Introduction
Postmenopausal osteoporosis is a degenerative
disease caused by lack of estrogen. Bone degeneration in menopausal
patients exceeds bone formation, resulting in loss of bone mass.
Osteoporosis can cause bone fragility and increase the risk of
fracture. Multiple drugs have been proved to be able to improve
bone strength in postmenopausal women. Those drugs include
bisphosphonates, vitamin D, vitamin K and teriparatide. The
combination of these drugs can effectively improve the bone
strength of some patients with osteoporosis (1).
Osteoblasts can synthesize non-collagenous bone
protein, which is known as osteocalcin (OC). The γ-carboxylation of
OC requires vitamin K. Carboxylated-type of osteocalcin (Gla-OC)
can effectively stimulate osteoblast to enhance bone mineralization
(2). It has been reported that
vitamin K can bind to steroid receptors to regulate gene
transcription in osteoblast (3,4). In
addition, vitamin K not only enhances bone formation, but also
inhibits bone degradation (5,6). Vitamin
K treatment can prevent bone loss and reduce the risk of fracture
in women with postmenopausal osteoporosis (7,8).
Injection of teriparatide increases the number of osteoblasts,
activates osteoblast activity, promotes osteoblast differentiation,
increases trabecular bone volume and strength, and effectively
stimulates bone formation (9,10).
Clinical trials have shown that teriparatide injection can increase
bone density and reduce fracture risk in women with postmenopausal
osteoporosis (11–14).
It has been reported that K-dependent
γ-carboxylation of osteocalcin may enhance the efficacy of
euproxine in bone repairing in rats with ovariectomy (15). Based on this, we hypothesized that
vitamin K combined with teriparatide can promote bone formation and
inhibit bone degradation, thereby improving bone metabolism. In
this study, vitamin K combined with teriparatide was used to treat
osteoporosis induced by ovariectomy in rats. The effects of the
combination therapy on bone mineral density, bone strength and bone
metabolic parameters were evaluated.
Materials and methods
Animal model
Fifty female Sprague Dawley rats (12 weeks old) were
purchased from Charles River Laboratories (Beijing, Chia). The rats
were separately raised in an SPF grade animal compartment with free
access to water and food. Rats were randomly divided into the sham,
ovariectomy (OVX), vitamin K (VK), teriparatine (TP), and vitamin K
and teriparatide (VK + TP) group (n=10 rats per group). No
significant difference in weight was found between the groups. Rats
in the OVX, VK, TP, and VK + TP groups were subjected to bilateral
ovariectomy through the back. Surgical incision was also made in
the same position of rats in the sham group but ovariectomy was not
performed.
The study was approved by the Ethics Committee of
the Second Affiliated Hospital of Kunming Medical University.
Drug administration
Rats in the VK and VK + TP groups were fed with
vitamin K-deficient feed supplemented with vitamin K (30 mg/kg/day)
and calcium (0.5/100 g feed). Rats in the other groups were fed
with vitamin K-deficient feed supplemented with calcium (0.5/100 g
feed). Rats in the TP and VK + TP groups were injected
subcutaneously with tropicotine hydrochloride (30 µg/kg body
weight), three times a week. Rats in the other groups were injected
subcutaneously with an equal volume of normal saline. After drug
treatment for 8 weeks, the rats were anesthetized by
intraperitoneal injection of pentobarbital sodium (50 mg/kg) to
collect blood (10 ml) from femoral vein. Blood samples were
centrifuged (800 × g for 20 min) to separate serum and serum was
stored at 4°C. The right femur was frozen at −80°C for bone density
and biomechanical detection. The left femur was stored at 70%
ethanol for bone histomorphometry.
Detection of bone metabolism parameters and bone
mineral density. Venous blood was collected and enzyme-linked
immunosorbent assay (ELISA) was used to detect Gla-OC (rat Gla-OC
kit; Takara, Otsu, Japan) and C-telopeptide of type I collagen
(CTX-I kit; RatLaps EIA, Boldon, UK) in serum. Rats were sacrificed
by cervical dislocation, bilateral femur was collected from each
rat, and soft tissue was removed. Dual-energy X-ray absorptiometry
was used to scan the right femur to detect the bone density of the
middle part of the femur and the position 1.5 mm from the growth
plate of the femoral condyle.
Biomechanical testing
Lateral diameter of right femur was measured with a
vernier caliper to calculate the area of the cross section of the
midline of the femur. The three-point bending test was performed
using a hydraulic servo fatigue testing machine (Servopulser;
Shimadzu, Kyoto, Japan). The loading point is in the middle of the
femur, with two fulcrums spaced 20 mm and a loading speed of 6
mm/sec. The load-deformation curve was recorded, and elastic load,
the maximum load and breaking load were calculated.
