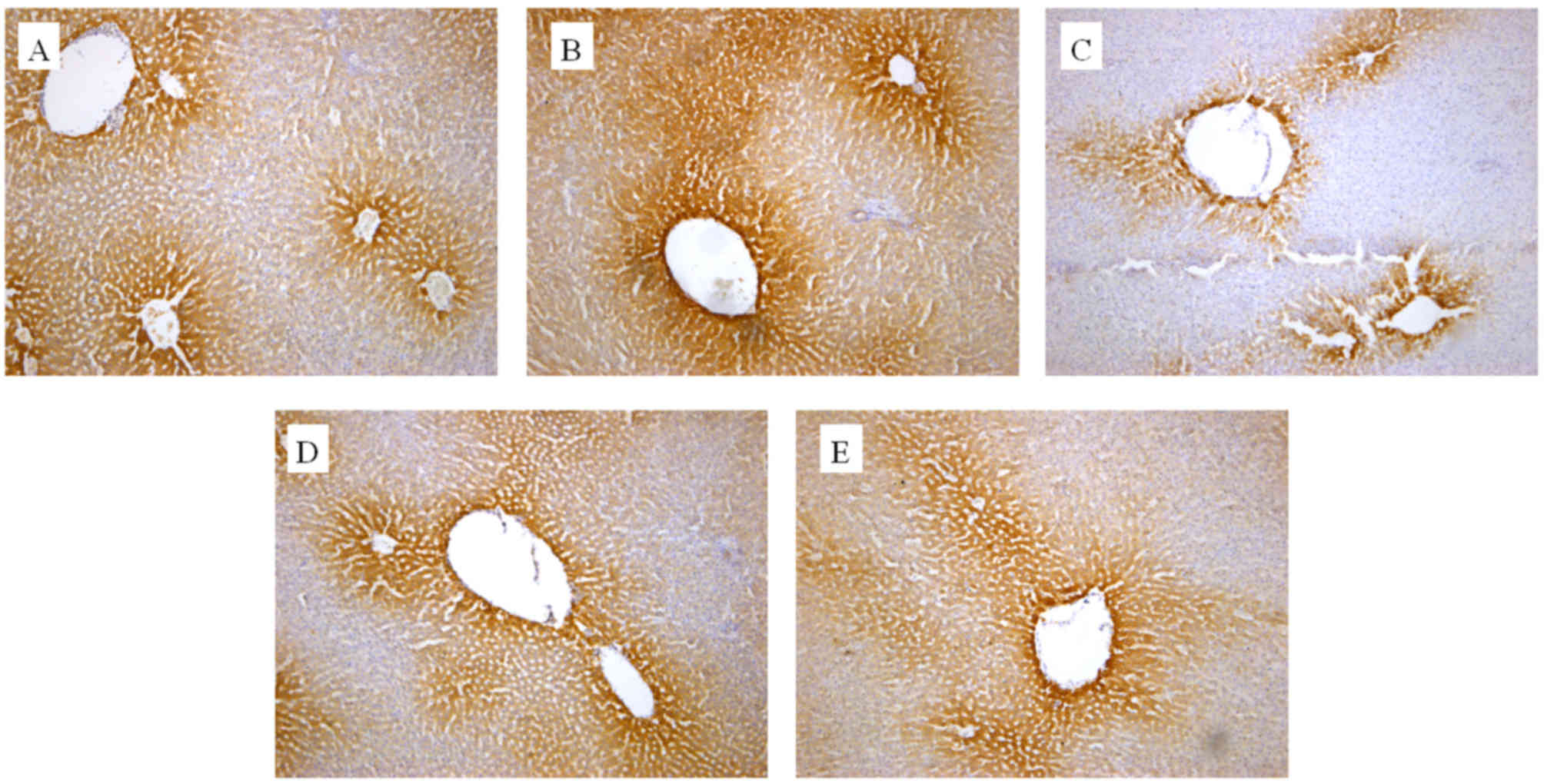
|
1
|
Xu Y, Qiu HQ, Liu H, Liu M, Huang ZY, Yang
J, Su YP and Yu CX: Effects of koumine, an alkaloid of Gelsemium
elegans Benth., on inflammatory and neuropathic pain models and
possible mechanism with allopregnanolone. Pharmacol Biochem Behav.
