Introduction
Gallbladder cancer (GBC) is one of the
intra-abdominal malignant tumors with the most unfavorable
prognosis (5-year overall survival rate of <10%) (1,2). The
only potentially curative therapy for GBC is surgical resection,
including simple cholecystectomy, radical or extended
cholecystectomy (sometimes accompanied by liver resection), bile
duct resection, and pancreaticoduodenectomy. However, few patients
undergo curative surgery because of distant metastasis or locally
excessive invasion. Understanding the mechanisms of metastasis of
GBC is, therefore, important to improve the survival of patients
with GBC.
CD44 (also called H-CAM) is a type I transmembrane
glycoprotein receptor that binds hyaluronic acid (3–5). The
functions of CD44 include cellular adhesion, lymphocyte activation,
homing, and migration. Further, CD44 can contribute to tumor
progression, drug resistance, and cancer stem cell function
(6). The gene encoding CD44
comprises 20 exons, with 10 in the variant region that generate
isoforms through alternative splicing (6,7). The
standard isoform is encoded by exons 1–5 and exons 16–20 and is the
isoform with the smallest molecular weight. Seven of the standard
exons encode the extracellular region, another exon is the
transmembrane region, and other two domains comprise the
cytoplasmic tail. Variant exons (exon 6–15, called as v1-v10) are
alternatively spliced and inserted into the extracellular standard
exons to generate variant isoforms. Accordingly, standard and
variant isoforms harbor the same antigenic region.
CD44 variant 9 (CD44v9) is associated with the
proliferation of cancer cells, inhibition of apoptosis,
chemoresistance, EMT, and poor prognosis. CD44v9 expression is
associated with the recurrence of primary early gastric cancer
(8) as well as with the poor
prognosis of patients with pancreatic cancer (9) or hepatocellular carcinoma (10). The CD44 variant isoform comprising
exons v8–10 promotes the uptake of cystine for synthesis of the
major antioxidant glutathione and contributes to the elimination of
excess reactive oxygen species (11), which increases resistance to
chemotherapy and radiotherapy. CD44v9 and CD44v6 interact with FAS,
a programmed cell death receptor that inhibits apoptosis induced by
its ligand FASL (12,13). Cancer cells that express CD44v9
resist chemotherapy by expressing the multidrug resistance protein
1 (14,15).
CD44 expression affects tumorigenicity or the EMT of
GBC cells; however, there are few reports on whether CD44 isoforms
are associated with these characteristics (16–18).
To fill this gap in our knowledge, the aim of the present study was
to investigate the relationship between the CD44 isoforms expressed
by the GBC cells with EMT, chemotaxis, and tumorigenicity.
Materials and methods
Human GBC cell line and culture
conditions
The human GBC cell line NOZ (19) was purchased from the Japanese
Collection of Research Bioresources (Osaka, Japan). NOZ cells were
cultured in a humidified atmosphere containing 5% CO2 at
37°C in DMEM/Ham's F-12 medium (Wako, Osaka, Japan) containing 10%
fetal bovine serum (FBS) (Gibco/Thermo Fisher Scientific, Waltham,
MA, USA) and 1% Antibiotic-Antimycotic 100× (Life Technologies,
Carlsbad, CA, USA).
Flow cytometry and cell sorting
Cell surface proteins were detected using an
allophycocyanin (APC)-conjugated anti-human CD44 monoclonal
antibody (clone BD105, catalog number 130-095-177; Miltenyi Biotec
GmbH, Bergisch Gladbach, Germany), a fluorescein isothiocyanate
(FITC)-conjugated anti-human CD44 standard monoclonal antibody
(CD44std; clone SFF-2, catalog number BMS113FI; Affymetrix Inc.,
Santa Clara, CA, USA), and an anti-human CD44v9 rat monoclonal
primary antibody (clone RV3, catalog number LKG-M001; Cosmo Bio
Co., Ltd., Tokyo, Japan). The secondary antibody used to detect
CD44v9 was an APC-conjugated anti-rat IgG2a mouse monoclonal clone
RG7/1.30 (catalog number 130-104-736; Miltenyi Biotec GmbH).
A FACS Aria II cell sorter and BD FACS Diva software
(BD Biosciences, San Jose, CA, USA) were used for cell sorting.
Monolayers of NOZ cells were dissociated using 0.025% Trypsin
containing 0.01% EDTA (Life Technologies) and suspended in PBS
containing 0.5% BSA. Cells were reacted with FITC or APC-conjugated
antibodies according to the manufacturer's instructions. These
cells were also stained with 7-aminoactinomycin D (7-AAD; Bio-Rad,
Richmond, CA, USA) to exclude dead cells. All experiments were
repeated at least five times. Cells were sorted to isolate
populations of CD44std+-CD44v9− cells (CD44s)
and CD44std−-CD44v9+ cells (CD44v), which
were each cultured to confirm viability and proliferation. The
sorted cells were used in the following study. To analyze
sequential expression of cell surface markers, cells were reacted
with primary antibodies and re-sorted after 1 or 2 weeks of
culture, which was repeated three times using the same
procedure.
Transwell migration assay and invasion
assay
The protocol used to analyze in vitro cell
migration was based on the transwell migration assay (Boyden
chamber assay) (20,21). Falcon cell culture inserts (Corning
Inc., Corning, NY, USA) with a porous membrane (pore-size 8
μm) were placed into a 24-well plate (Corning Inc.). The
attractant (medium containing 10% FBS) was added to the lower
chamber, and sorted NOZ cells (50,000 cells) were suspended in
serum-free medium and added to the upper chamber. The cells were
incubated at 37°C in an atmosphere containing 5% CO2 for
24 h. The migrated cells attached to the lower side of the membrane
were fixed and stained using a Differential Quik Stain kit
(Polysciences, Warminster, PA, USA). Cells remaining on the upper
side of membrane were removed with a cotton swab. The membranes
containing the migrated cells were dried, and the cells were
counted in three randomly selected fields (×200). The experiments
were performed in triplicate, and each experiment was independently
repeated three times.
Analysis of cell invasion was performed using a
Matrigel Transwell invasion assay (22). Cell culture inserts with porous
membranes (pore-size 8 μm) were coated overnight with 0.5
mg/ml Matrigel (Corning Inc.). The inserts were placed into a
24-well plate and the attractant (medium containing 10% FBS) was
added to the lower chamber and sorted NOZ cells (50,000 cells) were
suspended in serum-free medium and added to the upper chamber. The
cells were incubated at 37°C in an atmosphere containing 5%
CO2 for 24 h. The invaded cells attached to the lower
side of the membrane were fixed and stained using the Differential
Quik Stain kit (Polysciences). Cells remaining on the upper side of
the membrane were removed with a cotton swab, and the membranes
were dried. The number of invaded cells was counted in three
randomly selected fields (×200). The experiments were performed in
triplicate, and each experiment was independently repeated three
times.
