Introduction
Colorectal cancer is the third most common cancer
worldwide and the fourth leading cause of cancer-related death
(1). The patients diagnosed with
colorectal cancer often develop colorectal metastases and
approximately 80–90% of these patients are found with unresectable
metastatic liver disease (2).
Colorectal cancer is associated with the progressive accumulation
of mutations in oncogenes and tumor suppressor genes. These
tumor-specific mutations are the key to understand the cellular
processes underlying tumorigenesis and could be used as diagnostic
and therapeutic targets (3).
Ribosomal proteins (RPs) are one of the components
of ribosomes and there are approximately 80 RPs in eukaryotes
(4). RPs are known to stabilize
specific rRNA structures in mature ribosomal subunits and promote
correct folding of rRNAs during ribosomal assembly. However, not
only RPs have crucial role in protein biosynthesis, but recent
studies have also associated them to human congenital disorders and
cancers (5). For example, RPS4 has
been implicated in Turner's syndrome (6), and mutant RPS19 was found in
individuals with Diamond-Blackfan anemia (7). Hence, the functions of RPs that are
independent of protein biosynthesis are called extraribosomal
functions. The extraribosomal functions include transcription and
repair, RNA splicing and modification, cell growth and
proliferation, regulation of apoptosis and development (8). Moreover, knockdown of many individual
RPs resulted in p53 accumulation, cell death and defective
development (8,9). Various RPs have been found to be
overexpressed in many cancer cells and are associated with the
development and progression of malignant cancers (9). For instance, in gastric cancer cell
line, RPL6 promoted cell cycle progression and cancer cell
proliferation by upregulating the expression of cyclin E (10). Also, phosphorylated RPS3 activated
anti-apoptotic protein TRAF2 and enhanced radio-resistance in
non-small cell lung cancer cells (11).
Ribosomal protein L9 (RPL9) is a component of the
60S subunit that belongs to the L6P family of ribosomal proteins.
From what we have gathered, RPL9's extraribosomal function is not
known, but one of the studies have suggested that human RPL9
appears to be involved with the uncontrolled growth by promoting
stress-mediated survival and that RPL9 is more like
anti-apoptotic-encoding RP genes in yeast (12). In addition, there are reports that
RPL9 gene is overexpressed in colorectal cancer compared to normal
colon (5,13,14),
which provides the possibility that RPL9 might be involved in
tumorigenesis of colorectal cancer.
The aim of the present study was to define the
extraribosomal function of RPL9 in colorectal cancer progression.
Using RNA interference (RNAi) techniques, we targeted RPL9 gene in
colorectal cancer HCT116 and HT29 cells. We found that silencing of
RPL9 inhibited colorectal cancer cell growth in vitro as
well as in vivo and caused apoptotic cell death. The
anti-proliferative effects were induced through a common subset of
molecular alterations of 296 mRNA transcripts, including
downregulation of Id-1 (inhibitor of DNA binding-1) (15). Western blotting proved that the
decrease in Id-1 protein level by RPL9 knockdown was accompanied
with the reduction in IκBα phosphorylation, which suggests that
RPL9 is functionally associated with Id-1/NF-κB signaling pathway
and that the regulation of RPL9 expression may be a potential
therapeutic target in colorectal cancer treatment.
Materials and methods
Cell culture and siRNA transfection
Human colorectal cancer cell lines, HCT116 and HT29,
were purchased from the Korean Cell Line Bank (KCLB; Seoul,
Republic of Korea). Both HCT116 and HT29 cells were cultured in
RPMI-1640 medium (Corning Incorporated, Corning, NY, USA)
supplemented with 10% of fetal bovine serum (FBS; HyClone
Laboratories, Logan, UT, USA) and 1% of penicillin/streptomycin
solution (HyClone Laboratories), in a 37°C humidified incubator
with a mixture of 95% air and 5% CO2. The phenotypes of
these cell lines have been authenticated by the KCLB. Cells were
plated at 30% density 24 h before transfection. A total of 15 nM
siRNA was mixed with Lipofectamine 2000 (Invitrogen, Carlsbad, CA,
USA) in Opti-MEM (Invitrogen) and the medium was replaced 6 h after
transfection. RPL9 siRNA duplexes (si-RPL9) were chemically
synthesized by Ambion (Austin, TX, USA; siRNA ID# s226955). The
negative control siRNA (si-NC) that does not target any endogenous
transcript was used for control experiments. The sequences of si-NC
(Bioneer, Daejeon, South Korea) are as follows: 5′-ACGUGA
CACGUUCGGAGAA(UU)-3′ (sense) and 5′-UUCUCCGAAC GUGUCACGU-3′
(antisense).
Cell proliferation assay
Cell growth was measured using the Cell Counting
kit-8 (Dojindo Laboratories, Kumamoto, Japan) according to the
manufacturer's instructions. Log phase cells were trypsinized into
single cell suspension and HCT116 (2×103 cells) and HT29
(1×103 cells) were seeded into 96-well plates. After 24
h, cells were transfected as described above and after day 1, day
2, day 3 and day 4 of cell culture, the CCK-8 solution was added
into each well. After 2 h, OD value was measured at a wavelength of
450 nm using a VersaMax microplate reader (Molecular Devices,
Sunnyvale, CA, USA).
Quantitative real-time RT-PCR
(RT-qPCR)
The changes in target gene expression were detected
using RT-qPCR. Total RNA was isolated using TRIzol (Ambion) and
synthesized into cDNA by using first strand cDNA synthesis kit
(Takara Bio, Shiga, Japan) according to the manufacturer's
instruction. cDNAs were then amplified using corresponding pair of
primers (RPL9 forward, 5′-GCACAG TTATCGTGAAGG GC-3′ and RPL9
reverse, 5′-TTACCC CACCATTTGTCAA CC-3′; GAPDH forward,
5′-GGGAGCCAAAAGGGTCATCA TCTC-3′ and GAPDH reverse,
5′-CCATGCCAGTGAGCTT CCCGTTC-3′) synthesized by Macrogen (Seoul,
Republic of Korea). The relative quantification of mRNA was
measured by LightCycler 96 (Roche, Basel, Switzerland) according to
the manufacturer′s instructions and quantified using LightCycler 96
software version 1.1, comparing with the Ct (threshold cycle)
values of each target gene. The mRNA levels of GAPDH were used for
normalization.
Clonogenic assay
siRNA transfected cells were seeded into three
independent wells of 6-well culture plates (1×103
cells/well) and cultured at 37°C in 5% CO2. Cells were
maintained without medium change to let the viable cells propagate
to sizable colonies for quantification. The colonies were fixed
with methanol and then stained with 0.5% crystal violet for 30 min
at room temperature. The number of colonies formed in each well was
counted under the microscope and statistically analyzed.
Cell cycle analysis
Cells were cultured in 60-mm culture dishes and
harvested at 72 h after siRNA transfection. Cells were washed with
cold PBS, and then fixed 24 h with 70% cold ethanol at −20°C. Cells
were washed with cold PBS again and incubated in a dark condition
with propidium iodide (PI) staining solution containing RNase A (BD
Biosciences, San Diego, CA, USA) for 30 min at room temperature.
The cell cycle was measured by FACSVerse flow cytometry (BD
Biosciences) according to the manufacturer's instructions and
quantified using FlowJo software program.
Detection of apoptosis
Cells were cultured in 60-mm culture dishes and
harvested at 48 and 72 h after siRNA transfection. Apoptotic cells
were stained with Annexin V and PI using FITC Annexin V apoptosis
detection kit I (BD Biosciences) following the manufacturer's
instruction. The cell death was measured by FACSVerse flow
cytometry (BD Biosciences) and quantified using FlowJo software
program.
RNA sequencing
Total RNA was extracted 48 h after siRNA
transfection using the RNeasy mini kit (Qiagen, Valencia, CA, USA).
