Introduction
Head and neck squamous cell carcinoma (HNSCC) occurs
from the mucosa in the upper aerodigestive tract, including the
oral cavity, oropharynx, hypopharynx and larynx, and this disease
is the sixth most common cancer worldwide (1). Approximately 550,000 new patients are
diagnosed, and 30,000 patients die of this disease annually
(2). Due to the local recurrence
and distant metastasis of HNSCC, the overall survival of patients
with HNSCC has not improved in the last decade (3). Currently developed targeted molecular
therapies are not sufficiently efficacious in the management of
HNSCC (3). Therefore, improving
our understanding of the molecular mechanisms of HNSCC
aggressiveness is needed based on current genomic approaches.
MicroRNAs (miRNAs) are small noncoding RNAs (19–22
nucleotides in length) involved in the repression or degradation of
target RNA transcripts in a sequence-dependent manner (4). One of the unique features of miRNAs
is that a single miRNA regulates a vast number of protein-coding or
noncoding RNAs in human cells (5).
Thus, aberrant expression of miRNAs disrupts systematically
regulated RNA networks in cancer cells. In fact, accumulating
evidence has revealed that aberrant expression of miRNAs is deeply
involved in the pathogenesis of human cancers (6).
In miRNA biogenesis, precursor miRNA (pre-miRNA) is
cleaved in the cytoplasm, generating a miRNA duplex comprised of a
guide strand and passenger strand. The guide strand of miRNA is
thought to be incorporated into the RNA-induced silencing complex
(RISC) to target mRNAs, whereas the passenger strand of miRNA is
degraded and is not thought to have regulatory activity in cells
(7). However, in contrast to this
paradigm, we demonstrated that passenger strands of miRNAs, i.e.,
miR-144-5p, miR-139-3p, miR-150-3p and
miR-145-3p, were downregulated and acted as antitumor miRNAs
in several types of cancers (8–13).
Moreover, dual strands of pre-miR-145 (miR-145-5p and
miR-145-3p) coordinately target oncogenic MTDH and
UHRF1 in lung cancer and bladder cancer, respectively
(10,11). The involvement of passenger miRNA
strands and regulation of cancer networks by passenger miRNAs are
novel concepts in cancer research.
Analysis of the miRNA expression signature of HNSCC
by RNA sequencing revealed that miR-145-5p and
miR-145-3p were significantly downregulated in cancer
tissues. The guide strand miR-145-5p has been established as
an oncogene in several cancers, including HNSCC (14). However, the functional significance
of the passenger strand of miR-145 in HNSCC is still
unknown. The aims of this present study were to investigate the
antitumor function of miR-145-3p and to identify its target
oncogenic genes in HNSCC cells. Elucidation of the antitumor roles
of passenger strands of miRNAs and the cancer networks mediated by
these miRNAs may provide insights into the molecular pathogenesis
of HNSCC.
Materials and methods
Clinical HNSCC specimens, cell lines, and
cell culture
A total of 22 clinical tissue specimens were
collected from patients with HNSCC who underwent surgical resection
at Chiba university Hospital between 2008 and 2014. The
clinicopathological features of patients with HNSCC are summarized
in Table I. All patients in this
study provided informed consent, and the study protocol was
approved by the Institutional Review Board of Chiba University. TNM
classification and tumor stage were determined by the union for
International Cancer Control (UICC) (15).
 | Table IClinical features of 22 patients with
HNSCC. |
Table I
Clinical features of 22 patients with
HNSCC.
| No. | Age | Sex | Location | T | N | M | Stage |
Differentiation |
|---|
| 1 | 64 | F | Oral floor | 4a | 2c | 0 | IVA | Moderate |
| 2 | 73 | M | Tongue | 3 | 2b | 0 | IVA | Poor |
| 3 | 77 | M | Tongue | 2 | 2b | 0 | IVA | Poor |
| 4 | 63 | F | Oral floor | 2 | 2b | 0 | IVA | Basaloid SCC |
| 5 | 59 | M | Tongue | 1 | 2a | 0 | IVA | Moderate |
| 6 | 36 | F | Tongue | 3 | 1 | 0 | III | Moderate |
| 7 | 67 | M | Tongue | 3 | 0 | 0 | III | Moderate |
| 8 | 60 | F | Tongue | 2 | 1 | 0 | III | Well |
| 9 | 66 | M | Tongue | 2 | 0 | 0 | II | Moderate |
| 10 | 67 | M | Tongue | 2 | 0 | 0 | II | Poor to
moderate |
| 11 | 76 | F | Tongue | 1 | 0 | 0 | I | Well |
| 12 | 69 | M | Tongue | 1 | 0 | 0 | I | Well |
| 13 | 73 | F | Tongue | 1 | 0 | 0 | I | Well |
| 14 | 64 | M | Tongue | 1 | 0 | 0 | I | Well |
| 15 | 70 | M | Tongue | 1 | 0 | 0 | I | Well |
| 16 | 38 | M | Tongue | 1 | 0 | 0 | I | Well |
| 17 | 51 | M | Tongue | 1 | 0 | 0 | I | Well |
| 18 | 34 | F | Tongue | 1 | 0 | 0 | I | Poor |
| 19 | 70 | M | Tongue | 1 | 0 | 0 | I | Moderate |
| 20 | 71 | M | Tongue | 1 | 0 | 0 | I | Well |
| 21 | 82 | M | Oral floor | 1 | 0 | 0 | I | Well |
| 22 | 81 | M | Tongue | 1 | 0 | 0 | I | Extremely well |
In this study, we used the following human HNSCC
cells: SAS (derived from a primary lesion of tongue squamous cell
carcinoma) and HSC3 (derived from human lymph node metastasis of
tongue squamous cell carcinoma), as described previously.
Mature miRNA and small interfering RNA
(siRNA) transfection into HNSCC cells
The following RNA species were used in this study:
mature miRNAs, Pre-miR miRNA Precursors (hsa-miR-145-3p,
assay ID: PM 13036; hsa-miR-145-5p, assay ID: PM 11480),
negative control miRNA (assay ID: AM 17111) (both from Applied
Biosystems, Foster City, CA, USA), siRNA (Stealth Select RNAi
siRNA; si-MYO1B P/N: HSS106714 and HSS106716; Invitrogen,
Carlsbad, CA, USA). The transfection procedures were described
previously (16–20).
Quantitative real-time reverse
transcription polymerase chain reaction (qRT-PCR)
The procedure for PCR quantification was described
previously (16–19). TaqMan probes and primers for
MYO1B (P/N: Hs00362654_m1; Applied Biosystems) were
assay-on-demand gene expression products. Expression for
miR-145-3p (P/N: 002149; Applied Biosystems) and
miR-145-5p (P/N: 002278) was used to quantify the expression
levels of miRNAs according to the manufacturer's protocol. To
normalize the data for quantification of mRNA and miRNAs, we used
human GUSB (P/N: Hs99999908_m1), glyceraldehyde 3-phosphate
dehydrogenase (GAPDH) (P/N: Hs02758991_m1) and RNU48
(assay ID: 001006) (all from Applied Biosystems). The relative
expression levels were analyzed using the 2−ΔΔCT
method.
Cell proliferation, migration, and
invasion assays
Cell proliferation, migration and invasion assays
were described previously (16–19).
