Introduction
Goodpasture's syndrome (GPS) is an immuno disorder
characterized by pulmonary hemorrhage, glomerulonephritis and the
presence of the antiglomerular basement membrane (anti-GBM)
antibody (1,2). The incidence of GPS is <1
case/1,000,000 (3). Combination
therapy is recommended for the initial management of the disease,
including corticosteroid therapy, cytostatic therapy and
plasmapheresis. However, patients have poor prognosis with current
treatments and the mortality rate has been estimated as 11%
(4). The etiology of Goodpasture's
Syndrome remains unknown; however, the development of the disease
following environmental exposures including hydrocarbon fumes,
metallic dust or tobacco smoke has been reported (5). GPS occurs due to antibody and
T-lymphocyte reactivity to the NC1 domain of the α3 chain of type
IV collagen (6).
Women presenting with GPS in the early period of
pregnancy are uncommon and the present case study reports an
unusual case of GPS in the early stage of pregnancy. In the present
study, the patient received hemodialysis and methylprednisolone,
prior to succumbing due to sustained, irreversible renal failure.
The majority of pregnancies are terminated prior to term delivery
to prevent worsening of conditions (7,8). Thus,
current therapeutic strategies used to treat GPS should be revised
in order to improve the survival rate of pregnant patients with
GPS.
Case report
A 17-year-old female was hospitalized at the
Traditional Chinese Medicine Hospital of Guizhou (Guiyang, China)
on 9th December 2013 due to no urination and hemoptysis for 3 days
in the thirteenth week of pregnancy. The patient had history of
double lower limb swelling lasting from the onset of pregnancy,
which was treated with furosemide in Qianxi County Central
hospital. The patient's medical history prior to pregnancy was
normal; however, the patient had a 2-week history of cough prior to
admission and experienced an episode of brown-colored urine 1 week
later. The patient did not experience fever, dysuria or oliguria at
that time. The patient visited Qianxi County Central hospital
(Quianxi, China) on 21th October 2013 and was diagnosed with acute
renal insufficiency. Serum was analysed using centrifugation at
37°C for 5 min at 3,000 × g. The results were as follows: Urea
nitrogen, 34.98 mmol/l (normal range, 1.70–8.30 mmol/l);
creatinine, 1108.0 µmol/l (normal range, 41.0–73.0 µmol/l). Urine
analysis indicated that the urine was protein positive (+) and
examination under a light microscope indicated white blood cell
counts of 25/high power field which, indicated proteinuria and
pyuria. The patient received treatment with 40–100 mg intravenous
furosemide (Hunan Wuzhou Pharmaceutical Co., Ltd, Hunan, China) for
four days with a total dose of 240 mg in Qianxi County Central
hospital. The patient provided written, informed consent for
inclusion in the present case report.
Results of examination at the time of admission were
as follows: Respiratory rate, 20/min (normal range, 12–20/min);
pulse rate, 116/min (normal range, 60–100/min); and blood pressure,
146/106 mmHg (normal range, 90–129/60-84 mmHg).
Laboratory findings using a Sysmex XE-2100 automatic
blood cell analyzer as follows: Hemoglobin, 60 mg/dl (normal range,
115–150 mg/dl); white blood cell count, 14.3 k/mm3
(normal range, 3.5–9.5 k/mm3); platelet count, 181
k/mm3 (normal range, 125–350 k/mm3);
potassium, 5.84 mmol/l (normal range, 3.5–5.3 mmol/l);
cholinesterase, 2,334 U/l (normal range, 4,000–11,700 U/l);
bicarbonate, 21 mEq/l (normal range, 22–29 mEq/l); blood urea
nitrogen, 34.23 mmol/l and creatinine, 882.1 µmol/l (normal range,
1.70–8.30 mmol/l and 41.0–73.0 µmol/l). Serum protein levels were
all normal: Myoglobin, 34 ng/ml; B-type natriuretic peptide,
>5,000 pg/ml and D-dimer 1,660 ng/ml. Blood gas analysis
detected a urine pH of 7.293 (normal range, 7.35–7.45),
PCO2 of 24.5 mmHg (normal range, 35–45 mmHg), PO2 of
51.1 mmHg (85–100 mmHg), calcium of 0.5 mmol/l (1.1–1.3 mmol/l),
Be-1 of 4.7 mmol/l (−2.7–2.7) and urine protein of 1+. All
parameters were analysed using a Radiometer ABL90 micro blood gas
analyzer (Radiometer Medical ApS, Copenhagen, Denmark). The levels
of complements C3 and C4 were high at 195 and 57 mg/dl,
respectively (normal C3 range, 90–170 mg/dl; normal C4 range 12–36
mg/dl). Antiglomerular basement membrane antibodies were positive.