RNA extraction and quantitative real-time
PCR
Total RNA was extracted using a NucleoSpin RNA
column (Takara Bio Inc., Shiga, Japan), and cDNA was synthesized
using a PrimeScript II 1st Strand cDNA Synthesis kit (Takara Bio)
according to manufacturer's instructions. Quantitative real-time
PCR was performed using SYBR Premix Ex Taq II (Takara Bio) and an
Mx3000P real-time qPCR system (Agilent Technologies, Santa Clara,
CA, USA). The ΔΔCt method was used to calculate mRNA levels
(23). Glyceraldehyde-3-phosphate
dehydrogenase (GAPDH) mRNA was used as an internal control. Each
experiment was performed in triplicate and independently repeated
three times. The primer sequences were as follows: E-cadherin,
5′-GTCTGTCATGGAAGGTG CT-3′ (forward), 5′-TACGACGTTAGCCTCGTTC-3′
(reverse); Vimentin, 5′-AGTCCACTGAGTACCGGAGAC-3′ (forward),
5′-CATTTCACGCATCTGGCGTTC-3′ (reverse); ZEB1 (24), 5′-CAGCTTGATACCTGTGAATGGG-3′
(forward), 5′-TATCTGTGGTCGTGTGGGACT-3′ (reverse); ZEB2 (25), 5′-GCG ATGGTCATGCAGTCAG-3′
(forward), 5′-CAGGTGGCAG GTCATTTTCTT-3′ (reverse); ESRP1 (26), 5′-CAATATTG CCAAGGGAGGTG-3′
(forward), 5′-GTCCCCATGTGATG TTTGTG-3′ (reverse); GAPDH,
5′-AGCCTCAAGATCATCAGCAATGCC- 3′ (forward),
5′-TGTGGTCATGAGTCCTTCCACGAT-3′ (reverse); CD44
5′-AGCATCGGATTTGAGACCTG-3′ (forward), 5′-GTTGTTTGCTGCACAGATGG-3′
(reverse); CD44s (27),
5′-AAAGGAGCAGCACTTCAGGA-3′ (forward), 5′-TGTGTCTTGGTCTCTGGTAGC-3′
(reverse); CD44v9, 5′-ACCATCCAACAACTTCTACTCTGACA-3′ (forward),
5′-CCTTCAGAATGATTTGGGTCTCTT-3′ (reverse).
Tumorigenicity assay
Six-week-old female nude mice (BALB/c-nu, Charles
River Laboratories Japan, Yokohama, Japan) were used for the
tumorigenicity assay. Sorted NOZ cells were suspended in a 1:1
(v/v/) mixture of PBS and Matrigel (Corning Inc.). The mice were
anesthetized using sevoflurane, and the suspensions of sorted cells
(100–10,000 cells/0.1 ml) were injected subcutaneously into the
right and left lumbar regions. The mice were maintained under
standard conditions specified by institutional guidelines for
animal care. The mice that were injected with 10,000 cells were
sacrificed 4 weeks later, and tumor sizes and weights were
measured. Tumor volumes were calculated as follows: Tumor volume =
(long axis length in millimeters/2) × (short axis
length/2)2 × 4π/3. The other mice were observed for 8
weeks when palpable tumors were observed. Tumors were
histologically confirmed using hematoxylin and eosin (H&E)
staining.
Tissue samples and tissue microarray
(TMA) analysis
We collected a series of paraffin-embedded tissues
specimens from 52 patients with GBC who underwent surgery between
1997 and 2010 at the Toyama University Hospital, Japan. The tumors
were histologically diagnosed in the Department of Pathology. The
final stage of GBC was confirmed pathologically according to the
TNM Classification of Malignant Tumors, 7th edition. The ethics
committee of the University of Toyama approved this study.
The TMA comprised 1.0-mm cores of tissues from the
paraffin-embedded blocks of the surgical specimen described above.
Paraffin blocks containing tumor tissue were selected, and the most
representative areas encompassing the tumors were marked directly
on the blocks according to the corresponding HE-stained slides. The
array block was cut into sections that were placed onto glass
slides for H&E staining and immunohistochemical analyses
(28–30).
Immunohistochemical staining
The primary antibodies used for the
immunohistochemical staining were as follows: anti-cytokeratin
OSCAR polyclonal antibody (dilution 1:200, Abcam, Cambridge, UK)
and anti-human CD44v9 monoclonal antibody (clone RV3; dilution
1:200, Cosmo Bio Co., Ltd.), CD44std (clone SFF-2, Affymetrix
Inc.). The cancer cells were identified using the anti-cytokeratin
OSCAR antibody, then CD44v9 and CD44std expression was detected in
the membrane of cancer cells. The TMA score was calculated from
staining intensity and the distribution score. The criteria for
scoring were as follows: the staining distribution was scored as 0
(0–10%), 1 (11–50%), and 2 (51–100%) to indicate the percentage of
tumor cells present in one tissue section. Staining intensity was
scored as 0 (no staining of cancer cells), 1 (weak), 2 (moderate),
and 3 (marked). We defined CD44v9-high/low and CD44std-high/low
expression when the sum of the staining distribution and intensity
score was ≥4/≤3 and ≥3/≤2, respectively. Two researchers uninformed
of patient clinicopathological information independently scored the
tissues, and when there was discordance, the investigators
re-evaluated the data to assign a consensus score.
Statistical analysis
Data are expressed as the mean ± standard deviation
(SD). The Wilcoxon rank sum test and Fisher's exact test were used
to compare the differences between groups. Overall survival (OS)
was analyzed using the Kaplan-Meier method and the log-rank test.
Statistical analysis was performed using JMP Pro 11.2 (SAS
Institute Inc., Cary, NC, USA) and p-values <0.05 were
considered significant.
Results
The NOZ cell line comprises two
populations that express different CD44 isoforms
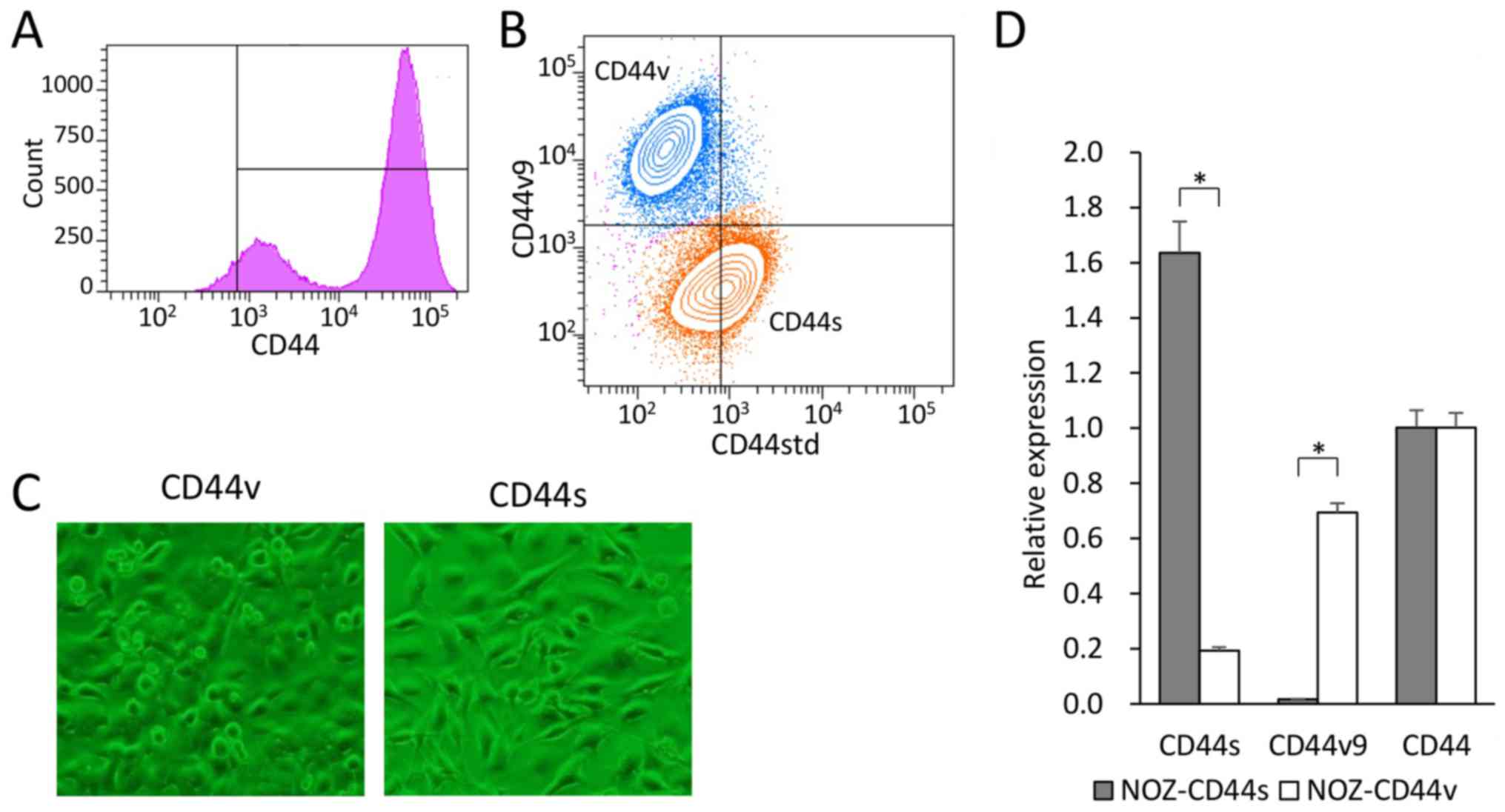
We used flow cytometry to analyze the cell surface
expression of CD44 in the GBC cell line NOZ. CD44 expression was
observed in 90% of the NOZ cells and showed a bimodal distribution
(Fig. 1A). Double-staining
analysis of CD44std and CD44v9 expression in the NOZ cell line
revealed two populations (Fig.