Bone tissue metrology
Bone tissue was fixed in 70% ethanol and embedded in
glycol methacrylate to prepare undecalcified sections for toluidine
blue staining. The tissues were observed and images were captured
using a fluorescence microscope (DM LB2; Leica, Wetzlar, Germany).
The measuring range was the secondary cancellous bone 1–4 mm below
the growth plate of metaphysis of proximal femur. Images were
randomly selected from the left, middle and right of the sections
and six images were captured for each specimen. The morphological
parameters of bone tissue were analyzed using pathological image
analysis software. Static parameters included bone volume (BV),
tissue volume (TV), and bone volume fraction (BV/TV). Dynamic
parameters included bone formation rate (BFR) (bone turnover
rate)/BV. Bone absorption parameters included number of osteoblast
(Ob), number of osteoblasts per unit area of bone trabecula, as
well as surface area of osteoclast (Oc), number of osteoclasts per
unit area of bone trabecula.
Statistical analysis
SPSS 19.0 software (Chicago, IL, USA) was used for
statistical analysis. Data are expressed as mean ± standard
deviation. Comparisons of indexes between the groups were performed
using the Student's t-test. P<0.05 was considered statistically
significant.
Results
Bone metabolism indexes
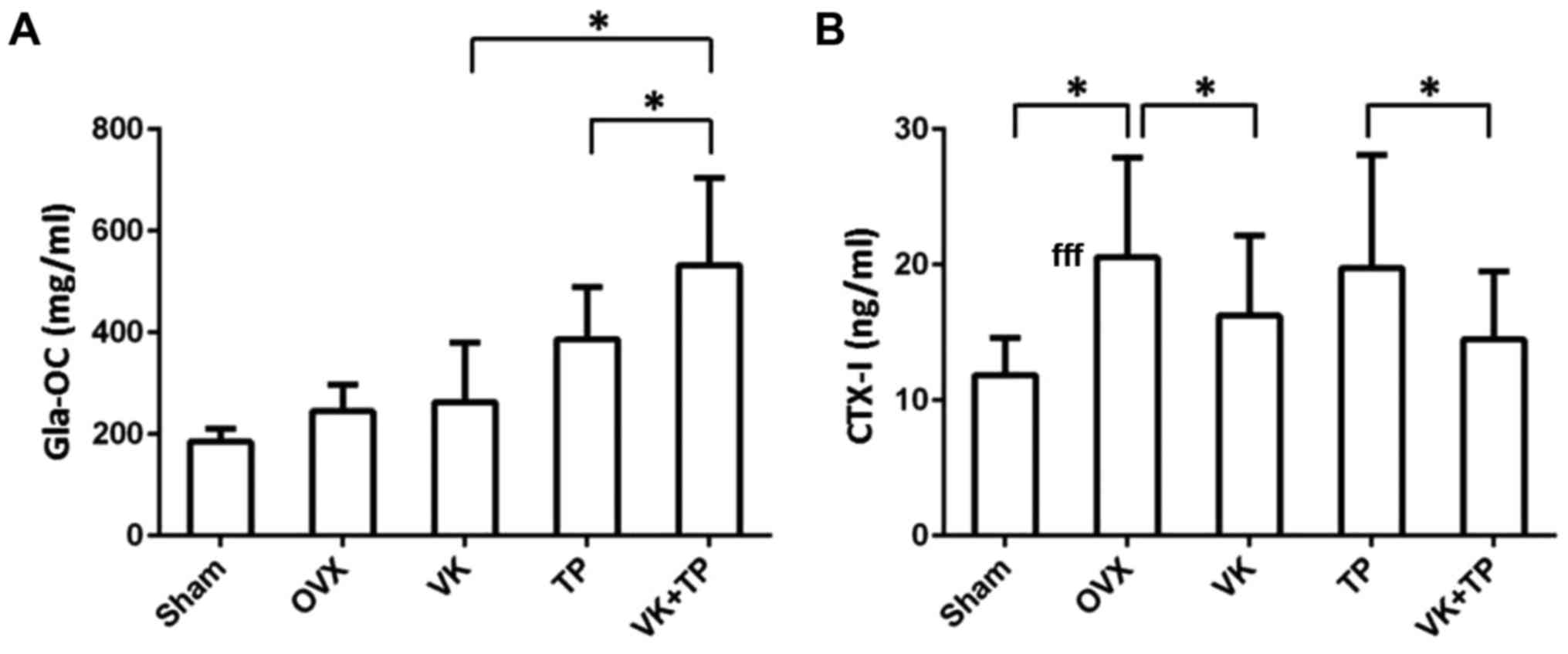
As shown in Fig. 1,
the level of bone formation marker Gla-OC in OVX group was higher
than that in the sham group. Gla-OC level in VK group was slightly
higher than that in OVX group, but the difference was not
statistically significant (P>0.05). Level of Gla-OC in TP group
was significantly higher than that in OVX group (P<0.05). Level
of Gla-OC in VK + TP group was significantly higher than that in VK
and TP groups, individually (P<0.05). Level of bone degradation
marker CTX-I was significantly higher in OVX group than in sham
group (P<0.05). Level of CTX-I in VK group was significantly
lower than that in OVX group (P<0.05). No significant difference
in CTX-I level was found between the TP and OVX groups (P>0.05).