Anti GBM antibody was positive. Antinuclear antibody, cytoplasmic
and perinuclear antineutrophil cytoplasmic antibodies (C-ANCA and
P-ANCA, respectively), hepatitis panel antibody and HIV antibody
tests were negative.
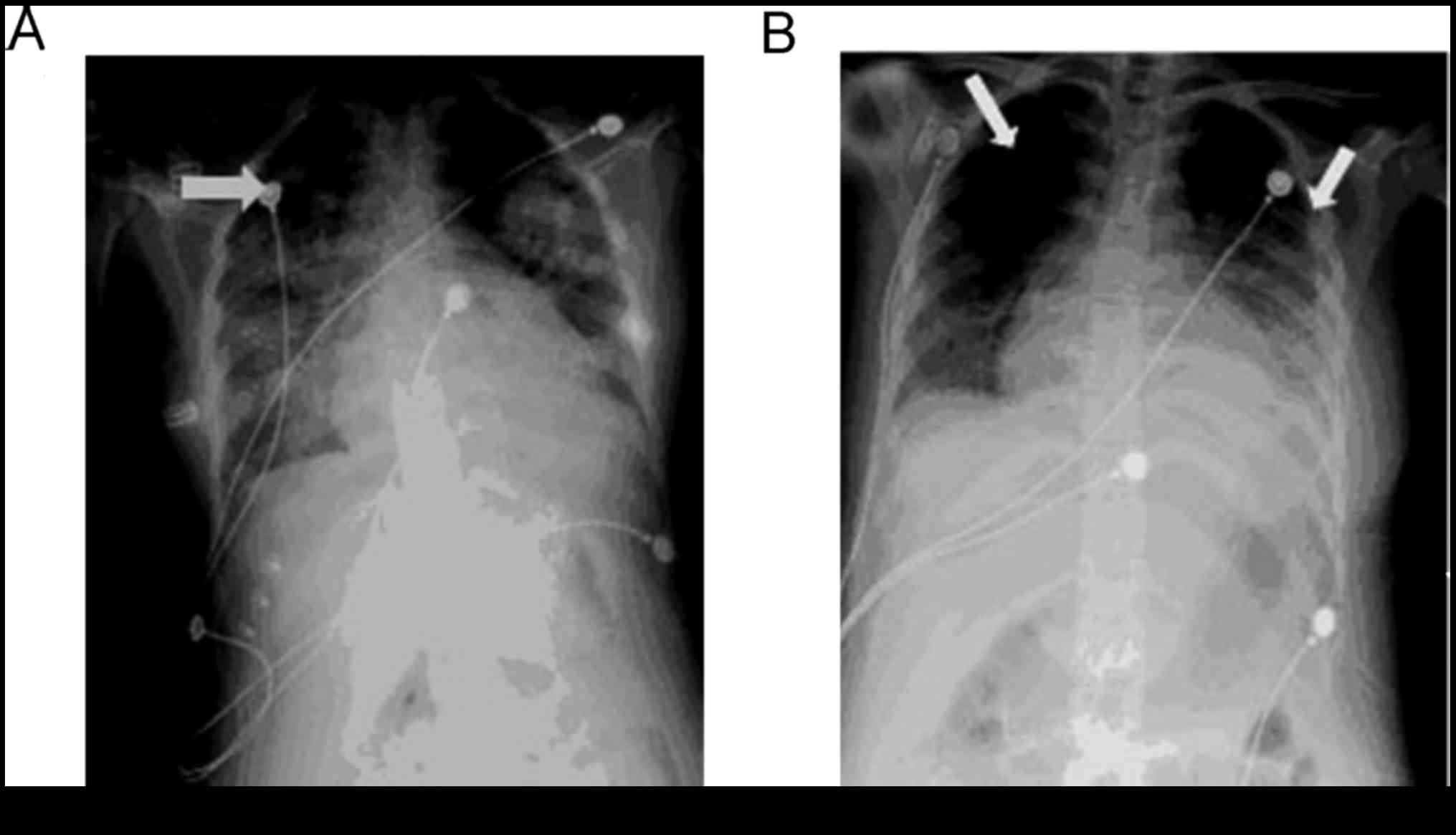
Chest X-ray (CXR) and computed tomography (CT) were
performed with a lead coat to protect the fetus on the 3rd day