1B). CD44-high cells accounted for approximately 70% of the NOZ
cells, which expressed CD44std+ and CD44v9−.
In contrast, CD44-low cells accounted for approximately 30% of the
cells, which expressed CD44v9+ and CD44std−.
We defined CD44s cells as CD44std+-CD44v9−,
and CD44v cells as CD44std−-CD44v9+. Each
group was sorted, collected, and used in the following
experiments.
The CD44s and CD44v cells were recultured to confirm
viability and proliferation. The NOZ-CD44s cells were
spindle-shaped, and the NOZ-CD44v cells were rounded with a spindle
or polygonal shape (Fig. 1C). The
expressed CD44 isoforms of the sorted cells were genetically
confirmed (Fig. 1D). The NOZ-CD44s
sorted cells expressed significantly higher and lower levels of
CD44s and CD44v9 mRNA compared with the NOZ-CD44v sorted cells. The
whole CD44 mRNA levels did not differ between CD44s and CD44v
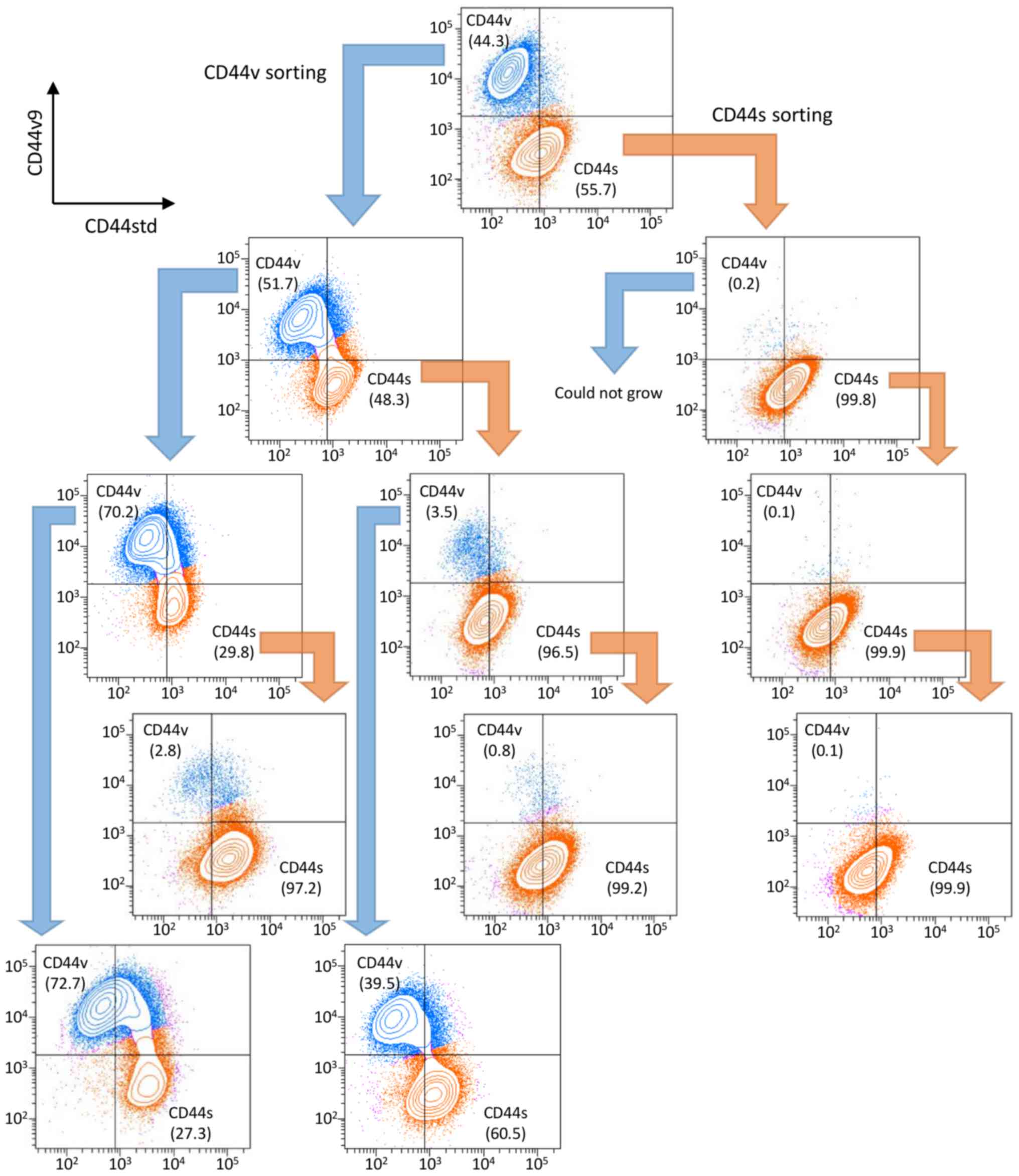
sorted cells. We analyzed the sequential expression of CD44
isoforms of the sorted cells. One week after sorting, cells were
restained and analyzed for CD44 isoform expression, and
sequentially repeated three times (Fig. 2). The NOZ-CD44s cells were
generated from CD44v cells (CD44v/CD44s range:
39.5–72.7/27.3–60.5), and NOZ-CD44v cells were not generated from
CD44s cells (CD44v/CD44s range 0.1–3.5/96.5–99.9), suggesting that
CD44s cells were derived from CD44v cells.