Incorporation of miR-145-3p or miR-145-5p
into the RISC by Ago2 immunoprecipitation
SAS cells were transfected with 10 nM miRNA by
reverse transfection. After 48 h, immunoprecipitation was performed
using a human Ago2 miRNA isolation kit (Wako, Osaka, Japan)
according to the manufacturer's protocol. Expression levels of
miR-145-3p or miR-145-5p were measured by qRT-PCR.
miRNA data were normalized to the expression of miR-150-5p (P/N:
PM10070; Applied Biosystems), which was not affected by
miR-145-3p and miR-145-5p transfection.
Western blot analysis
Cells were harvested and lysed 48 h after
transfection. Each cell lysate (50 µg of protein) was
separated using Mini-PROTEAN TGX gels (Bio-Rad, Hercules, CA, USA)
and transferred to polyvinylidene difluoride membranes.
Immunoblotting was performed with monoclonal anti-MYO1B
antibodies (1:250 dilution; HPA013607; Sigma-Aldrich, St. Louis,
MO, USA). Anti-glyceraldehyde 3-phosphate dehydrogenase (GAPDH)
antibodies (1:1,000 dilution; ab8245; Abcam, Cambridge, UK) were
used as an internal control. The procedures were described in our
previous studies (16–19).
Identification of putative genes
regulated by miR-145-3p in HNSCC cells
Specific genes regulated by miR-145-3p were
identified by a combination of in silico and genome-wide
gene expression analyses. Genes regulated by miR-145-3p were
listed using the TargetScan database. Oligo microarrays (Human GE
60K; Agilent Technologies) were used for gene expression analyses.
The microarray data were deposited into GEO (http://www.ncbi.nlm.nih.gov/geo/), with accession
number GSE82108. Upregulated genes in HNSCC were obtained from
publicly available data sets in GEO (accession no. GSE9638). To
identify signaling pathways regulated in silico, gene
expression data were analyzed using the KEGG pathway categories
with the GeneCodis program.
Regulation of targets downstream of MYO1B
in HNSCC
We investigated pathways regulated by MYO1B
in HNSCC cells. We analyzed gene expression using
si-MYO1B-transfected SAS cells. Microarray data were used
for expression profiling of si-MYO1B transfectants. The
microarray data were deposited into GEO (accession no. GSE100746).
We analyzed common downregulated genes using the GEO dataset.
Plasmid construction and dual-luciferase
reporter assay
The partial wild-type sequence of the MYO1B
3′-untranslated region (3′-UTR) was inserted between the
XhoI-PmeI restriction sites in the 3′-UTR of the
hRluc gene in the psiCHECK-2 vector (C8021; Promega, Madison, WI,
USA). Alternatively, we used sequences that were missing the
miR-145-3p target sites (position 88–94 or position
1117–1123). The synthesized DNA was cloned into the psiCHECK-2
vector. SAS cells were transfected with 20 ng of the vector, 20 nM
microRNAs, and 1 µl Lipofectamine 2000 in 100 µl
Opti-MEM (both from Invitrogen). The procedure of dual-luciferase
reporter assay was described previously (16–19).
Immunohistochemistry
Formalin-fixed, paraffin-embedded (FFPE) tissues
were used. Tissue sections were incubated overnight at 4°C with
anti-MYO1B antibodies diluted 1:300 (HPA013607;
Sigma-Aldrich). The procedure for immunohistochemistry was
described previously (21).
The Cancer Genome Atlas (TCGA)-HNSCC data
analysis
To explore the clinical significance of MYO1B
in HNSCC, we used the RNA sequencing database in TCGA (https://tcga-data.nci.nih.gov/tcga/). The gene
expression and clinical data were retrieved from cBioportal
(http://www.cbioportal.org/, the
provisional data downloaded July 1, 2017).
Statistical analysis
Relationships between two or three variables and
numerical values were analyzed using Mann-Whitney U tests or
Bonferroni-adjusted Mann-Whitney U tests. Spearman's rank tests
were used to evaluate the correlations between the expression of
miR-145-3p or miR-145-5p and target genes. Expert
StatView software (version 5.0; SAS Institute Inc., Cary, NC, USA)
was used for these analyses. Multivariate Cox proportional hazard
regression models were used to determine independent factors for
survival with JMP Pro 13.
Results
Expression levels of miR-145-5p and
miR-145-3p in HNSCC clinical specimens and cell lines
To confirm our miRNA expression signatures in HNSCC
by RNA sequencing, we validated the expression levels of
miR-145-5p and miR-145-3p in HNSCC clinical specimens
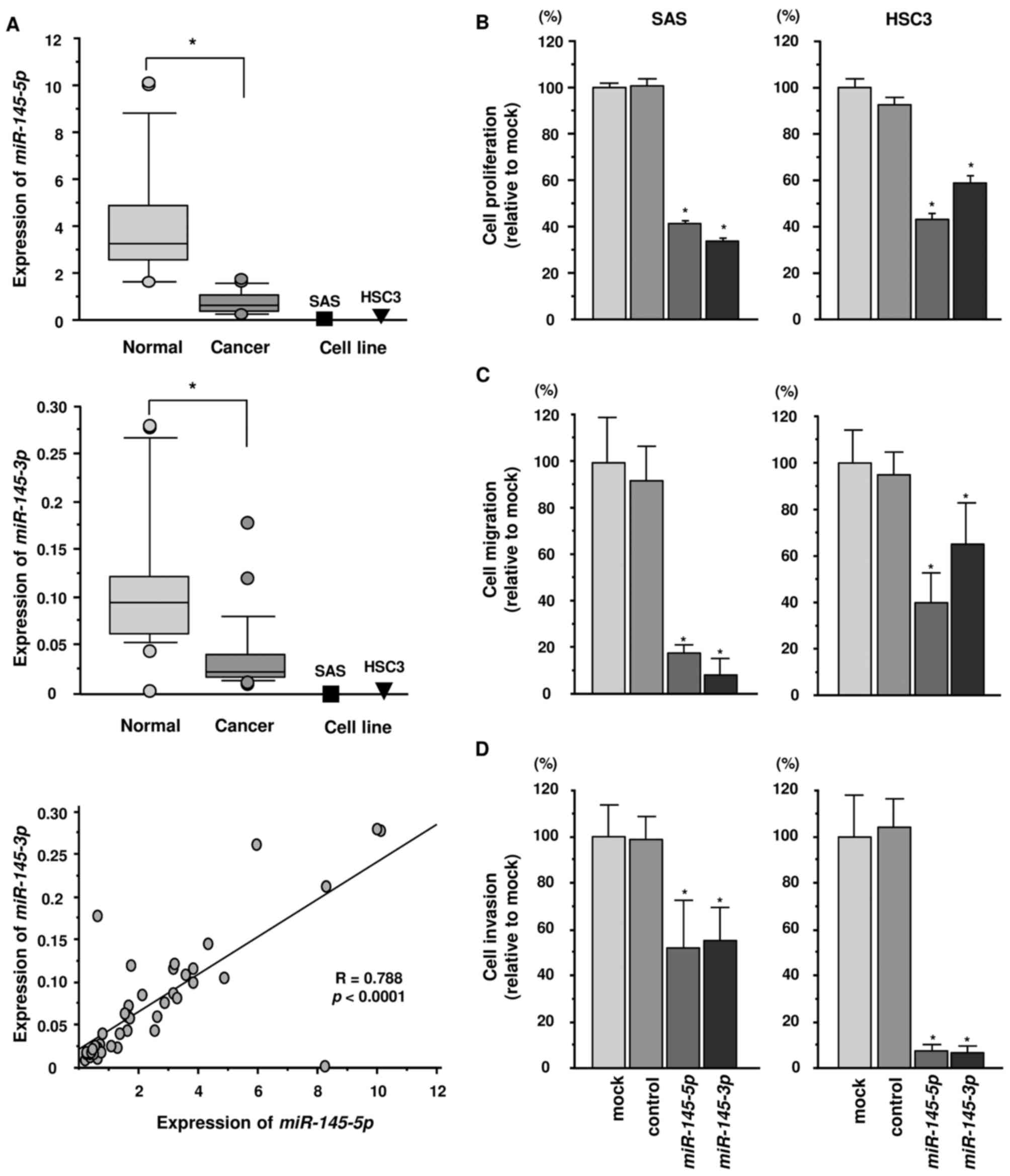
and cell lines. In Fig. 1, the
expression levels of miR-145-5p and miR-145-3p were
significantly reduced in cancer tissues compared with those in
corresponding adjacent noncancerous epithelium (p<0.0001)
(Fig. 1A). Additionally, the
expression levels of miR-145-5p and miR-145-3p in SAS
and HSC3 cells were markedly downregulated (Fig. 1A).
