Introduction
Vitiligo is a skin disorder that causes the skin to
lose its natural pigmentation (1).
It can develop at any age and there is no difference in prevalence
according to sex, skin type or race (2). The affected skin can lighten or turn
completely white. Some people develop a few patches while others
lose much more skin color. Usually there are no other signs or
local symptoms. Vitiligo can also affect other parts of the body
such us the hair, the eyes, the inside of the mouth or lips. Some
people develop low self-esteem, social anxiety or serious
depression (1,3,4).
Usually vitiligo represents a standalone phenomenon
but it can arise associated with other autoimmune conditions like
thyroiditis, alopecia areata or lupus (5).
Two types of vitiligo are recognized: non-segmental
(most common) and segmental. Non-segmental vitiligo is
characterized by the development of depigmentation areas on both
sides of the body, while segmental vitiligo is usually limited to
one side of the body with typical distribution patterns in the face
and trunk, which are useful for the differential diagnosis
(6–9).
The therapeutic options in vitiligo include: topical
potent or very potent corticosteroids, topical immunomodulators
(1,2,10);
narrow-band UVB phototherapy (11,12);
oral corticosteroid mini-pulse therapy (2), surgical treatment (skin grafting);
depigmentation with p-benzyloxyphenol, laser treatment, cryotherapy
(1,2), and long-term methotrexate (13). The side effects of those therapies
may include: Skin atrophy, striae, telangiectasia, acne, scales,
pustules, local endosymbiosis proliferation, contact allergy,
immunosuppression, weight gain, sleep disturbances and others cited
unpredictable effects (2,10,14–26), so
the treatment, as in other diseases, should be closely monitored
(27–31).
The evolution of vitiligo can not be predicted: Some
people see patches enlarge or new patches appear. On a rare
occasion, the skin may regain its lost color without treatment
(9,32,33).
Although the treatment options may seem numerous,
they are rarely efficient in the long run. In most cases, vitiligo
recurs over time. In addition, the uniqueness of each organism
causes each person to respond differently and unpredictably to
various treatments (34,35).
Plant extracts have been used for the treatment of
various diseases since ancient times, even in economic turn down
decades (36–39). PN, one of the most widely used spices
in the world, has also been used as medicine for centuries. In
recent years the unique pharmacological actions of these plants
have been explored. Among the phytochemicals in PN, the compound of
interest in this study is piperine, the main alkaloid, responsible
for the pungent taste (40). The
phenolic amides from PN have also shown an antioxidant capacity
superior to synthetic compounds. According to recent studies, PN
can stimulate melanocyte proliferation and bring back color to
depigmented skin (41), therefore
this could be a potential treatment for vitiligo.
A crude extract of PN fruits containing piperine was
shown to be more stimulatory that an equivalent concentration of
the pure compound, suggesting the presence of other active
components (42).
Materials and methods
The PN extract was obtained by Soxhlet extraction,
using chloroform as solvent. The black peppercorns originary from
India were purchased from the online store: www.pcfarm.ro. The powder resulted from grinding the
dried fruits (5 g of powder for each 25 ml of solvent) were
extracted for 24 h, until the complete depletion of the plant
product. The solvent was aftwerwards evaporated under vaccuum.
The CAMAG high performance thin layer cromatography
(HPTLC) system was used for thin layer cromatography analysis,
which helped to evaluate the variety of biological active compounds
found in the extract and to identify the alkaloid of interest,
piperine. The application of the samples on the silica gel plates
(stationary phase) was done automatically, using the automatic
sampler Linomat 5. The plates were developed using the automatic
developing chamber ADC2, in a mobile phase consisting in a mixture
of hexane, ethyl acetate and glacial acetic acid (3:1:0.1)
(43). Finally, using the TLC
Visualizer 2, the plates were evaluated using UV light (254
nm).
Using a standard piperine solution, we identified
the piperine in our extract (by use of the Rf value) and
approximately quantified its concentration.
