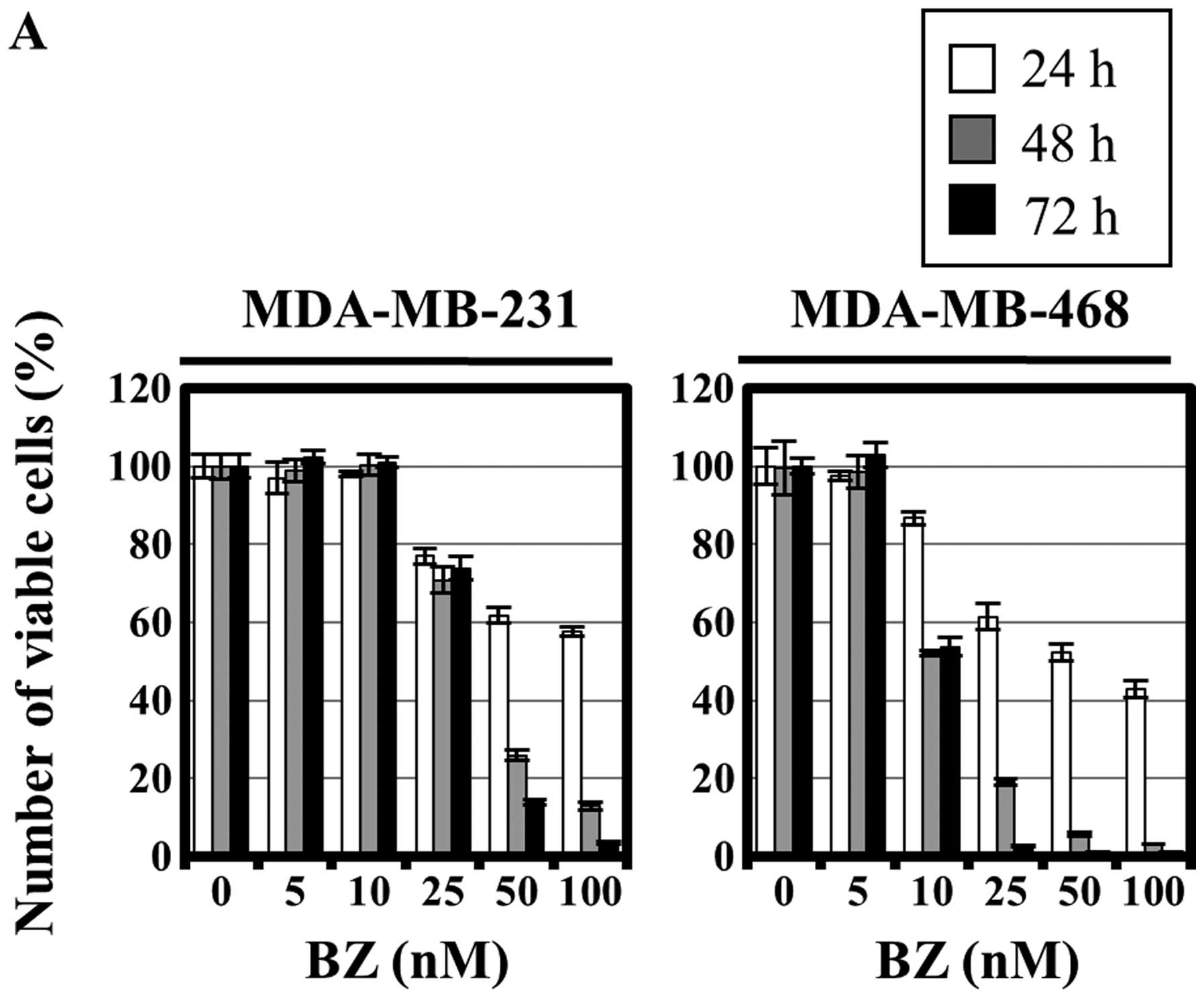
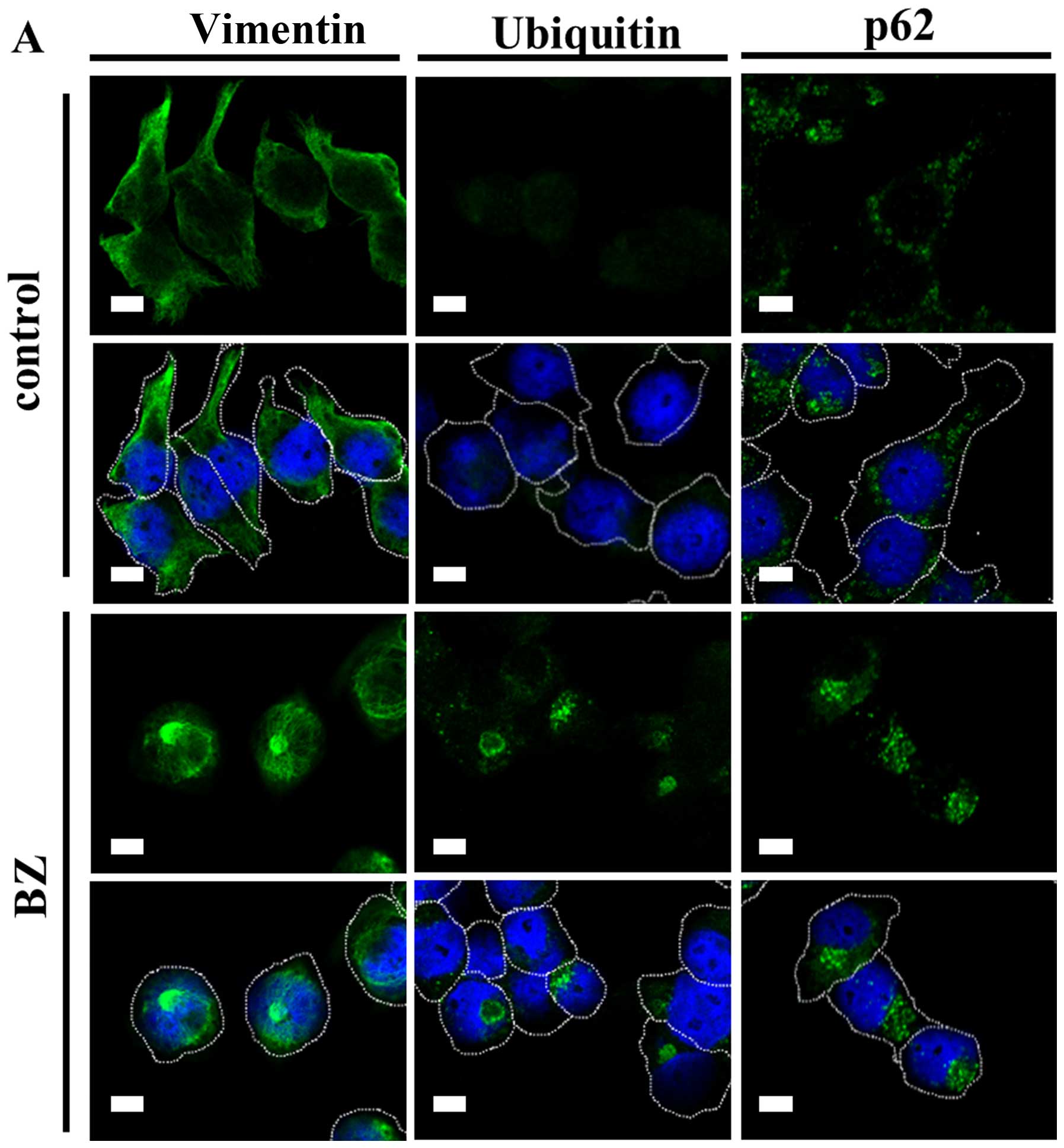
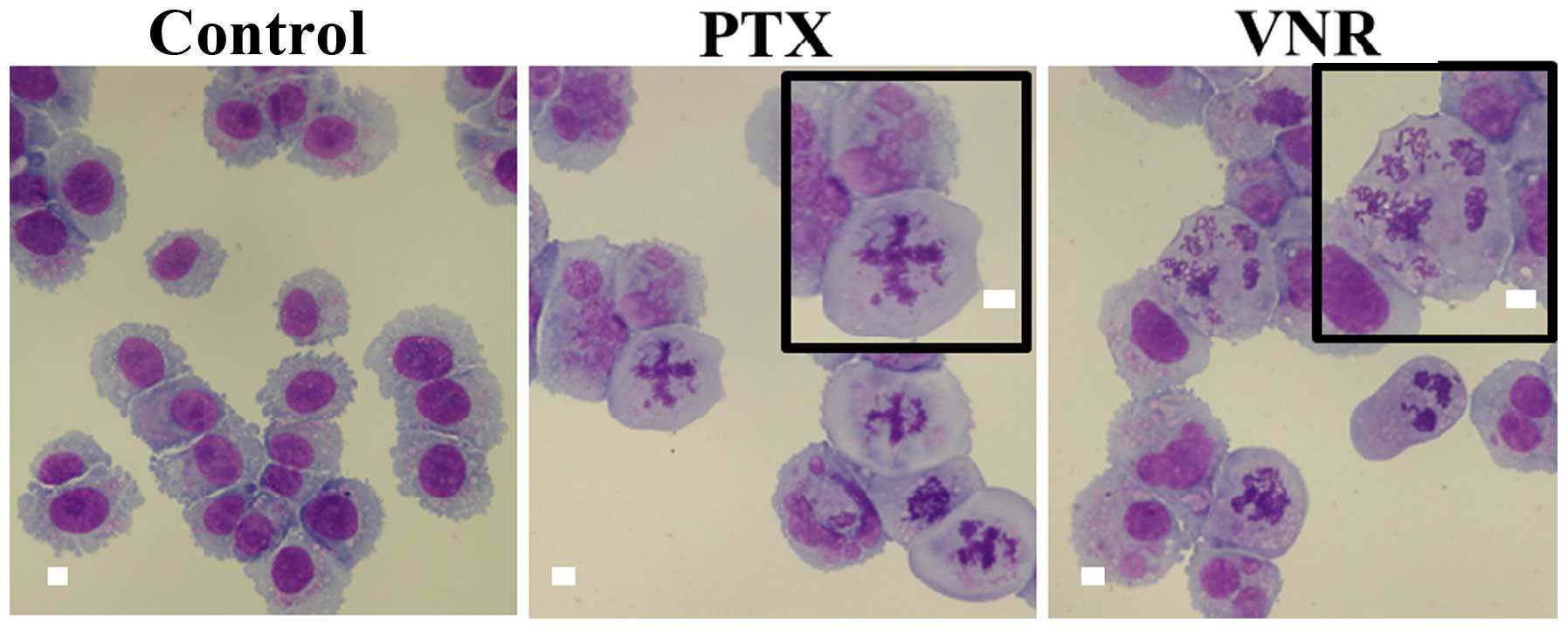
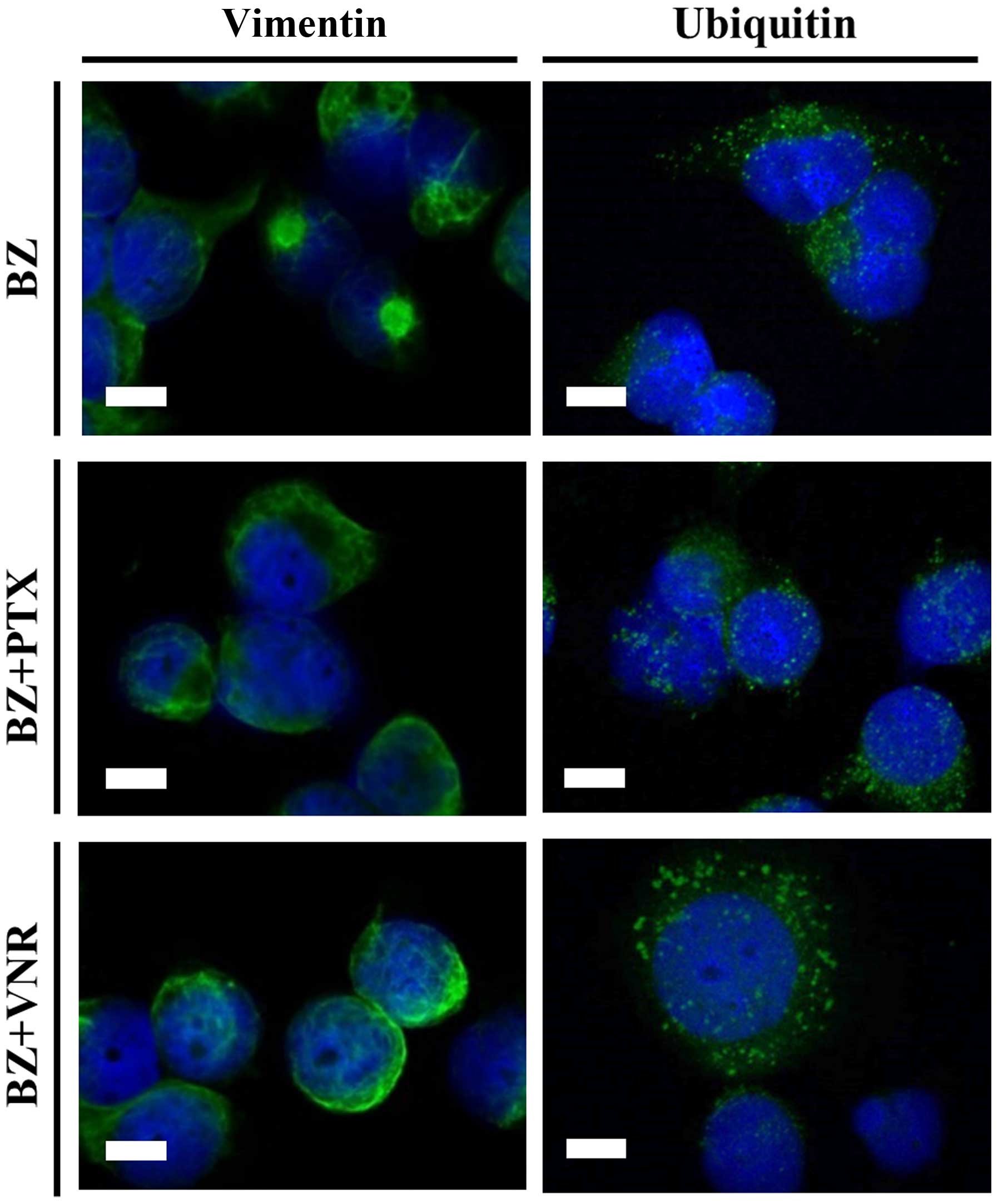
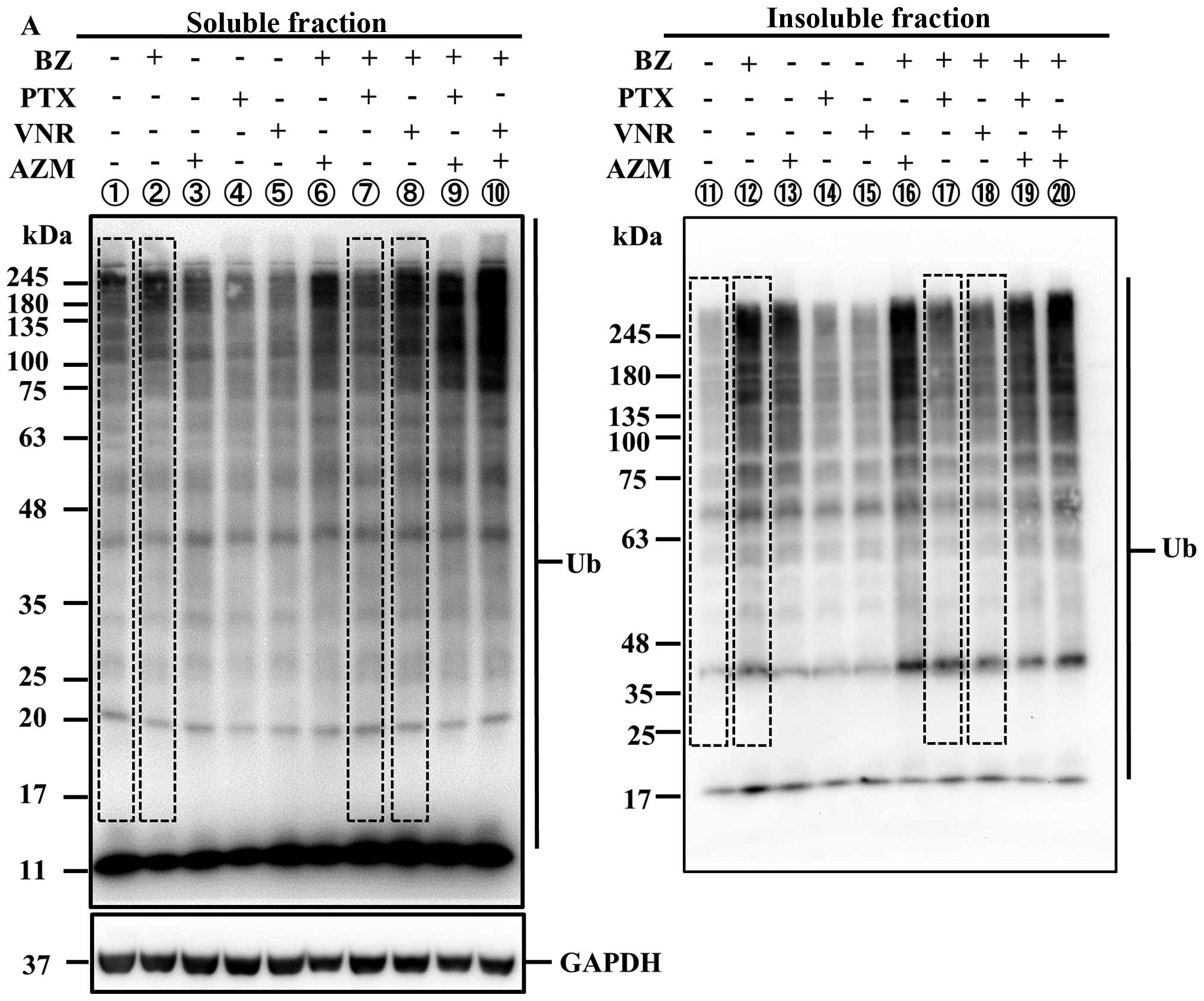