It is noteworthy that CTX-I level in TP group was higher than that
in VK group, but level of CTX-I in VK + TP group was significantly
lower than that in TP group (P<0.05).
Bone density
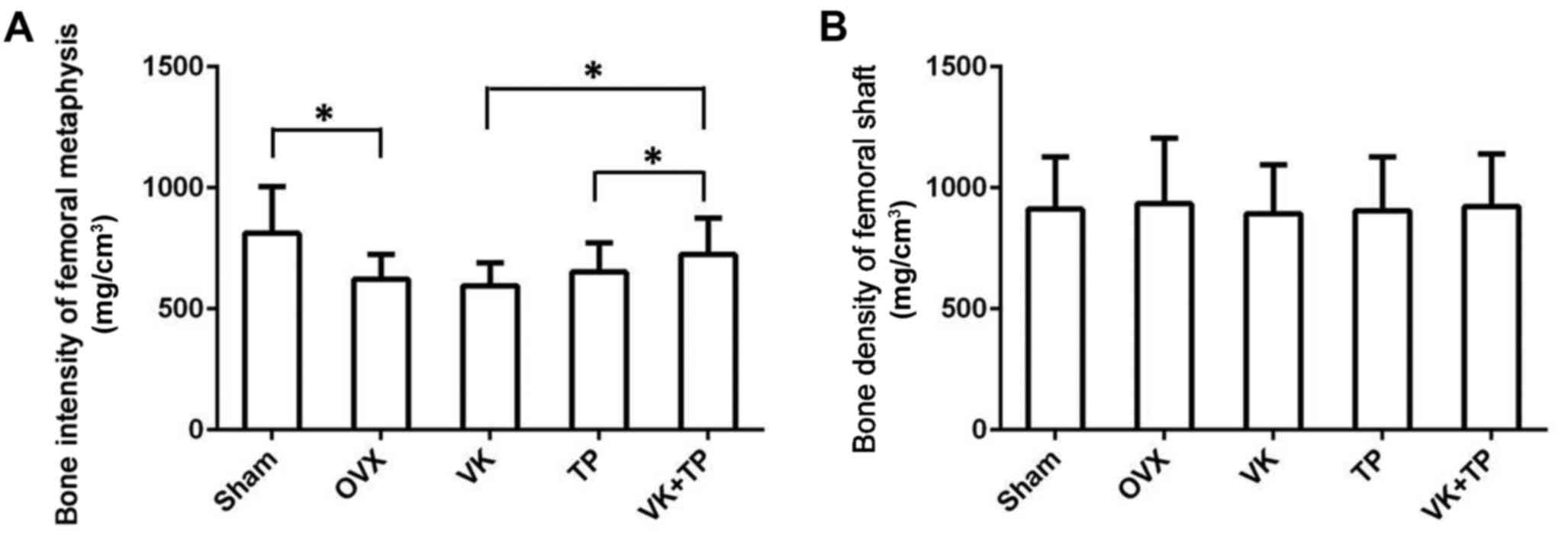
As shown in Fig. 2,
bone intensity of femoral metaphysis was lower in OVX group than
that in the sham group (P<0.05), suggesting the occurrence of
osteoporosis in ovariectomized rats. It is noteworthy that the bone
mineral density of the VK group did not change significantly, and
was slightly lower than that of the OVX group, the bone density of
the TP group was higher than that of the OVX group. Bone mineral
density of VK + TP group was significantly higher than that of the
VK and TP groups (P<0.05). By contrast, there was no significant
difference in bone density of the femoral shaft between groups
(P>0.05).