The antioxidant activity of the extract was
evaluated using the 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical
(44). This method consists in the
inactivation of the DPPH radical by an antioxidant, which is
measured spectrophotometrically, using a microplate reader. The
samples used were prepared with the use of 3 main solutions: S1,
standard piperine [2 mg/ml dimethyl sulfoxide (DMSO)]; S2, PN
extract (5 mg/ml DMSO); and S3, tyrosine (0.25 mg/ml
H2O). The tested samples were: Q1 (0.1 ml of S1); Q2
(0.1 ml of S1 mixed with 0.8 ml of S3); Q3 (0.1 ml of S2); Q4 (0.1
ml of S2 mixed with 0.8 ml of S3); Q5 (0.1 ml of DMSO). We used
ascorbic acid (Q6) as reference for the antioxidant activity. The
influence of tyrosine on the antioxidant activity was also
monitored (Q2, Q4) (Table I), since
it is a precursor of melanin, which means including it in our
ointments could be helpful.
 | Table I.RSC of piperine (Q1), extract (Q3),
DMSO (Q5), ascorbic acid (Q6), and the influence of tyrosine
association (Q2, Q4). |
Table I.
RSC of piperine (Q1), extract (Q3),
DMSO (Q5), ascorbic acid (Q6), and the influence of tyrosine
association (Q2, Q4).
| Item | Q1 | Q2 | Q3 | Q4 | Q5 | Q6 |
|---|
| RSC (%) | 7.14 | 7.87 | 21.24 | 26.19 | 1.46 | 78 |
The ointment base we used consisted of cetyl
alcohol, glycerin, sodium lauryl sulfate and water, to which we
added our active compounds (PN extract and pure piperine) and
preservatives (methyl parahydroxybenzoate and propyl
parahydroxybenzoate). To ensure that our biologically active
compounds reach the melanocytes, we also added an absorption
promoter. For this purpose we chose DMSO, since according to a
recent ex vivo permeability study, it has the best skin
penetration capacity and the least intense side effects (41).
The study was approved by the the Local Ethics
Committee (Comisia de Etica Universitara, no. 3/30 of May 2018;
Galați; Romania), and the in vivo testing took place only
after obtaining the informed consent from the patients involved in
the study.
We were able to skip the animal testing phase since
this was already done in a previous study, with conclusive results
(41). The testing was done in 3
human subjects, all female, aged between 40 and 70 years,
comprising 18 vitiligo plaques, localized mostly on the limbs and