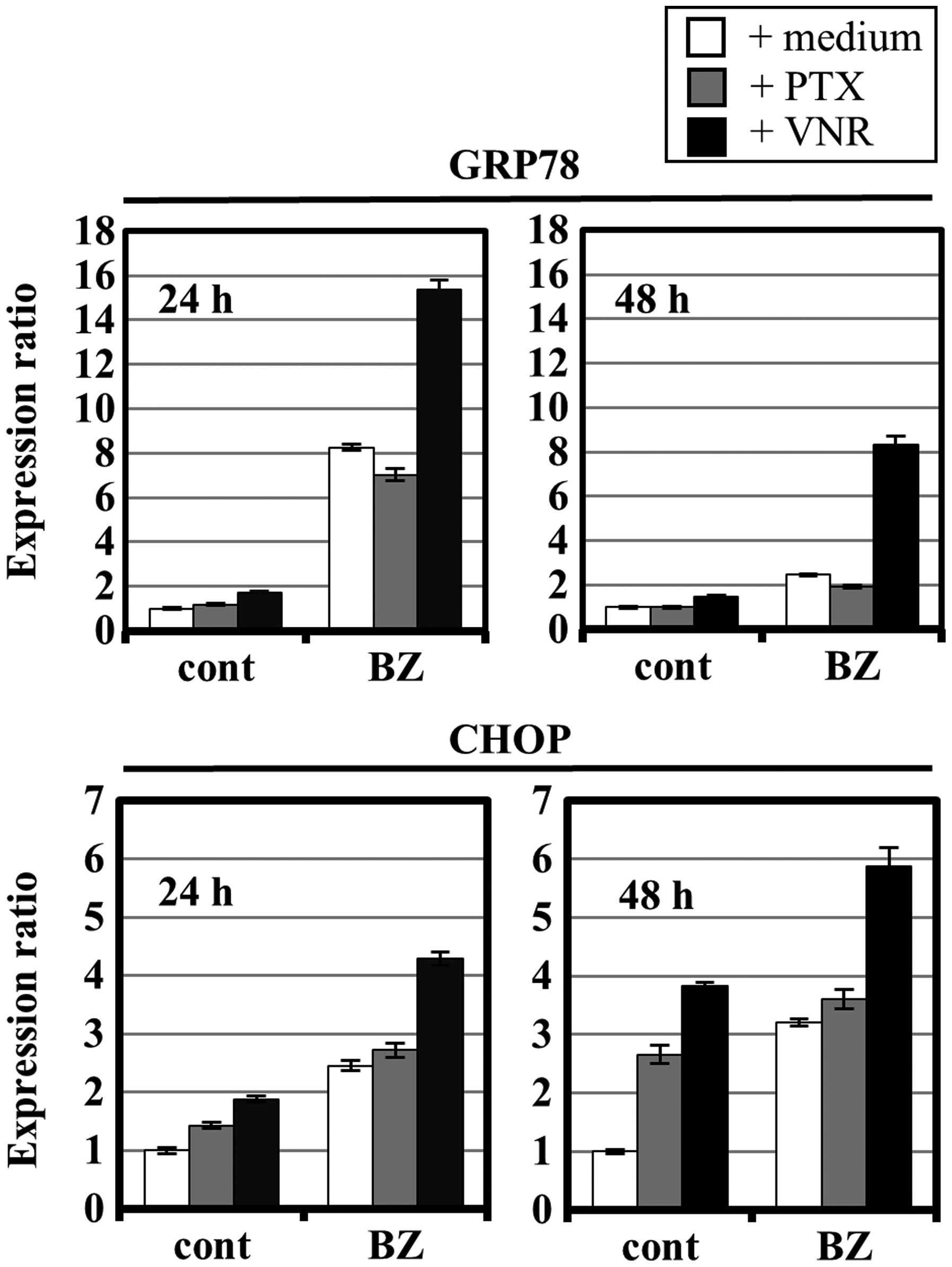
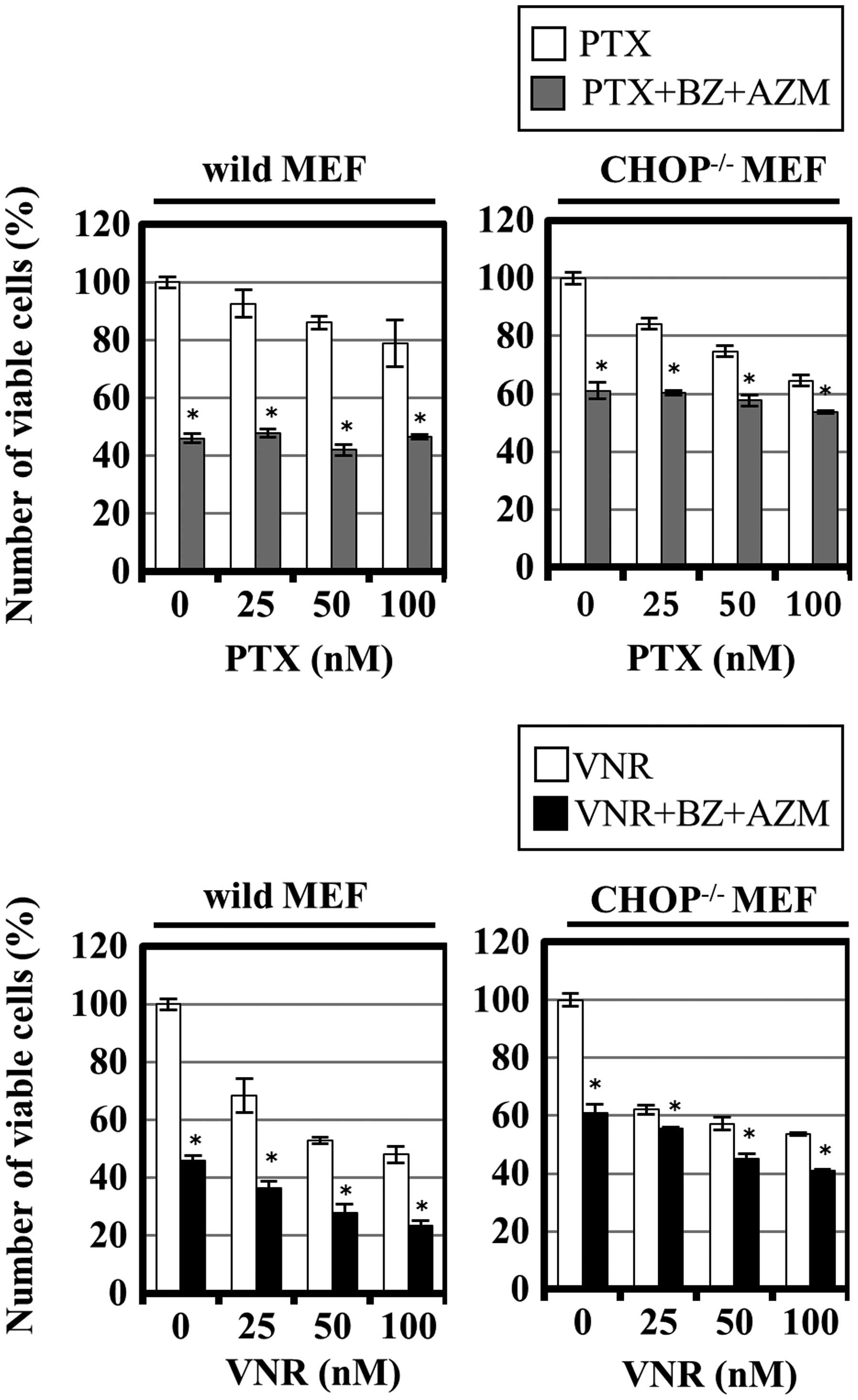
|
1
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Runowicz CD, Leach CR, Henry NL, Henry KS,
Mackey HT, Cowens-Alvarado RL, Cannady RS, Pratt-Chapman ML, Edge
SB, Jacobs LA, et al; American Cancer Society/American Society of
Clinical Oncology. Breast Cancer Survivorship Care Guideline. J
Clin Oncol. 34:611–635. 2016. View Article : Google Scholar
|
|
3
|
Veldhoen RA, Banman SL, Hemmerling DR,
Odsen R, Simmen T, Simmonds AJ, Underhill DA and Goping IS: The
chemotherapeutic agent paclitaxel inhibits autophagy through two
distinct mechanisms that regulate apoptosis. Oncogene. 32:736–746.
2013. View Article : Google Scholar
|
|
4
|
Wen J, Yeo S, Wang C, Chen S, Sun S, Haas
MA, Tu W, Jin F and Guan JL: Autophagy inhibition re-sensitizes
pulse stimulation-selected paclitaxel-resistant triple negative
breast cancer cells to chemotherapy-induced apoptosis. Breast
Cancer Res Treat. 149:619–629. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Liu S and Li X: Autophagy inhibition
enhances sensitivity of endometrial carcinoma cells to paclitaxel.