The quantity of the total RNA was evaluated using RNA
electropherograms (Bio-Rad Experion; Bio-Rad Laboratories,
Hercules, CA, USA); RNA quality was assessed based on the RNA
quality indicator (RQI). The total RNA from each sample with a RQI
value of 8.0 or higher was used. The resulting mRNA samples were
processed for sequencing libraries using the Illumina TruSeq
Stranded mRNA sample preparation kit (Illumina, San Diego, CA, USA)
according to the manufacturer's protocols. RNA sequencing was
performed using the Illumina HiSeq 2500 to generate
non-directional, paired-end 100-base-pair reads. Quality-filtered
reads were mapped to the human reference genome sequence hg19 (UCSC
Genome Bioinformatics, https://genome.ucsc.edu) using TopHat2 (http://ccb.jhu.edu/software/tophat). The relative
transcript abundance was estimated by counting the fragments per
kilo-base of the exon model per million mapped sequence reads
(FPKM), and differentially expressed genes were evaluated using the
cufflinks package (http://cole-trapnell-lab.github.io/cufflinks). The
significantly overlapping pathways and Gene Ontology categories
with differentially expressed genes were analyzed using DAVID
(http://david.abcc.ncifcrf.gov) and IPA
(ingenuity pathway analysis, www.ingenuity.com).
Western blot analysis
Cells were suspended in RIPA buffer (Thermo Fisher
Scientific, Inc., Rockford, IL, USA) containing 0.01% of a protease
and phosphatase inhibitor cocktail (Thermo Fisher Scientific) at 48
h after siRNA transfection. The amount of protein was quantified by
using the Pierce BCA protein assay kit (Thermo Fisher Scientific).
Nuclear-cytoplasmic fractionation was conducted using the NE-PER
Nuclear and Cytoplasmic Extraction Reagents kit (Thermo Fisher
Scientific) according to the manufacturer's protocol. Equal amounts
of total proteins were fractionated by SDS-PAGE on a 10% gel and
transferred to PVDF membranes (Roche, Basel, Switzerland). 0.1%
naphthol blue black (NBB) (Sigma-Aldrich, Seoul, Republic of Korea)
was used to stain PVDF membrane to confirm equal sample loading.
The membrane was blocked with 5% milk/Tris-buffered saline plus
Tween-20 (TBST) and incubated with primary antibodies against human