(Fig. 1). CXR identified diffuse
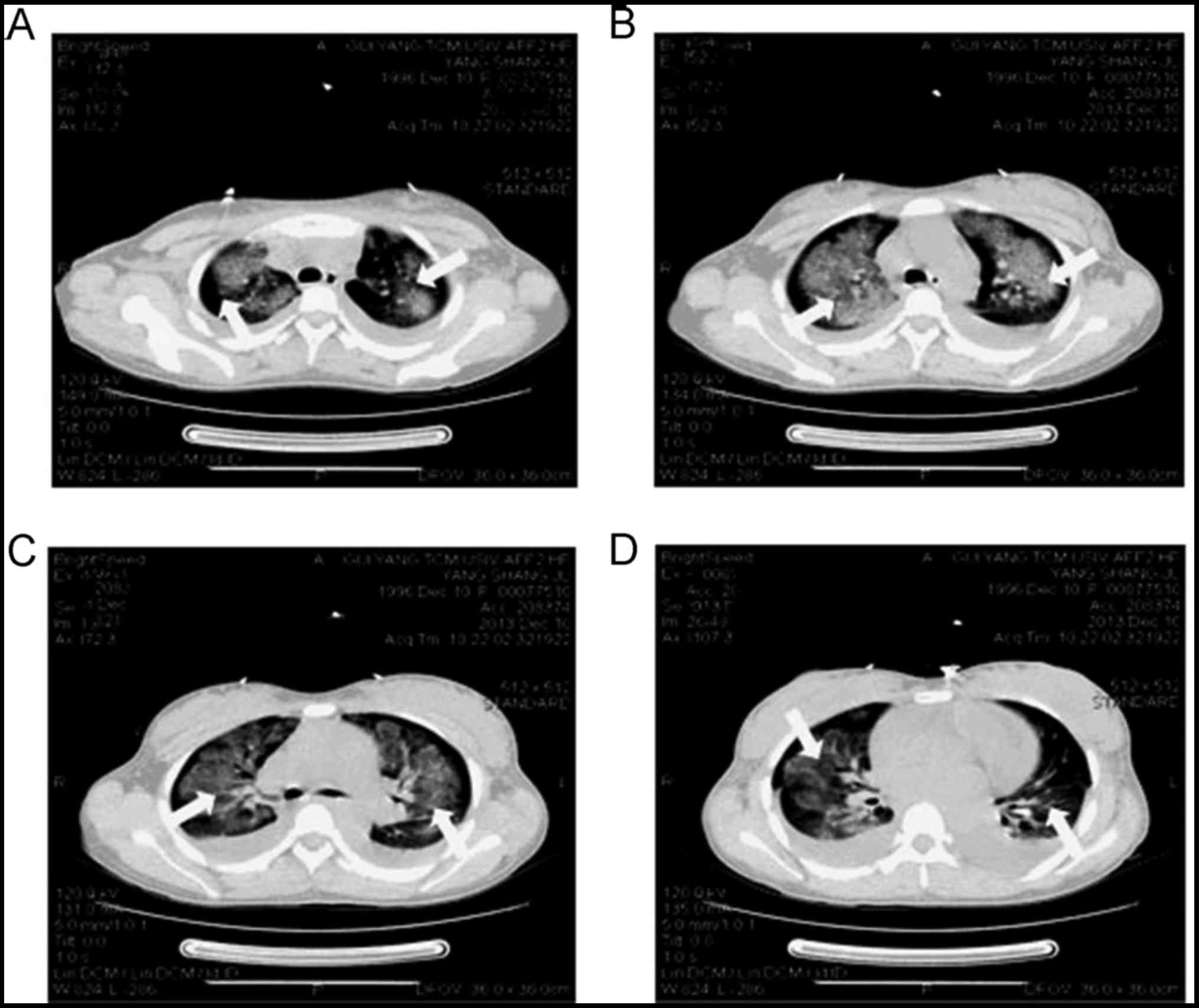
density parenchymal lesions with fibrosis in the lungs (Fig. 1A). Flake and cloud shaped lesions in
the lungs identified by CT exhibited high density at different
levels: The sternal angle level, aortic level, tracheal carina and
bronchus (Fig. 2).