neck. One of these subjects had segmental vitiligo while the other
two had generalized (non-segmental) vitiligo. We tested the
ointments alone and in association with a 40 µg/ml travoprost
solution, since periocular pigmentation was observed during recent
studies in patients treated with prostaglandin analogues for
glaucoma (45–48). We also monitored the influence of the
application technique on the effect by applying an occlusive
dressing on some of the affected skin patches. The ointments were
applied once a day, in the evening while the travoprost solution
was applied once a day, in the morning, to avoid interactions at
the application site. The testing spread over 12 weeks, evaluating
the results every 3 weeks.
Results
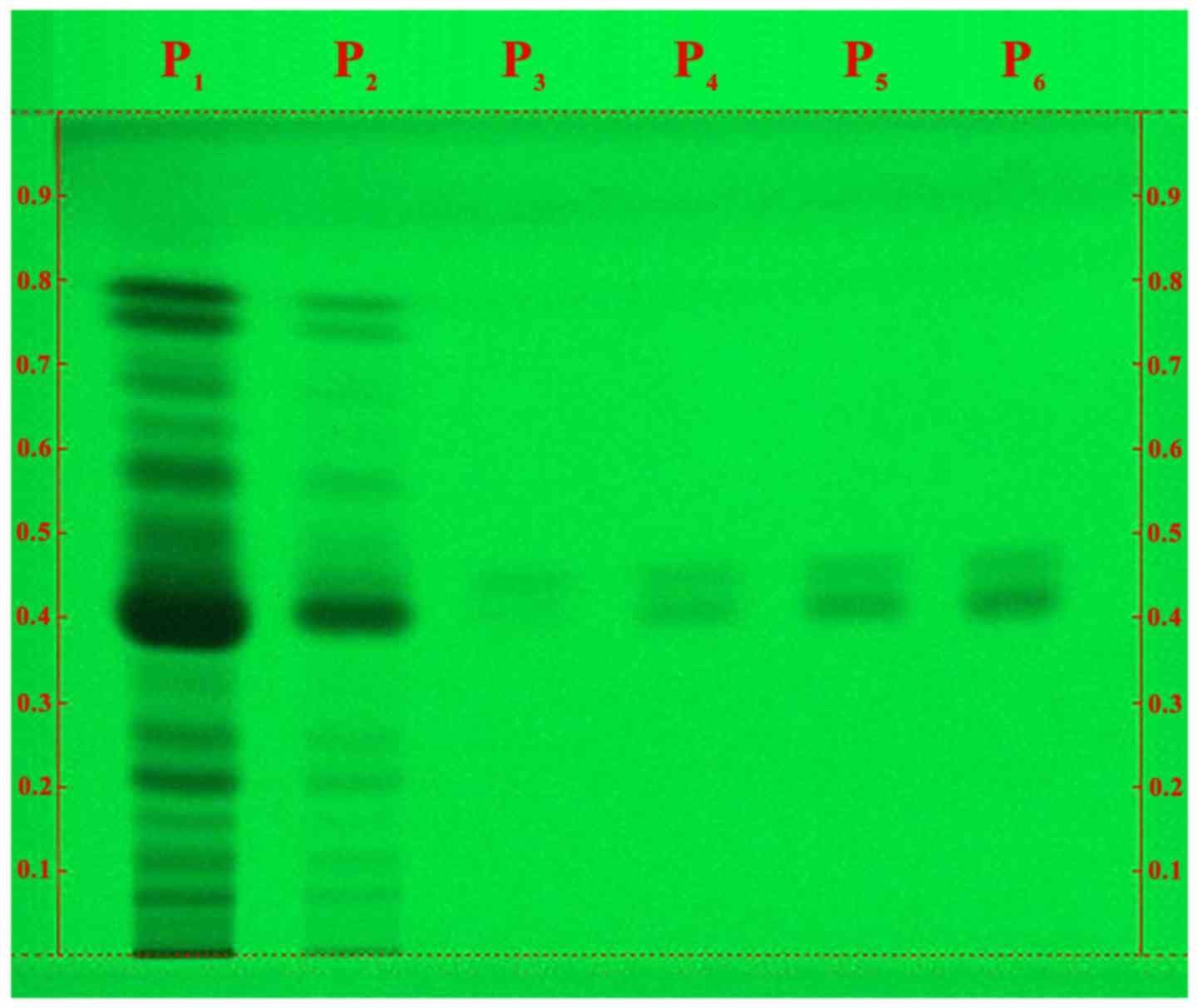
The HPTLC analysis of our extract in the optimized
solvent system is a qualitative and semi-quantitative chemical
testing of PN fruits for the presence of phytoconstituents. The
chromatogram revealed the presence of piperine alkaloid in our PN
extract, in a concentration of ~8%. A wide range of compounds other
than piperine can be observed in the extract (different spots in P1
and P2 samples; Fig. 1), when
compared with the P3-P6 samples, which only contain pure
piperine.
The antioxidant activity results are presented in
Table I. We have analyzed the
radical scavenging capacity of samples containing bioactive
compounds (pure piperine, PN extract and mixtures of these with
tyrosine) in various ratios to evaluate their synergism or
antagonism. By comparing the values in Table I, it can be noticed that the addition
of tyrosine to our samples increased the radical scavenging
capacity (RSC) value, both in the case of the PN extract and in the
case of pure piperine alone. Q4, contaning PN extract and tyrosine
has a greater RSC value than Q3, which contains just the PN
extract. Similar to this, Q2, containing piperine and tyrosine has
a greater RSC value than Q1, which only contains piperine. Dimethyl
sulfoxide (DMSO) doesn't have an important influence on the
antioxidant activity, since DMSO by itself (Q5) doesn't have a
great RSC value. Taking all these into consideration, we can
conclude that the tyrosine acts synergistically with the piperine
and the PN extract.