Id-1 (sc-133104), PARP-1 (sc-8007), NF-κB p65 (sc-372), Lamin B1
(sc-30264), β-actin (sc-47778) (all from Santa Cruz Biotechnology,
Santa Cruz, CA, USA), caspase-3 (#9662; Cell Signaling Technology,
Danvers, MA, USA), Phospho-IκBα (Ser32/36) (#9246; Cell Signaling
Technology), GAPDH (NB600-502; Novus Biologicals LLC, Littleton,
CO, USA), RPL9 (ab182556; Abcam, Cambridge, MA, USA). HRP goat
anti-mouse IgG, HRP goat anti-rabbit IgG and HRP rabbit anti-goat
IgG (Santa Cruz Biotechnology) were used as the secondary
antibodies. Immunoreactive bands were visualized with an LAS-3000
Imager (Fujifilm Corp., Tokyo, Japan).
Tumorigenic assay in nude mice
Tumor formation was assessed to define the effects
of RPL9 silencing on tumorigenicity in vivo. HCT116 cells
were transfected with si-NC or si-RPL9 using Lipofectamine 2000.
Twenty-four hours after transfection, cells were harvested by
trypsinization and then they were washed and resuspended in RPMI
mixed with Matrigel (Corning Incorporated). Cells
(1×106) transfected with si-NC and si-RPL9 were injected
subcutaneously into the left and right flank, respectively, of
4-week old male BALB/c nude mice (Orient Bio, Inc., Seongnam,
Korea). Tumor formation was monitored at 2-day intervals for 14
days after tumor injection. The tumor size was measured using a
vernier caliper and calculated as (width2 × length ×
1/2). The mice were then sacrificed and tumor weight of each mouse
was evaluated. All the animal experiments were performed in
accordance with the guidelines of IACUC (Institutional Animal Care
and Use Committee) in Yonsei University Health System with the
approval number 2015–0066.
Statistical analysis
All the statistical analysis was performed using
Student's t-test, except for in vivo experiment data which
was applied with Bootstrap t-test with 10,000 random repetitions.
All data are shown as means ± SEM. P-values ≤0.05, ≤0.01 and ≤0.001
were considered as statistically significant.
Results
RPL9 knockdown inhibits colorectal cancer
cell proliferation and long-term colony formation
To test the effect of RPL9 knockdown on colorectal
cancer cell survival, we transfected HCT116 and HT29 cells with