The patient was started on hemodialysis (B. Braun
hemodialysis machine, Hessen, Germany) and received 1 g/day
methylprednisolone (Pfizer, Inc., New York, NY, USA) intravenously
for 3 days Hemodialysis parameters: Following dilution,
displacement fluid, 2,000 ml/h; blood flow rate, 150–200 ml/min;
temperature, 38°C; ultrafiltration, 300–500 ml/h. The patient
underwent hemodialysis 6–8 h/day for five days.
A renal biopsy was taken 7 days following admission.
During the hospital stay, the patient's renal function improved;
urea levels were 16 mmol/l and serum creatinine was 320 µmol/l
(normal range, 1.70–8.30 mmol/l and 41.0–73.0 µmol/l). Samples were
centrifuged at 37°C for 5 min at 3,000 × g and parameters were
measured, The patient's urine volume was within the normal range of
250–400 ml/day. However, the patient's hemoglobin dropped to 45
mg/dl and a blood transfusion was required. Following this, the
patient's clinical condition improved: PO2 increased to
90 mmHg and hemoptysis disappeared 3 days following blood
transfusion.
Chest X-ray (CXR) taken 10 days following admission
identified a significant decrease in the diffuse density of
parenchymal lesions (Fig. 1B). A CT
scan was not performed at this point due to patient worries
regarding fetus safety. Anti GBM antibody became negative at this
point.
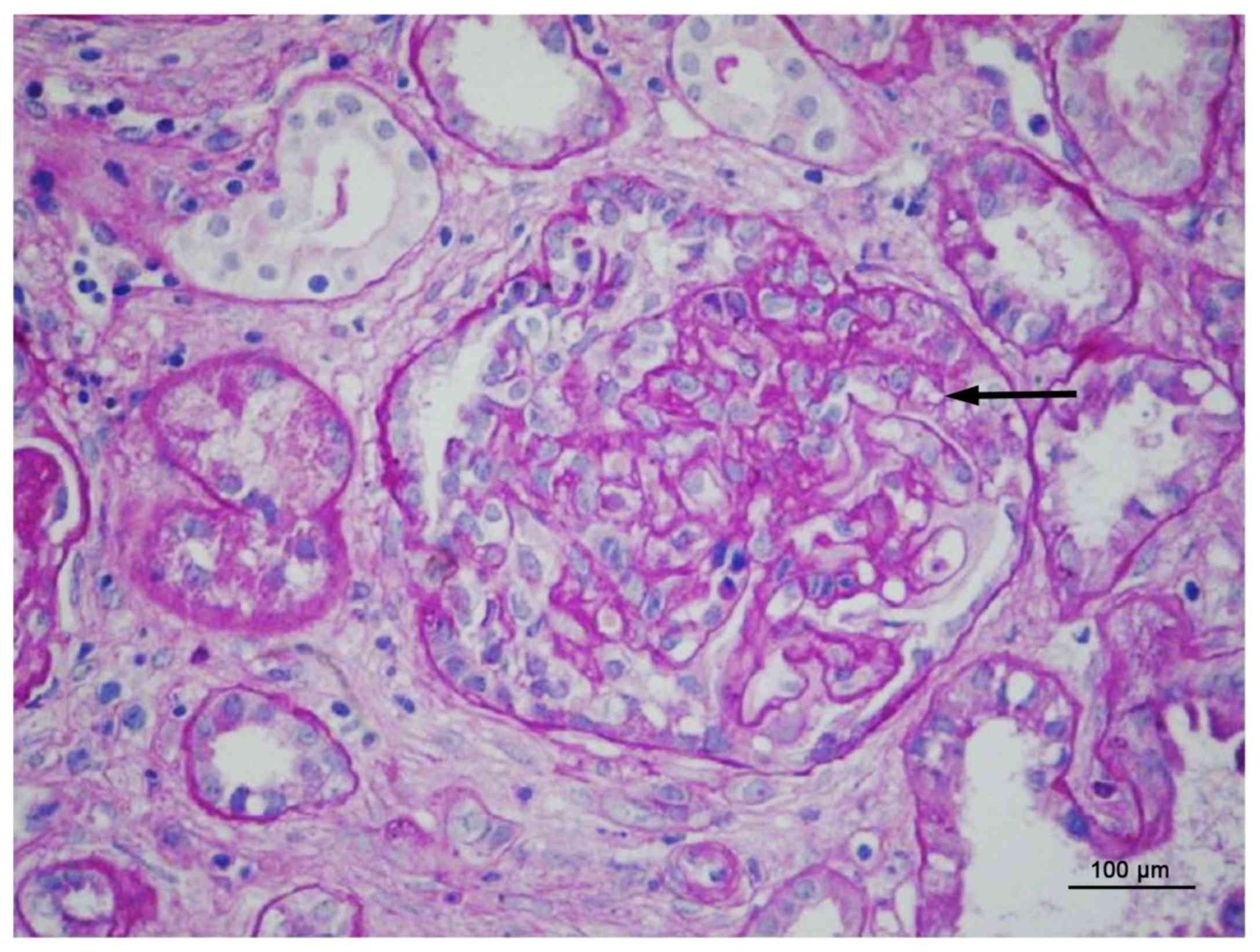
Renal biopsy was performed 7 days after admission.