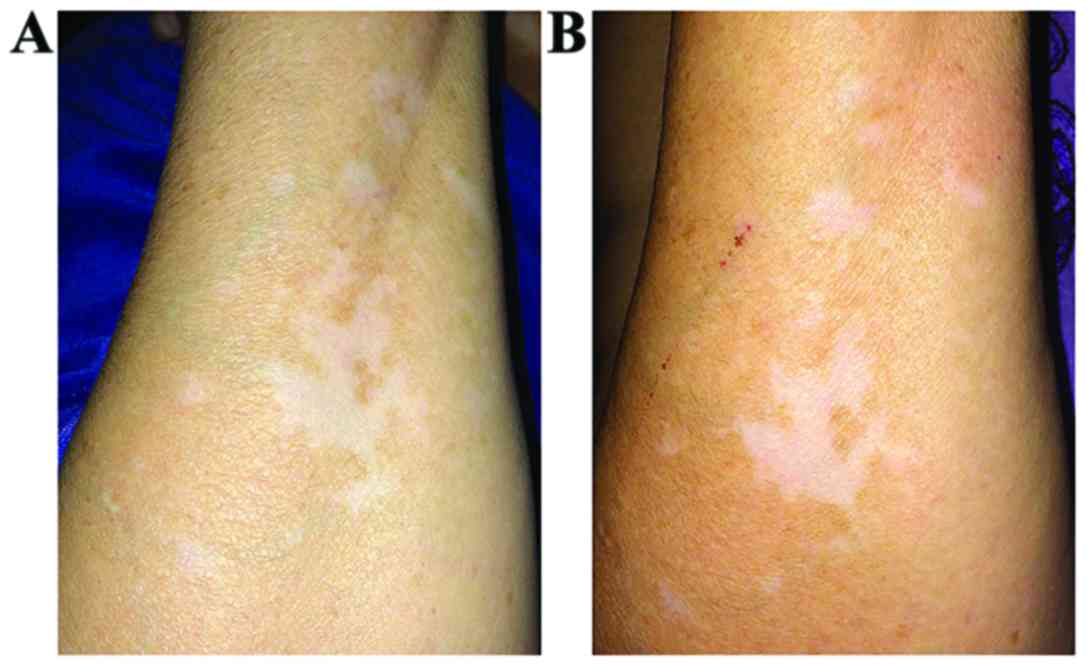
Concerning the in vivo testing results, both
of our ointments led to pigmentation of the affected skin areas.
The pattern and the speed of pigmentation were different, depending
on the substance and method of application. The most illustrative
results are presented in Figs.
2–6.
Discussion
Our study confirms the presence of piperine in PN
and its capacity to stimulate pigmentation in the skin. The HPTLC
results highlight the large variety of compounds that can be found
in PN, which is an important fact considering they could all have
therapeutic properties. Also, two Rf values can be
observed in the chromatograms, suggesting the presence of two
isomers in the pure piperine solution, both also present in our
extract.
The antioxidant capacity is relevant to our study
because of the implications of oxidative stress in the
physiopathology of vitiligo. The RSC of the PN extract was
approximately three times bigger than the one of pure piperine, due
to the other bioactive compounds from the extract. The association
of tyrosine seems to be beneficial, increasing the antioxidant
activity by 10% (when associated with piperine: Q2) to 20% (when
associated with the extract: Q4) (Table
I). Unfortunately, we were not able to include the tyrosine in
our ointments because of the pH incompatibility between tyrosine
(pH 5.0) on the one hand, and our extract (pH 9.0) and piperine (pH
9.5) on the other hand.
The in vivo testing results were
satisfactory, regarding both ointments. The ointment containing the
PN extract led to the fastest results, the pigmentation being
visible to the naked eye after only 3 weeks of application. This
suggests the presence of other biologically active compounds
playing an important role in the pigmentation process, a fact
highlighted by the HPTLC results and consistent with the findings
of previous studies (42).
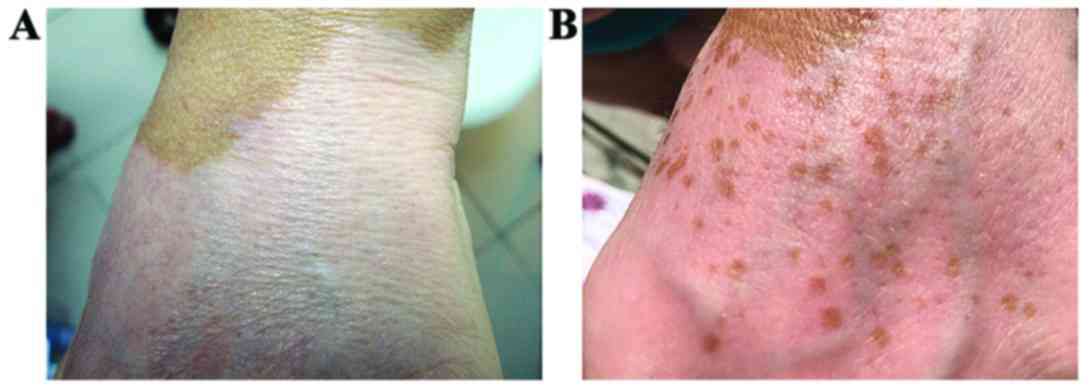
As to the pigmentation pattern, a difference between
the active compounds was noted: The PN extract led to pigmentation
islands while the piperine alone led to a diffuse pigmentation.
The association of the travoprost solution was
beneficial in both cases. Not only did it speed up the process but
it also changed the pigmentation pattern, especially when
associated with the extract. The travoprost solution alone caused a
diffuse pigmentation at first, visible after only 3 weeks, changing
the pattern to pigmentation islands after 9 weeks. When associated
with the extract ointment, both patterns of pigmentation were
observed.