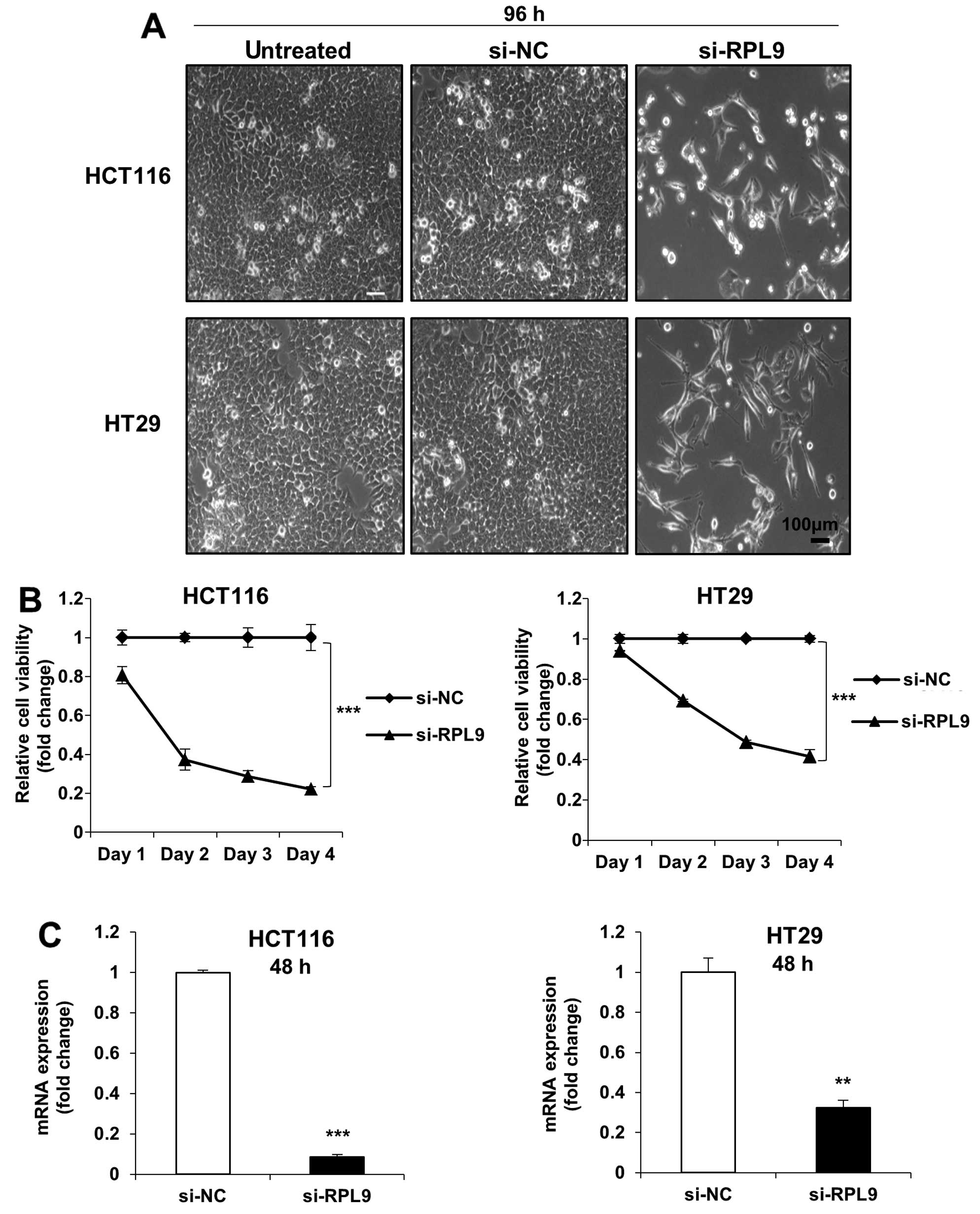
si-NC or si-RPL9. After 96 h, we found from microscopic observation
that the cell viability of RPL9 knockdown cells had decreased
notably (Fig. 1A). In addition, we
observed the maximum growth suppression of ~60–80% in both HCT116
and HT29 cells after 4 days of treatment (Fig. 1B). Concordance with the phenotypic
assay result, si-RPL9 effectively silenced target gene expression
in both the examined tumor cell lines (Fig. 1C). Consistent with the short-term
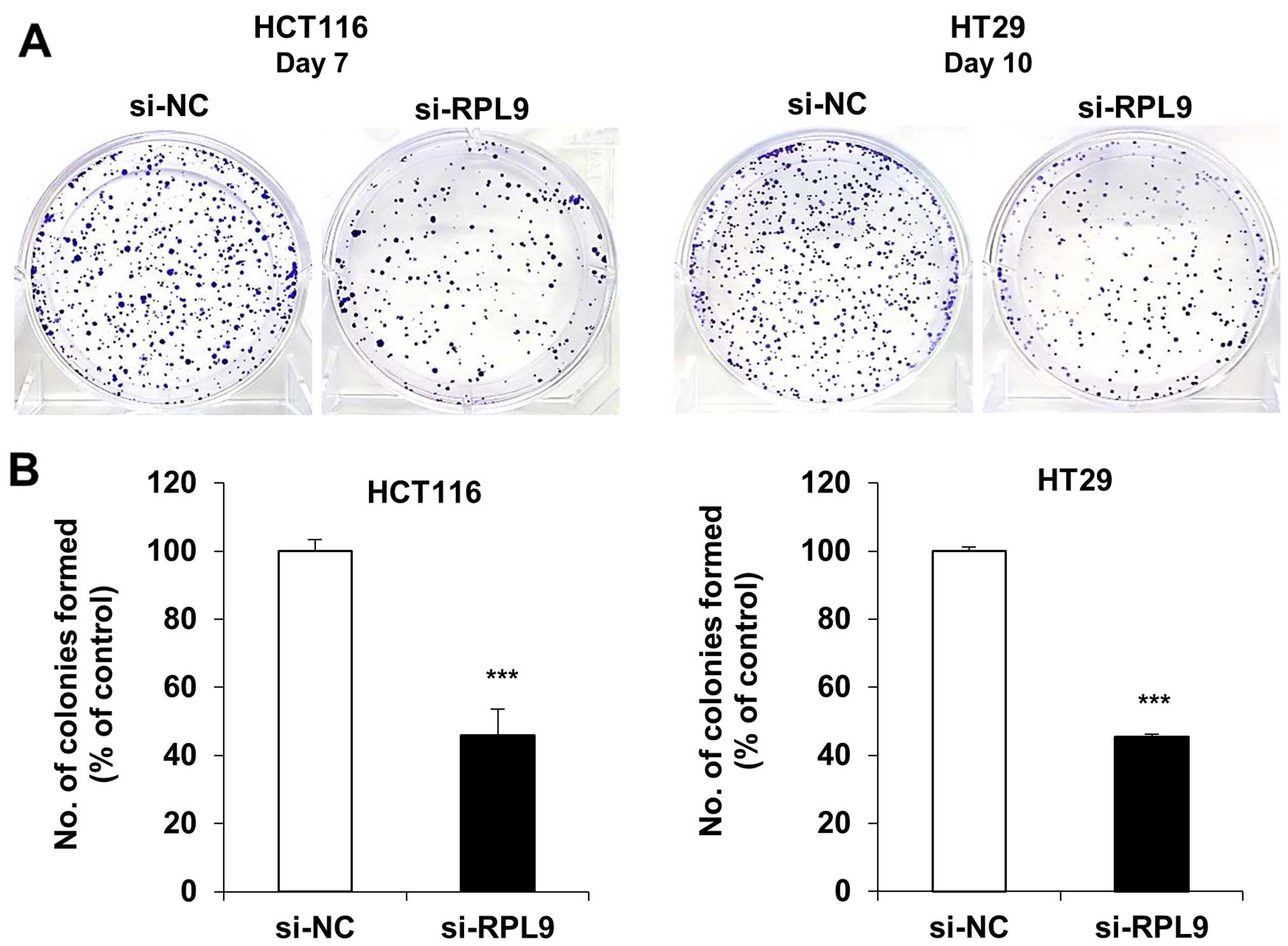
results, RPL9 knockdown effectively suppressed the long-term colony
formation of HCT116 and HT29 cells at day 7 and day 10,
respectively, by ~60% inhibition in both cell lines (Fig. 2). These data indicate that RPL9 is
functionally involved in colorectal cell growth.
Silencing of RPL9 induces apoptosis in
colorectal cancer cells
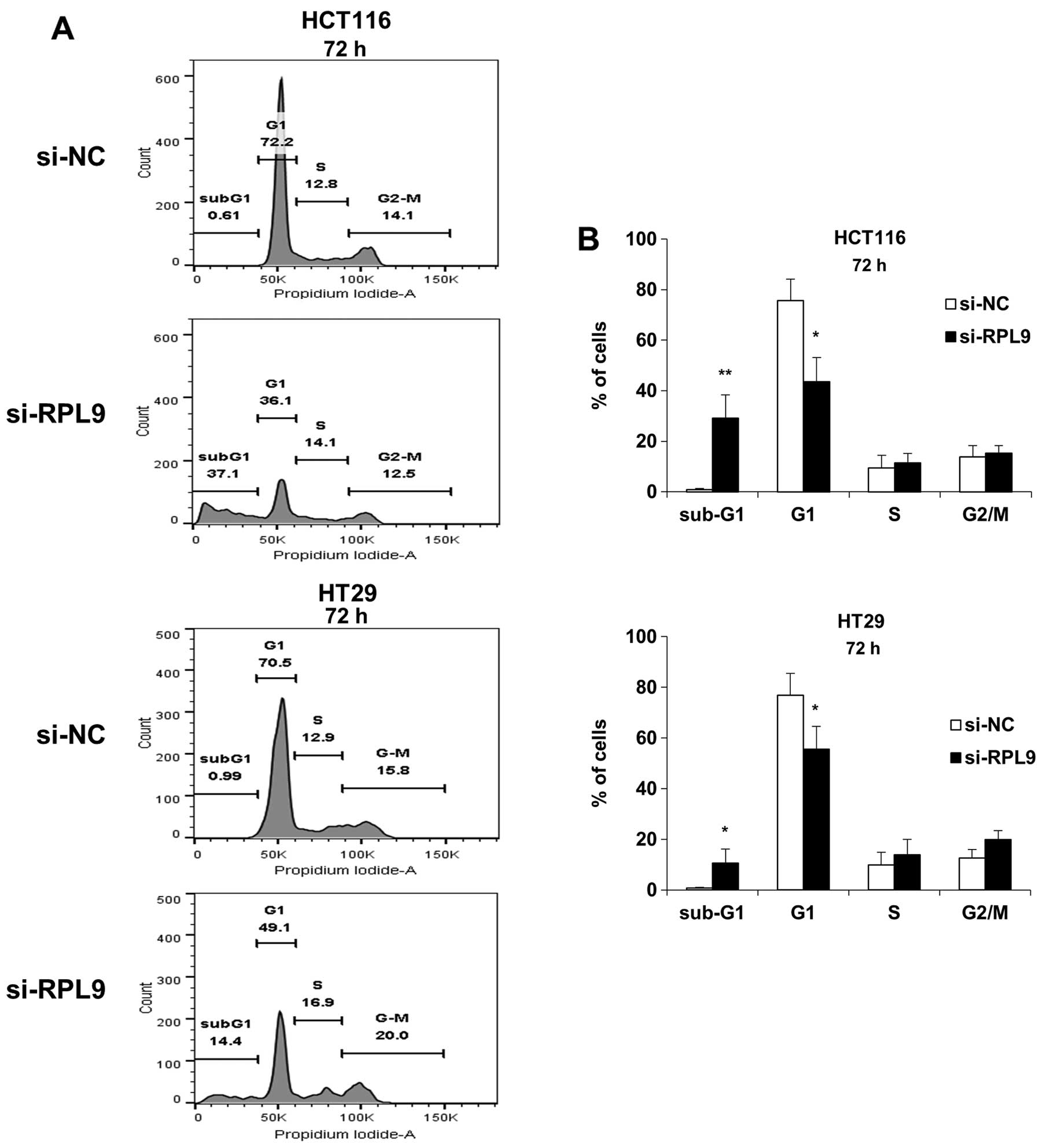
To further investigate the role of RPL9 in
colorectal cancer growth, the effect of RPL9 silencing on cell
cycle progression of HCT116 and HT29 cells were analyzed with flow
cytometry (Fig. 3A). It revealed
that, when compared to the control treatment, RPL9 knockdown cells
showed increased percentage of sub-G1 population and a concomitant
decrease in the G1-phase cells in both HCT116 and HT29 cells
(Fig. 3B). Since sub-G1 population
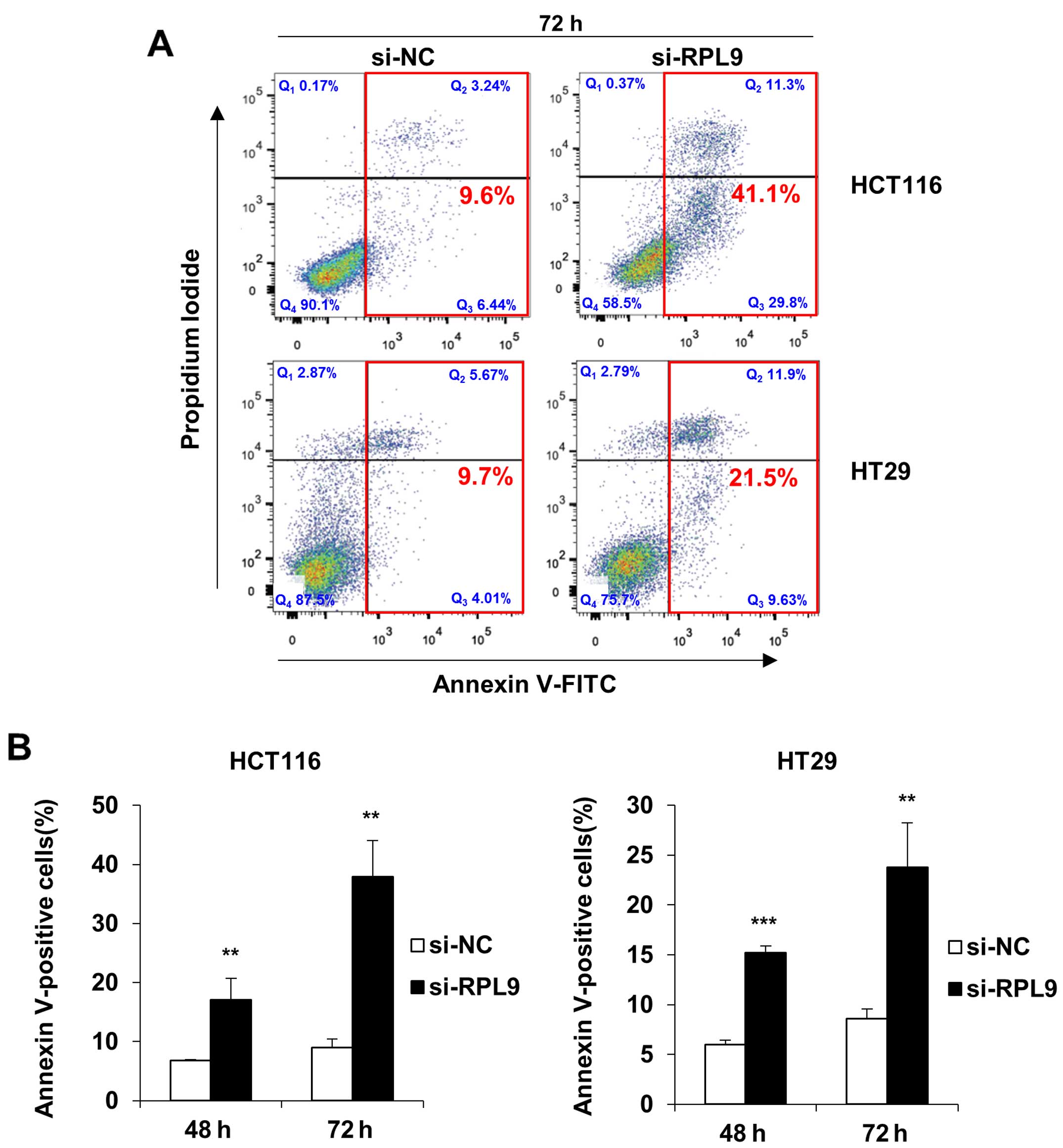
represents apoptotic cells, we then confirmed the phenomenon by
staining CRC cells with the apoptotic marker Annexin V using FACS.
Consequently, we affirmed that siRNA knockdown of RPL9
significantly induced the apoptotic cell death at 72 h after
transfection (Fig. 4A). Moreover,
the percentages of early-apoptotic cell populations (Q3 region)
plus late-apoptotic cell populations (Q2 region) in HCT116 and HT29
cells have increased in both 48 and 72 h after transfection
(Fig. 4B). Early apoptotic cells
are considered as cells that have intact plasma membrane which
expose phosphatidylserine (PS) on the surface and late apoptotic
cells have permeabilized plasma membrane (16). These results suggest that RPL9
knockdown induces apoptosis in colorectal cancer cells.