Biomechanics
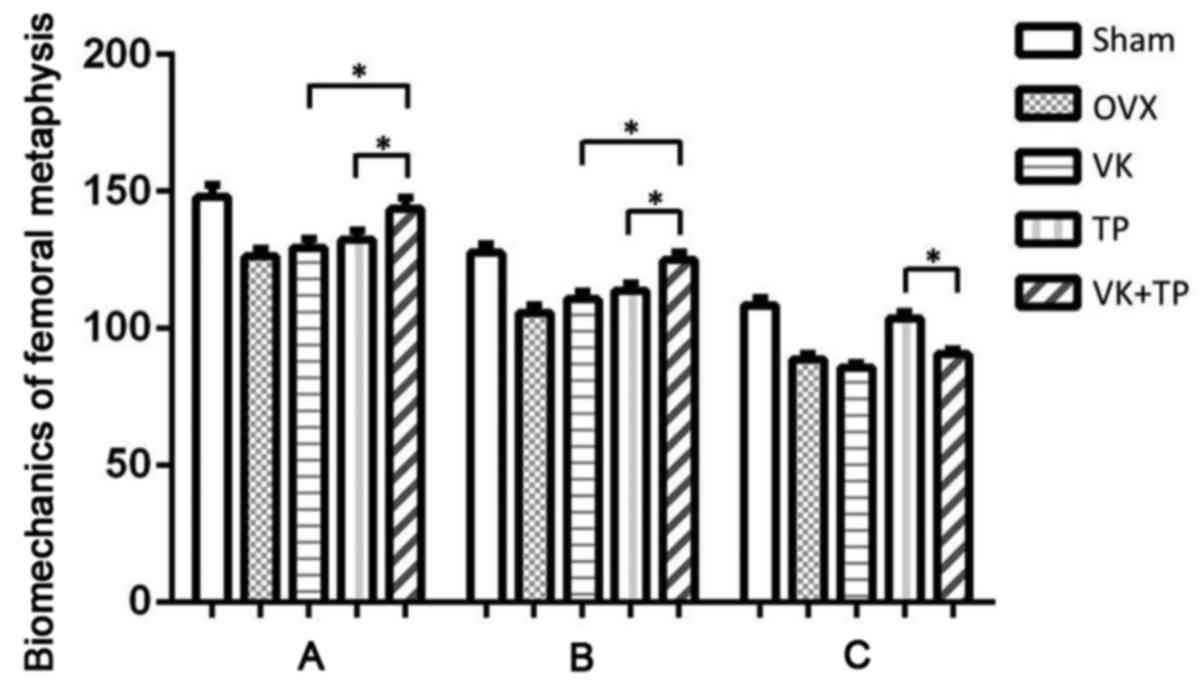
The elastic, maximum and breaking loads of femoral
metaphysis in OVX group were lower than those in Sham group
(P<0.05), suggesting severe osteoporosis in OVX group. The
elastic and maximum loads of the VK and TP groups and the breaking
loads of the TP groups were slightly higher than those of the OVX
group, while those of VK + TP group were significantly higher than
those of the OVX, VK and TP groups (P<0.05), suggesting the
combined use of VK and TP recovered osteoporosis in ovariectomized
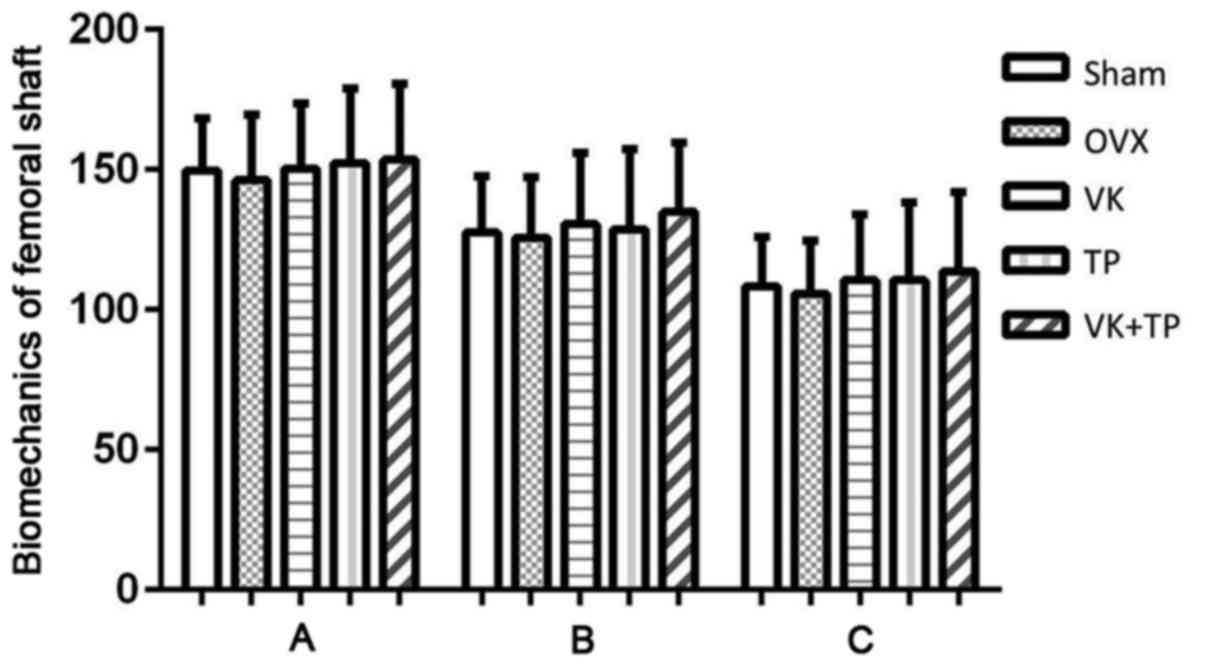
rats (Fig. 3). Similar to the
results of bone density of femoral shaft, no significant
differences in the biomechanical parameters (elastic, maximum and
fracture loads) of the femoral shaft were found between all groups
(Fig. 4).
