Introduction
Cerebral white matter injury (WMI) is a recognized
problem in premature newborns, particularly those with a very low
birth weight (VLBW) (1). In 2010,
the number of infants with a VLBW is likely to exceed several
million worldwide (2). Between
1990 and 1998, >85% of VLBW infants survived in the USA
(3,4). However, their likelihood of
long-term neurodevelopmental disability is increased compared with
newborns that have an average birth weight (4). Common disabilities exhibited by
newborns with a VLBW include spastic motor deficits (5–10% of
survivors) (5) and cognitive
socialization defects (25–50% of survivors) (6). These conditions can significantly
impair the patient's quality of life. Therefore, it is of great
clinical significance to study the treatment and potential
therapeutic targets of WMI in premature infants.
Oligodendrocyte precursor cells (OPCs) are
selectively vulnerable to WMI in premature infants (7). The targeted death of OPCs is
responsible for acute WMI mediated by lipid peroxidation in preterm
infants (8). The pathological
mechanisms underlying the death of OPCs are currently unknown;
however, specific events have been identified, including upstream
hypoxia and inflammation, and downstream microglia activation and
cytotoxicity of excitatory amino acids (9). A previous study revealed that
surviving OPCs often failed to differentiate, leading to a loss of
cellular functions due a decreasing production of compact myelin
(10). Myelination is a key
process during the maturation of OPCs; it supports neuronal growth,
is involved in neural transduction, and serves an important role in
recognition and motor function (11). Therefore, the maturation of OPCs
is the basis for normal myelination, while impaired migration and
differentiation of OPCs hinders myelination.
A previous study has demonstrated that extracellular
matrix (ECM) components, particularly chondroitin sulfate
proteoglycans (CSPGs), are associated with the regeneration and
plasticity of the central nervous system; removal of CSPGs was
revealed to facilitate axonal regeneration and functional recovery
(12). CSPGs are a mechanical
barrier that inhibit the migration, differentiation and recovery of
neural cells, and a chemical barrier that secretes inhibitors to
prevent the outgrowth of neuronal axons (13). Siebert and Osterhout (14) demonstrated that CSPGs inhibit OPC
process outgrowth and differentiation, thus indicating a direct
association between CSPGs and the differentiation of OPCs. However,
the mechanism by which this occurs remains unknown, and the effect
of CSPGs on the proliferation and migration of OPCs is unclear. The
potential interactions between CSPGs and other components of the
ECM, including laminin have not yet been identified.
In the present study, the role of CSPGs in the
proliferation, migration and differentiation of OPCs was
investigated. The interactive association between CSPGs and laminin
in the maturation and differentiation of OPCs was also examined.
The inhibitory effect of CSPGs on OPC differentiation was
identified to be associated with β1-integrin. The present study
provides novel insights into the underlying mechanisms of
myelination impairment in brain injuries in premature infants. This
may provide a basis to identify novel therapeutic targets.
Materials and methods
OPC culture
A total of 12 male postnatal day 1 Sprague Dawley
rats (mean weight, 10±2 g) were obtained from the Experimental
Animal Center of Sun Yat-sen University (Guangzhou, China). The
rats were housed in an environment with a standard 12 h light/dark
cycle and ad libitum access to food and water. On postnatal
day 2, the rats were sacrificed to obtain OPCs, which were cultured
as previously described (15).
The cerebral cortexes from the rats were cut into sections of ~1
mm3 in size, and then digested in minimal essential
media with 1.5 mg/ml papain and 60 µg/ml DNase I (both from
Gibco; Thermo Fisher Scientific, Inc., Waltham, MA, USA). Following
centrifugation at 300 × g for 5 min at 4°C, the cell suspension was
seeded in poly-L-lysine coated T75 culture flasks and cultured in
Dulbecco's modified Eagle's medium (DMEM) supplemented with 20%
fetal bovine serum (both from Gibco; Thermo Fisher Scientific,
Inc.). After 10 days, the flasks were placed in an agitator at 37°C
for 1.5 h to remove microglia. Following this, an additional
agitation was performed for 16 h at 37°C to detach OPCs, which were
subsequently seeded onto poly-D-lysine-coated plates and cultured
in OPC proliferation medium [OPCM; DMEM/F12 (Gibco; Thermo Fisher
Scientific, Inc.) supplemented with 2% B27 (Invitrogen; Thermo
Fisher Scientific, Inc.), 1% penicillin-streptomycin (Gibco; Thermo
Fisher Scientific, Inc.), 20 ng/ml platelet-derived growth factor
(PDGF)-AA and 20 ng/ml basic fibroblast growth factor (bFGF) (both
from Invitrogen; Thermo Fisher Scientific, Inc.)]. The flasks were
maintained at 37°C and half of the medium was changed every other
day. The present study was approved by the Ethics Committee of the
Children's Hospital of Fudan University (Shanghai, China).
OPC differentiation
To induce differentiation into mature
oligodendrocytes, the OPCs were seeded into a 24-well plate at a
concentration of 5×104 cells/ml per well. After 24 h the
cells were cultured in oligodendrocyte differentiation medium [OLM;
DMEM/F12 supplemented with 2% B27 (Invitrogen; Thermo Fisher
Scientific, Inc.), Triiodo-L-thyronine (T3; 40 ng/ml) and
L-Thyroxine (T4; 30 ng/ml) (both from Sigma-Aldrich; Merck KGaA,
Darmstadt, Germany)] for 5 days with the medium changed every
day.
Immunocytochemistry
The OPCs were fixed with 4% paraformaldehyde for 15
min at room temperature, and subsequently blocked in 2% bovine
serum albumin (BSA; Sigma-Aldrich; Merck KGaA) at 37°C for 2 h and
3% normal goat serum (Beyotime Institute of Biotechnology, Haimen,
China) for 1 h. The cells were then incubated with the following
primary antibodies overnight at 4°C: Rabbit polyclonal anti-PDGF
receptor (PDGFR)α (1:40; cat. no. SC-338; Santa Cruz Biotechnology,
Inc., Dallas, TX, USA), rabbit polyclonal anti-oligodendrocyte
transcription factor (Olig2; 1:200; cat. no. Ab81093; Abcam,
Cambridge, UK), mouse monoclonal anti-O4 (1:40; cat. no. O7139;
Sigma-Aldrich; Merck KGaA), mouse monoclonal anti-O1 (1:500; cat.
no. MAB1327; R&D Systems, Inc., Minneapolis, MN, USA) and
rabbit anti-myelin basic protein (MBP; 1:50; cat. no. Ab53294;
Abcam). After incubation with the primary antibodies, the cells
were washed twice with 0.1% Triton X-100 in phosphate-buffered
saline (PBS) for 10 min. Following this, the cells were incubated
with donkey anti-mouse IgG Alexa Fluor® 488 (cat. no.
A21202) or donkey anti-mouse IgG Alexa Fluor® 555 (cat.
no. A31572) secondary antibodies (both 1:200; Molecular Probes;
Thermo Fisher Scientific, Inc.) for 1 h at room temperature. The
nuclei were counterstained with 4′,6-diamidino-2-phenylindole
(DAPI) (1:1,000; cat. no. D1306; Invitrogen; Thermo Fisher
Scientific, Inc.) at room temperature for 30 min. The fluorescence
images were captured using a fluorescent microscope (magnification,
×200) (Olympus Corp., Tokyo, Japan).
Coverslip coating
Coverslips were coated overnight in poly-L-ornithine
(20 µg/ml; Sigma-Aldrich) and rinsed with sterile deionized
H2O the following day. The coverslips were subsequently
coated overnight in three different concentrations (1, 5 and 10
µg/ml) of laminin (cat. no. L2020; Sigma-Aldrich; Merck
KGaA), with CSPGs (cat. no. CC117; EMD Millipore, Billerica, MA,
USA) at varying concentrations (1, 2.5, 5 and 10 µg/ml) or
without CSPGs.
MTS assay
Cell proliferation was evaluated by an MTS assay
using the CellTiter96 Aqueous Cell Proliferation kit (Promega
Corp., Madison, WI, USA), which is a colorimetric method for
determining the number of viable cells. This solution contains a
tetrazolium compound, MTS, and an electron coupling reagent,
phenazine ethosulfate. Following precoating with poly-L-ornithine,
the coverslips were coated with laminin (10 µg/ml) in the
control group, and laminin and CSPGs (5 µg/ml) in the CSPGs
group. Following proliferation, the OPCM was removed at five
different time points (24, 48, 72, 96 and 120 h) and 20 µl
MTS reagent with 100 μl DMEM was added to each well. Cells were
incubated with MTS for 4 h at 37°C. The plates were placed in a
96-well microplate reader (FLUOstar Omega; BMG Labtech GmbH,
Ortenberg, Germany) and the absorbance was recorded at 490 nm.
EdU analysis
Cell proliferation was also measured by EdU analysis
using a Click-iT EdU Detection kit (Thermo Fisher Scientific, Inc.)
at 48 h following proliferation culture. Coverslips were coated
with laminin (10 µg/ml) in the control group, and with
laminin and low, medium and high (1, 5 and 10 µg/ml,
respectively) doses of CSPGs in the CSPGs groups. Briefly, the
cells were incubated with EdU for 16 h, and subsequently fixed with
4% paraformaldehyde at room temperature for 15 min. Following
fixation, the cells were washed with PBS and 3% BSA, followed by
incubation in PBS with 0.3% Triton X-100. The Click-iT mix solution
was added to the wells and the cells were kept in the dark for 30
min. Following washing with 0.1% Triton X-100 in PBS twice for 5
min at room temperature, the nuclei were counterstained with DAPI
at room temperature for 5 min. The images were captured using a
fluorescent microscope (magnification, ×200).