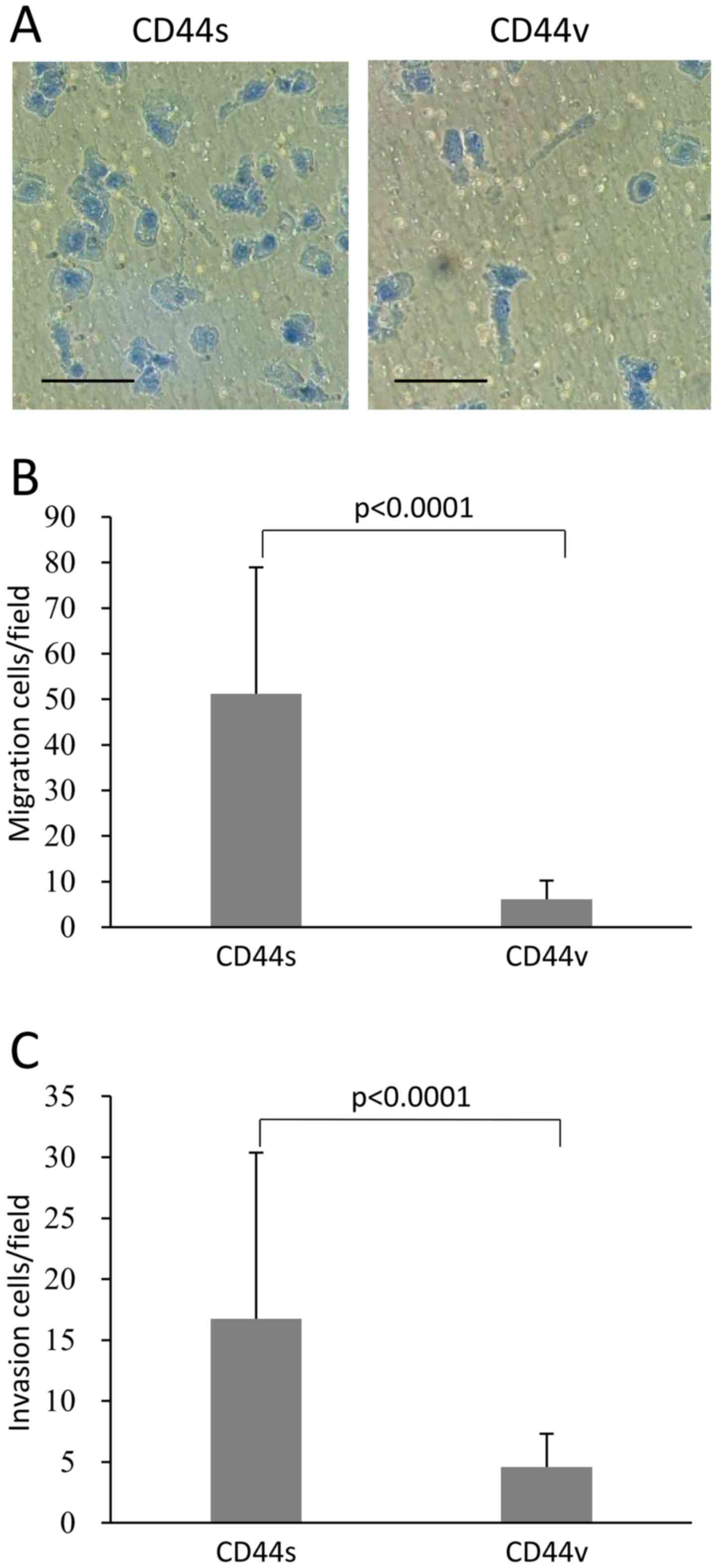
NOZ-CD44s cells have increased chemotaxis
activity and invasiveness compared with NOZ-CD44v cells
Analysis of chemotaxis in vitro revealed that
significantly more NOZ-CD44s cells migrated compared with NOZ-CD44v
cells (51.2±27.7 vs. 6.1±4.1 cells per field, p<0.0001)
(Fig. 3A and B). Similarly,
NOZ-CD44s cells were significantly more invasive compared with
NOZ-CD44v cells (16.7±13.6 cells vs. 4.6±2.7 cells per field,
p<0.0001) (Fig. 3C).
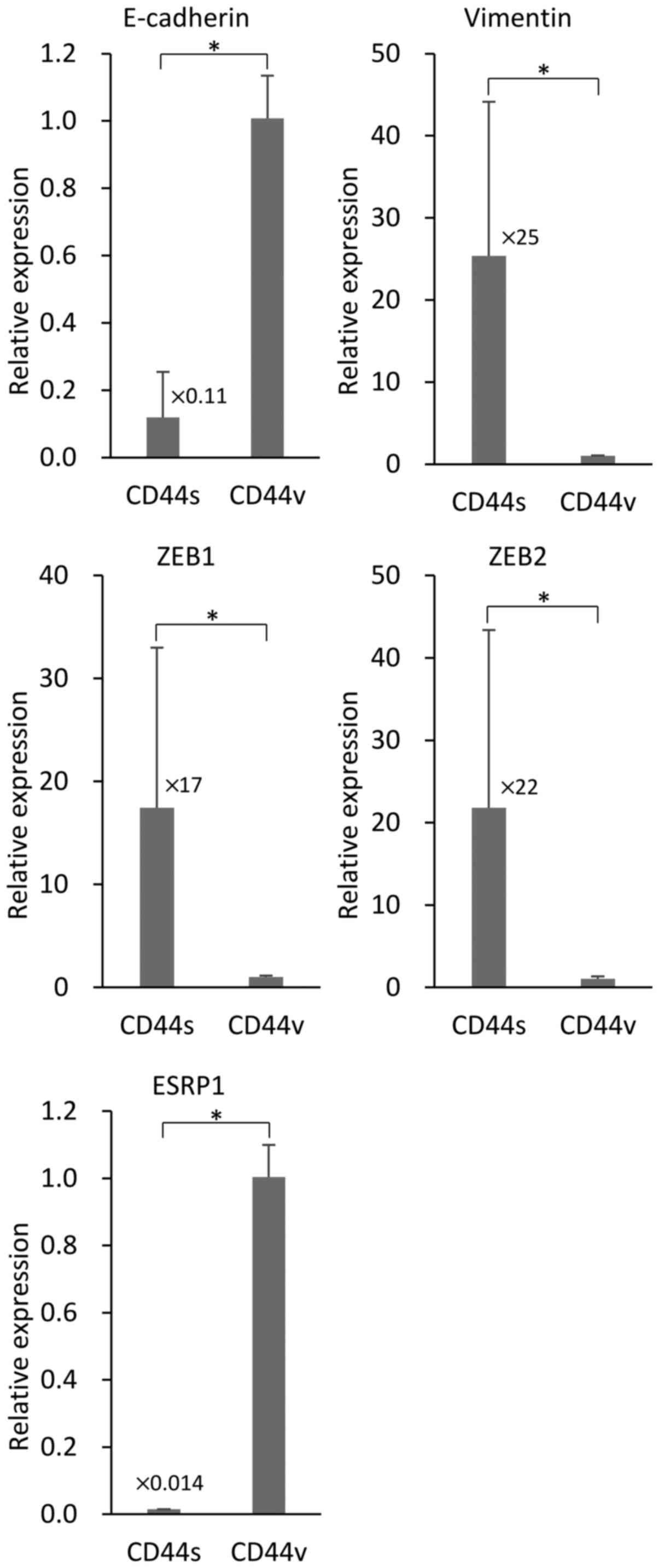
NOZ-CD44s cells exhibit a mesenchymal
phenotype
We investigated the association of the expression of
the different CD44s and CD44v isoforms expressed by NOZ cells on
EMT. We used RT-PCR to determine the levels of the mRNAs of the EMT
markers E-cadherin and vimentin in the sorted NOZ cells. NOZ-CD44s
expressed significantly higher levels of vimentin and lower levels
of E-cadherin compared with CD44v cells (Fig. 4). The transcription factors ZEB1
(δEF1) and ZEB2 (SIP-1) that control EMT, were highly expressed in
the NOZ-CD44s cells compared with NOZ-CD44v cells. The mRNA
encoding the splicing factor ESRP1 that controls the CD44 isoform
switch was expressed at lower levels in NOZ-CD44s cells compared
with NOZ-CD44v cells.