Applying the ointments under an occlusive dressing
did not bring as much benefit as we expected. It led to fast
pigmentation in the first 3 weeks but did not keep up the pace for
the rest of the testing period.
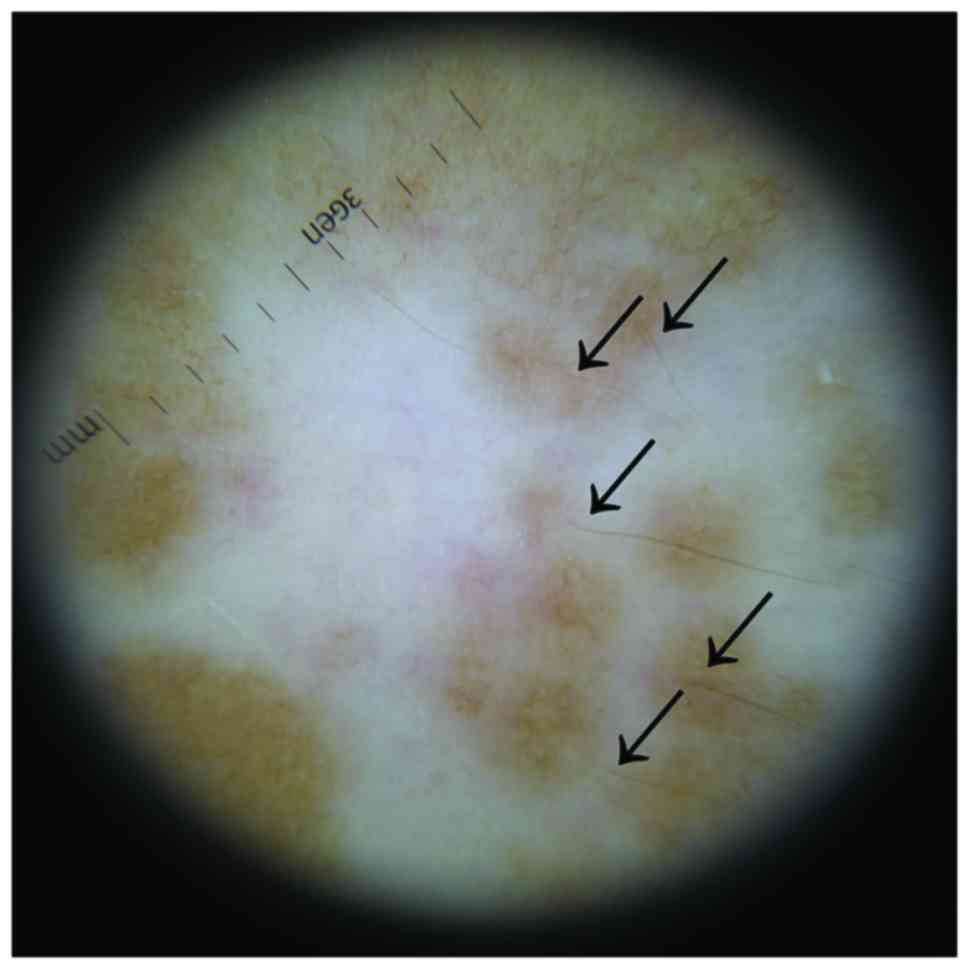
By analyzing the affected areas with a dermatoscope,
we were able to see that the pigmentation islands appeared around
the pigmented hairs (Fig. 4). This
is consistent with the scientific literature affirming that
re-pigmentation tends to occur mainly in those areas of skin where
there are still pigmented hairs, since this suggests the presence
of melanin reservoirs (49).
Regarding the subject with non-segmental vitiligo,
the results were almost the opposite. The difference between the
two ointments was not as noticeable and, moreover, the piperine
alone led to slightly better results than the extract. Also, they
both produced pigmentation islands, thus following the same
pattern. However, this could be the consequence of the patches of
affected skin being larger than the ones of the segmental vitiligo
subject.
The side effects of our ointments were easily
tolerated by our subjects. They described a slight burning
sensation at the first applications, especially when applying the
extract ointment under an occlusive dressing. Yet, one of the
subjects withdrew from the study because of the intense burning
sensation and the local redness and irritation. This might be
caused by the sensitive and hyper-reactive skin type of the
subject.
