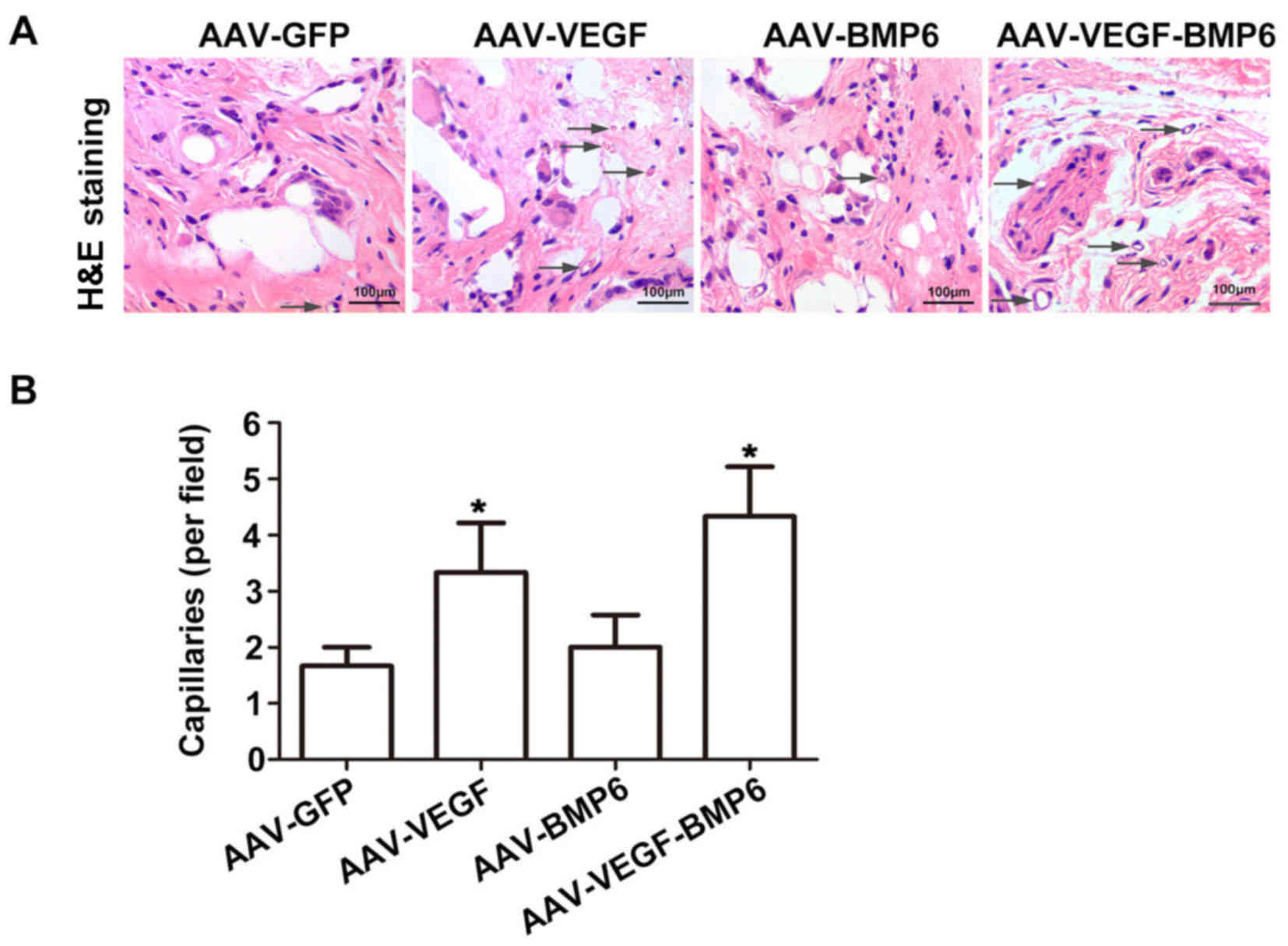
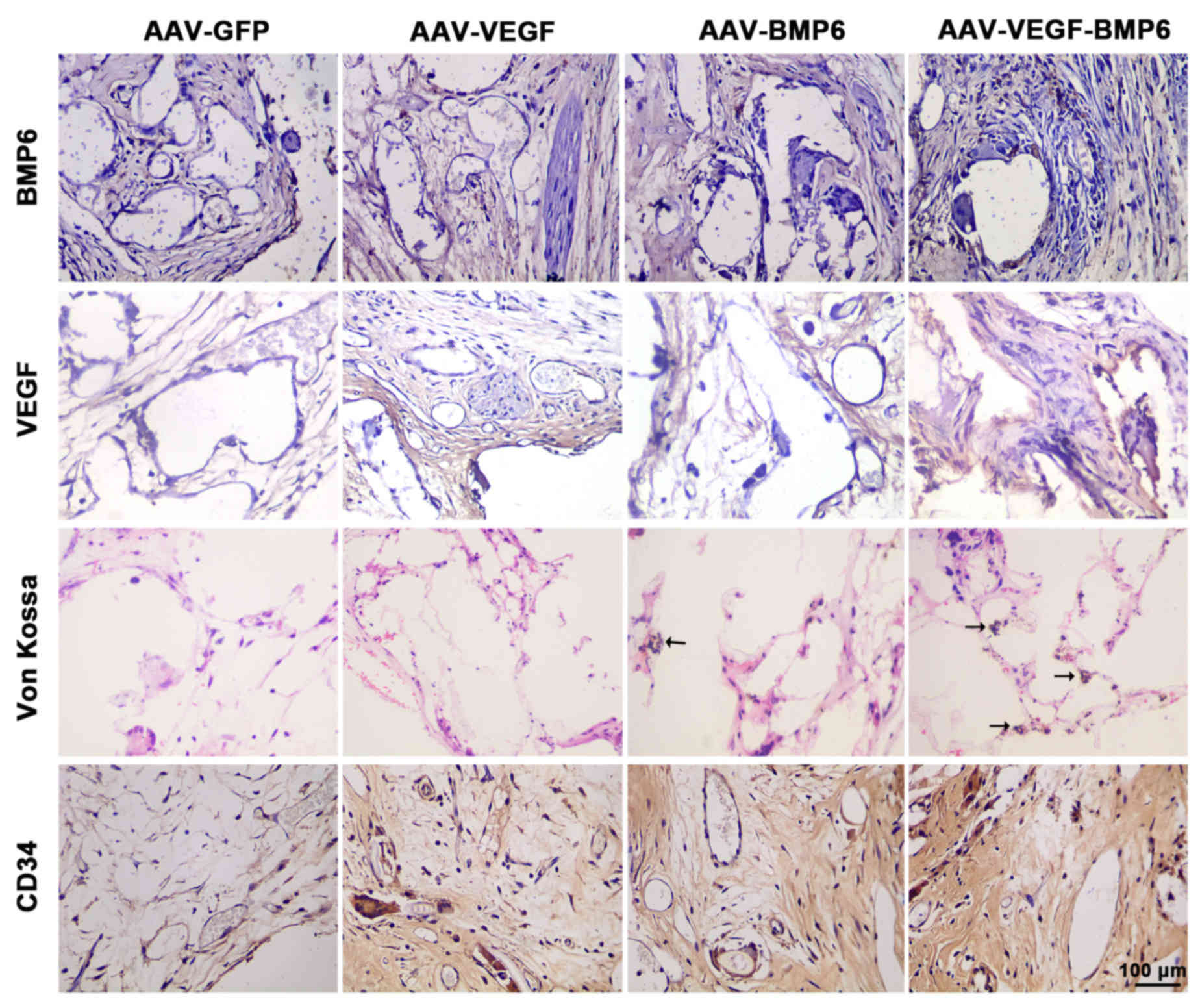
|
1
|
Chamberlain JR, Schwarze U, Wang PR,
Hirata RK, Hankenson KD, Pace JM, Underwood RA, Song KM, Sussman M,
Byers PH and Russell DW: Gene targeting in stem cells from
individuals with osteogenesis imperfecta. Science. 303:1198–1201.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Yang C, Yang S, Du J, Li J, Xu W and Xiong
Y: Experimental study of vascular endothelial growth factor gene
therapy for avascular necrosis of the femoral head. J Huazhong Univ
Sci Technolog Med Sci. 23:297–299, 316. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wilson CG, Martin-Saavedra FM, Vilaboa N
and Franceschi RT: Advanced BMP gene therapies for temporal and
spatial control of bone regeneration. J Dent Res. 92:409–417. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kuh SU, Zhu Y, Li J, Tsai KJ, Fei Q,
Hutton WC and Yoon ST: Can TGF-beta1 and rhBMP-2 act in synergy to
transform bone marrow stem cells to discogenic-type cells? Acta
Neurochir (Wien). 150:1073–1079. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Evans CH and Huard J: Gene therapy
approaches to regenerating the musculoskeletal system. Nat Rev
Rheumatol. 11:234–242. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Herberg S, Susin C, Pelaez M, Howie RN,
Moreno de Freitas R, Lee J, Cray JJ Jr, Johnson MH, Elsalanty ME,
Hamrick MW, et al: Low-dose bone morphogenetic protein-2/stromal
cell-derived factor-1β cotherapy induces bone regeneration in
critical-size rat calvarial defects. Tissue Eng Part A.