Spearman's rank test showed a positive correlation
between the expression levels of miR-145-5p and
miR-145-3p in clinical specimens (Fig. 1A).
Effects of ectopic expression of
miR-145-5p and miR-145-3p on cell proliferation, migration and
invasion in HNSCC cell lines
To validate the functional roles of
miR-145-3p and miR-145-5p, we carried out
gain-of-function assays using miRNA transfection into two HNSCC
cell lines (SAS and HSC3). XTT assays revealed that cell
proliferation was significantly inhibited in miR-145-3p and
miR-145-5p transfectants in comparison with mock or
miR-control transfectants (Fig.
1B). Similarly, migration assays showed that cell migration
activity was significantly inhibited in miR-145-3p and
miR-145-5p transfectants in comparison with mock and
miR-control transfectants (Fig.
1C). Matrigel invasion assays also demonstrated that cell
invasion activity was significantly inhibited in miR-145-3p
and miR-145-5p transfectants in comparison with mock and
miR-control transfectants (Fig.
1D).
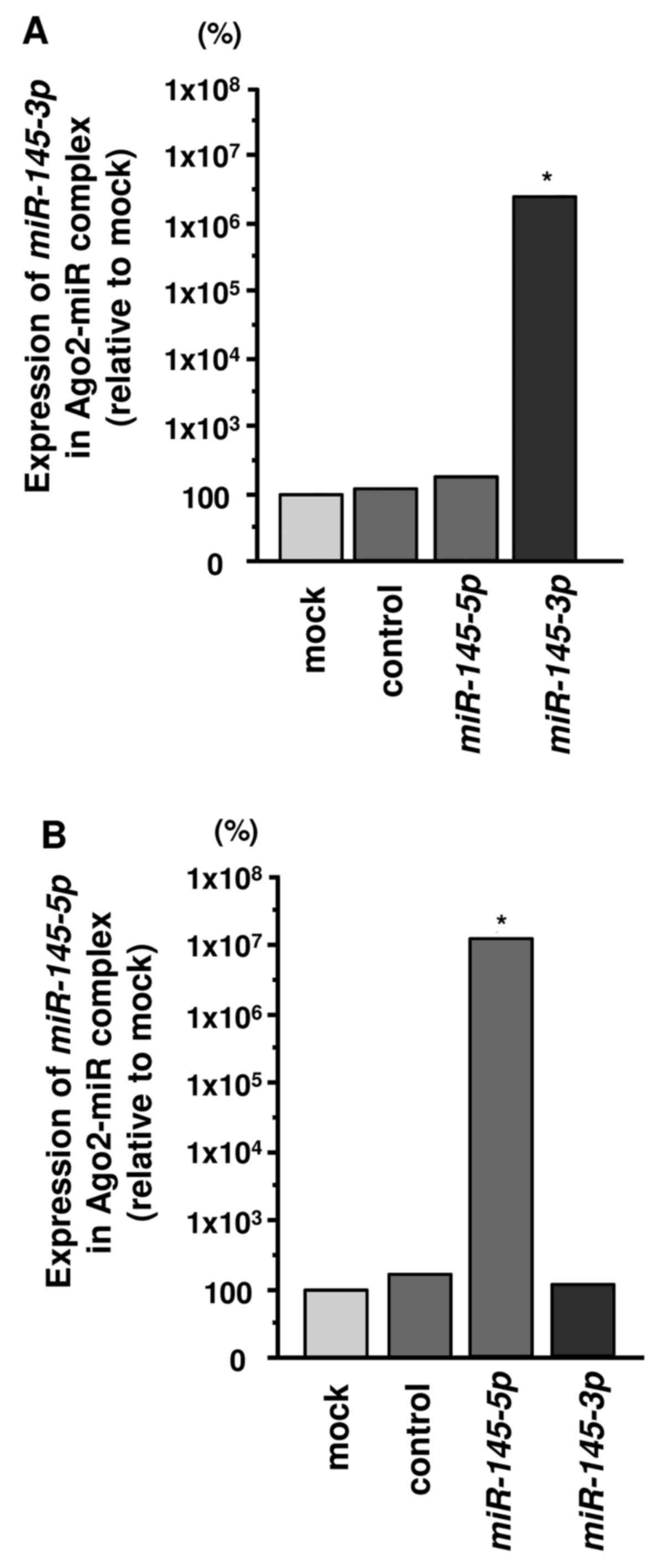
Incorporation of miR-145-3p into the RISC
in HNSCC cells
We hypothesized that the passenger strand
miR-145-3p may be incorporated into the RISC and exert
important effects in cancer cells. Accordingly, we performed
immunoprecipitation with antibodies targeting Ago2, which plays an
important role in the RISC. After transfection with
miR-145-3p or miR-145-5p, Ago2-bound miRNAs were
isolated, and qRT-PCR was carried out to determine whether
miR-145-3p and miR-145-5p bound to Ago2. After
transfection with miR-145-3p and immunoprecipitation by
anti-Ago2 antibodies, miR-145-3p levels were significantly
higher than those of mock- or miR-control-transfected cells and
those of miR-145-5p-transfected SAS cells (p<0.0001)
(Fig. 2A). Similarly, after
miR-145-5p transfection, miR-145-5p was detected by
Ago2 immunoprecipitation (p<0.0001) (Fig. 2B).