NOZ-CD44v cells are highly tumorigenic in
vivo
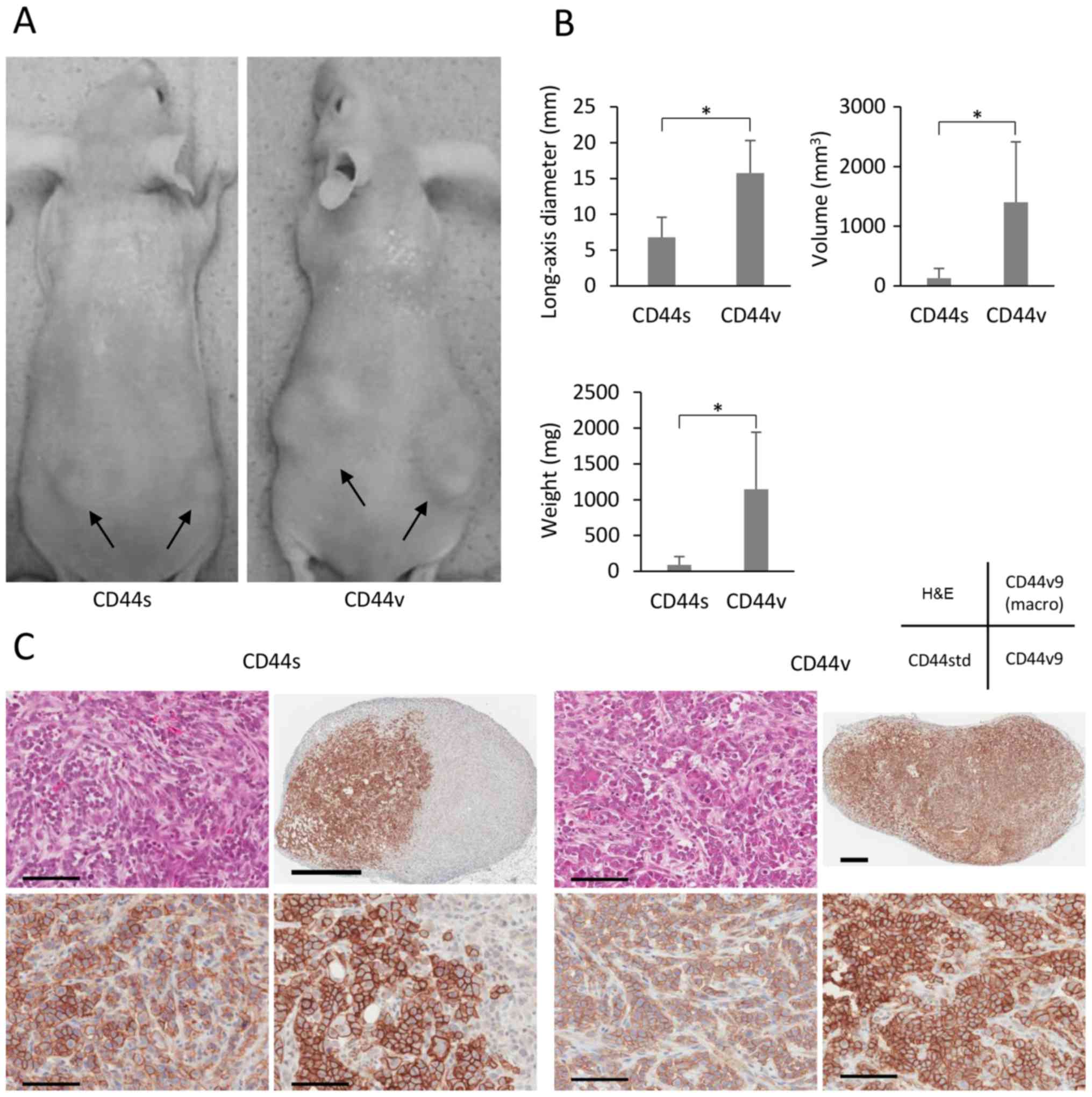
When we investigated the tumorigenicity of CD44s and
CD44v cells in nude mice, we found that the former were less
tumorigenic (Fig. 5A). Although
each cell population formed tumors at all sites where 10,000 cells
were injected, the respective long-axis diameters, tumor volumes,
and weights of the tumors formed by CD44s and CD44v cells were
6.8±2.8 and 15.7±4.5 mm, 131±160 and 1401±1013 mm3, and
0.09±0.12 and 1.15±0.80 g (all p<0.001). NOZ-CD44v cells (100
per site) formed tumors at 10 out of 12 sites, whereas NOZ-CD44s
cells did not generate tumors (Table
I). The tumors were pathologically confirmed using H&E
staining, and immunohistochemical analysis of CD44v9 expression
revealed that CD44s cells generated partly CD44v9-rich tumors
(Fig. 5C).
 | Table IIn vivo tumorigenicity
analysis in which NOZ-CD44s and CD44v cells were injected into nude
mice subcutaneously. |
Table I
In vivo tumorigenicity
analysis in which NOZ-CD44s and CD44v cells were injected into nude
mice subcutaneously.