20:1444–1453. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
White AP, Vaccaro AR, Hall JA, Whang PG,
Friel BC and McKee MD: Clinical applications of BMP-7/OP-1 in
fractures, nonunions and spinal fusion. Int Orthop. 31:735–741.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
La WG, Jin M, Park S, Yoon HH, Jeong GJ,
Bhang SH, Park H, Char K and Kim BS: Delivery of bone morphogenetic
protein-2 and substance P using graphene oxide for bone
regeneration. Int J Nanomed. 9 Suppl 1:S107–S116. 2014.
|
|
9
|
Wang YJ, Chen S, Deng C, Li F, Wang Y, Hu
X, Shi F and Dong N: MicroRNA-204 Targets Runx2 to Attenuate
BMP-2-induced osteoblast differentiation of human aortic valve
interstitial cells. J Cardiovasc Pharmacol. 66:63–71. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Mundy C, Gannon M and Popoff SN:
Connective tissue growth factor (CTGF/CCN2) negatively regulates
BMP-2 induced osteoblast differentiation and signaling. J Cell
Physiol. 229:672–681. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Hanada K, Solchaga LA, Caplan AI, Hering
TM, Goldberg VM, Yoo JU and Johnstone B: BMP-2 induction and
TGF-beta 1 modulation of rat periosteal cell chondrogenesis. J Cell
Biochem. 81:284–294. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Wang L, Park P, La Marca F, Than KD and
Lin CY: BMP-2 inhibits tumor-initiating ability in human renal
cancer stem cells and induces bone formation. J Cancer Res Clin
Oncol. 141:1013–1024. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Dohin B, Dahan-Oliel N, Fassier F and
Hamdy R: Enhancement of difficult nonunion in children with
osteogenic protein-1 (OP-1): Early experience. Clin Orthop Relat
Res. 467:3230–3238. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Takahashi T, Hatakeyama S and Machida T:
Ductal adenocarcinoma of the pancreas with psammomatous
calcification: Report of a case with immunohistochemical study for
bone morphogenetic protein. Pathol Int. 61:603–607. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Rahman MS, Akhtar N, Jamil HM, Banik RS
and Asaduzzaman SM: TGF-β/BMP signaling and other molecular events:
Regulation of osteoblastogenesis and bone formation. Bone Res.
3:150052015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Scharpfenecker M, van Dinther M, Liu Z,
van Bezooijen RL, Zhao Q, Pukac L, Löwik CW and Ten Dijke P: BMP-9
signals via ALK1 and inhibits bFGF-induced endothelial cell
proliferation and VEGF-stimulated angiogenesis. J Cell Sci.