Transwell assay
The migration of the OPCs was measured using a
Transwell assay. The Transwell polycarbonate membrane inserts
(Corning Inc., Corning, NY, USA) were put into a 24-well plate.
Firstly, the effect of CSPGs on OPC migration as a mechanical
barrier was investigated. The polycarbonate membrane was coated in
either 10 µg/ml laminin or laminin with 1, 5 or 10
µg/ml CSPGs. The cells were seeded into the upper chamber of
the Transwell plate (8×104 cells/well) and cultured with
200 µl OPCM/well without bFGF and PDGF-AA, whereas the
bottom chamber contained 600 µl OPCM/well with 20 ng/ml bFGF
and 20 ng/ml PDGF-AA to induce cell migration. Cells were allowed
to migrate for 4 h, and subsequently stained with 100 ng/ml DAPI at
room temperature for 5 min following fixation with 70% ethanol for
30 min. Non-migrated cells were removed with a cotton swab from the
upper chamber of the Transwell plate. The fluorescence of cells in
the bottom of the Transwell plate was captured using a fluorescent
microscope (magnification, ×200).
To investigate whether CSPGs functioned as a
chemical barrier to inhibit the migration of OPCs, CSPGs (5
µg/ml) and laminin (10 µg/ml) were coated onto the
polycarbonate membranes of a Transwell plate (group 1) or added
into culture medium in the bottom chamber of a Transwell plate
(group 2). In the control group the membrane was only coated with
laminin and there were no CSPGs in the culture medium. The
migration ratio of the CSPGs group was based on the percentage of
control group migration, which was set as 100%.
Morphological assay
Oligodendrocytes that were immunostained with mouse
monoclonal anti-O1 antibodies were divided into four types on the
basis of their morphology: Type I, primary processes shorter than
one cell body in length; type II, primary processes longer than one
cell body in length; type III, multiple processes with secondary
branching; and type IV, complex branching (tertiary or more) and
membranous processes. The cells were counted using ImageJ software
(version 1.48; National Institutes of Health, Bethesda, MD,
USA).
Western blot analysis
For protein extraction, the cells were lysed in cell
lysis buffer (140 mM NaCl, 10 mM Tris-HCl, 1% Triton X-100, 1 mM
ECTA and 1X protease inhibitor cocktail). The protein concentration
was determined using the Bradford reagent method. A total of 25
µg protein was loaded into each lane of a 10% sodium dodecyl
sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) gel, and
subsequently blotted onto a polyvinylidene difluoride membrane.
Following blocking with PBS with Tween-20 containing 5% nonfat dry
milk at room temperature for 1 h, the membrane was incubated with
primary antibodies directed against β1-integrin (1:200; cat. no.
AB1952; EMD Millipore) and glyceraldehyde 3-phosphate dehydrogenase
(GAPDH) (1:5,000; cat. no. 2118; Cell Signaling Technology, Inc.,
Danvers, MA, USA) overnight at 4°C. The membrane was then incubated
with peroxidase-linked immunoglobulin G secondary antibodies
(Thermo Fisher Scientific, Inc.). The proteins were visualized