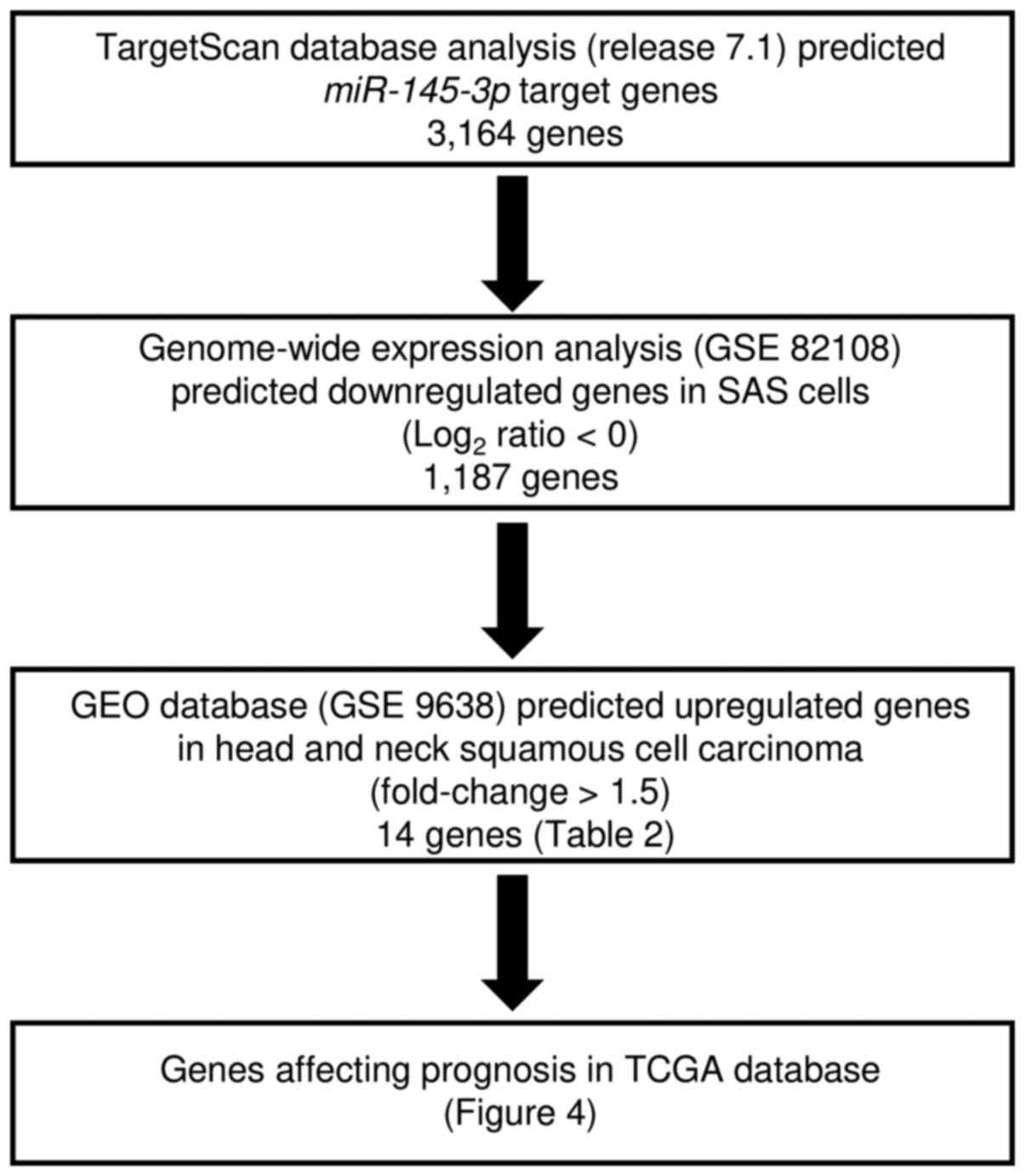
Identification of putative targets of
miR-145-3p regulation in HNSCC cells
We performed in silico and gene expression
analyses to identify genes targeted by miR-145-3p for
regulation (Fig. 3). First, we
selected putative miR-145-3p target genes using the
TargetScan database and identified 3,164 genes. Next, we performed
comprehensive gene expression analysis using miR-145-3p
transfectants of SAS, with negative control miRNA transfectants
serving as controls (accession no. GSE 82108). A total of 1,187
genes were commonly downregulated (log2 ratio<0). The
gene set was then analyzed with a publicly available gene
expression data set in GEO (accession no. GSE9638), and genes
upregulated in HNSCC were chosen (fold-change >1.5). A total of
14 genes were identified as candidate targets of miR-145-3p
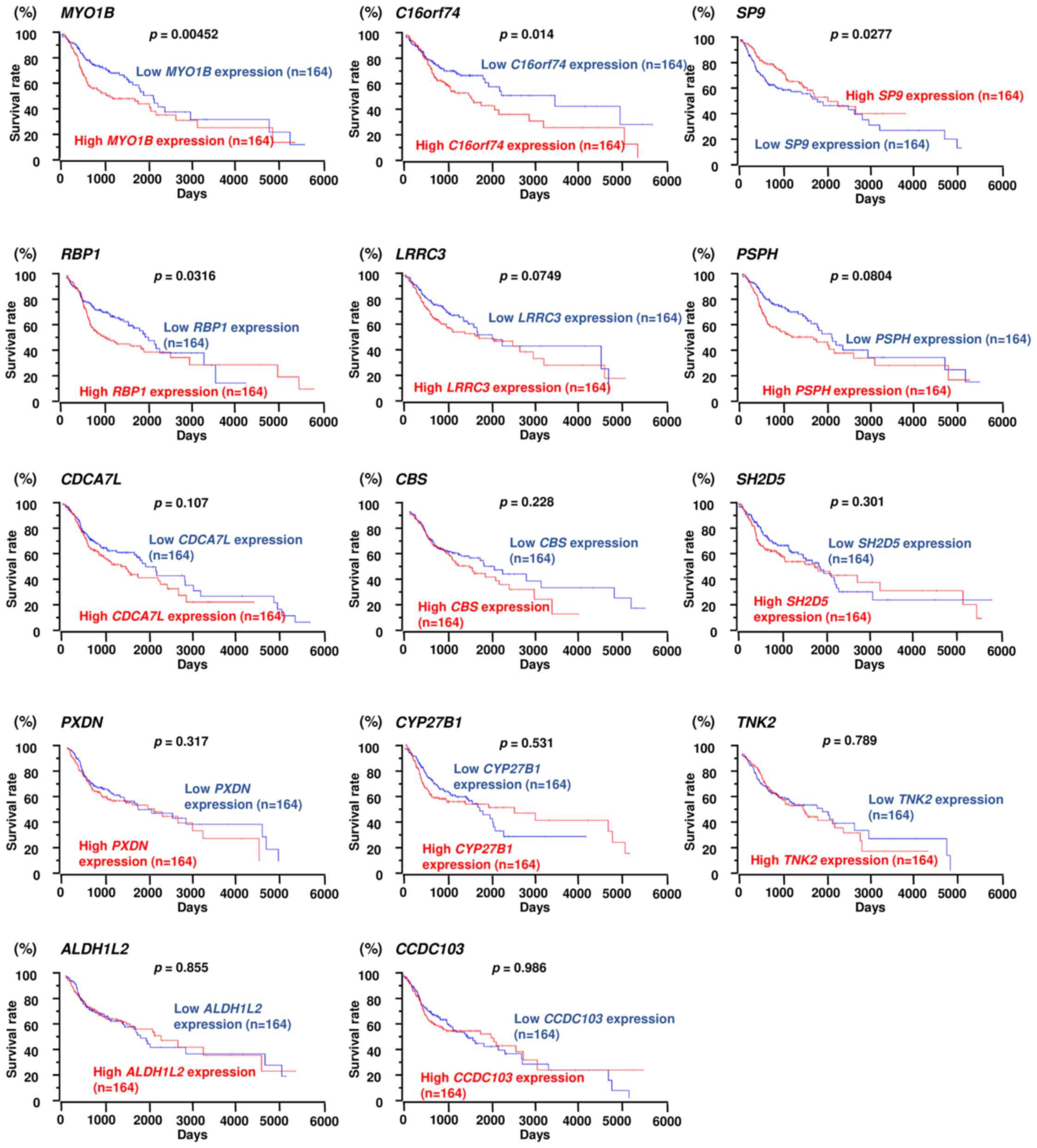
regulation (Table II). Next,
these genes were validated with TCGA database, and we investigated
the correlations between survival rates and target genes with high
or low expression. In this study, 3 genes (MYO1B,
C16orf74 and RBP1) were selected as genes that
affected the patient's overall survival (Table II and Fig. 4). Among them, MYO1B was
found to have the greatest effect on the overall survival rate
(p=0.00452). In this study, we focused on MYO1B as a
candidate target gene of miR-145-3p regulation and
investigated the functional roles of HNSCC cells.