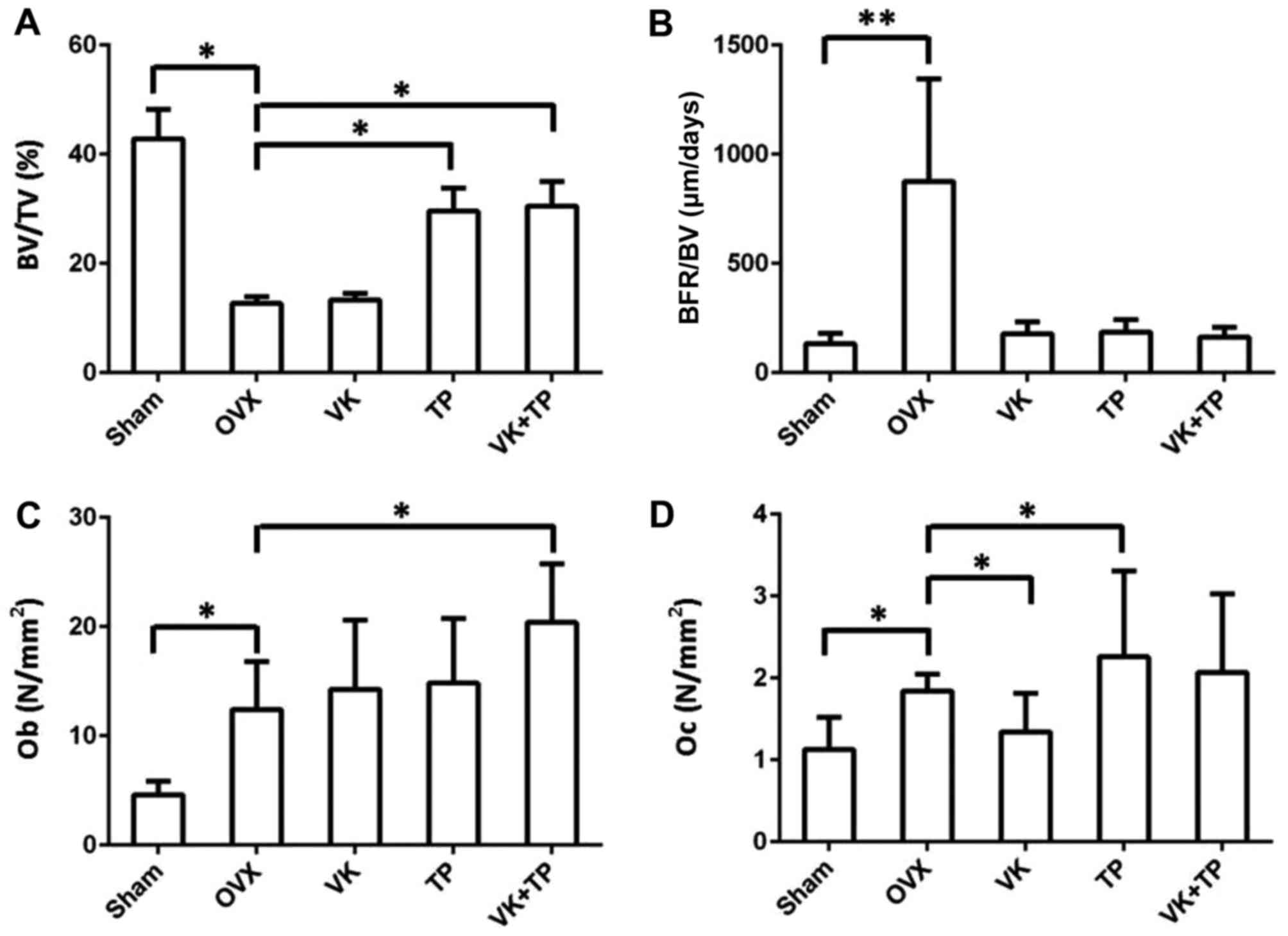
Bone histology
Changes in the static, dynamic, and bone resorption
parameters are shown in Fig. 5. For
the static parameters, bone volume fraction (BV/TV) in OVX group
was significantly lower than that in sham group (P<0.01),
suggesting that osteoporosis in ovariectomized rats. Compared to
the OVX group, BV/TV of the TP group was significantly increased
(P<0.05), but there was no significant change in VK group. By
contrast, compared to OVX group, BV/TV was significantly increased
in the VK + TP group (P<0.05), but no significant difference was
found in BV/TV between the VK + TP and TP groups. For the dynamic
parameters, the bone formation rate (BFR/BV) of OVX group was
significantly higher than that of the sham, VK, TP and VK + TP
groups (P<0.01). BFR/BV of the VK and TP groups was higher than
that of sham group, and BFR/BV of VK + TP group was slightly higher
than that of the sham group. Concerning bone absorption parameters,
Ob was significantly higher in OVX group than in the sham group
(P<0.05). Ob of the VK and TP groups was higher than that of the
OVX group, but the difference was not statistically significant
(P>0.05). Ob of VK + TP group was significantly higher than that
of OVX group (P<0.05). Oc of OVX group was significantly higher
than that of the sham group (P<0.05). Oc of VK group was
significantly lower than that of the OVX group, and the Oc of the
TP group was significantly higher than that of OVX group, while Oc
of the VK + TP group was not significantly different from that of
the OVX group (P<0.05).
Discussion
In this study, ovariectomy was performed to
construct a rat model of osteoporosis. Effects of vitamin K and
teriparatide monotherapy and combination therapy on bone metabolic
parameters, bone mineral density, biomechanics and bone
histomorphology were compared. Compared with monotherapy, vitamin K
combined with teriparatide significantly increased the level of
bone formation marker Gla-OC in serum and the number of
osteoblasts. In addition, vitamin K combined with teriparatide
increased the bone intensity of femoral metaphyseal and improved
femoral tissue metrological parameters BV/TV and BFR. Results
showed that the efficacy of vitamin K combined with teriparatide in