using an Enhanced Chemiluminescence Western Blotting Detection kit
(GE Healthcare, Chicago, IL, USA) according to the manufacturer's
protocol. The western blot analysis results were quantified using
ImageJ software (the National Institutes of Health).
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR) analysis
RT-qPCR was performed to detect the expression of
β1-integrin mRNA. Total RNA extraction was performed using a TRIzol
reagent (Thermo Fisher Scientific, Inc.) according to the
manufacturer's protocol. A total of 2 µg of total RNA
extracted from the OPCs was subjected to RT. Synthesis of cDNA was
performed using the One-Step RT-PCR kit (Takara Bio, Inc., Otsu,
Japan) according to the manufacturer's protocol. SYBR-Green (Toyobo
Life Science, Osaka, Japan) RT-qPCR amplification and real-time
fluorescence detection were performed using an ABI 7300 Real-Time
PCR system (Thermo Fisher Scientific, Inc.) according to the
manufacturer's protocol. The thermocycling conditions were as
follows: 95°C for 30 sec; 40 cycles of 95°C for 15 sec, 60°C for 30
sec and 72°C for 45 sec; and 72°C for 10 min. Relative gene
expression was calculated using the 2−ΔΔCq method
(16). The primers used for
β1-integrin were as follows: Forward, 5′-GAGAACCACAGAAGTTTACA-3′
and reverse, 5′-CTGAAGTAATCCTCCTCATTTCA-3′. The relative expression
levels were normalized to expression of endogenous GAPDH. The
primers used for GAPDH were: Forward, 5′-GCAAGAGAGAGGCCCTCAG-3′ and
reverse, 5′- TGTGAGGGAGATGCTCAGTG-3′.
Statistical analysis
Raw data were analyzed using SPSS version 20.0
software (IBM Corp., Armonk, NY, USA) and presented as the mean ±
standard deviation. The statistical significance of differences
between two groups was determined by one-way analysis of variance
followed by a post hoc Fisher's least significant difference test
was used for the comparisons of multiple groups. The interactive
role between laminin and CSPGs was determined by factor analysis.
P<0.05 was considered to indicate a statistically significant
difference.
Results
Characterization of OPCs
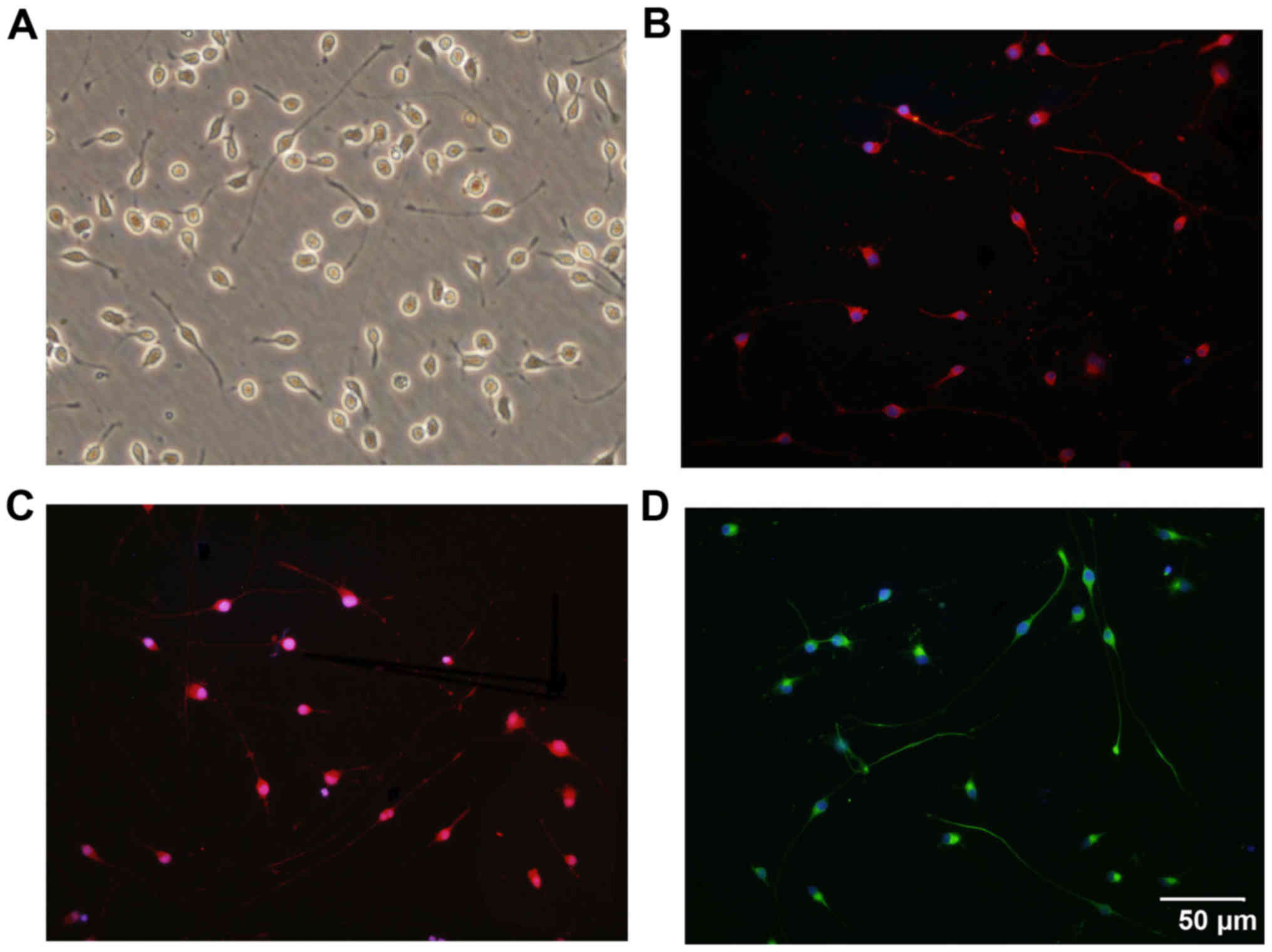
Following isolation and purification, OPCs were oval
in shape and demonstrated a typical bipolar morphology (Fig. 1A). Similar morphology was observed
through immunostaining for OPC biomarkers, including PDGFRα
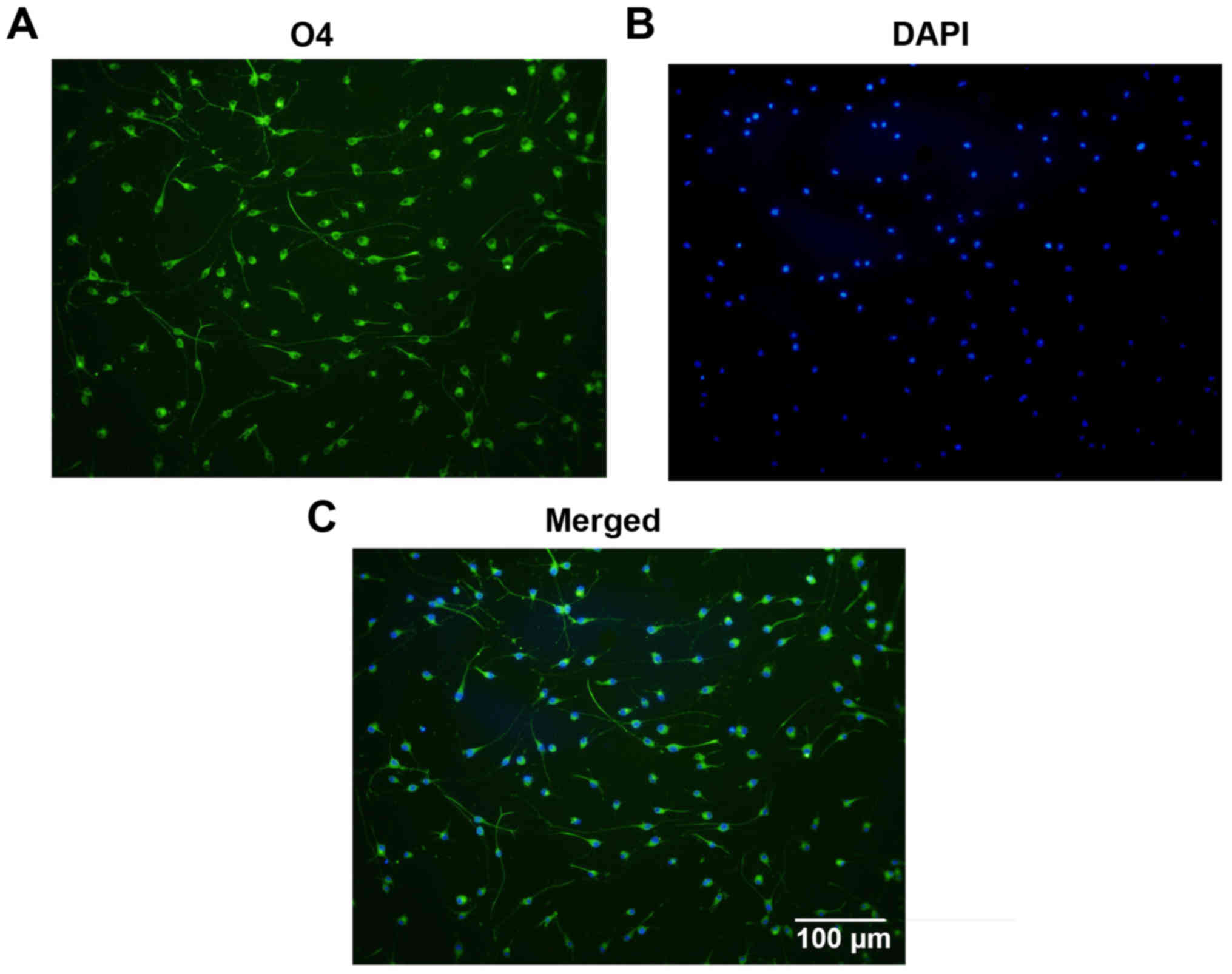
(Fig. 1B), Olig2 (Fig. 1C) and O4 (Fig. 1D). Statistically, 97.3±1.22% of
the cells were immunoreactive to O4, which indicates the high
purity of the cultured OPCs (Fig.
2). Subsequently, the differentiation capacity of the OPCs was
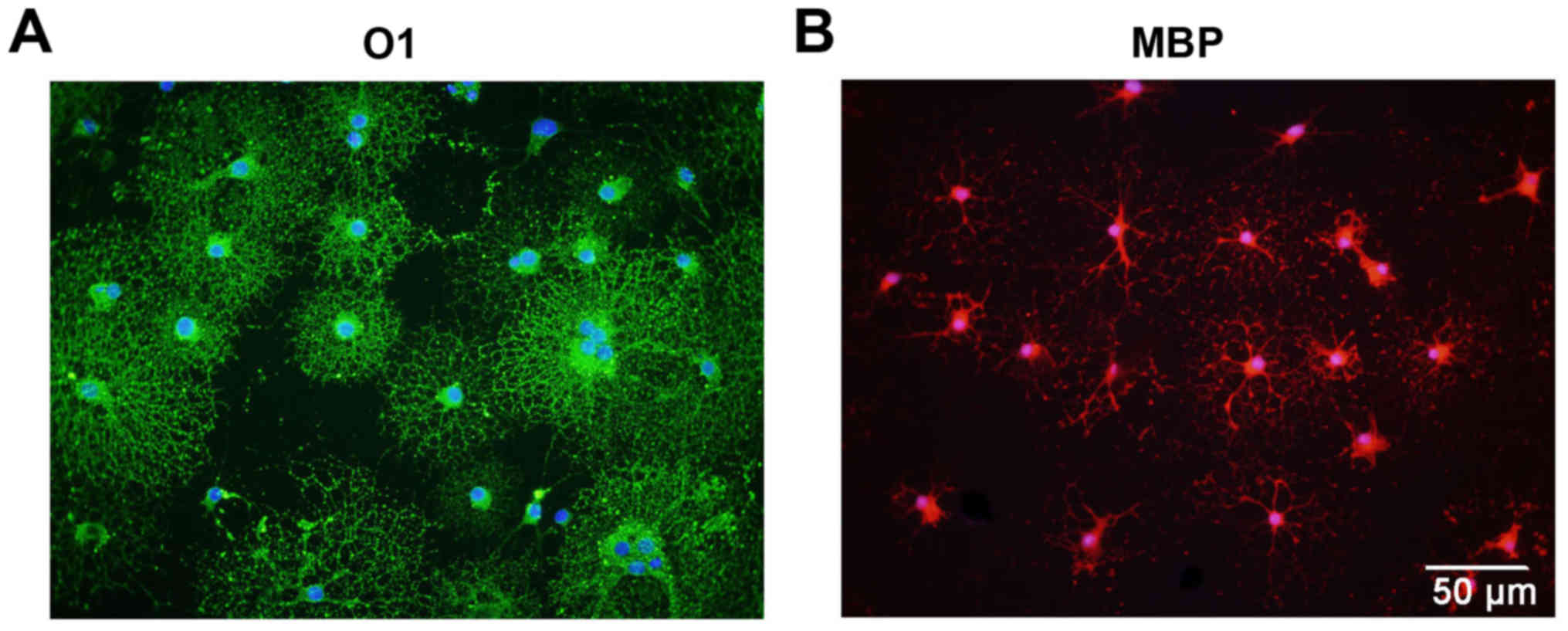
determined. Following treatment with OLM, the OPCs differentiated
into mature oligodendrocytes with a spider web-like morphology
(Fig. 3). Immunocytochemistry
data demonstrated that these differentiated cells expressed the
mature oligodendrocyte markers O1 (Fig. 3A) and MBP (Fig. 3B). Collectively, these results
indicate that cultured OPCs possess the capacity to generate mature
oligodendrocytes.