 | Table IIPutative targets of miR-145-3p
regulation in HNSCC cells. |
Table II
Putative targets of miR-145-3p
regulation in HNSCC cells.
| Gene symbol | Gene name | Conserved site
count | SAS
miR-145-3p transfection | HNSCC
fold-change | Prognosis (high vs.
low) p-value |
|---|
| MYO1B | Myosin IB | 2 | −1.49 | 1.72 | 0.00452 |
| C16orf74 | Chromosome 16 open
reading frame 74 | 1 | −0.88 | 1.97 | 0.014 |
| SP9 | Sp9 transcription
factor | 1 | −0.97 | 2.38 | 0.0277a |
| RBP1 | Retinol binding
protein 1, cellular | 1 | −1.2 | 2.6 | 0.0316 |
| LRRC3 | Leucine rich repeat
containing 3 | 2 | −0.86 | 1.54 | 0.0749 |
| PSPH | Phosphoserine
phosphatase | 1 | −0.8 | 1.95 | 0.0804 |
| CDCA7L | Cell division cycle
associated 7-like | 1 | −0.88 | 1.71 | 0.107 |
| CBS |
Cystathionine-β-synthase | 2 | −1.29 | 1.54 | 0.228 |
| SH2D5 | SH2 domain
containing 5 | 1 | −1.87 | 2.34 | 0.301 |
| PXDN | Peroxidasin homolog
(Drosophila) | 1 | −0.94 | 1.63 | 0.317 |
| CYP27B1 | Cytochrome P450,
family 27, subfamily B, polypeptide 1 | 2 | −0.86 | 2.65 | 0.531 |
| TNK2 | Tyrosine kinase,
non-receptor, 2 | 1 | −1.08 | 2.3 | 0.789 |
| ALDH1L2 | Aldehyde
dehydrogenase 1 family, member L2 | 1 | −1.19 | 2.3 | 0.855 |
| CCDC103 | Coiled-coil domain
containing 103 | 1 | −1.77 | 2.03 | 0.986 |
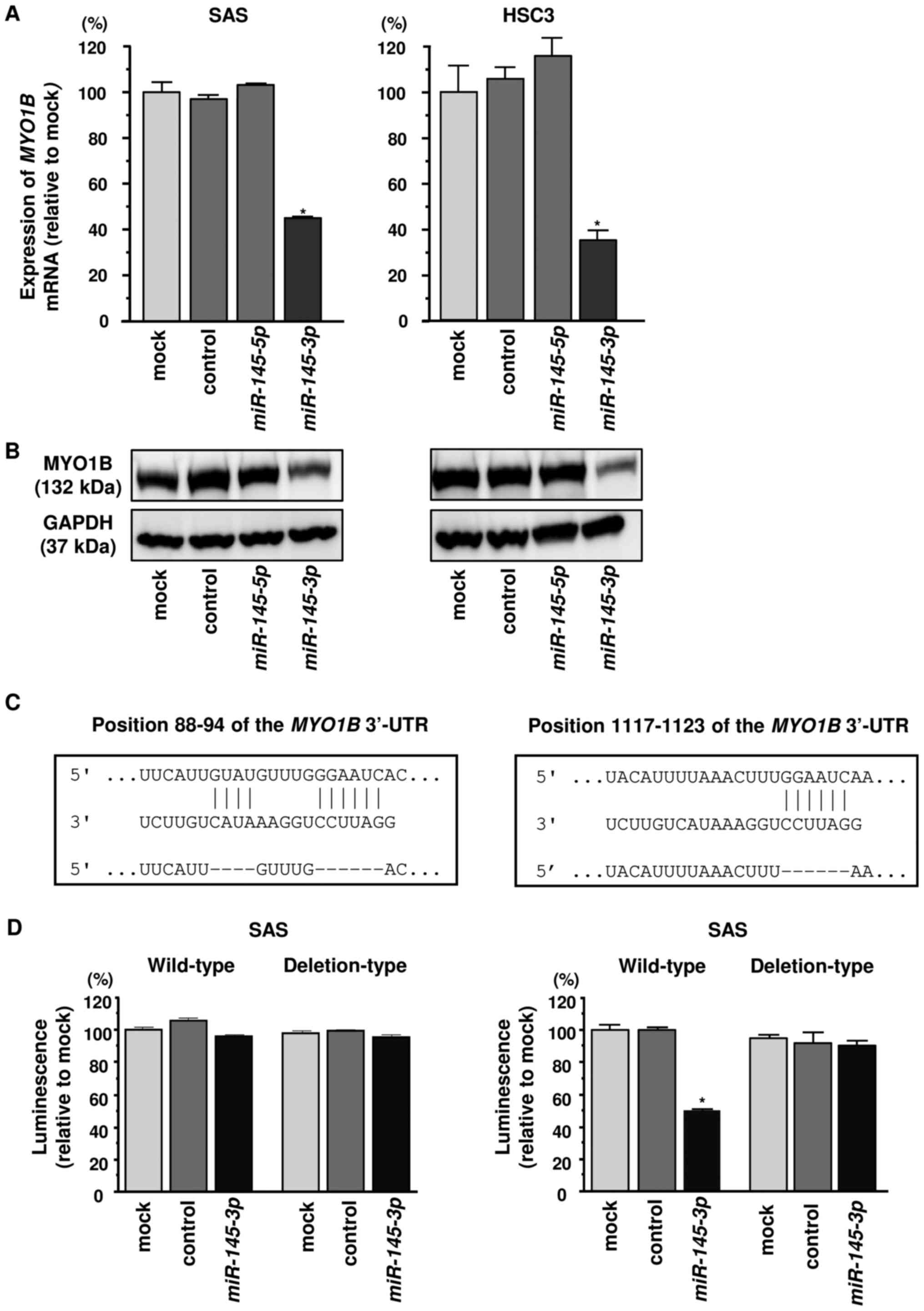
Direct regulation of MYO1B by miR-145-3p
in HNSCC cells
Next, we investigated whether the expression of
MYO1B decreased in miR-145-3p-transfected HNSCC
cells. MYO1B mRNA levels were significantly reduced by
miR-145-3p transfection compared with the mock or
miR-control transfectants (Fig.
5A). Furthermore, MYO1B protein levels were also reduced
by miR-145-3p transfection compared with mock or miR-control
transfectants (Fig. 5B). In
contrast, miR-145-5p transfectants did not show altered
expression of MYO1B mRNA or protein (Fig. 5A and B).
We then carried out luciferase reporter assays with
a vector that included the 3′-UTR of MYO1B to confirm that
miR-145-3p directly regulated MYO1B in a
sequence-dependent manner. TargetScan Human database predicted that
there were two binding sites for miR-145-3p in the 3′-UTR of
MYO1B (positions 88–94 and 1117–1123) (Fig. 5C). Cotransfection with
miR-145-3p and vectors significantly reduced luciferase
activity in comparison with those in mock and miR-control
transfectants in position 1117–1123 of the MYO1B 3′-UTR
(Fig. 5D).