the treatment of rats with osteoporosis is better than that of
monotherapy. The combination treatment can promote bone formation
by increasing the number of osteoblasts.
Rats that underwent ovariectomy were usually used as
an animal model of osteoporosis. The model has good
reproducibility, high bone turnover rate, accelerated bone
formation and degradation, which can mimic postmenopausal
osteoporosis in women caused by the lack of estrogen (16). In this study, ovariectomy resulted in
the loss of femoral metaphyseal minerals and decreased mechanical
strength, but did not affect the bone density and mechanical
strength of the femoral shaft. This is consistent with previous
studies whereby ovarian resection led to loss of cancellous bone
(metaphyseal) without affecting the cortical bone (backbone)
(17). In addition, the present
study also found that ovariectomy increased the serum levels of
Gla-OC and CTX-I, indicating accelerated bone turnover in rats with
osteoporosis.
Following treatment with vitamin K, levels of Gla-OC
were increased, CTX-I levels were decreased, the number of
osteoblasts was increased, while the number of osteoclasts was
decreased in rats with osteoporosis, indicating that vitamin K
promotes bone formation and inhibits bone degradation, which is
consistent with previous studies (5,6,18,19).
Teriparatide can increase the number of osteoblasts in bone
trabecula and activate cell activity in rats with osteoporosis,
thereby promoting bone formation and increasing cancellous bone
mass (20,21). The findings of this study showed that
teriparatide was able to promote bone formation by increasing serum
levels of Gla-OC and number of osteoblasts. The mechanism of the
effects of teriparatide on bone degradation remains unclear.
Administration of teriparatide in ovariectomized rats did not alter
the level of CTX-I, but increased the number of osteoclasts.
Compared with vitamin K or teriparatide monotherapy,
vitamin K combined with teriparatide significantly increased the
serum levels of Gla-OC and the number of osteoblasts, indicating
that vitamin K combined with teriparatide can promote bone
formation. Following treatment with vitamin K, the serum CTX-I
level was reduced and the number of osteoclasts was also reduced.