| Number of cells
injected | Tumor incidence
|
|---|
| 100 | 1000 | 10000 |
|---|
| CD44s | 0/12 | 4/12 | 12/12 |
| CD44v | 10/12 | 11/12 | 12/12 |
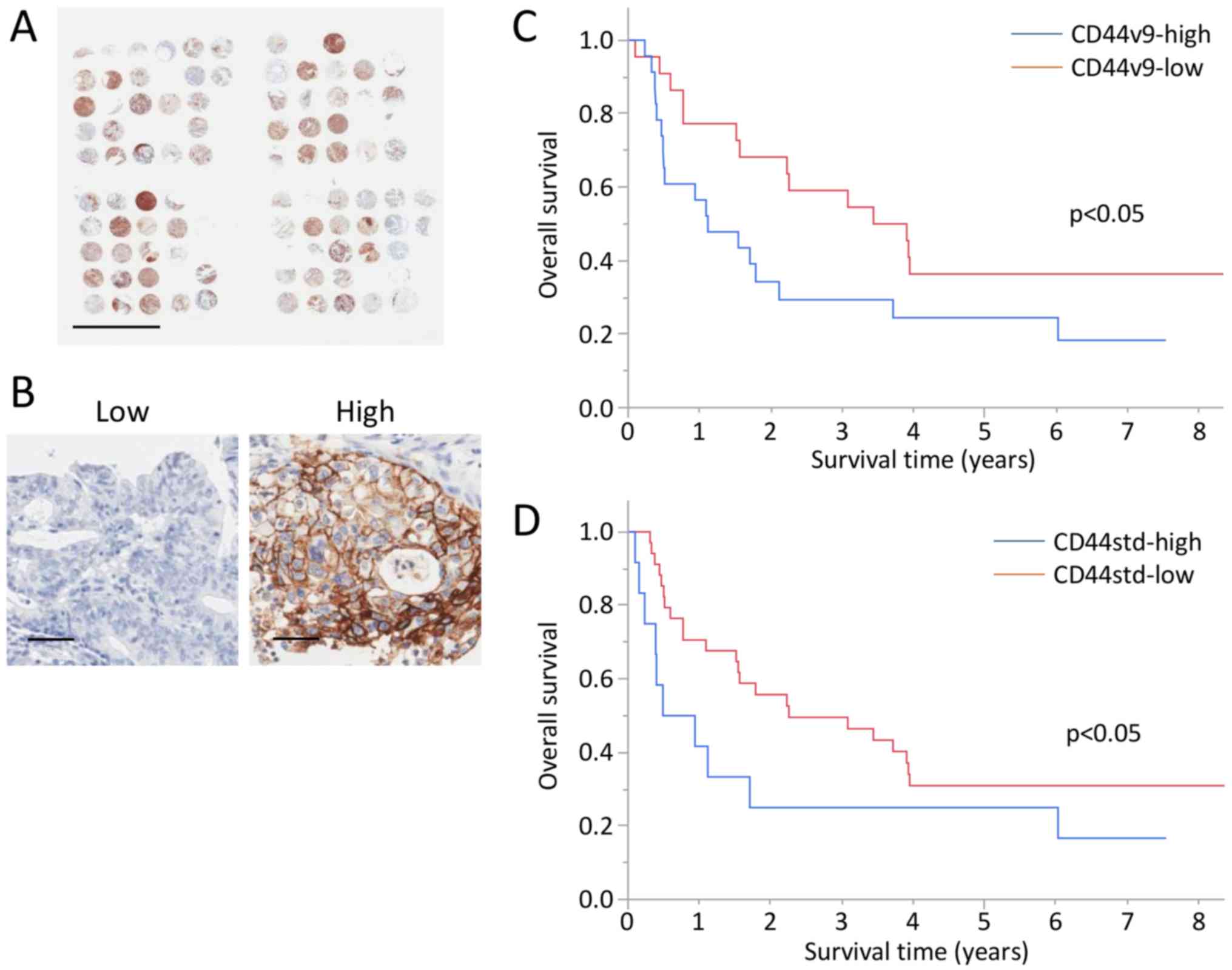
CD44v9 expression associates with poor
prognosis
A TMA of GBC was performed, and CD44v9 and CD44std
expression was analyzed in 45 and 47 tumors from patients with GBC
(Fig. 6A and Table II). Immunohistochemical analysis
detected high levels of CD44v9 and CD44std expression in 23 and 12
tumors (Fig. 6B). No significant
differences were detected in age, sex, serum CA19-9 levels, and
pathological T, N, ly, and v factors associated with the expression
of CD44v9 and CD44std. CD44v9 expression was significantly
associated with serum CEA levels. Whereas, CD44std expression was,
significantly associated with poorly differentiation and distant
metastasis.
 | Table IICD44v9 and CD44std expression in
tumors from patients with GBC. |
Table II
CD44v9 and CD44std expression in
tumors from patients with GBC.
| Factors | CD44v9
| p-value | CD44std
| p-value |
|---|
Low
(N=22) | High
(N=23) | Low
(N=22) | High
(N=23) |
|---|
| Age | 71.1±10.7 | 69.2±11.5 | 0.566 | 70.9±12.0 | 68.0±8.2 | 0.450 |
| Sex | | | 0.928 | | | 0.702 |
| Male | 6 (27%) | 6 (26%) | | 10 (29%) | 2 (17%) | |
| Female | 16 (73%) | 17 (74%) | | 25 (71%) | 10 (83%) | |
| CEA (ng/ml) | 4.1±3.1 | 12.9±20.9 | 0.032 | 7.2±11.9 | 13.2±22.7 | 0.243 |
| CA19-9 (U/ml) | 249.4±609.2 | 161.4±327.3 | 0.549 | 237.5±515.3 | 133.8±346.4 | 0.521 |
|
Differentiation | | | 0.588 | | | 0.024 |
| Well/Mod | 21 (95%) | 17 (89%) | | 33 (97%) | 6 (67%) | |
| Poor | 1 (5%) | 2 (11%) | | 1 (3%) | 3 (33%) | |
| pT | | | 0.850 | | | 0.261 |
| 1–2 | 13 (62%) | 13 (59%) | | 22 (65%) | 5 (45%) | |
| 3–4 | 8 (38%) | 9 (41%) | | 12 (35%) | 6 (55%) | |
| pN | | | 0.790 | | | 0.733 |
| Negative | 11 (55%) | 13 (59%) | | 18 (56%) | 8 (67%) | |
| Positive | 9 (45%) | 9 (41%) | | 14 (44%) | 4 (33%) | |
| M | | | 0.153 | | | 0.045 |
| Negative | 16 (73%) | 12 (52%) | | 24 (69%) | 4 (33%) | |
| Positive | 6 (27%) | 11 (48%) | | 11 (31%) | 8 (67%) | |
| ly | | | 0.208 | | | 0.161 |
| Negative | 11 (52%) | 6 (29%) | | 16 (47%) | 2 (20%) | |
| Positive | 10 (48%) | 15 (71%) | | 18 (53%) | 8 (80%) | |
| v | | | 0.059 | | | 0.074 |
| Negative | 15 (71%) | 9 (43%) | | 22 (65%) | 3 (30%) | |
| Positive | 6 (29%) | 12 (57%) | | 12 (35%) | 7 (70%) | |
The 3-year OS rates of the CD44v9-high, CD44v9-low,
CD44std-high, and CD44std-low groups were 29.4, 59.1, 25.0, and
49.5%, respectively (Fig. 6C and
D).
Discussion
In the present study, we showed that the GBC cell
line NOZ comprises two populations that expressed different CD44
isoforms. The major population (CD44s) expressed
CD44std+/CD44v9−. The CD44s cells showed the
mesenchymal phenotype, and they showed strong chemotactic and
invasive abilities. On the other hand, they showed weakly
tumorigenic ability. The minor population (CD44v) expressed
CD44std−/CD44v9+. This population showed the
epithelial phenotype, and it showed weakly chemotactic and invasive
abilities. However, it showed highly tumorigenic ability. These
isoforms of CD44 in the NOZ sorted cells were also genetically
identified. CD44v cells generated CD44s cells in vitro,
although the xenotransplantation study indicated that CD44v cells
were generated from CD44s cells in vivo. In patients with
GBC, we demonstrated that both phenotypes CD44v9high and
CD44stdhigh were significantly associated with poor
prognosis. CD44v9high was significantly associated with
high CEA levels, and CD44stdhigh was significantly
associated with poor differentiation and distant metastasis.