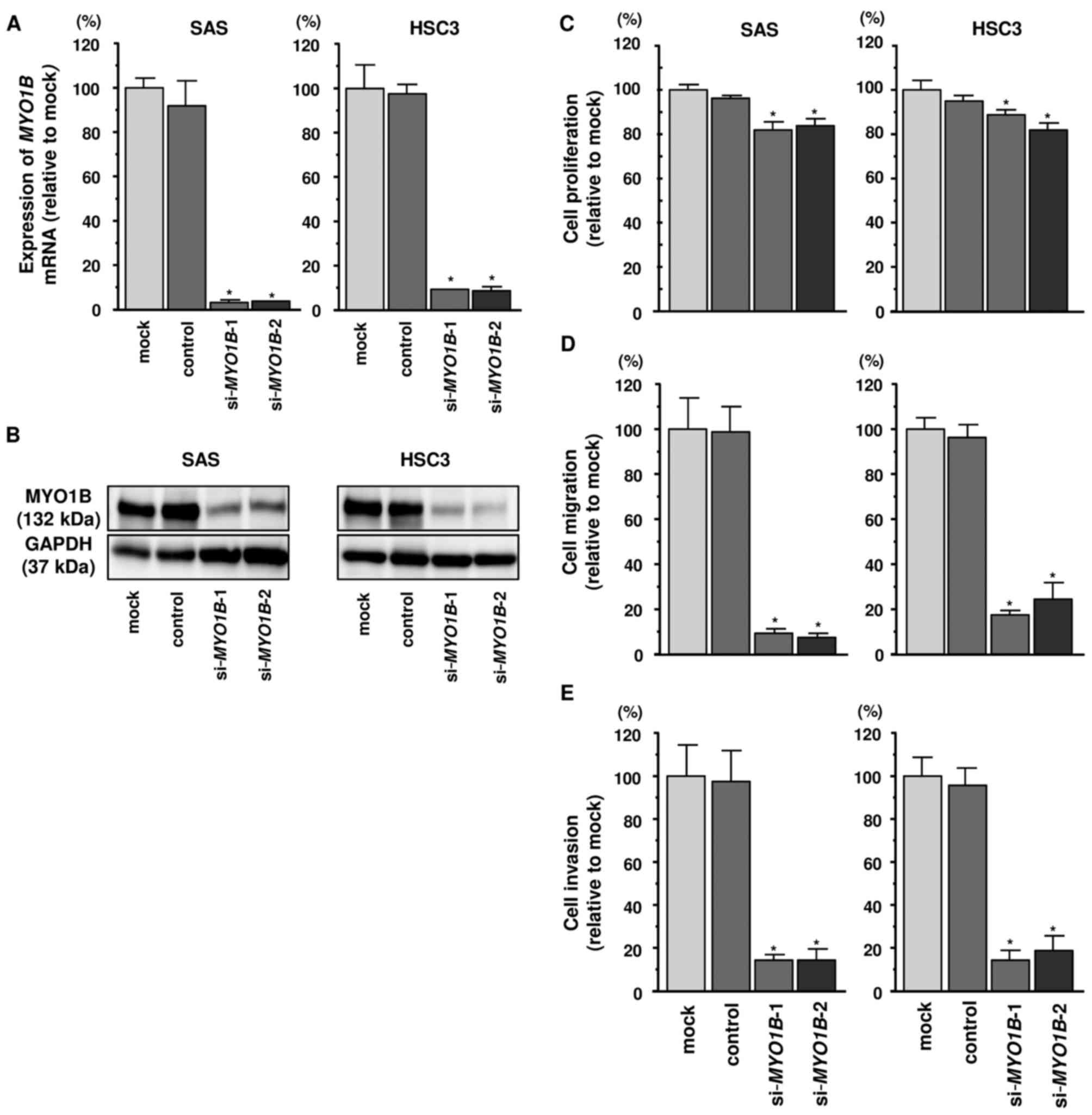
Effects of MYO1B knockdown on cell
proliferation, migration, and invasion in HNSCC cell lines
A loss-of-function assay using siRNA was performed
to examine the function of MYO1B in HNSCC cell lines. The
expression levels of MYO1B mRNA and protein were reduced by
si-MYO1B in HNSCC cell lines (Fig. 6A and B). Furthermore, we
investigated effects of MYO1B knockdown on cell
proliferation, migration, and invasion in HNSCC cell lines. Cancer
cell proliferation was significantly reduced in si-MYO1B
transfectants in comparison with that in mock- or miR
control-transfected cell lines (Fig.
6C). Additionally, migration activities were significantly
suppressed in si-MYO1B transfectants in comparison with that
in mock- or miR control-transfected cell lines (Fig. 6D). Invasion activity was also
significantly inhibited in si-MYO1B transfectants in
comparison with that in mock- or miR control-transfected cell lines
(Fig. 6E).
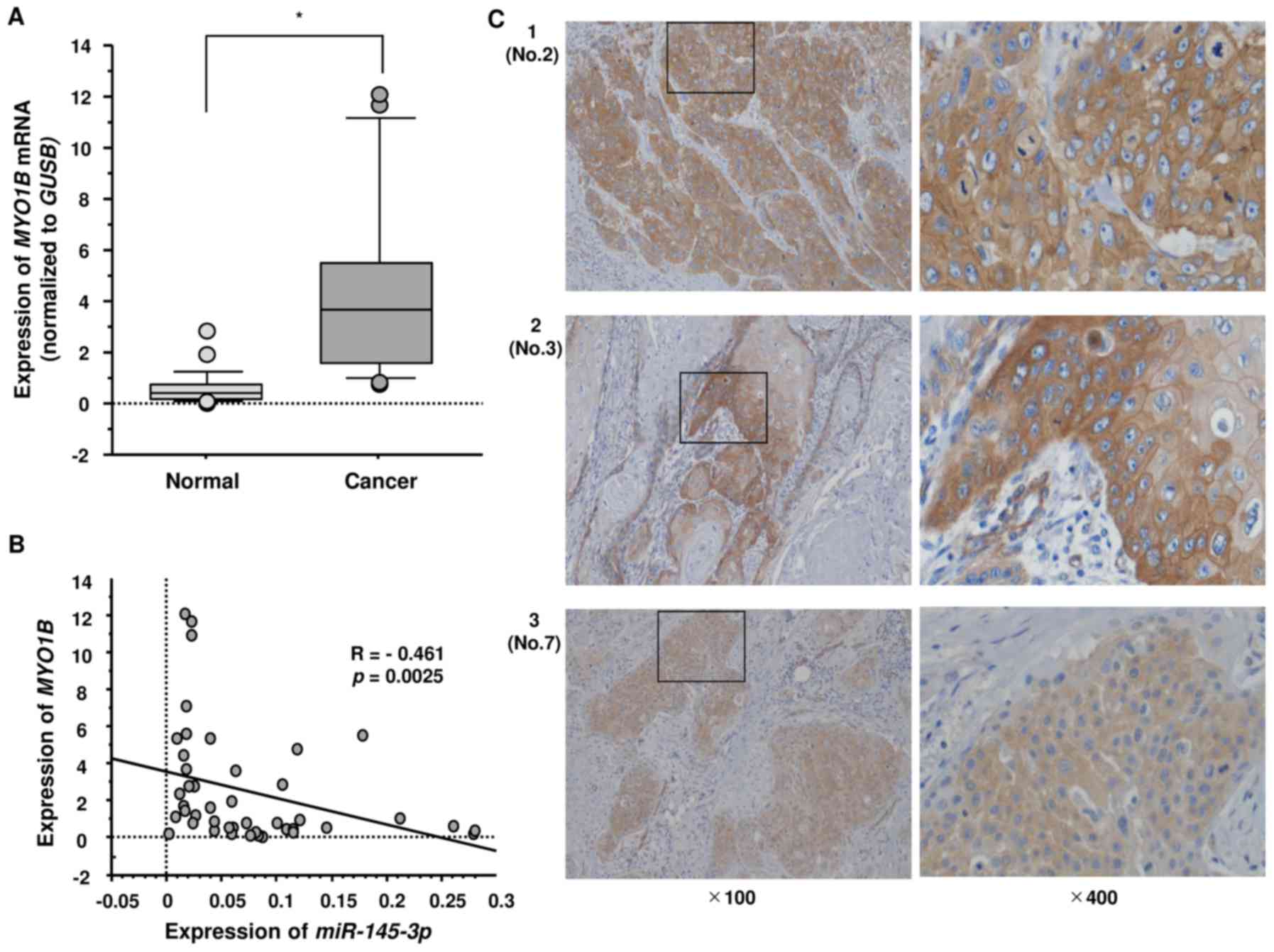
Expression of MYO1B in HNSCC clinical
specimens
Next, we investigated the mRNA expression levels of
MYO1B in 22 HNSCC clinical specimens by qRT-PCR.