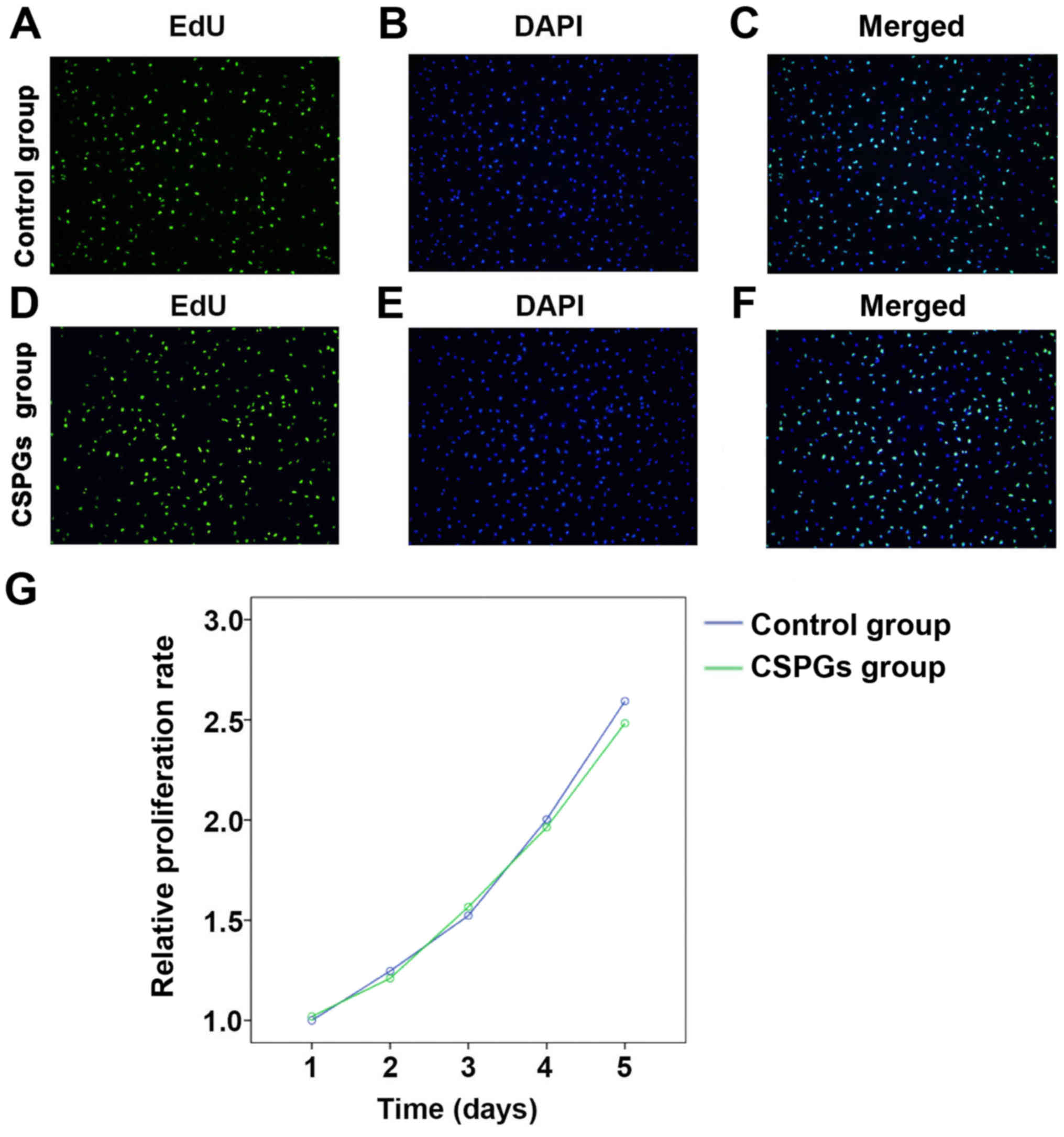
CSPGs do not affect the proliferation of
OPCs
To determine whether the presence of CSPGs affected
the proliferation of OPCs, an EdU analysis and MTS assay were
performed (Fig. 4). No
significant differences (data not shown) were identified between
the control group (Fig. 4A–C) and
the CSPGs group (Fig. 4D–F) using
EdU analysis. Statistical analysis demonstrated that the ratio of
proliferative cells was 68.9±5.59% in the control group, whereas
the ratios in the low, medium and high dose groups of CSPGs were
63.8±4.88, 60.7±10.41 and 62.4±7.24%, respectively (data not
shown). Similar results were obtained with the MTS assay (Fig. 4G). No significant difference was
identified between the control and the CSPG group at five different
time points following the treatment of OPCs with CSPGs. These
results suggest that CSPG treatment does not alter the
proliferation pattern of cultured OPCs in vitro.
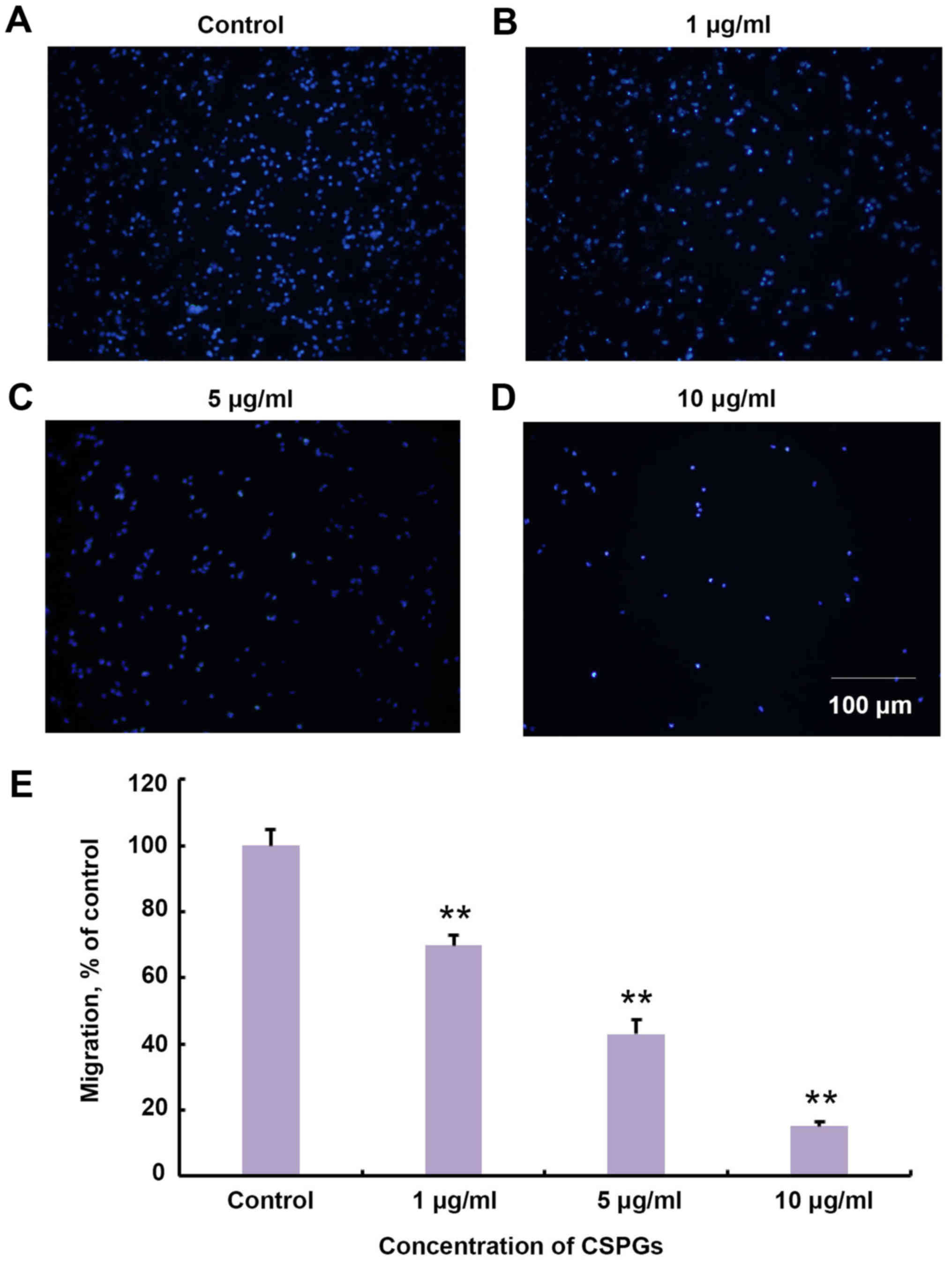
CSPGs significantly reduce the migration
of OPCs
It was investigated whether treatment with CSPGs
changed the migration of OPCs in vitro. The Transwell assay
revealed that the migration of OPCs was impaired in a
dose-dependent manner with increasing concentrations of CSPGs
(Fig. 5). The migration ratio was
significantly decreased compared with the control group at all
concentrations of CSPGs investigated (P<0.01). The migration
ratio was 68.9±3.1% in the presence of 1 µg/ml CSPGs,
43.0±4.3% with 5 µg/ml CSPGs (P<0.01 vs. the control
group), and 15.0±1.5% with 10 µg/ml CSPGs (P<0.01 vs. the
control group).