The present study has limitations due to the small
number of patients and lesions and the absence of controls. In the
future, larger batches of patients should be included. This was
just the start point for further studies and observations on this
novel, simple and cheap possible treatment for vitiligo; these
first observations regarding the synthesis, pharmacologic content
and clinical effects have to be reported. We believe that our study
contains promising results that should be investigated further,
especially taking into consideration the current context, in which
identifying plant compounds that are active in diseases such as
vitiligo is of great importance.
Although the results may not be permanent, PN and
piperine could represent a less aggressive treatment alternative
for vitiligo than the ones that are currently used. However,
further studies are necessary to establish certain details such as
how long does the pigmentation last, what happens once the
application is interrupted or in what way could the skin type or
color influence the results.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
RMD prepared and analyzed the extract. ODB performed
the ointment formulation. BM was involved in all the stages of the
study, prepared the ointments and was a major contributor in
writing the manuscript. ALT examined the test subjects and
evaluated the in vivo effects. All authors contributed to
the conception and design of the study, as well as revising it. All
authors read and approved the final manuscript to be published and
agreed to be accountable for all aspects of the work in ensuring
that questions related to the accuracy or integrity of any part of
the work are appropriately investigated and resolved.
Ethics approval and consent to
participate
The study was approved by the Local Ethics
Commission of Comisia de Etica Universitara (CEU no. 3/30 of May
2018; Galați, Romania), and written informed consent for
participation in the study was obtained from all patients.
Patient consent for publication
Written informed consent for the publication of the
images was provided by all participants.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Gawkrodger DJ, Ormerod AD, Shaw L,
Mauri-Sole I, Whitton ME, Watts MJ, Anstey AV, Ingham J and Young
K: Therapy Guidelines and Audit Subcommittee, British Association
of Dermatologists; Clinical Standards Department, Royal College of
Physicians of London; Cochrane Skin Group; Vitiligo Society:
Guideline for the diagnosis and management of vitiligo. Br J
Dermatol. 159:1051–1076. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Speeckaert R and van Geel N: Vitiligo: An
update on pathophysiology and treatment options. Am J Clin
Dermatol. 18:733–744. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Nicolaidou E, Antoniou C, Stratigos A and
Katsambas AD: Narrowband ultraviolet B phototherapy and 308-nm
excimer laser in the treatment of vitiligo: A review. J Am Acad
Dermatol. 60:470–477. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Whitton ME, Ashcroft DM and González U:
Therapeutic interventions for vitiligo. J Am Acad Dermatol.
59:713–717. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Tatu AL and Ionescu MA: Multiple
autoimmune syndrome type III-thyroiditis, vitiligo and alopecia
areata. Acta Endo Buc. 13:124–125. 2017. View Article : Google Scholar
|
|
6
|
Taïeb A and Picardo M: Clinical practice.
Vitiligo. N Engl J Med. 360:160–169. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kim DY, Oh SH and Hann SK: Classification
of segmental vitiligo on the face: Clues for prognosis. Br J
Dermatol. 164:1004–1009. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
van Geel N, Bosma S, Boone B and
Speeckaert R: Classification of segmental vitiligo on the trunk. Br
J Dermatol. 170:322–327. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Mazereeuw-Hautier J, Bezio S, Mahe E,
Bodemer C, Eschard C, Viseux V, Labreze C, Plantin P, Barbarot S,
Vabres P, et al: Groupe de Recherche Clinique en Dermatologie
Pédiatrique (GRCDP): Segmental and nonsegmental childhood vitiligo
has distinct clinical characteristics: A prospective observational
study. J Am Acad Dermatol. 62:945–949. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ho N, Pope E, Weinstein M, Greenberg S,
Webster C and Krafchik BR: A double-blind, randomized,
placebo-controlled trial of topical tacrolimus 0·1% vs. clobetasol
propionate 0·05% in childhood vitiligo. Br J Dermatol. 165:626–632.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Anbar TS, Westerhof W, Abdel-Rahman AT and
El-Khayyat MA: Evaluation of the effects of NB-UVB in both
segmental and non-segmental vitiligo affecting different body
sites. Photodermatol Photoimmunol Photomed. 22:157–163. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Cavalié M, Ezzedine K, Fontas E, Montaudié
H, Castela E, Bahadoran P, Taïeb A, Lacour JP and Passeron T:
Maintenance therapy of adult vitiligo with 0.1% tacrolimus
ointment: A randomized, double blind, placebo-controlled study. J
Invest Dermatol. 135:970–974. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Garza-Mayers AC and Kroshinsky D: Low-dose
methotrexate for vitiligo. J Drugs Dermatol. 16:705–706.