The function of RPL9 correlates with
Id-1/NF-κB signaling
To explore the molecular basis of the growth
inhibition caused by RPL9 silencing, we compared the global gene
expression profiles of RPL9 knockdown HCT116 and HT29 cells to
those of control cells transfected with si-NC using next-generation
RNA sequencing. The global gene expression analysis, defined as at
least a 2.0-fold change, revealed that RPL9-specific knockdown
resulted in the upregulation and downregulation of 918 RNA
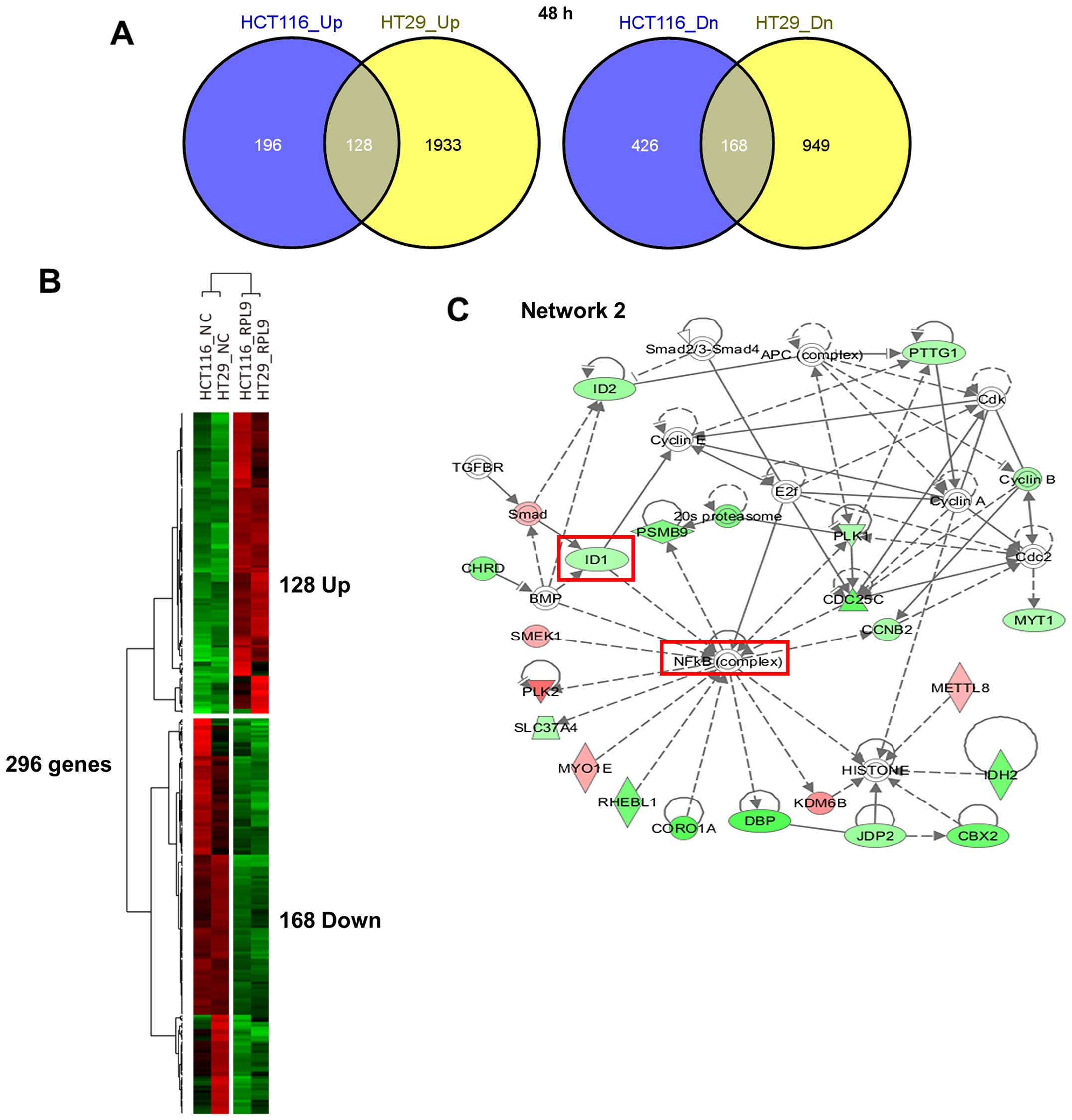
transcripts in HCT116 and 3178 transcripts in HT29 cells (Fig. 5A). Comparing these two gene sets
generated a statistically significant overlap of 296 genes (128
upregulated and 168 downregulated genes) which are considered as a
common RPL9 knockdown signature (Fig.
5B). Subsequent IPA showed that the 269 mRNA transcripts were
functionally enriched in the top five networks (Table I). In particular, as shown in
Fig. 5C, we found that the
expression level of Id-1, which is involved in carcinogenesis
(17) and NF-κB activation
(18), was downregulated in mRNA
level by RPL9 silencing. Consistent with our observation, it was
previously demonstrated that Id-1 silenced colorectal cancer cells
resulted in decreased proliferation and increased apoptotic rate
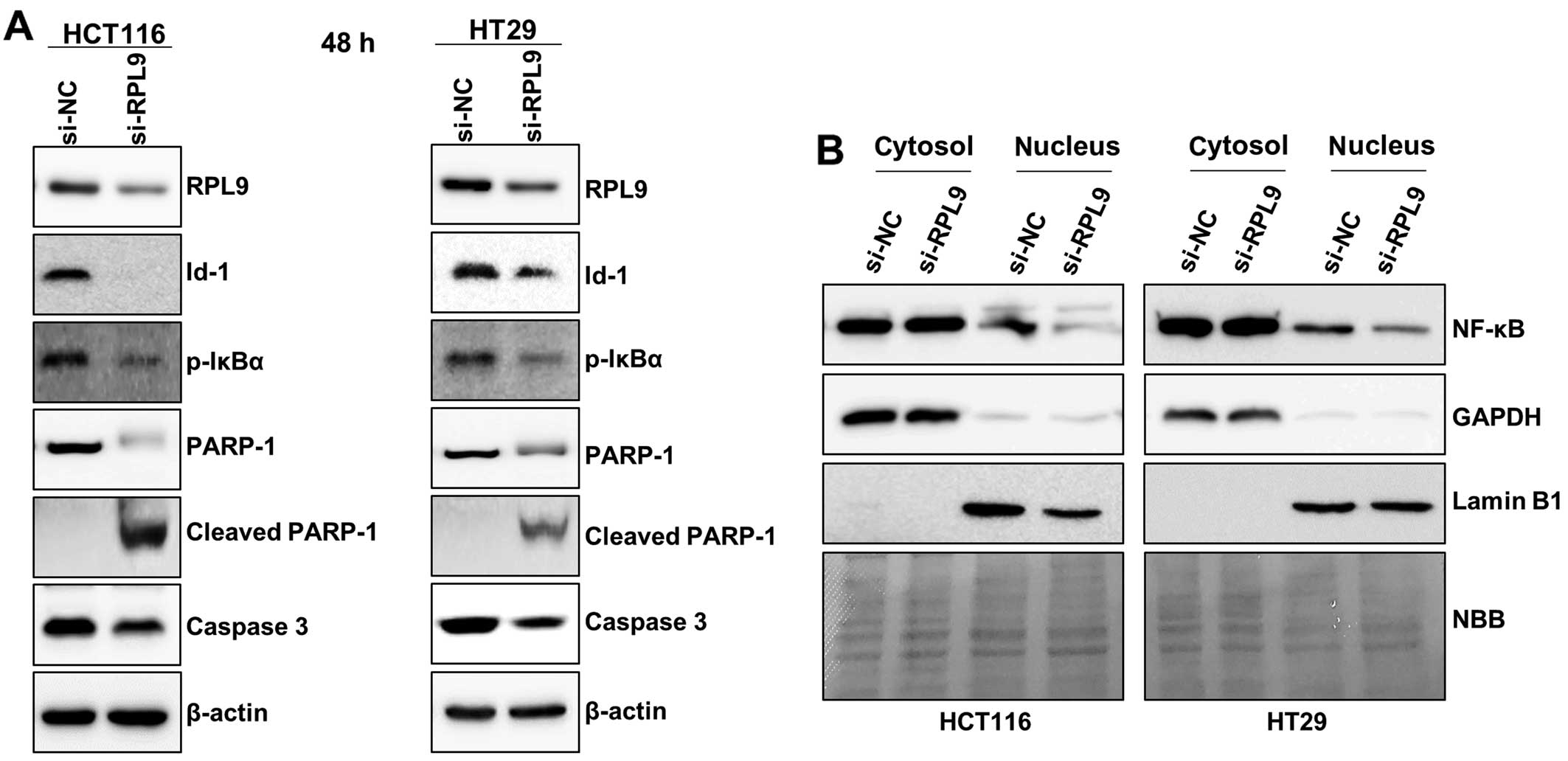
(15). Subsequent western blot
analysis confirmed that the protein level of Id-1 was decreased in
RPL9 knockdown HCT116 and HT29 cells when compared to the control
(Fig. 6A). In addition, a
concomitant decrease in pro-caspase 3, which activate apoptotic
signaling, and PARP-1, a nuclear enzyme essential for genomic
stability, were observed (Fig.
6A). Meanwhile, IκBα is an inhibitory protein that binds with
NF-κB in the cytosol making NF-κB into an inactivated state.