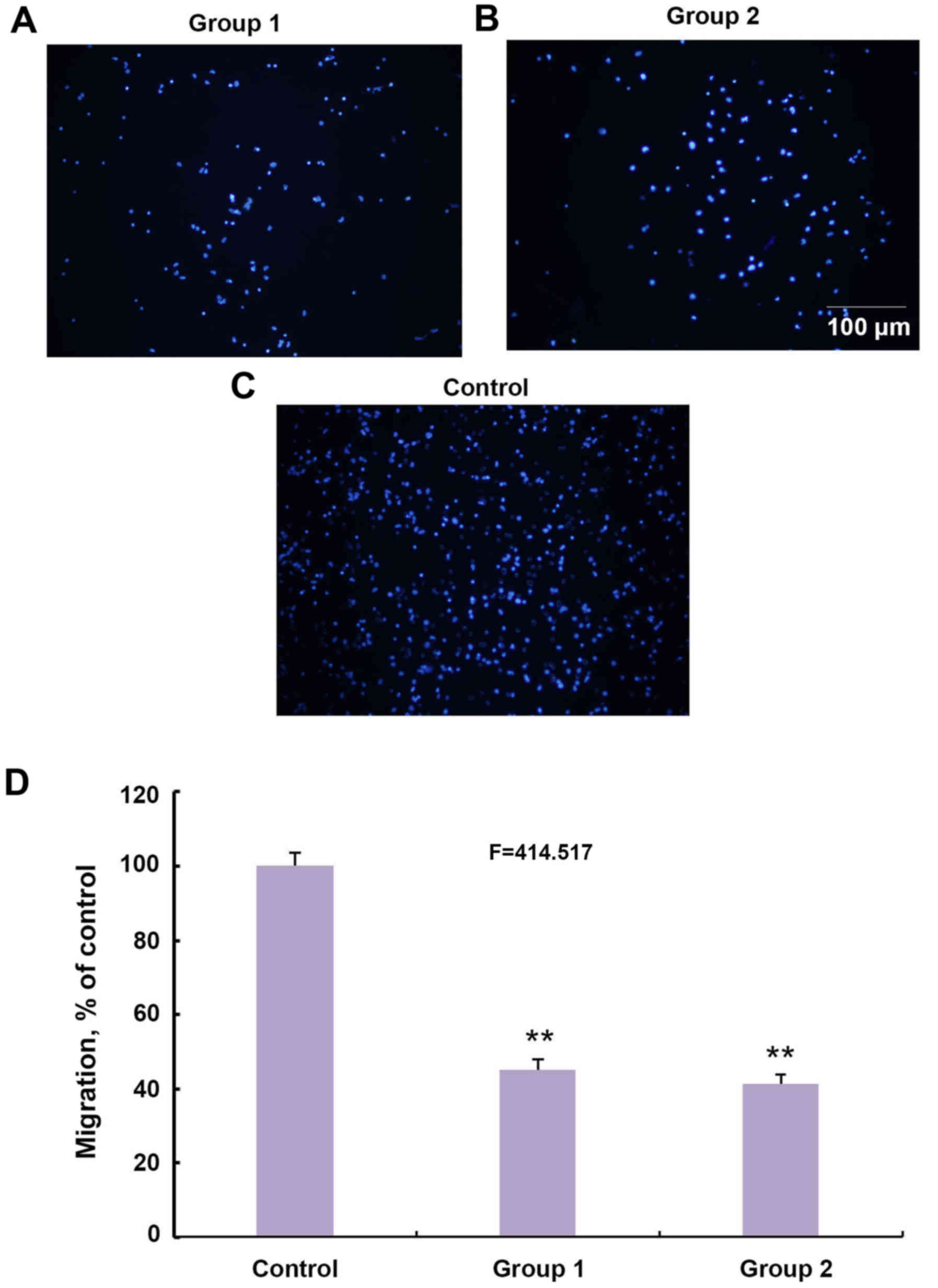
It was subsequently investigated whether a different
pattern of CSPGs would affect the migration of OPCs. CSPGs were
added to the membrane of the Transwell plate in group 1 (Fig. 6A) and in the culture medium of the
bottom chamber in group 2 (Fig.
6B). The number of migrated cells in the two groups was
significantly lower than the control group (Fig. 6C and D). The ratio of migrated
cells in groups 1 and 2 decreased to 45.1±2.8 and 41.3±2.6% of the
control group, respectively (F=414.517; P<0.01) (Fig. 6D). Collectively, these results
indicate that CSPGs possess the capacity to inhibit the migration
of OPCs as either a mechanical or chemical barrier.
CSPGs inhibit the maturation of OPCs
It was investigated whether CSPGs alter the
maturation of OPCs. It was observed that 6 h following the
culturing of OPCs in OPCM, untreated cells (Fig. 7A) had longer processes compared
with the CSPG-treated cells (Fig.
7B). This inhibitory effect was ameliorated at 24 h following
culture in OPCM (data not shown). To induce the differentiation of
OPCs, OPCM was replaced with OLM. It was revealed that untreated
cells expressed O1 (Fig. 7C) and
MBP (Fig. 7D), and exhibited the
morphology of a complex network, indicating the complete maturation
of the oligodendrocytes. By contrast, the CSPG-treated cells had
shorter, sparse processes and more suspended cells were observed
(Fig. 7E–H). However, the
expression of MBP (Fig. 7E) and
O1 (Fig. 7G) was still observed
in the CSPG-treated cells, indicating that the effect CSPGs had on
OPCs maturation was not due to the arrest of cell development at
the preoligodendrocyte stage.
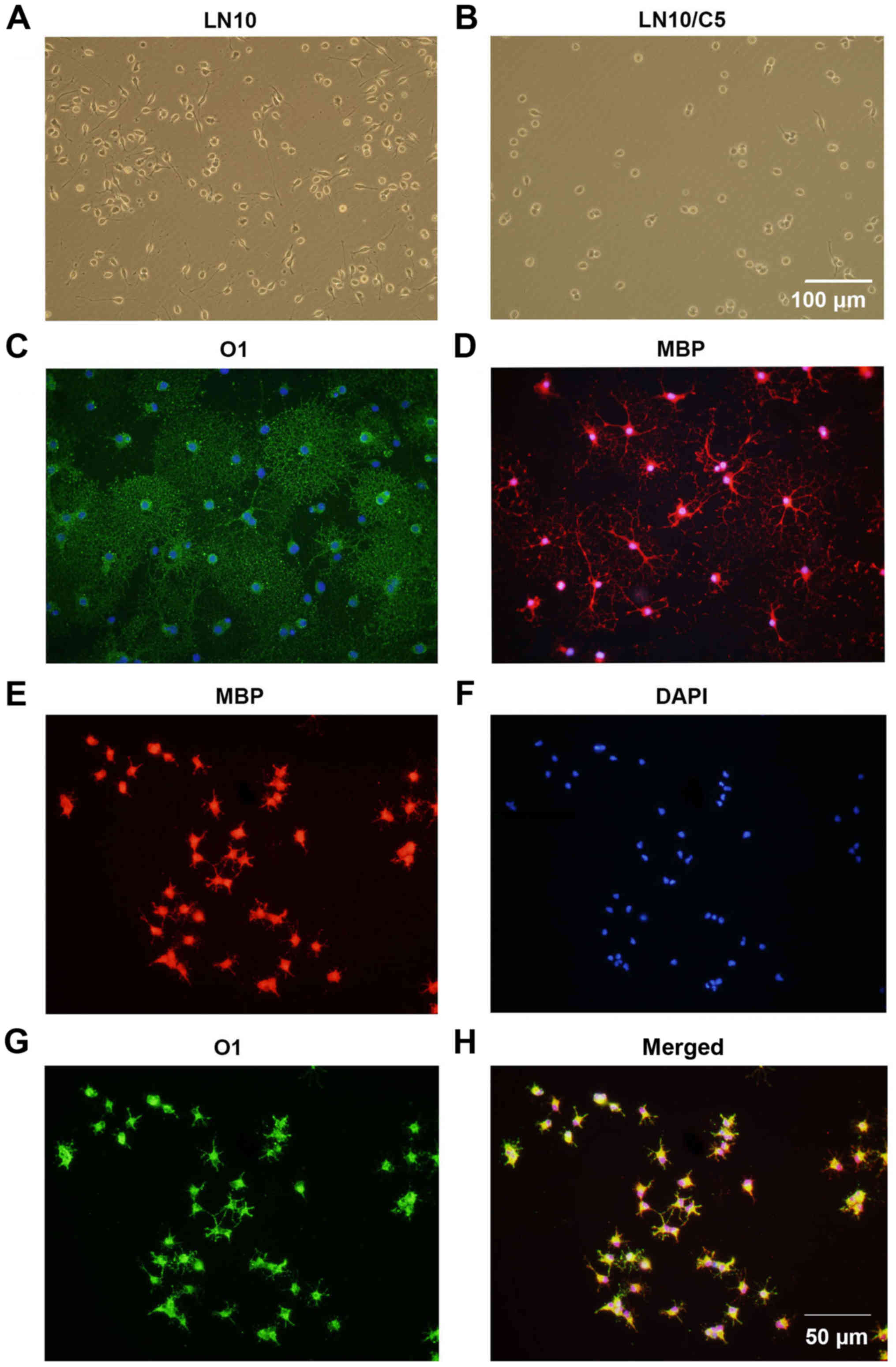 | Figure 7CSPGs inhibit the outgrowth of
processes in oligodendrocytes differentiated from OPC cultures. The
processes in (A) untreated cells were longer than those in (B)
CSPG-treated cells 6 h following culture in OPC proliferation
medium. Scale bar, 100 µm. The untreated cells expressed (C)
O1 and (D) MBP, with mature processes at 5 days following
differentiation. The CSPG-treated cells had shorter processes, but
still expressed (E) MBP, (F) DAPI and (G) O1. (H) Merged images.