Int J Oncol. 46:2399–2408. 2015.PubMed/NCBI
|
|
6
|
Wojcik S: Crosstalk between autophagy and
proteasome protein degradation systems: Possible implications for
cancer therapy. Folia Histochem Cytobiol. 51:249–264. 2013.
View Article : Google Scholar
|
|
7
|
Mizushima N: Autophagy: Process and
function. Genes Dev. 21:2861–2873. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Levine B and Kroemer G: Autophagy in the
pathogenesis of disease. Cell. 132:27–42. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Korolchuk VI, Menzies FM and Rubinsztein
DC: Mechanisms of cross-talk between the ubiquitin-proteasome and
autophagy-lysosome systems. FEBS Lett. 584:1393–1398. 2010.
View Article : Google Scholar
|
|
10
|
Kirkin V, McEwan DG, Novak I and Dikic I:
A role for ubiquitin in selective autophagy. Mol Cell. 34:259–269.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Degenhardt K, Mathew R, Beaudoin B, Bray
K, Anderson D, Chen G, Mukherjee C, Shi Y, Gélinas C, Fan Y, et al:
Autophagy promotes tumor cell survival and restricts necrosis,
inflammation, and tumorigenesis. Cancer Cell. 10:51–64. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Mathew R, Karp CM, Beaudoin B, Vuong N,
Chen G, Chen HY, Bray K, Reddy A, Bhanot G, Gelinas C, et al:
Autophagy suppresses tumorigenesis through elimination of p62.
Cell. 137:1062–1075. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Karantza-Wadsworth V, Patel S, Kravchuk O,
Chen G, Mathew R, Jin S and White E: Autophagy mitigates metabolic
stress and genome damage in mammary tumorigenesis. Genes Dev.
21:1621–1635. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Guo JY, Chen HY, Mathew R, Fan J,
Strohecker AM, Karsli-Uzunbas G, Kamphorst JJ, Chen G, Lemons JM,
Karantza V, et al: Activated Ras requires autophagy to maintain
oxidative metabolism and tumorigenesis. Genes Dev. 25:460–470.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kawaguchi T, Miyazawa K, Moriya S, Ohtomo
T, Che XF, Naito M, Itoh M and Tomoda A: Combined treatment with
bortezomib plus bafilomycin A1 enhances the cytocidal effect and
induces endoplasmic reticulum stress in U266 myeloma cells:
Crosstalk among proteasome, autophagy-lysosome and ER stress. Int J
Oncol. 38:643–654. 2011.
|
|
16
|
Komatsu S, Miyazawa K, Moriya S, Takase A,
Naito M, Inazu M, Kohno N, Itoh M and Tomoda A: Clarithromycin
enhances bortezomib-induced cytotoxicity via endoplasmic reticulum
stress-mediated CHOP (GADD153) induction and autophagy in breast
cancer cells. Int J Oncol. 40:1029–1039. 2012.
|
|
17
|
Moriya S, Che XF, Komatsu S, Abe A,
Kawaguchi T, Gotoh A, Inazu M, Tomoda A and Miyazawa K: Macrolide
antibiotics block autophagy flux and sensitize to bortezomib via
endoplasmic reticulum stress-mediated CHOP induction in myeloma
cells. Int J Oncol. 42:1541–1550. 2013.PubMed/NCBI
|
|
18
|
Meusser B, Hirsch C, Jarosch E and Sommer
T: ERAD: The long road to destruction. Nat Cell Biol. 7:766–772.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Hetz C: The unfolded protein response:
Controlling cell fate decisions under ER stress and beyond. Nat Rev
Mol Cell Biol. 13:89–102. 2012.PubMed/NCBI
|
|
20
|
Smith MH, Ploegh HL and Weissman JS: Road
to ruin: Targeting proteins for degradation in the endoplasmic
reticulum. Science. 334:1086–1090. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Brodsky JL: Cleaning up: ER-associated
degradation to the rescue. Cell. 151:1163–1167. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Sano R and Reed JC: ER stress-induced cell
death mechanisms. Biochim Biophys Acta. 1833.3460–3470. 2013.
|
|
23
|
Oyadomari S and Mori M: Roles of
CHOP/GADD153 in endoplasmic reticulum stress. Cell Death Differ.
11:381–389. 2004. View Article : Google Scholar
|
|
24
|
Tabas I and Ron D: Integrating the
mechanisms of apoptosis induced by endoplasmic reticulum stress.
Nat Cell Biol. 13:184–190. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Verfaillie T, Garg AD and Agostinis P:
Targeting ER stress induced apoptosis and inflammation in cancer.