However, when IκBα becomes phosphorylated it dissociates from
NF-κB. Then the activated NF-κB translocates into the nucleus where
it binds to the specific sequences of DNA causing changes in cell
function such as promoting cell survival (19). We observed that the protein level
of p-IκBα is decreased by RPL9 silencing (Fig. 6A), and NF-κB protein level also
decreased in nucleic fraction of the RPL9 knockdown colorectal
cancer cells (Fig. 6B). Since the
expression level of housekeeping proteins may vary depending on the
experimental condition (20,21),
equal sample loading was confirmed by naphthol blue black staining
of the western blot membrane (Fig.
6B). These findings indicate that the function of RPL9 is
associated with Id-1/NF-κB signaling pathway in colorectal cancer
cells.
 | Table ITop 5 gene networks from Ingenuity
Pathway Analysis. |
Table I
Top 5 gene networks from Ingenuity
Pathway Analysis.
| Network ID | Score | # of focus
genes | Genes in network
and their top functions |
|---|
| 1 | 54 | 30 | FBLN1↓,
LAMC2↑, LAMB3↑, LAMA3↑, ERK1/2, DGKA↑,
GLIPR2↓, NUCB2↓, TRIB1↑, VLDL-cholesterol,
ACAT2↓, RAB26↓, ITGA7↓, SHB↑,
AKAP12↑, FHL1↓, SMAD1/5, BCAR3↑, atypical
protein kinase C, Sos, TIPARP↑, DAP↑, DAPK2↓,
CA2↓, WWC1↑, AURK↓, AURKB↓,
KLHL21↑, KIF20A↓, MAD2L1↓, KIF2C↓,
NDC8O↓, SPC24↓, NUF2↓, SPC25↓
Cell cycle, cellular assembly and organization, DNA replication,
recombination and repair |
| 2 | 33 | 24 | TGFBR,
Smad↑, ID1↓, NFkb (complex), CHRD↓, BMP,
ID2↓, cyclin E, SMEK1↑, PLK2↑,
SLC37A4↓, MY01E↑, RHEBL1↓, COR01A↓,
DBP↓, JDP2↓, CBX2↓, KDM6B↑, HISTONE,
IDH2↓, METTL8↑, CCNB2↓, CDC25C↓,
PLK1↓, 20s proteasome↓, PSMB9↓, E2f,
Smad2/3-Smad4, APC (complex), PTTG1↓, Cdk, cyclin B↓,
cyclin A, Cdc2, MYT1↓
Cell cycle, DNA replication, recombination and repair, organismal
survival |
| 3 | 30 | 23 | IFRD1↑,
DES↓, Pak, ARHGEF6↓, Dynamin, ERK, lgG2a, Alpha
tubulim, KIF22↓, TMEM173↓, STMN1↓,
ZFP36L1↑, CHAD↓, LIMA1↑, G-Actin↑,
MKNK2↑, CaMKII, NFAT (complex), SDC4↑, Nfat (family),
KLF6↑, Fcer1, calcineurin protein(s), DUSP4↑, Cg,
MAP2K1/2, PDGFA↑, CYR61↑, GUSB↓,
HERPUD1↓, CTSV↓, DNASE2↓, H2AFZ↓,
HPSE↓, CENPA↓
Organismal functions, organismal injury and abnormalities, tissue
morphology |
| 4 | 27 | 22 | NRGN↓,
thymidine kinase↓, Rxr, FABP6↓, Rar, ITGB3BP↓,
CRABP2↓, T3-TR-RXR, SLC01B3↑, Nr1h, PEPCK↓,
PCK2↓, C/ebp, GOT, ANGPTL4↑, Growth hormone,
PPP1R15A↑, STAT5a/b, JINK1/2, thyroid hormone receptor,
UCP2↓, Cbp/p300, PTPase↓, SLC22A17↓,
ACAP1↑, Akt, ATF3↑, LOC102724788/PRODH↓,
SEMA7A↑, PLAGL1↑, death receptor↑,
TNFRSF10B↑, TP5313↑, TNFRSF1D↑, HDL
cholesterol
Cell death and survival, lipid metabolism, molecular transport |
| 5 | 25 | 22 | MMP17↓,
Fibrinogen, Ck2, LDL↓, PSRC1↓, PCSK9↓, Pdgf
(complex)↑, Ap1, F3↑, Creb, PTPN6↓,
phosphatase↓, RNASEH2A↓, PTPRM↑,
EPHX2↓, PPF1A4↓, Gsk3, Creb, Ap1, FSH,
DUSP14↑, CFLAR↑, Pkc(s), Pka, Insulin,
ATP2A3↓, 14-3-3, PP2A, MIDN↑, Histone H1↓,
H1FX↓, PLC↓, ADRB, Lh, PTP4A1↑,
LOC81691↓, CRYM↓
Neurological disease, psychological disorders, post-translational
modification |
Silencing of RPL9 delays tumor growth
kinetics in vivo
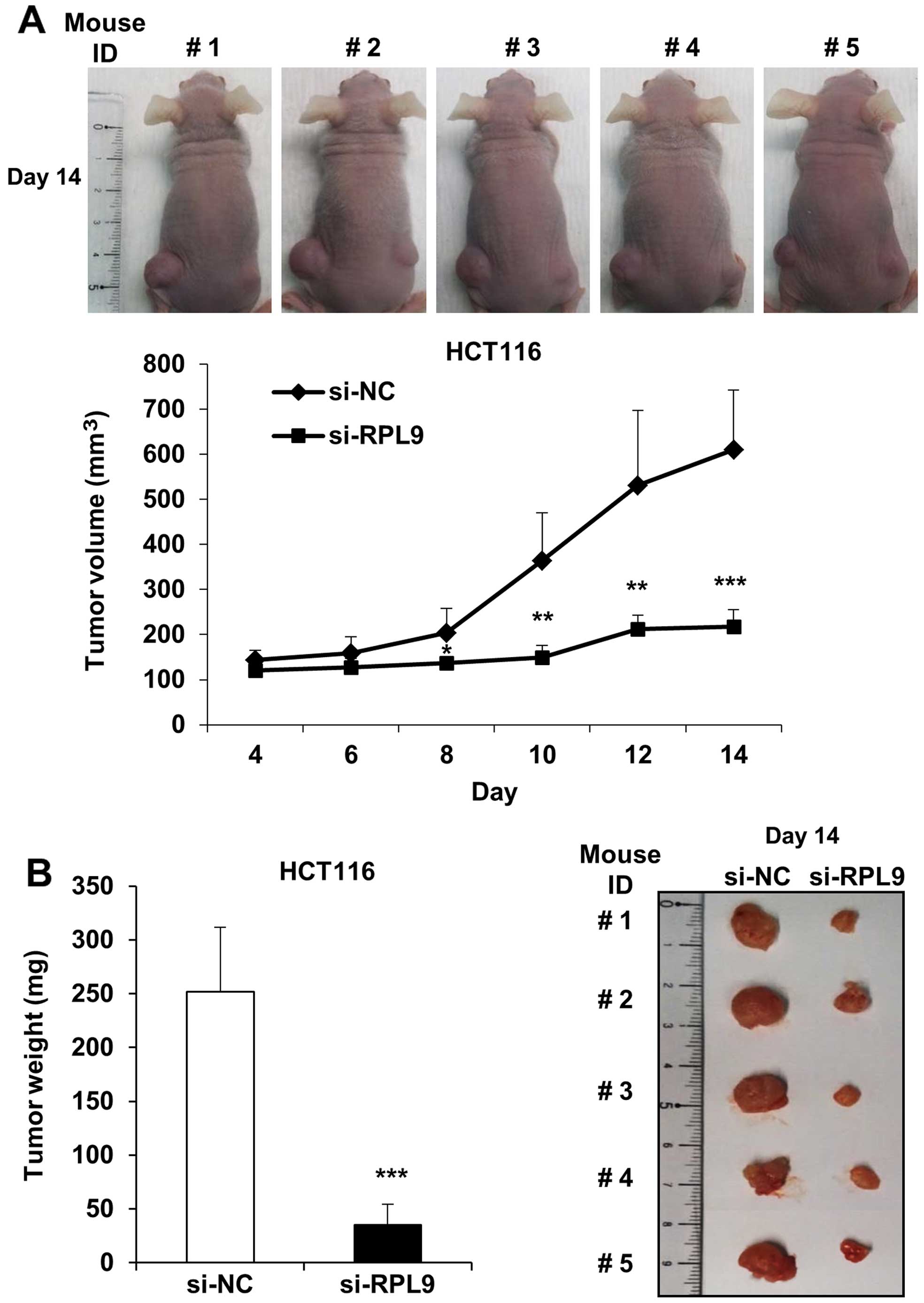
To further validate the effects of RPL9 knockdown on
tumorigenesis of colorectal cancer, tumor formation assay was
performed in BALB/c nude mice. si-NC and si-RPL9 transfected HCT116
cells were subcutaneously inoculated into the left and right flank,
respectively. Tumor sizes were measured in 2-day interval for 14
days. Eight days after the inoculation, tumor sizes between the
control and RPL9 knock-down showed definite difference and that the
control tumor sizes were larger (P<0.05) (Fig. 7A). After 14 days, a significant
difference (P<0.001) was apparent in size and weight between the
control and RPL9 knock-down (Fig.