MYO1B was significantly upregulated in HNSCC tumor tissues
(Fig. 7A). Spearman's rank test
showed a negative correlation between the expression of
MYO1B and miR-145-3p (p=0.0025, R=−0.461) (Fig. 7B). Furthermore, we also examined
the expression levels of MYO1B in HNSCC clinical specimens
by immunostaining. MYO1B was strongly expressed in several
cancer tissues (Fig. 7C: 1,
patient no. 2; 2, no. 3; 3, no. 7 in Table I).
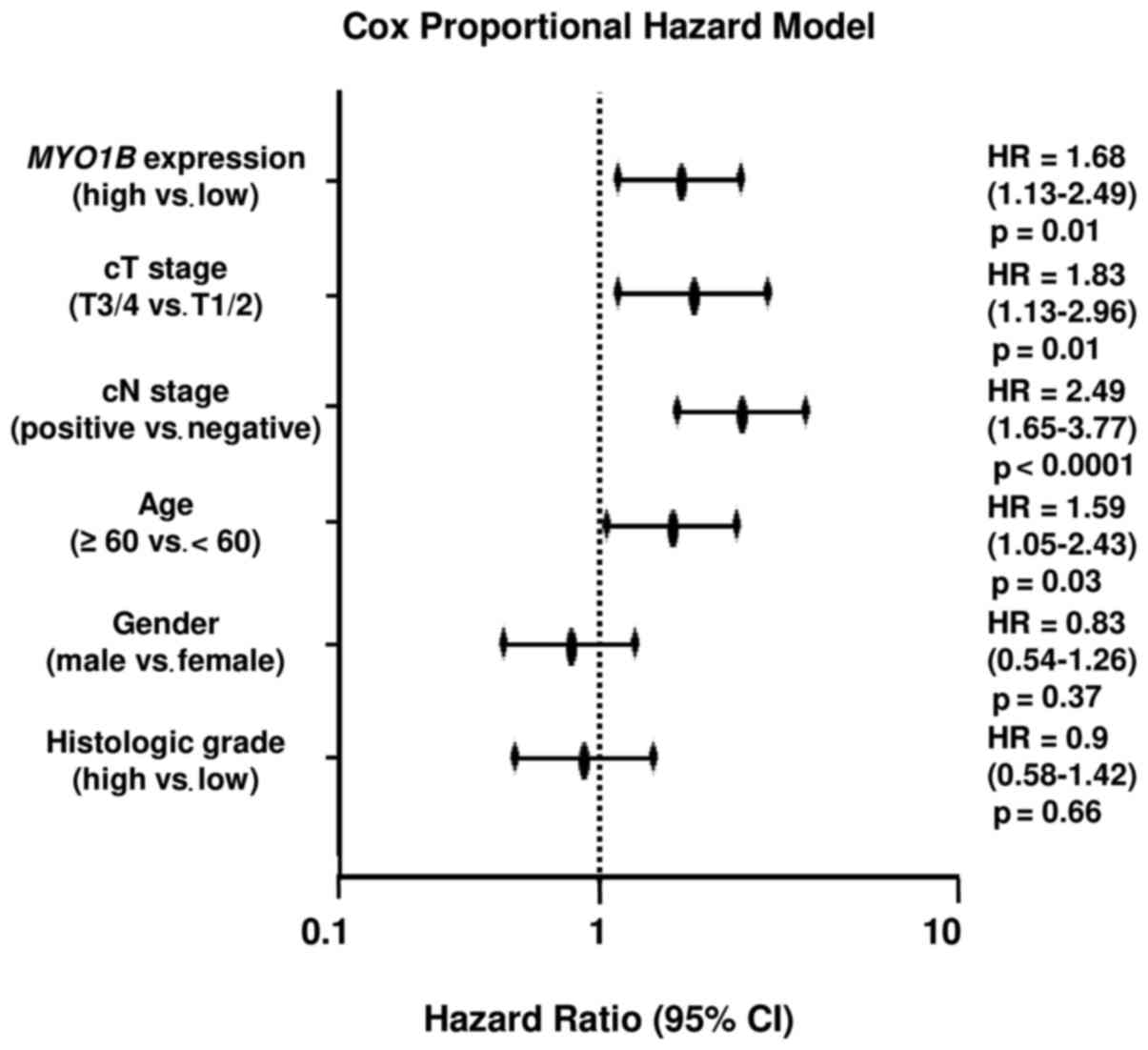
Correlation between MYO1B expression and
clinicopathological characteristics in prognostic prediction in
HNSCC specimens
We collected clinical data from TCGA database and
analyzed clinicopathological factors and expression of MYO1B
as a prognostic predictive factor. The multivariate cox
proportional hazards model was used to validate independent
predictors for overall survival, including MYO1B expression,
clinical T stage, clinical N stage, age, sex and histologic grade.
As a result, high expression of MYO1B was an independent
predictive factor for survival [hazard ratio (HR), 1.68; 95%
confidence interval (CI), 1.13–2.49; p=0.01] (Fig. 8).
Downstream genes affected by silencing of
MYO1B in SAS cells
Finally, we performed genome-wide gene expression
analysis using si-MYO1B in SAS cells to investigate which
genes were mediated by MYO1B signaling. A SurePrint G3 Human
GE 60K v3 microarray was used for genome-wide expression analysis.
We submitted the raw data to the GEO database (accession no.
GSE100746). In this study, we focused on significantly
downregulated genes by both si-MYO1B-1 and si-MYO1B-2
transfection (log2 [si-MYO1B/mock] <−1.5).