Compared with trepiparone monotherapy, vitamin K combined with
teriparatide reduced the serum level of CTX-I and the number of
osteoclasts. These results suggest that both vitamin K monotherapy
and in combination with teriparatone can inhibit bone
degradation.
Additionally, the administration of teriparatide in
rats with osteoporosis significantly increased bone density,
maximum load and breaking load, which is consistent with previous
studies (22,23). By contrast, vitamin K can enhance
fracture load, but cannot increase bone density (24). Vitamin K mainly enhances bone
strength by increasing Gla-OC levels and therefore does not affect
bone density (25). Compared to
vitamin K monotherapy, vitamin K combined with teriparatide can
significantly improve the bone density of femoral metaphysis.
Compared with teriparatide monotherapy, vitamin K combined with
teriparatide did not significantly improve bone density. However,
compared with vitamin K or teriparatide monotherapy, vitamin K
combined with teriparatide significantly increased the maximum and
breaking loads. These results suggest that vitamin K combined with
teriparatide can improve bone density and strength. Vitamin K
monotherapy can reduce the femoral elastic load. Similarly,
compared with teriparatide monotherapy, vitamin K combined with
teriparatide also reduces the femoral elastic load. This suggests
that vitamin K can enhance the flexibility of cancellous bone.
Therefore, vitamin K combined with teriparatide, not only enhances
bone strength, but also improves bone mass.
In conclusion, vitamin K combined with teriparatide
can promote bone formation, and improve bone density and strength.
Vitamin K combined with teriparatide combined can effectively
prevent fractures in women with postmenopausal osteoporosis.
However, future studies are required to elucidate the molecular
mechanism of the effects of vitamin K combined with teriparatide on
bone metabolism.
References
|
1
|
Orimo H, Nakamura T, Fukunaga M, Ohta H,
Hosoi T, Uemura Y, Kuroda T, Miyakawa N, Ohashi Y and Shiraki M;
A-TOP (Adequate Treatment of Osteoporosis) research group, :
Effects of alendronate plus alfacalcidol in osteoporosis patients
with a high risk of fracture: The Japanese Osteoporosis
Intervention Trial (JOINT) − 02. Curr Med Res Opin. 27:1273–1284.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Kobayashi M, Hara K and Akiyama Y: Effects
of vitamin K2 (menatetrenone) and alendronate on bone mineral
density and bone strength in rats fed a low-magnesium diet. Bone.
35:1136–1143. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Tabb MM, Sun A, Zhou C, Grün F, Errandi J,
Romero K, Pham H, Inoue S, Mallick S, Lin M, et al: Vitamin K2
regulation of bone homeostasis is mediated by the steroid and
xenobiotic receptor SXR. J Biol Chem. 278:43919–43927. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Igarashi M, Yogiashi Y, Mihara M, Takada
I, Kitagawa H and Kato S: Vitamin K induces osteoblast
differentiation through pregnane X receptor-mediated
transcriptional control of the Msx2 gene. Mol Cell Biol.
27:7947–7954. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Akiyama Y, Hara K, Kobayashi M, Tomiuga T
and Nakamura T: Inhibitory effect of vitamin K2 (menatetrenone) on
bone resorption in ovariectomized rats: A histomorphometric and
dual energy X-ray absorptiometric study. Jpn J Pharmacol. 80:67–74.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Hara K, Akiyama Y, Nakamura T, Murota S
and Morita I: The inhibitory effect of vitamin K2 (menatetrenone)
on bone resorption may be related to its side chain. Bone.
16:179–184. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Inoue T, Fujita T, Kishimoto H, Makino T,
Nakamura T, Nakamura T, Sato T and Yamazaki K: Randomized
controlled study on the prevention of osteoporotic fractures (OF
study): A phase IV clinical study of 15-mg menatetrenone capsules.
J Bone Miner Metab. 27:66–75. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Flore R, Ponziani FR, Di Rienzo TA, Zocco
MA, Flex A, Gerardino L, Lupascu A, Santoro L, Santoliquido A, Di
Stasio E, et al: Something more to say about calcium homeostasis:
The role of vitamin K2 in vascular calcification and osteoporosis.
Eur Rev Med Pharmacol Sci. 17:2433–2440. 2013.PubMed/NCBI
|
|
9
|
Zhang K, Zhang FJ, Zhao WJ, Xing GS, Bai X
and Wang Y: Effects of parathyroid hormone-related protein on
osteogenic and adipogenic differentiation of human mesenchymal stem
cells. Eur Rev Med Pharmacol Sci. 18:1610–1617. 2014.PubMed/NCBI
|
|
10
|
Rosen CJ: The role of parathyroid hormone
in the management of osteoporosis. Horm Res. 64 Suppl 2:81–85.