7). The results show that silencing of RPL9 significantly
suppresses the growth of human CRC xenografts in vivo.
Discussion
Ribosomal proteins are known as the components of
ribosomes which play a role in protein biosynthesis. However, in
many types of human cancer cells, some of these ribosomal proteins
are ectopically expressed compared to normal cells. For example,
the mRNA expression of S8, L12, L23a, L27 and L30 ribosomal
proteins was upregulated in human hepatocellular carcinoma
(22). RPL34 was overexpressed in
non-small cell lung cancer cells (23), and RPS13 was found to promote the
growth and cell cycle progression of gastric cancer cells through
inhibiting p27Kip1 expression (9). Reversely, it was demonstrated that
RPS29 induces apoptosis in non-small cell lung cancer cells by
downregulating Bcl-2, Bcl-XL and survivin and upregulating p53 and
Bax (24). Recent evidence
suggests that ribosomal proteins are not only involved in the
protein synthesis but also in the development of cancer, and it is
likely that these proteins exhibit secondary functions called
extraribosomal functions (25).
In colorectal cancer cells, the overexpression of
ribosomal protein L9 was found (5,13,14),
yet, there is no report on the function of RPL9 in human cancers.
In this study we aimed at finding the extraribosomal function of
RPL9 in colorectal cancer cells. We used RNAi system to knockdown
RPL9 and observed strong inhibition in cell proliferation and
colony formation of HCT116 and HT29, which demonstrates that RPL9
is important for the growth of colorectal cancer. Also, we found an
increased sub-G1 population in cell cycle analysis and likewise, a
significant increase of apoptotic cells. Furthermore, in
vivo mouse model showed that silencing of RPL9 delayed tumor
growth. The global gene expression profiles of RPL9 knockdown
HCT116 and HT29 cells to those of control cells by next-generation
RNA sequencing showed common knockdown signature of 296 genes.
These data suggest that RPL9 is differentially involved in protein
synthesis, but does not influence entire cellular protein synthesis
in a non-specific manner. Therefore, silencing of RPL9 does not end
up downregulating the protein synthesis as a whole but it is rather
gene-specific and protein-specific (26).
Of all the genes that were dysregulated by RPL9
knockdown, we focused on finding the particular molecule that might
be involved in inducing apoptosis in colorectal cancer cells. Thus,
in network 2 of the IPA analysis, we found an oncogenic Id-1
(15) was downregulated. Id-1, a
helix-loop-helix (HLH) protein, is known to functionally involve in
cell growth, senescence and differentiation. Increased Id-1
expression has been found in over 20 types of human cancers
including colorectal (27), breast
(28), prostate (29) and cervical cancers (30). Many studies suggested that Id-1
plays a key role in cancer progression and associated it as a
prognostic marker. According to a study, Id-1 knockdown colorectal
cancer cells were decreased in proliferation, migration and
invasion and increased in apoptosis rate (15). Therefore, overexpression of Id-1
protects cancer cells against apoptosis, but decreasing the Id-1
expression would lead cancer cells to apoptosis. We found by
western blotting that the protein level of Id-1 was decreased in
RPL9 knockdown HCT116 and HT29 cells compared to the control. This
finding shows that silencing of RPL9 decreased Id-1 protein
expression in a mechanism we have not explored and may have induced
apoptosis in CRC cells. In addition, Id-1 has been shown to promote
cell survival by activating PI3K/Akt/NF-κB signaling pathway in
esophangeal cancer and it has been suggested that it may be one of
the upstream regulators of NF-κB (18,31).
NF-κB is a transcription factor that translocates the nucleus and
mediates the transcription of a vast array of proteins involved in
cell survival and proliferation, and anti-apoptotic factors
(32). Hence, we investigated the
change in NF-κB activity in RPL9 knockdown HCT116 and HT29 cells
and observed that protein level of p-IκBα was decreased and NF-κB
protein level decreased in the nuclear fraction of RPL9 silenced
cells. These results reveal that the downregulation of RPL9 cause
inactivation of Id-1 and NF-κB, which leads to apoptosis of
colorectal cancer cells.
In Fig. 6B, Lamin
B1, a nuclear loading control, shows slight decrease in RPL9
knockdown HCT116 cell. The reason for this may be that Lamin B1 is
involved in apoptosis and often cleaved by caspases or becomes
degraded (33,34). Depending on the experimental
conditions, the expression level of a particular housekeeping gene
may change. Hence, there are studies that argue that reversible
protein staining dye can be used advantageously over housekeeping
protein detection for quality or equal loading control in western
blotting (20,21). Therefore, we stained the
transferred membrane with naphthol blue black and confirmed equal
sample loading of western blotting.
In the present study, we demonstrated for the first
time that silencing of RPL9 by RNAi suppresses the proliferation
and induces apoptosis in colorectal cancer cells by in vitro
and in vivo assays. Also, we found that the downregulation
of RPL9 induces the inactivation of Id-1/NF-κB signaling axis.
Taken together, our findings suggest that RPL9 may play an
important role in promoting the malignant growth of colorectal
cancer cells and that targeting of RPL9 might be an attractive
option for exploiting a next-line molecular therapy of colorectal
cancer.
Acknowledgements
The present study was financially supported by the
grants from the Korean Health Technology R&D Project, Ministry
of Health & Welfare (A121982) and the Mid-career Researcher
Program of the Ministry of Education, Science and Technology
(NRF-2014R1A2A1A11054162), Republic of Korea. This study was also
supported by the National Research Foundation of Korea (NRF) Grant
funded by the Korean Government (MSIP) (NRF-2014R1A5A2010008).
References
|
1
|
Marmot SM, Atinmo T, Byers T, Chen J,
Hirohata T, Jackson A, James WPT, Kolonel L, Kumanyika S, et al:
Food, Nutrition, Physical Activity, and the Prevention of Cancer: A
Global Perspective. WCRF/AICR report. pp. 280–288. 2007
|
|
2
|
Benson AB III, Venook AP, Bekaii-Saab T,
Chan E, Chen YJ, Cooper HS, Engstrom PF, Enzinger PC, Fenton MJ,
Fuchs CS, et al; National Comprehensive Cancer Network. Colon
cancer, version 3.2014. J Natl Compr Canc Netw. 12:1028–1059.