120:964–972. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Simic P, Culej JB, Orlic I, Grgurevic L,
Draca N, Spaventi R and Vukicevic S: Systemically administered bone
morphogenetic protein-6 restores bone in aged ovariectomized rats
by increasing bone formation and suppressing bone resorption. J
Biol Chem. 281:25509–25521. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Mizrahi O, Sheyn D, Tawackoli W, Kallai I,
Oh A, Su S, Da X, Zarrini P, Cook-Wiens G, Gazit D and Gazit Z:
BMP-6 is more efficient in bone formation than BMP-2 when
overexpressed in mesenchymal stem cells. Gene Ther. 20:370–377.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Street J, Bao M, deGuzman L, Bunting S,
Peale FV Jr, Ferrara N, Steinmetz H, Hoeffel J, Cleland JL,
Daugherty A, et al: Vascular endothelial growth factor stimulates
bone repair by promoting angiogenesis and bone turnover. Proc Natl
Acad Sci USA. 99:pp. 9656–9661. 2002; View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Moens S, Goveia J, Stapor PC, Cantelmo AR
and Carmeliet P: The multifaceted activity of VEGF in
angiogenesis-Implications for therapy responses. Cytokine Growth
Factor Rev. 25:473–482. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Gerber HP, Vu TH, Ryan AM, Kowalski J,
Werb Z and Ferrara N: VEGF couples hypertrophic cartilage
remodeling, ossification and angiogenesis during endochondral bone
formation. Nat Med. 5:623–628. 1999. View
Article : Google Scholar : PubMed/NCBI
|
|
22
|
Ferrara N: VEGF-A: A critical regulator of
blood vessel growth. Eur Cytokine Netw. 20:158–163. 2009.PubMed/NCBI
|
|
23
|
Joensuu K, Uusitalo L, Alm JJ, Aro HT,
Hentunen TA and Heino TJ: Enhanced osteoblastic differentiation and
bone formation in co-culture of human bone marrow mesenchymal
stromal cells and peripheral blood mononuclear cells with exogenous
VEGF. Orthop Traumatol Surg Res. 101:381–386. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ramazanoglu M, Lutz R, Rusche P, Trabzon
L, Kose GT, Prechtl C and Schlegel KA: Bone response to biomimetic
implants delivering BMP-2 and VEGF: An immunohistochemical study. J
Craniomaxillofac Surg. 41:826–835. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zhang W, Zhu C, Wu Y, Ye D, Wang S, Zou D,
Zhang X, Kaplan DL and Jiang X: VEGF and BMP-2 promote bone
regeneration by facilitating bone marrow stem cell homing and
differentiation. Eur Cells Mater. 27:1–12. 2014. View Article : Google Scholar
|
|
26
|
Kempen DH, Lu L, Heijink A, Hefferan TE,
Creemers LB, Maran A, Yaszemski MJ and Dhert WJ: Effect of local
sequential VEGF and BMP-2 delivery on ectopic and orthotopic bone
regeneration. Biomaterials. 30:2816–2825. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Peng H, Usas A, Olshanski A, Ho AM,
Gearhart B, Cooper GM and Huard J: VEGF improves, whereas sFlt1
inhibits, BMP2-induced bone formation and bone healing through
modulation of angiogenesis. J Bone Miner Res. 20:2017–2027. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Pittenger MF, Mackay AM, Beck SC, Jaiswal
RK, Douglas R, Mosca JD, Moorman MA, Simonetti DW, Craig S and
Marshak DR: Multilineage potential of adult human mesenchymal stem
cells. Science. 284:143–147. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Büning H, Perabo L, Coutelle O,
Quadt-Humme S and Hallek M: Recent developments in adeno-associated
virus vector technology. J Gene Med. 10:717–733. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Shi ZB and Wang KZ: Effects of recombinant
adeno-associated viral vectors on angiopoiesis and osteogenesis in
cultured rabbit bone marrow stem cells via co-expressing hVEGF and
hBMP genes: A preliminary study in vitro. Tissue Cell. 42:314–321.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2012.
View Article : Google Scholar
|
|
32
|
Xin CW, Zheng LW, Lei LC, Ma L, Ehrbar M,
Weber FE and Zwahlen RA: Effect of different rhBMP-2 and TG-VEGF
ratios on the formation of heterotopic bone and neovessels. Biomed
Res Int. 2014:5715102014.PubMed/NCBI
|
|
33
|
Ali RR, Reichel MB, Thrasher AJ, Levinsky
RJ, Kinnon C, Kanuga N, Hunt DM and Bhattacharya SS: Gene transfer
into the mouse retina mediated by an adeno-associated viral vector.
Hum Mol Genet. 5:591–594. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Merten OW, Gény-Fiamma C and Douar AM:
Current issues in adeno-associated viral vector production. Gene
Ther. 12 Suppl 1:S51–S61. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Grieger JC, Choi VW and Samulski RJ:
Production and characterization of adeno-associated viral vectors.