2017.PubMed/NCBI
|
|
14
|
Radakovic-Fijan S, Fürnsinn-Friedl AM,
Hönigsmann H and Tanew A: Oral dexamethasone pulse treatment for
vitiligo. J Am Acad Dermatol. 44:814–817. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Tatu AL, Ionescu MA, Clatici VG and
Cristea VC: Bacillus cereus strain isolated from Demodex
folliculorum in patients with topical steroid-induced
rosaceiform facial dermatitis. An Bras Dermatol. 91:676–678. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Tatu AL: Topical steroid induced facial
rosaceiform dermatitis. Acta Endo Buc. 12:232–233. 2016. View Article : Google Scholar
|
|
17
|
Kubiak K, Sielawa H, Chen W and Dzika E:
Endosymbiosis and its significance in dermatology. J Eur Acad
Dermatol Venereol. 32:347–354. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Tatu AL, Ionescu MA and Nwabudike LC:
Contact allergy to topical mometasone furoate confirmed by
rechallenge and patch test. Am J Ther. 25:e497–e498.
2018.PubMed/NCBI
|
|
19
|
Tatu AL, Clatici V and Cristea VC:
Isolation of Bacillus simplex strain from Demodex
folliculorum and observations about Demodicosis
spinulosa. Clin Exp Dermatol. 41:818–820. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Tatu AL, Ionescu MA and Cristea VC:
Demodex folliculorum associated Bacillus pumilus in
lesional areas in rosacea. Indian J Dermatol Venereol Leprol.
83:610–611. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Gambichler T, Rüddel I, Hessam S, Bechara
FG, Stockfleth E and Schmitz L: Altered epigenetic pathways and
cell cycle dysregulation in healthy appearing skin of patients with
koebnerized squamous cell carcinomas following skin surgery. J Eur
Acad Dermatol Venereol. Feb 25–2018.(Epub ahead of print). doi:
10.1111/jdv.14887. View Article : Google Scholar
|
|
22
|
Tatu AL and Nwabudike LC: Rosacea-like
demodicosis (but not primary demodicosis) and papulo pustular
rosacea may be two phenotypes of the same disease - a microbioma,
therapeutic and diagnostic tools perspective. J Eur Acad Dermatol
Venereol. Jun 29–2018.(Epub ahead of print). doi:
10.1111/jdv.15166. View Article : Google Scholar
|
|
23
|
Tatu AL: Nasal spinulosis. J Cutan Med
Surg. 21:3392017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Tatu AL and Cristea VC: Pityriasis
folliculorum of the back thoracic area: Pityrosporum, keratin
plugs, or demodex involved? J Cutan Med Surg. 21:4412017.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Tatu AL and Cristea VC: Unilateral
blepharitis with fine follicular scaling. J Cutan Med Surg.
21:4422017. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Tatu AL, Nwabudike LC; Reply to Happle R.,
; et al: Koebners sheep in Wolfs clothing: does the isotopic
response exists as a distinct phenomenon? J Eur Acad Dermatol
Venereol. Feb 28–2018.(Epub ahead of print). doi:
10.1111/jdv.14900.
|
|
27
|
Negrei C, Căruntu C, Ginghină O,
Burcea-Dragomiroiu GT, Toderescu CD and Boda D: Qualitative and
quantitative determination of methotrexate polyglutamates in
erythrocytes by high performance liquid chromatography. Rev Chim.
66:607–610. 2015.
|
|
28
|
Negrei C, Ginghină O, Căruntu C,
Burcea-Dragomiroiu GT, Jinescu G and Boda D: Investigation
relevance of methotrexate polyglutamates in biological systems by
high performance liquid chromatography. Rev Chim. 66:766–768.
2015.
|
|
29
|
Batani A, Brănișteanu DE, Ilie MA, Boda D,
Ianosi S, Ianosi G and Caruntu C: Assessment of dermal papillary
and microvascular parameters in psoriasis vulgaris using in vivo
reflectance confocal microscopy. Exp Ther Med. 15:1241–1246.
2018.PubMed/NCBI
|
|
30
|
Ion R and Boda D: Porphyrin-based
supramolecular nanotubes generated by aggregation processes. Rev
Chim. 59:205–207. 2008.
|
|
31
|
Boda D and Ion RM: Synthesis, spectral and
photodynamic properties of lithium phthalocyanine. Rev De Chim.