101:504–514. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Editorial Committee of Chinese Materia
Medica, . the Administration Bureau of Traditional Chinese
Medicine: Chinese Materia Medica (ZhonghuaBencao). Shanghai Science
and Technology: Shanghai. 6:1–215. 2000.
|
|
3
|
Rujjanawate C, Kanjanapothi D and Panthong
A: Pharmacological effect and toxicity of alkaloids from gelsemium
elegans benth. J Ethnopharmacol. 89:91–95. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Yang F, Lu Y, Li Y, Meng ZQ and Liang NS:
Experimental research on antineoplastic effect of extract from
Gelsemiu Elegans Benth. Guangxi Zhong Yi Yao. 1:51–53. 2004.(In
Chinese).
|
|
5
|
Yang CS, Chhabra SK, Hong JY and Smith TJ:
Mechanisms of inhibition of chemical toxicity and carcinogenesis by
diallyl sulfide (DAS) and related compounds from garlic. J Nutr.
131:1041S–1045S. 2001.PubMed/NCBI
|
|
6
|
Ronis MJJ, Lindros KO and Ingelman S: The
cytochrome P450 2E subfamilyIoannides C: Cytochromes P450,
metabolic and toxicological aspects. CRC Press; Boca Raton, FL: pp.
211–239. 1996
|
|
7
|
Bradford BU, Kono H, Isayama F, Kosyk O,
Wheeler MD, Akiyama TE, Bleye L, Krausz KW, Gonzalez FJ, Koop DR
and Rusyn I: Cytochrome P450 CYP2E1, but not nicotinamide adenine
dinucleotide phosphate oxidase, is required for ethanol-induced
oxidative DNA damage in rodent liver. Hepatology. 41:336–344. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Gallagher EP, Kunze KL, Stapleton PL and
Eaton DL: The kinetics of aflatoxin B1 oxidation by human
cDNA-expressed and human liver microsomal cytochromes P450 1A2 and
3A4. Toxicol Appl Pharmacol. 141:595–606. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wang Y, Wu S, Chen Z, Zhang H and Zhao W:
Inhibitory effects of cytochrome P450 enzymes CYP1A2, CYP2A6,
CYP2E1 and CYP3A4 by extracts and alkaloids of gelsemium elegans
roots. J Ethnopharmacol. 166:66–73. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Dourado DF, Fernandes PA and Ramos MJ:
Glutathione transferase classes alpha, pi, and mu: GSH activation
mechanism. J Phys Chem B. 114:12972–12980. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Townsend DM, Manevich Y, He L, Hutchens S,
Pazoles CJ and Tew KD: Novel role for glutathione S-transferase pi.
regulator of protein S-glutathionylation following oxidative and
nitrosative stress. J Biol Chem. 284:436–445. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Lu TL, Su LL, Ji D, Gu W and Mao CQ:
Interaction between CYP450 enzymes and metabolism of traditional
chinese medicine as well as enzyme activity assay. Zhongguo Zhong
Yao Za Zhi. 40:3524–3529. 2015.(In Chinese). PubMed/NCBI
|
|
13
|
Wang Y, Wu S, Li D and Wang H: Uniform
design research on the compatibility toxicity of Gelsemium Elegans
Benth and Mussaenda Pubescens. Chin J Mod Appl Pharm. 2:150–154.
2016.
|
|
14
|
Lu XH, Wang YH, Wang HS, Li DS and Wu SS:
Study on the HPLC fingerprint of chemical constituents in
dichloromethane extraction of gelsemium elegans. Shizhen Guo Yi Guo
Yao. 5:1055–1057. 2016.(In Chinese).
|
|
15
|
Yu HM, Lu XH, Wang YH and Wu SS:
Establishment of determination method of total triterpenoids in
Mussaendapubescens. J Liaoning Univ TCM. 2:40–42. 2015.
|
|
16
|
Spencer WE and Christensen MJ: Multiplex
relative RT-PCR method for verification of differential gene
expression. Bio Techniques. 27:1044–1046. 1999.
|
|
17
|
Habig WH, Pabst MJ and Jacoby WB:
Glutathiene S-transferases. The first enzymatic step in the
mercapturic acid formation. J Biol Chem. 249:7130–7139.
1974.PubMed/NCBI
|
|
18
|
Autrup H: Genetic polymorphisms in human
xenobiotica metabolizing enzymes as susceptibility factors in toxic
response. Mutat Res. 464:65–76. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Li L, Li W, Kim YH and Lee YW: Chlorella
vulgaris extract ameliorates carbon tetrachloride-induced acute
hepatic injury in mice. Exp Toxicol Pathol. 65:73–80. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Truong Q, Bradley SA, Doss GA, Kumar S and
Reddy VB: Glutathione S-transferase catalyzed desulfonylation of a
sulfonylfuropyridine. Drug Metab Dispos. 38:108–114. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Vancutsem PM and Babish JG: In vitro and
in vivo study the effects of enrofloxacin on hepatic cytochrome
P450. potential for drug interactions. Vet-Hum Toxicol. 38:254–256.
1996.PubMed/NCBI
|