It is important to clarify whether CD44 represents
particular CD44 isoform or pan-CD44. Each variant isoform possesses
specific epitopes, although most epitopes of the CD44 standard
isoform are included in the variant isoforms. Numerous anti-CD44
antibodies react with CD44 variant isoforms as well as the CD44
standard isoform, although these isoforms are associated with
different phenotypes. Thus, the isoforms of CD44 (or pan-CD44)
should be identified when studies using CD44-specific antibodies
are planned. We show here that the GBC cell line NOZ comprised
populations of CD44 standard and variant 9 isoforms that were
associated with different phenotypes, although flow cytometry using
the anti-CD44 antibody detected both isoforms. We then used
isoform-specific antibodies CD44std and CD44v9 to show that the
CD44s and CD44v cells expressed the respective cognate
isoforms.
CD44 isoforms are associated with EMT. It is
reported that pancreatic cancer cells that express CD44v9 showed
epithelial phenotype (14), and
hepatocellular carcinomas that express CD44 standard isoform showed
the mesenchymal phenotype (7,31).
Further, breast cancer cells are reported to show isoform switch
from variant isoform to the standard isoform through EMT (32), ESRP1 is critical for regulating the
isoform switch (32,33), and signaling through AKT, which is
activated by the CD44 standard isoform, is essential for driving
EMT (32). Moreover, CD44 isoform
switch occurs in colorectal cancer cells (27). When ESRP1 is silenced, the CD44
variant isoform is replaced by the standard isoform that promotes
EMT. Further, the CD44 standard isoform is essential for EMT,
because CD44 knockdown inhibits vimentin expression (32). In cancers of the biliary tract,
expression of the CD44 standard isoform is associated with
well-differentiated cancers (16),
and CD44v6 isoforms are associated with moderate-poorly
differentiated cancers and Stage IV tumors (17,34).
Our TMA analysis, however, showed that CD44std expression was
associated with poorly differentiated cancers and distant
metastasis, which was compatible with the mesenchymal phenotype of
the CD44s showed in the in vitro studies.
Our findings described above that the CD44 standard
and variant 9 isoforms were associated with mesenchymal and
epithelial phenotypes, respectively, are consistent with a switch
from the CD44 variant to the standard isoform through EMT. The
levels of the mRNAs encoding the transcription factors ZEB1 and
ZEB2 were increased in cells that expressed the CD44 standard
isoform that was associated with promoting EMT. Further, the levels
of the mRNA encoding the splicing factor ESRP1 were lower in cells
that expressed the CD44 standard isoform, which triggered the
CD44-isoform switch and then promoted EMT. Further, we showed that
the CD44s cells were generated from CD44v cells under the normal
culture condition. There may, therefore be an auto-trigger which
induces the CD44 isoform switch through NOZ cell progression.
CD44 is associated with tumorigenicity of many
cancers such as the breast (35),
colon, head and neck, and pancreas (36). In cholangiocarcinoma,
CD44+CD24+EpCAMhigh cells exhibit
high tumorigenic potential (37).
However, these studies did not analyze CD44 isoforms. In contrast,
analyses using xenotransplantation of mice reveal that colon
(15) and breast cancer (33) cells that express CD44v9 are highly
tumorigenic. Further, pancreatic cancer cells express CD44v9 when
they re-enter mitosis (14).
Clinical studies reveal that CD44v9 expression is associated with
the recurrence of early gastric cancer (8) and with poor OS and recurrence-free
survival of patients with hepatocellular carcinoma (10). Moreover, RT-PCR analyses show that
increased CD44v9 expression in pancreatic cancer tissues correlates
with lymph node metastasis, liver metastasis, TNM stage
progression, and decreased overall survival rate (9). Although the relationship between
CD44v9 expression and tumorigenicity is unknown, CD44v9 was
proposed to activate the Ras/Rac1/RhoA pathway, leading to the
migration, growth, invasion, and survival of tumor cells (37–39).
In the present study, CD44v9 cells had higher tumorigenicity in
terms of tumor burden and tumor incidence compared with CD44s
cells. Further, both tumors generated from CD44v9 and CD44s cells
expressed CD44v9 immunohistochemically. Although the mechanism by
which the CD44s generated CD44v9-rich tumors is unclear, it is
suggesting that a switch from the standard to variant isoform is
induced through engraftment or the progression of tumor
formation.
In conclusion, expression of the CD44 standard
isoform was associated with a mesenchymal phenotype, increased
chemotaxis, increased invasiveness, and lower tumorigenicity. In
contrast, expression of the CD44 variant 9 isoform was associated
with an epithelial phenotype, decreased chemotaxis, decreased
invasiveness, and increased tumorigenicity. These findings suggest
that these CD44 isoforms play different roles in cancer progression
and metastasis. Therefore, investigating the potential role and
mechanism of the isoform switch may lead to the development of
novel therapeutic interventions or diagnostic agents that target
splicing or specific isoforms that will improve the treatment of
patients with GBC.
Acknowledgments
We thank Mr. Masahiko Kawahara for his technical
assistance.
References
|
1
|
Misra S, Chaturvedi A, Misra NC and Sharma
ID: Carcinoma of the gallbladder. Lancet Oncol. 4:167–176. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Gourgiotis S, Kocher HM, Solaini L,
Yarollahi A, Tsiambas E and Salemis NS: Gallbladder cancer. Am J
Surg. 196:252–264. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Hofmann M, Rudy W, Zöller M, Tölg C, Ponta
H, Herrlich P and Günthert U: CD44 splice variants confer
metastatic behavior in rats: Homologous sequences are expressed in
human tumor cell lines. Cancer Res. 51:5292–5297. 1991.PubMed/NCBI
|
|
4
|
Sneath RJS and Mangham DC: The normal
structure and function of CD44 and its role in neoplasia. Mol
Pathol. 51:191–200. 1998. View Article : Google Scholar
|
|
5
|
Goodison S, Urquidi V and Tarin D: CD44
cell adhesion molecules. Mol Pathol. 52:189–196. 1999. View Article : Google Scholar
|
|
6
|
Zöller M: CD44: Can a cancer-initiating
cell profit from an abundantly expressed molecule? Nat Rev Cancer.
11:254–267. 2011. View
Article : Google Scholar : PubMed/NCBI
|
|
7
|
Okabe H, Ishimoto T, Mima K, Nakagawa S,
Hayashi H, Kuroki H, Imai K, Nitta H, Saito S, Hashimoto D, et al:
CD44s signals the acquisition of the mesenchymal phenotype required
for anchorage-independent cell survival in hepatocellular
carcinoma. Br J Cancer. 110:958–966. 2014. View Article : Google Scholar :
|
|
8
|
Hirata K, Suzuki H, Imaeda H, Matsuzaki J,
Tsugawa H, Nagano O, Asakura K, Saya H and Hibi T: CD44 variant 9
expression in primary early gastric cancer as a predictive marker
for recurrence. Br J Cancer. 109:379–386. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Li Z, Chen K, Jiang P, Zhang X, Li X and
Li Z: CD44v/CD44s expression patterns are associated with the
survival of pancreatic carcinoma patients. Diagn Pathol. 9:792014.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kakehashi A, Ishii N, Sugihara E, Gi M,
Saya H and Wanibuchi H: CD44 variant 9 is a potential biomarker of
tumor initiating cells predicting survival outcome in hepatitis C
virus-positive patients with resected hepatocellular carcinoma.
Cancer Sci. 107:609–618. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ishimoto T, Nagano O, Yae T, Tamada M,
Motohara T, Oshima H, Oshima M, Ikeda T, Asaba R, Yagi H, et al:
CD44 variant regulates redox status in cancer cells by stabilizing
the xCT subunit of system xc(-) and thereby promotes tumor growth.