65:1271–1274. 2014.
|
|
32
|
Wolff K, Goldsmith LA, Katz SI, Gilchrest
BA, Paller AS and Leffell DJ: Fitzpatricks Dermatology in General
Medicine. 7th. McGraw Hill Medical; USA: pp. 616–621. 2008
|
|
33
|
Ortonne JP: Vitiligo and other disorders
of hypopigmentation. Dermatology. Bolognia JL, Jorizzo JL and
Rapini RP: 2nd. Mosby Elsevier; Simi Valley, CA, USA: pp. 913–920.
2008
|
|
34
|
Tatu AL and Nwabudike LC:
Metoprolol-associated onset of psoriatic arthropathy. Am J Ther.
24:e370–e371. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Tatu AL and Nwabudike LC: Bullous
reactions associated with COX-2 inhibitors. Am J Ther.
24:e477–e480. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Buzia OD, Fasie V, Mardare N, Diaconu C,
Gurau G and Tatu AL: Formulation, preparation, physico-chimical
analysis, microbiological peculiarities and therapeutic challenges
of extractive solution of Kombucha. Rev Chim Buchar. 69:720–724.
2018.
|
|
37
|
Zălaru C, Crişan C, Călinescu I, Moldovan
Z, Ţârcomnicu I, Litescu S, Tatia R, Moldovan L, Boda D and Iovu M:
Polyphenols in Coreopsis tinctoria Nutt. fruits and the
plant extracts antioxidant capacity evaluation. Open Chem.
12:858–867. 2014.
|
|
38
|
Ionescu C, Țârcomnicu I, Ionescu MA,
Nicolescu TO, Boda D and Nicolescu F: Identification and
characterization of the methanolic extract of hellebrigenin
3-acetate from hellebori rhizomes. Mass spectrometry. Rev Chim.
1:972–975. 2014.
|
|
39
|
Raţiu MP, Purcărea I, Popa F, Purcărea VL,
Purcărea TV, Lupuleasa D and Boda D: Escaping the economic turn
down through performing employees, creative leaders and growth
driver capabilities in the Romanian pharmaceutical industry.
Farmacia. 59:119–130. 2011.
|
|
40
|
Meghwal M and Goswami TK: Chemical
composition, nutritional, medicinal and functional properties of
black pepper: A review. Open Access Sci Rep. 1:1722012.
|
|
41
|
Faas L, Venkatasamy R, Hider RC, Young AR
and Soumyanath A: In vivo evaluation of piperine and synthetic
analogues as potential treatments for vitiligo using a sparsely
pigmented mouse model. Br J Dermatol. 158:941–950. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Lin Z, Liao Y, Venkatasamy R, Hider RC and
Soumyanath A: Amides from Piper nigrum L. with dissimilar
effects on melanocyte proliferation in-vitro. J Pharm Pharmacol.
59:529–536. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Hamrapurkar PD, Jadhav Kavita and Sandip
Zine: Quantitative estimation of piperine in Piper nigrum Piper
longum using high performance thin layer chromatography. J Appl
Pharm Sci. 1:117–120. 2011.
|
|
44
|
Bozin B, Mimica-Dukic N, Samojlik I, Goran
A and Igic R: Phenolics as antioxidants in garlic (Allium
sativum L., Alliaceae). Food Chem. 111:925–929. 2008.
View Article : Google Scholar
|
|
45
|
Choi YM, Diehl J and Levins PC: Promising
alternative clinical uses of prostaglandin F2α analogs: Beyond the
eyelashes. J Am Acad Dermatol. 72:712–716. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Kapur R, Osmanovic S, Toyran S and Edward
DP: Bimatoprost-induced periocular skin hyperpigmentation:
Histopathological study. Arch Ophthalmol. 123:1541–1546. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Feletti F, Vincenzi C, Pazzaglia M and
Tosti A: Periocular pigmentation associated with use of travoprost
for the treatment of alopecia areata of the eyelashes. J Eur Acad
Dermatol Venereol. 21:421–423. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Anbar TS, El-Ammawi TS, Abdel-Rahman AT
and Hanna MR: The effect of latanoprost on vitiligo: a preliminary
comparative study. Int J Dermatol. 54:587–593. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Menon SM, Sharma YK, Bansal P and
Ghadgepatil SS: Restoration of pigmentation by follicular unit
extraction transplant in three cases of focal vitiligo recalcitrant
to therapy including with previous nonculture
melanocyte-keratinocyte transplant. Int J Trichology. 8:87–88.
2016. View Article : Google Scholar : PubMed/NCBI
|