Scale bar, 50 µm. CSPG, chondroitin sulfate proteoglycan;
OPC, oligodendrocyte precursor cell; MBP, myelin basic protein;
DAPI, 4′,6-diamidino-2-phenylindole. |
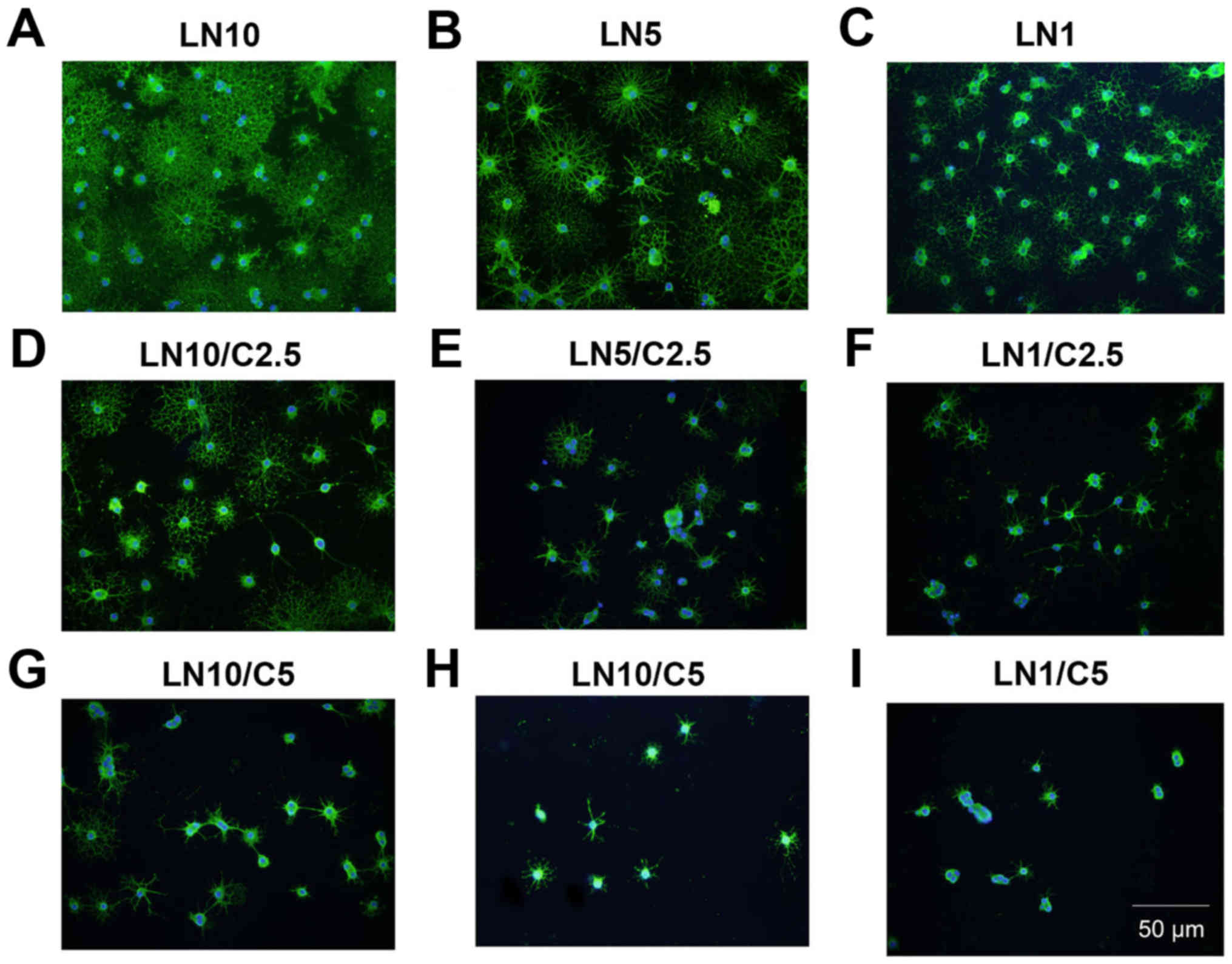
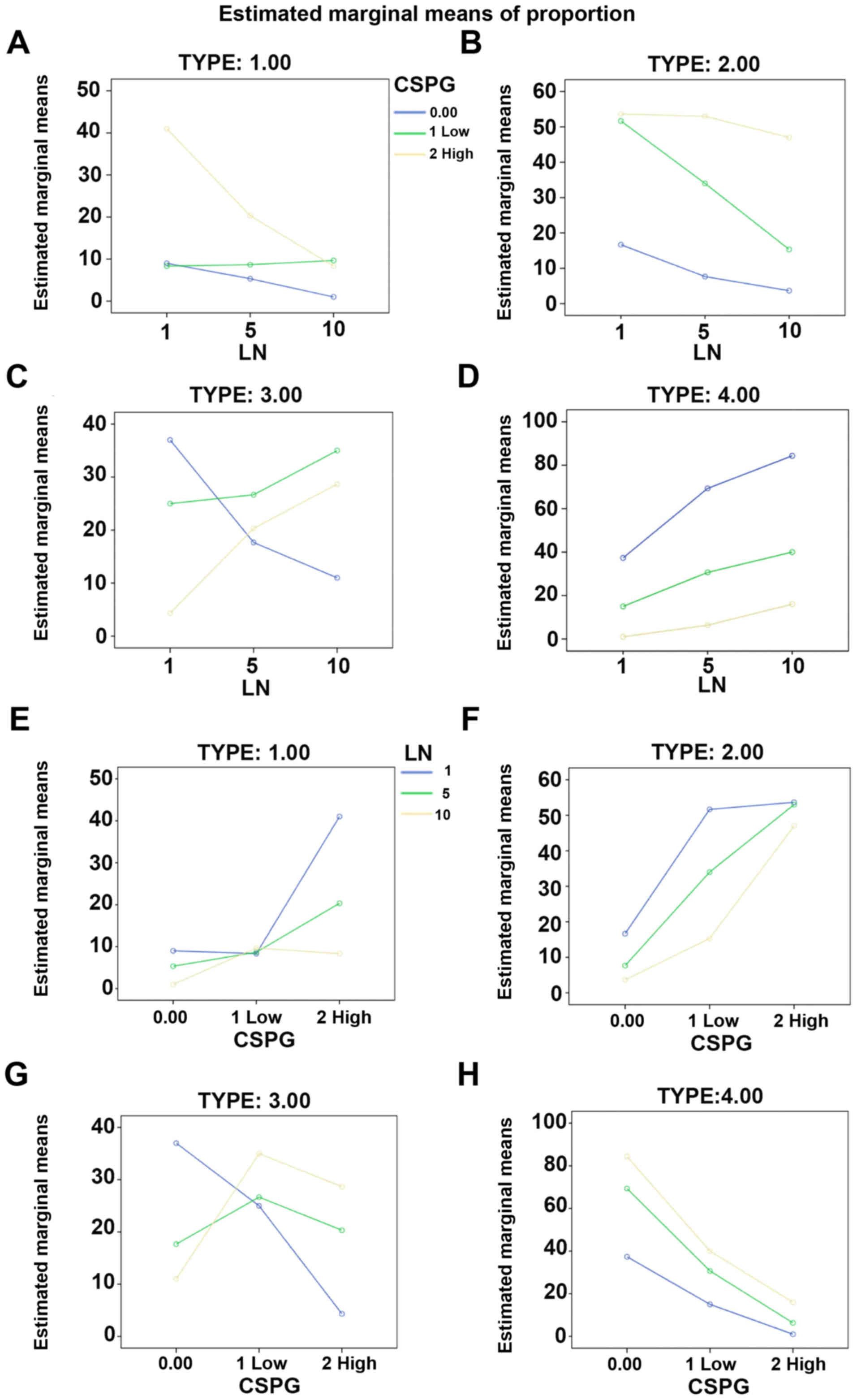
Laminin has a counteractive effect on
CSPGs and the differentiation of OPCs
It was investigated whether laminin has a
counteractive effect on the differentiation of OPCs treated with
CSPGs. Coverslips were coated with three different concentrations
of laminin (1, 5 and 10 µg/ml), and CSPGs at two different
concentrations (2.5 and 5 µg/ml) or without CSPGs. As
revealed in Table I, when
compared with the cells treated with 10 µg/ml of laminin
without CSPGs, the lower doses (5 and 1 µg/ml) of
laminin-treated cells demonstrated the differentiation into all
four types of oligodendrocytes. Morphologically, more spider
web-like cells with more complex and longer processes were observed
in the presence of laminin; this effect increased with the higher
doses of laminin (Fig. 8A–C) and
was observed most prominently with 10 µg/ml laminin. With
the co-application of CSPGs and laminin the process outgrowth of
OPCs was inhibited (Fig. 8D–F);
the higher doses of CSPGs had more prominent inhibitory effects on
oligodendrocyte process outgrowth (Fig. 8G–I). However, the cells treated
with 1 µg/ml laminin and 2.5 µg/ml CSPGs did not
follow this pattern; type II and IV cells increased, whilst type I
and III cells decreased (Table I
and Fig. 9). Furthermore, an
increase in laminin concentration had the opposite effect as an
increase in CSPGs, instead resulting in an increase in the ratio of
type III and IV cells, and a decrease in the ratio of type I and II
cells. These results indicate that laminin treatment facilitates
the process outgrowth of oligodendrocytes on CSPGs, as more type IV
cells are present. This further contributes to the hypothesis that
laminin reduces the impairment of maturation of oligodendrocytes
derived from OPCs by CSPGs. The interactive role of laminin and
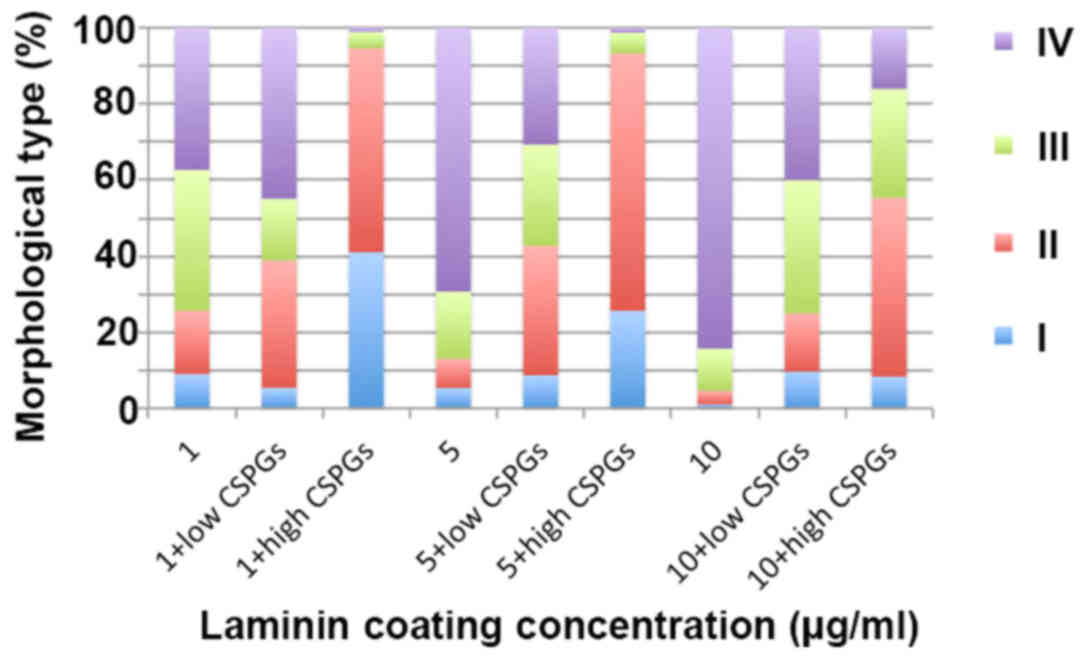
CSPGs was analyzed based on different cell subtypes (Fig. 10 and Table II). The data indicated that
different concentrations of CSPGs and laminin affected the
differentiation into each of the four cell subtypes differently.