Nat Protoc. 1:1412–1428. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Roedersheimer M, West J, Huffer W, Harral
J and Benedict J: A bone-derived mixture of TGF beta-superfamily
members forms a more mature vascular network than bFGF or TGF-beta
2 in vivo. Angiogenesis. 8:327–338. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Samee M, Kasugai S, Kondo H, Ohya K,
Shimokawa H and Kuroda S: Bone morphogenetic protein-2 (BMP-2) and
vascular endothelial growth factor (VEGF) transfection to human
periosteal cells enhances osteoblast differentiation and bone
formation. J Pharmacol Sci. 108:18–31. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Sukul M, Nguyen TB, Min YK, Lee SY and Lee
BT: Effect of local sustainable release of BMP2-VEGF from
Nano-cellulose loaded in sponge biphasic calcium phosphate on bone
regeneration. Tissue Eng Part A. 21:1822–1836. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Cao QS, Zhang T and Zhang J: Correlation
analysis of STAT3 and VEGF expression and eosinophil infiltration
in nasal polyps. Eur Arch Otorhinolaryngol. 272:1955–1960. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Kanczler JM, Ginty PJ, White L, Clarke NM,
Howdle SM, Shakesheff KM and Oreffo RO: The effect of the delivery
of vascular endothelial growth factor and bone morphogenic
protein-2 to osteoprogenitor cell populations on bone formation.
Biomaterials. 31:1242–1250. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Jiang J, Wang SH, Chai F, Ai CC and Chen
SY: The study on vascularisation and osteogenesis of BMP/VEGF
co-modified tissue engineering bone in vivo. RSC Adv.
6:41800–41808. 2016. View Article : Google Scholar
|
|
42
|
Zhang C, Wang KZ, Qiang H, Tang YL, Li Q,
Li M and Dang XQ: Angiopoiesis and bone regeneration via
co-expression of the hVEGF and hBMP genes from an adeno-associated
viral vector in vitro and in vivo. Acta Pharmacol Sin. 31:821–830.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Yung LM, Sánchez-Duffhues G, Ten Dijke P
and Yu PB: Bone morphogenetic protein 6 and oxidized Low-density
lipoprotein synergistically recruit osteogenic differentiation in
endothelial cells. Cardiovasc Res. 108:278–287. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Wei Z, Salmon RM, Upton PD, Morrell NW and
Li W: Regulation of bone morphogenetic protein 9 (BMP9) by
redox-dependent proteolysis. J Biol Chem. 289:31150–31159. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Friedman MS, Long MW and Hankenson KD:
Osteogenic differentiation of human mesenchymal stem cells is
regulated by bone morphogenetic protein-6. J Cell Biochem.
98:538–554. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Cui F, Wang X, Liu X, Dighe AS, Balian G
and Cui Q: VEGF and BMP-6 enhance bone formation mediated by cloned
mouse osteoprogenitor cells. Growth Factors. 28:306–317. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Derubeis AR and Cancedda R: Bone marrow
stromal cells (BMSCs) in bone engineering: Limitations and recent
advances. Ann Biomed Eng. 32:160–165. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Xiong J, Menicanin D, Zilm PS, Marino V,
Bartold PM and Gronthos S: Investigation of the cell surface
proteome of human periodontal ligament stem cells. Stem Cells Int.
2016:19471572016. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Hung SC, Chen NJ, Hsieh SL, Li H, Ma HL
and Lo WH: Isolation and characterization of size-sieved stem cells
from human bone marrow. Stem Cells. 20:249–258. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Bongio M, van den Beucken JJJP,
Leeuwenburgh SCG and Jansen JA: Development of bone substitute
materials: From ‘biocompatible’ to ‘instructive’. J Mater Chem.
20:8747–8759. 2010. View Article : Google Scholar
|
|
51
|
Nugroho RWN, Odelius K, Hoglund A and
Albertsson AC: Highlighting the importance of surface grafting in
combination with a Layer-by-Layer approach for fabricating advanced
3D Poly(L-lactide) microsphere scaffolds. Chem Mater. 28:3298–3307.
2016. View Article : Google Scholar
|
|
52
|
Madhu V, Li CJ, Dighe AS, Balian G and Cui
Q: BMP-non-responsive
Sca1+CD73+CD44+ mouse bone marrow
derived osteoprogenitor cells respond to combination of VEGF and
BMP-6 to display enhanced osteoblastic differentiation and ectopic
bone formation. PLoS One. 9:e1030602014. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Fiedler J, Leucht F, Waltenberger J, Dehio
C and Brenner RE: VEGF-A and PlGF-1 stimulate chemotactic migration
of human mesenchymal progenitor cells. Biochem Biophys Res Commun.
334:561–568. 2005. View Article : Google Scholar : PubMed/NCBI
|