2014.PubMed/NCBI
|
|
3
|
Sjöblom T, Jones S, Wood LD, Parsons DW,
Lin J, Barber TD, Mandelker D, Leary RJ, Ptak J, Silliman N, et al:
The consensus coding sequences of human breast and colorectal
cancers. Science. 314:268–274. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Taylor DJ, Devkota B, Huang AD, Topf M,
Narayanan E, Sali A, Harvey SC and Frank J: Comprehensive molecular
structure of the eukaryotic ribosome. Structure. 17:1591–1604.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lai MD and Xu J: Ribosomal proteins and
colorectal cancer. Curr Genomics. 8:43–49. 2007. View Article : Google Scholar
|
|
6
|
Fisher EM, Beer-Romero P, Brown LG, Ridley
A, McNeil JA, Lawrence JB, Willard HF, Bieber FR and Page DC:
Homologous ribosomal protein genes on the human X and Y
chromosomes: Escape from X inactivation and possible implications
for Turner syndrome. Cell. 63:1205–1218. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Draptchinskaia N, Gustavsson P, Andersson
B, Pettersson M, Willig TN, Dianzani I, Ball S, Tchernia G, Klar J,
Matsson H, et al: The gene encoding ribosomal protein S19 is
mutated in Diamond-Blackfan anaemia. Nat Genet. 21:169–175. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Warner JR and McIntosh KB: How common are
extraribosomal functions of ribosomal proteins? Mol Cell. 34:3–11.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Guo X, Shi Y, Gou Y, Li J, Han S, Zhang Y,
Huo J, Ning X, Sun L, Chen Y, et al: Human ribosomal protein S13
promotes gastric cancer growth through down-regulating
p27Kip1. J Cell Mol Med. 15:296–306. 2011. View Article : Google Scholar
|
|
10
|
Gou Y, Shi Y, Zhang Y, Nie Y, Wang J, Song
J, Jin H, He L, Gao L, Qiao L, et al: Ribosomal protein L6 promotes
growth and cell cycle progression through upregulating cyclin E in
gastric cancer cells. Biochem Biophys Res Commun. 393:788–793.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yang HJ, Youn H, Seong KM, Jin YW, Kim J
and Youn B: Phosphorylation of ribosomal protein S3 and
antiapoptotic TRAF2 protein mediates radioresistance in non-small
cell lung cancer cells. J Biol Chem. 288:2965–2975. 2013.
View Article : Google Scholar :
|
|
12
|
Eid R, Sheibani S, Gharib N, Lapointe JF,
Horowitz A, Vali H, Mandato CA and Greenwood MT: Human ribosomal
protein L9 is a Bax suppressor that promotes cell survival in
yeast. FEMS Yeast Res. 14:495–507. 2014. View Article : Google Scholar
|
|
13
|
Zhang XL, Gao F, Li W, Tang WZ and Zhang
S: Serial analysis of gene expression in adenocarcinoma samples and
normal colonic mucosa in a Chinese population. Genet Mol Res.
14:12903–12911. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Sim EUH, Bong IPN, Balraj P, Tan SK, Jamal
R, Sagap I, Nadeson S, Rose IM and Lim PKM: A preliminary study of
differentially expressed genes in Malaysian colorectal carcinoma
cases. J Biosci. 17:19–37. 2006.
|
|
15
|
Lai X, Liao J, Lin W, Huang C, Li J, Lin
J, Chen Q and Ye Y: Inhibitor of DNA-binding protein 1 knockdown
arrests the growth of colorectal cancer cells and suppresses
hepatic metastasis in vivo. Oncol Rep. 32:79–88. 2014.PubMed/NCBI
|
|
16
|
Poon IK, Hulett MD and Parish CR:
Molecular mechanisms of late apoptotic/necrotic cell clearance.
Cell Death Differ. 17:381–397. 2010. View Article : Google Scholar
|
|
17
|
Meteoglu I, Meydan N and Erkus M: Id-1:
Regulator of EGFR and VEGF and potential target for colorectal
cancer therapy. J Exp Clin Cancer Res. 27:692008. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ling MT, Wang X, Ouyang XS, Xu K, Tsao SW
and Wong YC: Id-1 expression promotes cell survival through
activation of NF-kappaB signalling pathway in prostate cancer
cells. Oncogene. 22:4498–4508. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Gilmore TD: Introduction to NF-kappaB:
Players, pathways, perspectives. Oncogene. 25:6680–6684. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Aldridge GM, Podrebarac DM, Greenough WT
and Weiler IJ: The use of total protein stains as loading controls:
An alternative to high-abundance single-protein controls in
semi-quantitative immunoblotting. J Neurosci Methods. 172:250–254.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Romero-Calvo I, Ocón B, Martínez-Moya P,
Suárez MD, Zarzuelo A, Martínez-Augustin O and de Medina FS:
Reversible Ponceau staining as a loading control alternative to
actin in Western blots. Anal Biochem. 401:318–320. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kondoh N, Shuda M, Tanaka K, Wakatsuki T,
Hada A and Yamamoto M: Enhanced expression of S8, L12, L23a, L27
and L30 ribosomal protein mRNAs in human hepatocellular carcinoma.
Anticancer Res. 21(4A): 2429–2433. 2001.PubMed/NCBI
|
|
23
|
Yang S, Cui J, Yang Y, Liu Z, Yan H, Tang
C, Wang H, Qin H, Li X, Li J, et al: Over-expressed RPL34 promotes
malignant proliferation of non-small cell lung cancer cells. Gene.
576:421–428. 2016. View Article : Google Scholar
|
|
24
|
Khanna N, Sen S, Sharma H and Singh N: S29
ribosomal protein induces apoptosis in H520 cells and sensitizes
them to chemotherapy. Biochem Biophys Res Commun. 304:26–35. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Naora H and Naora H: Involvement of
ribosomal proteins in regulating cell growth and apoptosis:
Translational modulation or recruitment for extraribosomal
activity? Immunol Cell Biol. 77:197–205. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Bee A, Brewer D, Beesley C, Dodson A,
Forootan S, Dickinson T, Gerard P, Lane B, Yao S, Cooper CS, et al:
siRNA knockdown of ribosomal protein gene RPL19 abrogates the
aggressive phenotype of human prostate cancer. PLoS One.
6:e226722011. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zhao ZR, Zhang ZY, Zhang H, Jiang L, Wang
MW and Sun XF: Overexpression of Id-1 protein is a marker in
colorectal cancer progression. Oncol Rep. 19:419–424.
2008.PubMed/NCBI
|
|
28
|
Lin CQ, Singh J, Murata K, Itahana Y,
Parrinello S, Liang SH, Gillett CE, Campisi J and Desprez PY: A
role for Id-1 in the aggressive phenotype and steroid hormone
response of human breast cancer cells. Cancer Res. 60:1332–1340.
2000.PubMed/NCBI
|
|
29
|
Ouyang XS, Wang X, Lee DT, Tsao SW and
Wong YC: Over expression of ID-1 in prostate cancer. J Urol.
167:2598–2602. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Schindl M, Oberhuber G, Obermair A,
Schoppmann SF, Karner B and Birner P: Overexpression of Id-1
protein is a marker for unfavorable prognosis in early-stage
cervical cancer. Cancer Res. 61:5703–5706. 2001.PubMed/NCBI
|
|
31
|
Li B, Cheung PY, Wang X, Tsao SW, Ling MT,
Wong YC and Cheung AL: Id-1 activation of PI3K/Akt/NFkappaB
signaling pathway and its significance in promoting survival of
esophageal cancer cells. Carcinogenesis. 28:2313–2320. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Vlahopoulos SA, Cen O, Hengen N, Agan J,
Moschovi M, Critselis E, Adamaki M, Bacopoulou F, Copland JA,
Boldogh I, et al: Dynamic aberrant NF-κB spurs tumorigenesis: A new
model encompassing the microenvironment. Cytokine Growth Factor
Rev. 26:389–403. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Broers JL and Ramaekers FC: The role of
the nuclear lamina in cancer and apoptosis. Adv Exp Med Biol.
773:27–48. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Goldman RD, Gruenbaum Y, Moir RD, Shumaker
DK and Spann TP: Nuclear lamins: Building blocks of nuclear
architecture. Genes Dev. 16:533–547. 2002. View Article : Google Scholar : PubMed/NCBI
|