These results indicate that laminin has a counteractive effect on
CSPGs.
 | Table IComposition of the subtypes of
oligodendrocytes in the presence of different concentrations of
CSPGs and LN. |
Table I
Composition of the subtypes of
oligodendrocytes in the presence of different concentrations of
CSPGs and LN.
| CSPGs
(µg/ml) | LN
(µg/ml) | Subtype of
oligodendrocyte
|
|---|
| I | II | III | IV |
|---|
| 0 | 10 | 1.00±1.00 | 3.67±1.15 | 11.00±4.58 | 84.33±4.04 |
| 5 | 5.33±1.15a | 7.67±1.15 | 17.67±2.52b | 69.33±3.79b |
| 1 | 9.00±2.00b | 16.7±2.08b | 37.00±1.00b | 37.33±3.06b |
| 2.5 | 10 | 9.67±2.52b | 15.33±1.53b | 35.00±4.00b | 40.00±4.36b |
| 5 | 8.67±2.52b | 34.00±5.29b | 26.67±2.08b | 30.67±4.04b |
| 1 | 8.33±1.52b | 51.67±2.08b | 25.00±3.00b | 15.00±1.00b |
| 5 | 10 | 8.33±1.52b | 47.00±4.00b | 28.67±2.31b | 16.00±3.60b |
| 5 | 20.33±2.52b | 53.00±2.64b | 20.33±2.08b | 6.33±1.53b |
| 1 | 41.33±3.60b | 53.66±4.72b | 4.33±1.53b | 1.00±1.00b |
 | Table IIDistribution of the different
subtypes of oligodendrocytes. |
Table II
Distribution of the different
subtypes of oligodendrocytes.
| Subtype of
oligodendrocyte | Source | Sum of squares
(type III) | Mean square | F-value | P-value |
|---|
| I | LN | 786.074 | 393.037 | 82.264 | <0.0001 |
| CSPGs | 1,643.185 | 821.593 | 171.96 | <0.0001 |
| CSPGs*LN | 951.259 | 237.815 | 49.775 | <0.0001 |
| II | LN | 1,568.296 | 784.148 | 81.119 | <0.0001 |
| CSPGs | 7,964.963 | 3,982.481 | 411.98 | <0.0001 |
| CSPGs*LN | 759.259 | 189.815 | 19.636 | <0.0001 |
| III | LN | 57.407 | 28.704 | 3.708 | 0.0450 |
| CSPGs | 568.074 | 284.037 | 36.694 | <0.0001 |
| CSPGs*LN | 2,126.593 | 531.648 | 68.682 | <0.0001 |
| IV | LN | 3,844.667 | 1,922.333 | 186.70 | <0.0001 |
| CSPGs | 14,364.222 | 7,182.111 | 697.54 | <0.0001 |
| CSPGs*LN | 917.778 | 229.444 | 22.284 | <0.0001 |
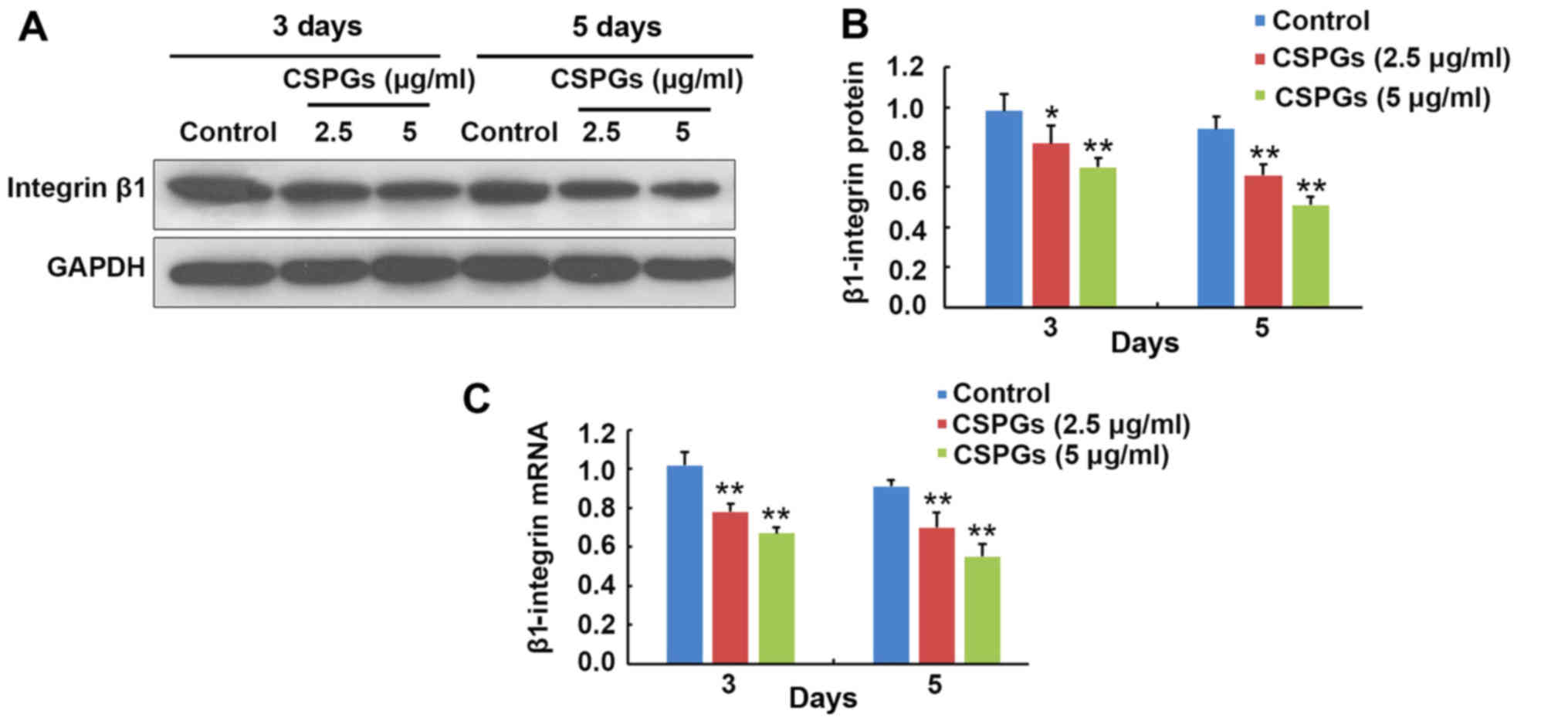
Inhibition of OPC maturation by CSPGs is
associated with β1-integrin
At 3 and 5 days following OPCs differentiation, the
expression level of β1-integrin was determined (Fig. 11). At 3 days following
differentiation, the protein expression of β1-integrin was
decreased from 0.98±0.09 to 0.82±0.09 (P<0.05) and 0.70±0.05
(P<0.01) when treated with 2.5 and 5 µg/ml CSPGs,
respectively (Fig. 11A and B).
Similarly, the protein expression of β1-integrin was reduced from
0.89±0.06 to 0.66±0.06 (P<0.01) and 0.51±0.04 (P<0.01) in the
presence of 2.5 and 5 µg/ml CSPGs at 5 days following
differentiation (Fig. 11A and
B). The β1-integrin mRNA level was decreased from 1.02±0.07 to
0.78±0.04 (P<0.01) and 0.67±0.03 (P<0.01) when treated with
2.5 and 5 µg/ml CSPGs, respectively, at 3 days following
differentiation, and reduced from 0.91±0.03 to 0.70±0.08
(P<0.01) and 0.55±0.07 (P<0.01) at 5 days following
differentiation (Fig. 11C). The
levels of β1-integrin protein and mRNA were consistently decreased
with increasing treatment with CSPGs, demonstrating that the
inhibitory effect of CSPGs may be associated with the alteration of
β1-integrin expression.