4% Formaldehyde fixation (pH=7.0) was performed for the tissues at
room temperature for 5 h. Hematoxylin and eosin staining was
performed at room temperature for ~30 min on embedded tissues and
sections were 1 mm thick.
It revealed cellular crescents in all 20 glomeruli
observed in the biopsy specimen (Fig.
3). Light microscopic analysis of 27 glomeruli revealed 16
glomerulosclerosis. The remaining glomeruli consisted of 2 cellular
crescents and 7 cell fibrous crescents with the formation of
fracture and shrinkage in capillaries. The 2 glomerular mesangial
cells with no crescent exhibited no evident hyperplasia and tropic
complex red protein deposition. One of the two glomerular mesangial
cells with no crescent exhibited ischemia and crinkle, and granular
degeneration was identified in epithelial cells.
Granular degeneration was detected in the renal
tubular epithelial cells and visible epithelial shedding and
regeneration were detected in some renal tubular ectasia. Part of
the tubular basement membrane thickened and lumen narrowing was
identified. Multifocal inflammatory cell infiltration and mild
fibrosis were observed in the kidney matter with thickening
arteriolar walls and narrow lumens.
Once the patient was started on furosemide and
methylprednisolone the pregnancy continued. However, whether these
drugs administered during pregnancy increases the likelihood of the
fetus surviving remains unclear.
The patient was discharged at 16 weeks gestation, 11
days following the start of treatment and ultrasound confirmed
survival of the fetus at this point.
The patient attended irregular follow-up in Qianxi
County hospital. Renal function declined following discharge with
the results of laboratory tests indicating urea levels of 58.19
mmol/l and creatinine levels of 1,625 µmol/l (normal range,
1.70–8.30 mmol/l and 41.0–73.0 µmol/l) during the second trimester
and the patient finally succumbed four weeks following
discharge.
Discussion
GPS is a rare autoimmune disease with a low
incidence rate of <1/1,000,000 (9). The primary clinical manifestation of
GPS includes pulmonary hemorrhage, crescentic glomerulonephritis
and the presence of anti-glomerular basement membrane antibodies
(10). To date, there have been few
case reports of GPS occurring during pregnancy (11). Therefore, it remains controversial
whether the termination of pregnancy and current therapeutic
strategies used to treat GPS increase the chances of full term
pregnancy being achieved and improve survival rates in pregnant
patients.
Few studies have described cases of GPS in women
during different periods of pregnancy. To date, there have been
only 4 studies investigating the effect of pregnancy on GPS. Nair
et al (12) described a
pregnant woman diagnosed with GPS at 13 weeks. This patient
underwent termination at 15 weeks and received plasma exchange and
treatment with steroids. Renal failure was resolved following
treatment. Muqeet et al (13)
reported that a patient with GPS experienced a rapid recovery
following spontaneous abortion and speculated that the termination
of pregnancy may be responsible (13). However, Deubner et al
(14) postulated the hypothesis that
gravidity may serve a useful role in the elimination of anti-GBM
antibody titer but the relative effect of gravidity on anti-GBM
antibodies remains unknown. The placenta may be an adsorptive
surface for the autoantibody and may ameliorate the symptoms. The
study described the case of a pregnant female with acute renal
failure whom survived following delivery. The patient's accelerated
decline in renal function postpartum may have been due to removal
of the placenta. The patient experienced a full-term pregnancy and
gave birth to a healthy infant (14). Thus, the effect of gravidity factors
on anti-GBM antibody should be observed in future clinical studies,
including the number of weeks at which previous pregnancies were
terminated and complications of pregnancy.