2005.PubMed/NCBI
|
|
11
|
Neer RM, Arnaud CD, Zanchetta JR, Prince
R, Gaich GA, Reginster JY, Hodsman AB, Eriksen EF, Ish-Shalom S,
Genant HK, et al: Effect of parathyroid hormone (1–34) on fractures
and bone mineral density in postmenopausal women with osteoporosis.
N Engl J Med. 344:1434–1441. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Miyauchi A, Matsumoto T, Sugimoto T,
Tsujimoto M, Warner MR and Nakamura T: Effects of teriparatide on
bone mineral density and bone turnover markers in Japanese subjects
with osteoporosis at high risk of fracture in a 24-month clinical
study: 12-month, randomized, placebo-controlled, double-blind and
12-month open-label phases. Bone. 47:493–502. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Boonen S, Marin F, Mellstrom D, Xie L,
Desaiah D, Krege JH and Rosen CJ: Safety and efficacy of
teriparatide in elderly women with established osteoporosis: Bone
anabolic therapy from a geriatric perspective. J Am Geriatr Soc.
54:782–789. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Miyauchi A, Matsumoto T, Shigeta H,
Tsujimoto M, Thiebaud D and Nakamura T: Effect of teriparatide on
bone mineral density and biochemical markers in Japanese women with
postmenopausal osteoporosis: A 6-month dose-response study. J Bone
Miner Metab. 26:624–634. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Shimizu T, Takahata M, Kameda Y, Hamano H,
Ito T, Kimura-Suda H, Todoh M, Tadano S and Iwasaki N: Vitamin K-
dependent carboxylation of osteocalcin affects the efficacy of
teriparatide (PTH(1–34)) for skeletal repair. Bone. 64:95–101.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Turner RT, Van der Steenhoven JJ and Bell
NH: The effects of ovariectomy and 17 beta-estradiol on cortical
bone histomorphometry in growing rats. J Bone Miner Res. 2:115–122.
1987. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kalu DN: The ovariectomized rat model of
postmenopausal bone loss. Bone Miner. 15:175–191. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Koshihara Y and Hoshi K: Vitamin K2
enhances osteocalcin accumulation in the extracellular matrix of
human osteoblast in vitro. J Bone Miner Res. 12:431 4381997.
View Article : Google Scholar
|
|
19
|
Iwasaki Y, Yamato H, Murayama H, Takahashi
T, Ezawa I, Kurokawa K and Fukagawa M: Menatetrenone prevents
osteoblast dysfunction in unilateral sciatic neurectomized rats.
Jpn J Pharmacol. 90:88–93. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Hori M, Uzawa T, Morita K, Noda T,
Takahashi H and Inoue J: Effect of human parathyroid hormone
(PTH(1–34)) on experimental osteopenia of rats induced by
ovariectomy. Bone Miner. 3:193–199. 1988.PubMed/NCBI
|
|
21
|
Fox J, Miller MA, Newman MK, Metcalfe AF,
Turner CH, Recker RR and Smith SY: Daily treatment of aged
ovariectomized rats with human parathyroid hormone (1–84) for 12
months reverses bone loss and enhances trabecular and cortical bone
strength. Calcif Tissue Int. 79:262–272. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Iwaniec UT, Mosekilde L, Mitova-Caneva NG,
Thomsen JS and Wronski TJ: Sequential treatment with basic
fibroblast growth factor and PTH is more efficacious than treatment
with PTH alone for increasing vertebral bone mass and strength in
osteopenic ovariectomized rats. Endocrinology. 143:2515–2526. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Alexander JM, Bab I, Fish S, Müller R,
Uchiyama T, Gronowicz G, Nahounou M, Zhao Q, White DW, Chorev M, et
al: Human parathyroid hormone 1–34 reverses bone loss in
ovariectomized mice. J Bone Miner Res. 16:1665–1673. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Matsumoto T, Miyakawa T and Yamamoto D:
Effects of vitamin K on the morphometric and material properties of
bone in the tibiae of growing rats. Metabolism. 61:407–414. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Iwamoto J, Sato Y, Takeda T and Matsumoto
H: Bone quality and vitamin K2 in type 2 diabetes: Review of
preclinical and clinical studies. Nutr Rev. 69:162–167. 2011.
View Article : Google Scholar : PubMed/NCBI
|