Cancer Cell. 19:387–400. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Mielgo A, van Driel M, Bloem A, Landmann L
and Günthert U: A novel antiapoptotic mechanism based on
interference of Fas signaling by CD44 variant isoforms. Cell Death
Differ. 13:465–477. 2006. View Article : Google Scholar
|
|
13
|
Nagano O, Okazaki S and Saya H: Redox
regulation in stem-like cancer cells by CD44 variant isoforms.
Oncogene. 32:5191–5198. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kiuchi S, Ikeshita S, Miyatake Y and
Kasahara M: Pancreatic cancer cells express CD44 variant 9 and
multidrug resistance protein 1 during mitosis. Exp Mol Pathol.
98:41–46. 2015. View Article : Google Scholar
|
|
15
|
Kimura Y, Goi T, Nakazawa T, Hirono Y,
Katayama K, Urano T and Yamaguchi A: CD44 variant exon 9 plays an
important role in colon cancer initiating cells. Oncotarget.
4:785–791. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kalekou H and Miliaras D:
Immunohistochemical study of microvessel density, CD44 (standard
form), p53 protein and c-erbB2 in gallbladder carcinoma. J
Gastroenterol Hepatol. 19:812–818. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yanagisawa N, Mikami T, Mitomi H, Saegusa
M, Koike M and Okayasu I: CD44 variant overexpression in
gallbladder carcinoma associated with tumor dedifferentiation.
Cancer. 91:408–416. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yamaguchi A, Zhang M, Goi T, Fujita T,
Niimoto S, Katayama K and Hirose K: Expression of variant CD44
containing variant exon v8-10 in gallbladder cancer. Oncol Rep.
7:541–544. 2000.PubMed/NCBI
|
|
19
|
Homma S, Hasumura S, Nagamori S and Kameda
H: Establishment and characterization of a human gall bladder
carcinoma cell line NOZ. Hum Cell. 1:95–97. 1988.In Japanese.
PubMed/NCBI
|
|
20
|
Kramer N, Walzl A, Unger C, Rosner M,
Krupitza G, Hengstschläger M and Dolznig H: In vitro cell migration
and invasion assays. Mutat Res. 752:10–24. 2013. View Article : Google Scholar
|
|
21
|
Valster A, Tran NL, Nakada M, Berens ME,
Chan AY and Symons M: Cell migration and invasion assays. Methods.
37:208–215. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Long J, Luo G, Liu C, Cui X, Satoh K, Xiao
Z, Zhang B, Xu J, Ni Q, Li M, et al: Development of a unique mouse
model for pancreatic cancer lymphatic metastasis. Int J Oncol.
41:1662–1668. 2012.PubMed/NCBI
|
|
23
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
24
|
Eger A, Aigner K, Sonderegger S, Dampier
B, Oehler S, Schreiber M, Berx G, Cano A, Beug H and Foisner R:
DeltaEF1 is a transcriptional repressor of E-cadherin and regulates
epithelial plasticity in breast cancer cells. Oncogene.
24:2375–2385. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Comijn J, Berx G, Vermassen P, Verschueren
K, van Grunsven L, Bruyneel E, Mareel M, Huylebroeck D and van Roy
F: The two-handed E box binding zinc finger protein SIP1
downregulates E-cadherin and induces invasion. Mol Cell.
7:1267–1278. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Ishii H, Saitoh M, Sakamoto K, Kondo T,
Katoh R, Tanaka S, Motizuki M, Masuyama K and Miyazawa K:
Epithelial splicing regulatory proteins 1 (ESRP1) and 2 (ESRP2)
suppress cancer cell motility via different mechanisms. J Biol
Chem. 289:27386–27399. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Mashita N, Yamada S, Nakayama G, Tanaka C,
Iwata N, Kanda M, Kobayashi D, Fujii T, Sugimoto H, Koike M, et al:
Epithelial to mesenchymal transition might be induced via CD44
isoform switching in colorectal cancer. J Surg Oncol. 110:745–751.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Sekine S, Shimada Y, Nagata T, Moriyama M,
Omura T, Watanabe T, Hori R, Yoshioka I, Okumura T, Sawada S, et
al: Prognostic significance of aquaporins in human biliary tract
carcinoma. Oncol Rep. 27:1741–1747. 2012.PubMed/NCBI
|
|
29
|
Sekine S, Shimada Y, Nagata T, Sawada S,
Yoshioka I, Matsui K, Moriyama M, Omura T, Osawa S, Shibuya K, et
al: Role of aquaporin-5 in gallbladder carcinoma. Eur Surg Res.
51:108–117. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Fukuoka J, Fujii T, Shih JH, Dracheva T,
Meerzaman D, Player A, Hong K, Settnek S, Gupta A, Buetow K, et al:
Chromatin remodeling factors and BRM/BRG1 expression as prognostic
indicators in non-small cell lung cancer. Clin Cancer Res.
10:4314–4324. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Mima K, Okabe H, Ishimoto T, Hayashi H,
Nakagawa S, Kuroki H, Watanabe M, Beppu T, Tamada M, Nagano O, et
al: CD44s regulates the TGF-β-mediated mesenchymal phenotype and is
associated with poor prognosis in patients with hepatocellular
carcinoma. Cancer Res. 72:3414–3423. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Brown RL, Reinke LM, Damerow MS, Perez D,
Chodosh LA, Yang J and Cheng C: CD44 splice isoform switching in
human and mouse epithelium is essential for epithelial-mesenchymal
transition and breast cancer progression. J Clin Invest.
121:1064–1074. 2011. View
Article : Google Scholar : PubMed/NCBI
|
|
33
|
Yae T, Tsuchihashi K, Ishimoto T, Motohara
T, Yoshikawa M, Yoshida GJ, Wada T, Masuko T, Mogushi K, Tanaka H,
et al: Alternative splicing of CD44 mRNA by ESRP1 enhances lung
colonization of metastatic cancer cell. Nat Commun. 3:8832012.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Gu MJ and Jang BI: Clinicopathologic
significance of Sox2, CD44 and CD44v6 expression in intrahepatic
cholangiocarcinoma. Pathol Oncol Res. 20:655–660. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Han Z, Chen Z, Zheng R, Cheng Z, Gong X
and Wang D: Clinicopathological significance of CD133 and CD44
expression in infiltrating ductal carcinoma and their relationship
to angiogenesis. World J Surg Oncol. 13:562015. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Visvader JE and Lindeman GJ: Cancer stem
cells in solid tumours: Accumulating evidence and unresolved
questions. Nat Rev Cancer. 8:755–768. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Wang M, Xiao J, Shen M, Yahong Y, Tian R,
Zhu F, Jiang J, Du Z, Hu J, Liu W, et al: Isolation and
characterization of tumorigenic extrahepatic cholangiocarcinoma
cells with stem cell-like properties. Int J Cancer. 128:72–81.
2011. View Article : Google Scholar
|
|
38
|
Wang SJ and Bourguignon LYW: Role of
hyaluronan-mediated CD44 signaling in head and neck squamous cell
carcinoma progression and chemoresistance. Am J Pathol.
178:956–963. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Akasaka J, Uekuri C, Shigetomi H, Koike M
and Kobayashi H: Hepatocyte nuclear factor (HNF)-1β and its
physiological importance in endometriosis. Biomed Rep. 1:13–17.
2013.PubMed/NCBI
|