The present study describes the case of a pregnant
female diagnosed with GPS that experienced acute renal failure
during the thirteenth week of pregnancy. The patient exhibited an
improvement of renal function following regular hemodialysis and
treatment with methylprednisolone. Although GPS was controlled
without the patient undergoing a termination of pregnancy, there
was no evidence of the effect of terminating pregnancy on the
development of GPS. The results and conclusions of the present
study may be difficult to extrapolate; therefore further studies
are required to enable reliable analysis.
Meanwhile, assessments of the prognosis of GPS
during pregnancy and delivery have attracted the attention of
gynecologists. Currently, the primary therapeutic strategy used to
treat GPS is plasma exchange, which may effectively remove the
pathogenic antibodies in the blood and alleviate symptoms of the
disease (15). Additionally,
methylprednisolone serves an important role in inhibiting the
formation of immune antibodies. The severity of the clinical
symptoms and renal function depend on the antibody titer.
Furthermore, it was demonstrated that patients with ANCA
seropositivity exhibit more unfavorable prognosis (16).
The patient in the present study exhibited
hemoptysis and acute renal dysfunction. C-ANCA and P-ANCA were
negative and GBM was positive. Chest X-ray and CT scanning were
applied to detect GPS with protective measures and the patient
underwent relevant treatment. The renal function of the patient
improved following regular treatment and the GBM turned negative
without termination of pregnancy being performed. However, the
patient did not insist on follow-up and regular treatment following
transfer to a local hospital. The final clinical outcome of the
present study was failure and the patient succumbed.
Factors determining the prognoses of patients
include the level of C-ANCA, P-ANCA and GBM antibodies in the
blood, clinical symptoms and early detection and the present study
demonstrates that the length of follow-up time is important in
determining patient prognosis. Even if immune antibodies turn
negative, renal function should continue to be monitored,
particularly during pregnancy. Levels of creatinine and urea
nitrogen should be regarded as prognostic indicators and should be
used to determine the treatment course.
The present study describes a case of renal failure
with positive anti-GBM antibodies, which is more likely to be
present in patients with progression and recurrence of renal or
pulmonary disease. Previous studies investigating pregnant patients
with GPS demonstrated that they may exhibit atypical presentation
of antibodies in pregnancy, which may delay diagnosis and treatment
(12–14). Due to its diverse clinical
manifestations, gynecologists should consider the possibility of
this disease when patients present with hemoptysis and renal
function damage during pregnancy. Levels of anti-GBM and p-ANCA
antibodies should also be checked. In the present study, a biopsy
was taken to verify the disease and clinical symptoms improved
following regular treatment. Winbeck et al (17) reported that early detection, early
diagnosis and early treatment may improve the prognosis of patients
with GPS. This was also determined in a study by Prabhakaran et
al (18).
The patient in the present study exhibited clinical
improvement following treatment with hemodialysis and
methylprednisolone even as pregnancy progressed. However, the
patient underwent irregular follow-up, which may be responsible for
the decline in renal function of the patient and the endpoint of
mortality. Thus, close long-term follow-up is recommended for
pregnant patients with GPS.
Furthermore, pregnancy-associated complications were
not monitored in the present study. Therefore, more data should be
collected during the prepartum, intrapartum and postpartum periods.
The incidence of fetal and maternal morbidity and mortality in
women with GPS remain significant in recent years (5). Therefore, for women with GPS who want
to conceive or continue with pregnancy, long-term hemodialysis
should be recommended and the period of treatment determined by
renal function. Appropriate contraceptive and pre-pregnancy
counseling should be offered to patients with GPS of childbearing
age. Improved collaboration between the obstetrics and intensive
care units are also required to improve maternal survival rates and
quality of life.
In conclusion, the present study reports a case of a
17-year-old primigravida who presented with Goodpasture's syndrome
during week 13 of pregnancy. The patient was treated with
hemodialysis and methylprednisolone as pregnancy progressed and the
patient's condition improved 11 days following treatment. However,
the patient discharged themselves for personal reasons and
succumbed at 20 weeks gestation.
Acknowledgements
This study was supported by the Guizhou Maternal and
Child Health Hospital [grant no. 2013 (2020); Guiyang, Guizhou,
China].