Discussion
WMI is the most common cause of brain injury in
preterm infants (17). OPCs are
selectively vulnerable to WMI in premature infants (18). CSPGs act as a mechanical barrier
to inhibit the migration, differentiation and recovery of neural
cells (19). However, the
mechanism underlying the correlation between CSPGs and the
differentiation, migration and differentiation of OPCs remains
unclear. In the present study, the role of CSPGs in inhibiting the
migration and differentiation of OPCs was examined. This effect was
counteracted by the presence of laminin, revealing the interaction
between CSPGs and laminin in the maturation of OPCs. The
involvement of β1-integrin signaling in the CSPG-induced inhibition
of the differentiation of OPCs was also investigated. The results
identified a novel mechanism of WMI in premature infants and may
provide a novel candidate as a potential therapeutic target.
There are currently 18 identified subtypes of
integrin; oligodendrocytes express several different types of
integrin and the expression of these receptors is developmentally
regulated (20). The interaction
between laminin-2 and the integrin that is expressed on an
oligodendrocyte is an important part of the formation of the myelin
membrane, which is required to cover the axon and form the myelin
sheath (21). The present study
demonstrated that laminin could potentially offset the inhibitory
effect CSPGs have on myelination in oligodendrocytes. Among all the
integrin subtypes expressed in oligodendrocytes, α6β1 is the only
known laminin-2 receptor, serving a critical role in the
myelination of oligodendrocytes (20). The intracellular proteins
correlated with β1-integrin include integrin-linked protein kinase,
focal adhesion kinase, protein kinase B, tyrosine-protein kinase
Fyn, ras-related C3 botulinum toxin substrate 1 (Rac1), cell
division control protein 42 homolog (cdc42) and Rho factor. These
proteins are involved in the reconstruction of the cytoskeleton and
are therefore important in regulating the morphological development
of oligodendrocytes (22). Taking
into consideration that OPCs are the progenitors of
oligodendrocytes and mainly express β1-integrin, which is of great
significance in the differentiation of OPCs, it was hypothesized
that the β1-integrin signaling pathway was the main pathway that
regulates the effect of laminin and CSPGs on the differentiation of
OPCs. This hypothesis was supported by the results of the present
study, as an alteration in β1-integrin protein and mRNA expression
levels were observed in CSPG-treated OPCs.
CSPGs are an important component of the ECM, which
have been previously reported to regulate the differentiation and
migration of multipotent neural precursor cells through the
integrin signaling pathway (23).
The results of the present study indicate that CSPGs also regulate
similar events in OPCs. CSPGs inhibited the differentiation of
OPCs, including the outgrowth of processes in oligodendrocytes.
However, the expression of O1 and MBP in CSPG-treated cells was
still observed. This revealed that the inhibitory effect of CSPGs
on oligodendrocyte maturation was not due to their suspension at a
precursor stage, but the inhibition of the processes extension.
Following treatment with CSPGs it was observed that the OPCs were
more detached and had fewer processes. This meant that CSPGs only
inhibited the process of extension, but did not cause the death of
OPCs. Further investigation is required to determine whether
detachment is due to the impaired capacity for process outgrowth or
the competition between laminin and adhesion molecules.
Furthermore, detachment occurred when bFGF and PDGF-AA were
replaced by T3 and T4. Therefore, it was hypothesized that these
growth factors may protect OPCs from CSPGs treatment.
Yu et al (24) identified that laminin was required
for morphogenesis in the radial axonal sorting of Schwann cells,
mediated by the ErbB-cdc42 and β1-integrin-Rac1 signaling pathways.
As Rac1 is the downstream factor of β1-integrin, laminin may
regulate this process in Schwann cells through the β1-integrin-Rac1
signaling pathway. A similar situation may occur in OPCs based on
the previous finding that laminin regulates oligodendrocyte
production in the subventricular zone (25). Further studies investigating the
underlying mechanism of this are required to progress understanding
of the role of CSPGs in OPC migration and differentiation, and the
possibility for a novel therapeutic target for the treatment of WMI
in premature infants.
Acknowledgments
The present study was supported by the National
Natural Science Foundation of China (grant no. 81070520).
References
|
1
|
SanGiovanni JP, Allred EN, Mayer DL,
Stewart JE, Herrera MG and Leviton A: Reduced visual resolution
acuity and cerebral white matter damage in very-low-birthweight
infants. Dev Med Child Neurol. 42:809–815. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Muglia LJ and Katz M: The enigma of
spontaneous preterm birth. N Engl J Med. 362:529–535. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
du Plessis AJ: Neurology of the newborn
infant. Preface. Clin Perinatol. 36:xi–xiii. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Wilson-Costello D, Friedman H, Minich N,
Fanaroff AA and Hack M: Improved survival rates with increased
neurodevelop-mental disability for extremely low birth weight
infants in the 1990s. Pediatrics. 115:997–1003. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Groenendaal F, Termote JU, van der
Heide-Jalving M, van Haastert IC and de Vries LS: Complications
affecting preterm neonates from 1991 to 2006: What have we gained?
Acta Paediatr. 99:354–358. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Msall ME: Central nervous system
connectivity after extreme prematurity: Understanding autistic
spectrum disorder. J Pediatr. 156:519–521. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Volpe JJ, Kinney HC, Jensen FE and
Rosenberg PA: The developing oligodendrocyte: Key cellular target
in brain injury in the premature infant. Int J Dev Neurosci.
29:423–440. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Back SA, Luo NL, Mallinson RA, O'Malley
JP, Wallen LD, Frei B, Morrow JD, Petito CK, Roberts CT Jr, Murdoch
GH, et al: Selective vulnerability of preterm white matter to
oxidative damage defined by F2-isoprostanes. Ann Neurol.
58:108–120. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Khwaja O and Volpe JJ: Pathogenesis of
cerebral white matter injury of prematurity. Arch Dis Child Fetal
Neonatal Ed. 93:F153–F161. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Billiards SS, Haynes RL, Folkerth RD,
Borenstein NS, Trachtenberg FL, Rowitch DH, Ligon KL, Volpe JJ and
Kinney HC: Myelin abnormalities without oligodendrocyte loss in
periventricular leukomalacia. Brain Pathol. 18:153–163. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Back SA, Luo NL, Borenstein NS, Levine JM,
Volpe JJ and Kinney HC: Late oligodendrocyte progenitors coincide
with the developmental window of vulnerability for human perinatal
white matter injury. J Neurosci. 21:1302–1312. 2001.PubMed/NCBI
|
|
12
|
Galtrey CM and Fawcett JW: The role of
chondroitin sulfate proteoglycans in regeneration and plasticity in
the central nervous system. Brain Res Brain Res Rev. 54:1–18. 2007.
View Article : Google Scholar
|
|
13
|
Carulli D, Laabs T, Geller HM and Fawcett
JW: Chondroitin sulfate proteoglycans in neural development and
regeneration. Curr Opin Neurobiol. 15:116–120. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Siebert JR and Osterhout DJ: The
inhibitory effects of chondroitin sulfate proteoglycans on
oligodendrocytes. J Neurochem. 119:176–188. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
O'Meara RW, Ryan SD, Colognato H and
Kothary R: Derivation of enriched oligodendrocyte cultures and
oligodendrocyte/neuron myelinating co-cultures from post-natal
murine tissues. J Vis Exp. 54:33242011.
|
|
16
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
17
|
Back SA and Rosenberg PA: Pathophysiology
of glia in perinatal white matter injury. Glia. 62:1790–1815. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Volpe JJ, Kinney HC, Jensen FE and
Rosenberg PA: Reprint of 'The developing oligodendrocyte: Key
cellular target in brain injury in the premature infant'. Int J Dev
Neurosci. 29:565–582. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Sirko S, von Holst A, Wizenmann A, Götz M
and Faissner A: Chondroitin sulfate glycosaminoglycans control
proliferation, radial glia cell differentiation and neurogenesis in
neural stem/progenitor cells. Development. 134:2727–2738. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
O'Meara RW, Michalski JP and Kothary R:
Integrin signaling in oligodendrocytes and its importance in CNS
myelination. J Signal Transduct. 2011:3540912011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Buttery PC and ffrench-Constant C:
Laminin-2/integrin interactions enhance myelin membrane formation
by oligodendrocytes. Mol Cell Neurosci. 14:199–212. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Olsen IM and Ffrench-Constant C: Dynamic
regulation of integrin activation by intracellular and
extracellular signals controls oligodendrocyte morphology. BMC
Biol. 3:252005. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Gu WL, Fu SL, Wang YX, Li Y, Lü HZ, Xu XM
and Lu PH: Chondroitin sulfate proteoglycans regulate the growth,
differentiation and migration of multipotent neural precursor cells
through the integrin signaling pathway. BMC Neurosci. 10:1282009.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Yu WM, Chen ZL, North AJ and Strickland S:
Laminin is required for Schwann cell morphogenesis. J Cell Sci.
122:929–936. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Relucio J, Menezes MJ, Miyagoe-Suzuki Y,
Takeda S and Colognato H: Laminin regulates postnatal
oligodendrocyte production by promoting oligodendrocyte progenitor
survival in the subventricular zone. Glia. 60:1451–1467. 2012.
View Article : Google Scholar : PubMed/NCBI
|