Cancer Lett. 332:249–264. 2013. View Article : Google Scholar
|
|
26
|
Li J, Ni M, Lee B, Barron E, Hinton DR and
Lee AS: The unfolded protein response regulator GRP78/BiP is
required for endoplasmic reticulum integrity and stress-induced
autophagy in mammalian cells. Cell Death Differ. 15:1460–1471.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Johnston JA, Ward CL and Kopito RR:
Aggresomes: A cellular response to misfolded proteins. J Cell Biol.
143:1883–1898. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kawaguchi Y, Kovacs JJ, McLaurin A, Vance
JM, Ito A and Yao TP: The deacetylase HDAC6 regulates aggresome
formation and cell viability in response to misfolded protein
stress. Cell. 115:727–738. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Olzmann JA and Chin LS: Parkin-mediated
K63-linked polyubiquitination: A signal for targeting misfolded
proteins to the aggresome-autophagy pathway. Autophagy. 4:85–87.
2008. View Article : Google Scholar
|
|
30
|
Lee JY, Koga H, Kawaguchi Y, Tang W, Wong
E, Gao YS, Pandey UB, Kaushik S, Tresse E, Lu J, et al: HDAC6
controls autophagosome maturation essential for ubiquitin-selective
quality-control autophagy. EMBO J. 29:969–980. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Rodriguez-Gonzalez A, Lin T, Ikeda AK,
Simms-Waldrip T, Fu C and Sakamoto KM: Role of the aggresome
pathway in cancer: Targeting histone deacetylase 6-dependent
protein degradation. Cancer Res. 68:2557–2560. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kopito RR: Aggresomes, inclusion bodies
and protein aggregation. Trends Cell Biol. 10:524–530. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Brüning A and Jückstock J: Misfolded
proteins: From little villains to little helpers in the fight
against cancer. Front Oncol. 5:472015.PubMed/NCBI
|
|
34
|
Moriya S, Komatsu S, Yamasaki K, Kawai Y,
Kokuba H, Hirota A, Che XF, Inazu M, Gotoh A, Hiramoto M, et al:
Targeting the integrated networks of aggresome formation,
proteasome, and autophagy potentiates ER stress-mediated cell death
in multiple myeloma cells. Int J Oncol. 46:474–486. 2015.
|
|
35
|
Komatsu S, Moriya S, Che XF, Yokoyama T,
Kohno N and Miyazawa K: Combined treatment with SAHA, bortezomib,
and clarithromycin for concomitant targeting of aggresome formation
and intracellular proteolytic pathways enhances ER stress-mediated
cell death in breast cancer cells. Biochem Biophys Res Commun.
437:41–47. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Yu L, McPhee CK, Zheng L, Mardones GA,
Rong Y, Peng J, Mi N, Zhao Y, Liu Z, Wan F, et al: Termination of
autophagy and reformation of lysosomes regulated by mTOR. Nature.
465:942–946. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Zaarur N, Meriin AB, Bejarano E, Xu X,
Gabai VL, Cuervo AM and Sherman MY: Proteasome failure promotes
positioning of lysosomes around the aggresome via local block of
microtubule-dependent transport. Mol Cell Biol. 34:1336–1348. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Mackeh R, Perdiz D, Lorin S, Codogno P and
Poüs C: Autophagy and microtubules - new story, old players. J Cell
Sci. 126:1071–1080. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Olzmann JA, Li L and Chin LS: Aggresome
formation and neurodegenerative diseases: Therapeutic implications.
Curr Med Chem. 15:47–60. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Nawrocki ST, Carew JS, Pino MS, Highshaw
RA, Andtbacka RH, Dunner K Jr, Pal A, Bornmann WG, Chiao PJ, Huang
P, et al: Aggresome disruption: A novel strategy to enhance
bortezomib-induced apoptosis in pancreatic cancer cells. Cancer
Res. 66:3773–3781. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Helfand BT, Chang L and Goldman RD:
Intermediate filaments are dynamic and motile elements of cellular
architecture. J Cell Sci. 117:133–141. 2004. View Article : Google Scholar
|
|
42
|
Perlson E, Michaelevski I, Kowalsman N,
Ben-Yaakov K, Shaked M, Seger R, Eisenstein M and Fainzilber M:
Vimentin binding to phosphorylated Erk sterically hinders enzymatic
dephosphorylation of the kinase. J Mol Biol. 364:938–944. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Pérez-Sala D, Oeste CL, Martínez AE,
Carrasco MJ, Garzón B and Cañada FJ: Vimentin filament organization
and stress sensing depend on its single cysteine residue and zinc
binding. Nat Commun. 6:72872015. View Article : Google Scholar : PubMed/NCBI
|