References
|
1
|
Moulis G, Huart A, Guitard J, Fortenfant F
and Chauveau D: IgA-mediated anti-glomerular basement membrane
disease: An uncommon mechanism of Goodpasture's syndrome. Clin
Kidney J. 5:545–548. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Wang A, Wang Y, Wang G, Zhou Z, Xun Z and
Tan X: Mesangial IgA deposits indicate pathogenesis of
anti-glomerular basement membrane disease. Mol Med Rep.
5:1212–1214. 2012.PubMed/NCBI
|
|
3
|
Lahmer T and Heemann U: Anti-glomerular
basement membrane antibody disease: A rare autoimmune disorder
affecting the kidney and the lung. Autoimmun Rev. 12:169–173. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Stojkovikj J, Zejnel S, Gerasimovska B,
Gerasimovska V, Stojkovic D, Trajkovski M, Angelovska I,
Debreslioska A and Jovanovski S: Goodpasture syndrome diagnosed one
year and a half after the appearance of the first symptoms (case
report). Open Access Maced J Med Sci. 4:683–687. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Proskey AJ, Weatherbee L, Easterling RE,
Greene JA Jr and Weller JM: Goodpasture's syndrome: A report of
five cases and review of the literature. Am J Med. 48:162–173.
1970. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ramaswami A, Kandaswamy T, Rajendran T,
Aung H, Jacob CK, Zinna HS and Telesinge PU: Goodpasture's syndrome
with positive C-ANCA and normal renal function: A case report. J
Med Case Rep. 2:2232008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Nair S, George J, Kumar S, Gracious N and
Das M: A case of Goodpasture's syndrome complicating pregnancy with
dialysis requiring renal failure responding to plasmapheresis and
termination of pregnancy. Ren Fail. 35:1173–1175. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Shah A, Bailey E and Hughes S:
Goodpasture's syndrome, haemodialysis and pregnancy. Br J Hosp Med
(Lond). 68:48–49. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Bolton WK: Goodpasture's syndrome. Kidncy
Int. 50:1753–1766. 1996. View Article : Google Scholar
|
|
10
|
Salama AD, Levy JB, Lightstone L and Pusey
CD: Goodpasture's disease. Lancet. 358:917–920. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Al-Harbi A, Malik GH, Al-Mohaya SA and
Akhtar M: Anti-glomerularn basement membrane antibody disease
presenting as acute renal failure during pregnancy. Saudi J Kidney
Dis Transpl. 14:516–521. 2003.PubMed/NCBI
|
|
12
|
Nair S, George J, Kumar S, Gracious N and
Das M: Acase of Goodpasture's syndrome complicating pregnancy with
dialysis requiring renal failure responding to plasmapheresis and
termination of pregnancy. Ren Fail. 35:1173–1175. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Adnan M Muqeet, Morton J, Hashmi S, Mujeeb
S Abdul, Kem W and Cowley BJ: Anti-GBM of pregnancy: Acute renal
failure resolved after spontaneous abortion, plasma exchange,
hemodialysis, and steroids. Case Rep Nephrol.
2014:2437462014.PubMed/NCBI
|
|
14
|
Deubner H, Wagnild JP, Wener MH and Alpers
CE: Glomerulonephritis with anti-glomerular basement membrane
antibody during pregnancy: Potential role of the placenta in
amelioration of disease. Am J Kidney Dis. 25:330–335. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Jara LJ, Vera-Lastra O and Calleja MC:
Pulmonary-renal vasculitic disorders: Differential diagnosis and
management. Curr Rheumatol Rep. 5:107–115. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ang C, Savige J, Dawborn J, Miach P, Heale
W, Clarke B and Sinclair RS: Anti-glomerular basement membrane
(GBM)-antibody-mediated disease with normal renal function. Nephrol
Dial Transplant. 13:935–939. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Winbeck K, Bruckmaier K, Etgen T, von
Einsiedel HG, Röttinger M and Sander D: Transient ischemic attack
and stroke can be differentiated by analyzing early
diffusionweighted imaging signal intensity changes. Stroke.
35:1095–1099. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Prabhakaran S, Chong JY and Sacco RL:
Impact of abnormal diffusion weighted imaging results on short-term
outcome following transient ischemic attack. Arch Neurol.
64:1105–1109. 2007. View Article : Google Scholar : PubMed/NCBI
|