MYO1B was the most significantly downregulated gene,
indicating that the array data were worthy of evaluation. Genes
significantly downregulated by silencing of MYO1B are listed
in Table III. Among MYO1B
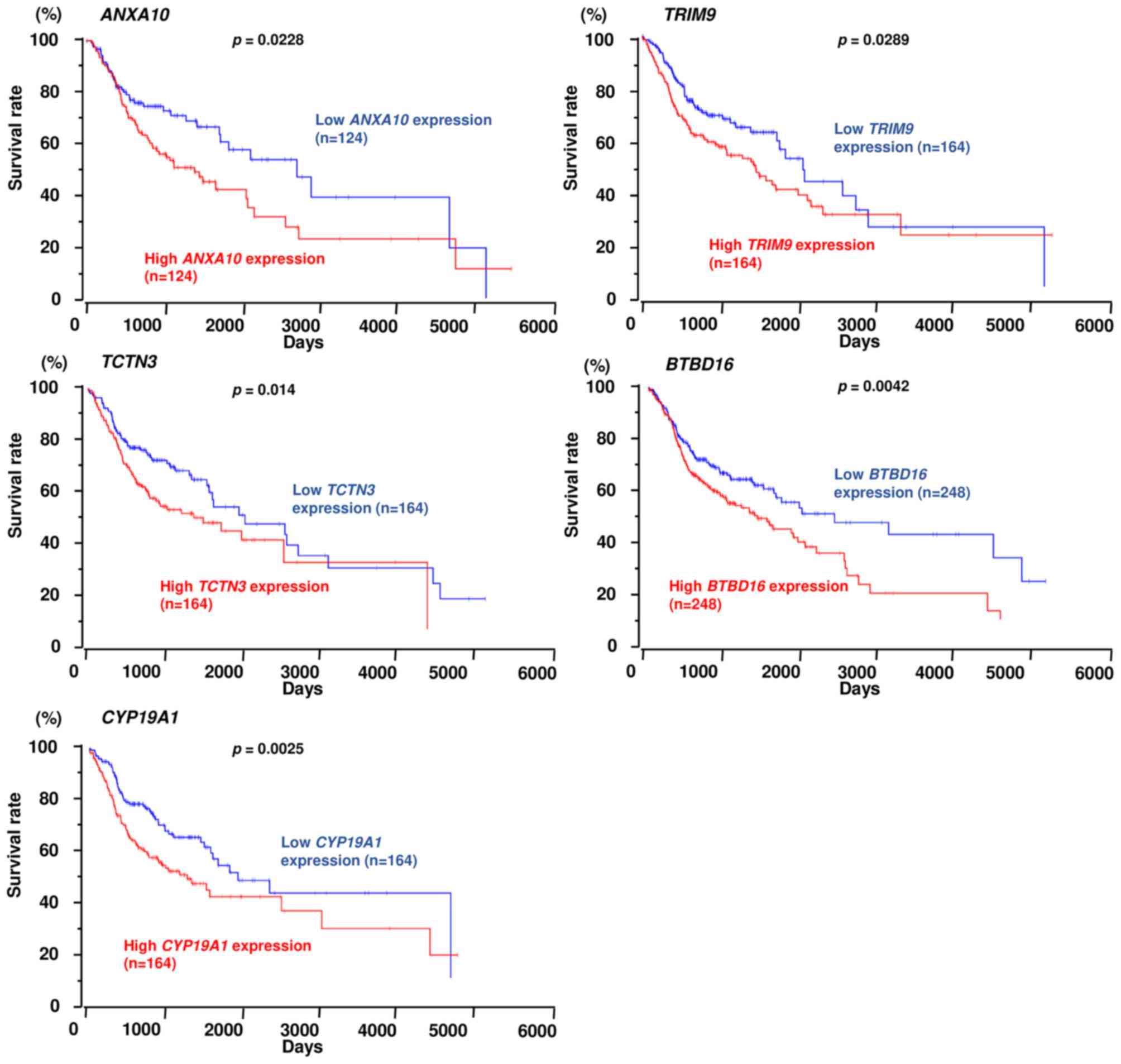
downstream genes, expression of 5 genes (ANXA10,
TRIM9, TCTN3, BTBD16 and CYP19A1) was
significantly associated with poor prognosis in patients with HNSCC
based on TCGA database (Fig.
9).
 | Table IIIIdentification of MYO1B
downstream genes in HNSCC cells. |
Table III
Identification of MYO1B
downstream genes in HNSCC cells.
| Gene symbol | Gene name | Log2
(si-MYO1B-1/mock) | Log2
(si-MYO1B-2/mock) | Average
Log2 (si-MYO1B/mock) |
|---|
| MYO1B | Myosin IB | −4.007414 | −4.668526 | −4.337970 |
| ANXA10 | Annexin A10 | −3.842131 | −2.8575826 | −3.349857 |
| MATN3 | Matrilin 3 | −4.224010 | −2.0824907 | −3.153250 |
| SOHLH1 | Spermatogenesis and
oogenesis specific basic helix-loop-helix 1 | −4.337910 | −1.8513346 | −3.094622 |
| SMAD1-AS1 | SMAD1 antisense RNA
1 | −3.191749 | −2.902154 | −3.046952 |
| KRT6B | Keratin 6B, type
II | −3.626921 | −2.0426638 | −2.834793 |
| KLK13 | Kallikrein-related
peptidase 13 | −3.540325 | −1.9546604 | −2.747493 |
| PAX6 | Paired box 6 | −2.130839 | −3.0853565 | −2.608098 |
| C5orf66-AS1 | C5orf66 antisense
RNA 1 | −2.704701 | −2.1148643 | −2.409783 |
| PDGFRB | Platelet-derived
growth factor receptor, β polypeptide | −2.886416 | −1.8465691 | −2.366492 |
| HSD17B2 | Hydroxysteroid
(17-β) dehydrogenase 2 | −2.325892 | −2.3811436 | −2.353518 |
| SP140 | SP140 nuclear body
protein | −2.470807 | −2.1072135 | −2.289010 |
| OR9G4 | Olfactory receptor,
family 9, subfamily G, member 4 | −2.296291 | −2.1278794 | −2.212085 |
| FOXD3-AS1 | FOXD3 antisense RNA
1 (head to head) | −1.846461 | −2.4128325 | −2.129647 |
| MAGEB17 | Melanoma antigen
family B, 17 | −2.394958 | −1.7456088 | −2.070283 |
| AMDHD1 | Amidohydrolase
domain containing 1 | −2.223687 | −1.916799 | −2.070243 |
| IGFBP1 | Insulin-like growth
factor binding protein 1 | −2.512259 | −1.5926342 | −2.052446 |
| MMP1 | Matrix
metallopeptidase 1 (interstitial collagenase) | −1.821691 | −2.2728753 | −2.047283 |
| EN1 | Engrailed homeobox
1 | −1.834606 | −2.220468 | −2.027537 |
| FGF13-AS1 | FGF13 antisense RNA
1 | −2.349053 | −1.6906263 | −2.019840 |
| ZC3H12D | Zinc finger
CCCH-type containing 12D | −2.232335 | −1.8071643 | −2.019749 |
| KRT6A | Keratin 6A, type
II | −2.307169 | −1.6317264 | −1.969448 |
| FAM196B | Family with
sequence similarity 196, member B | −1.855253 | −2.031945 | −1.943599 |
| DNMT3B | DNA
(cytosine-5-)-methyltransferase 3β | −1.530055 | −2.3343146 | −1.932185 |
| LIN28A | Lin-28 homolog A
(C. elegans) | −2.292716 | −1.5482489 | −1.920483 |
| ZNF501 | Zinc finger protein
501 | −1.788270 | −2.042201 | −1.915235 |
| REC114 | REC114 meiotic
recombination protein | −2.230243 | −1.5574937 | −1.893868 |
| TRIM9 | Tripartite motif
containing 9 | −2.078308 | −1.691343 | −1.884826 |
| ZBED3-AS1 | ZBED3 antisense RNA
1 | −2.140241 | −1.6105609 | −1.875401 |
| PHKA2-AS1 | PHKA2 antisense RNA
1 | −2.028810 | −1.7067645 | −1.867787 |
| ZDHHC22 | Zinc finger,
DHHC-type containing 22 | −2.044774 | −1.6702744 | −1.857524 |
| SCAND2P | SCAN domain
containing 2 pseudogene | −1.924617 | −1.7586662 | −1.841642 |
| SPRR1B | Small proline-rich
protein 1B | −1.967544 | −1.7028618 | −1.835203 |
| SLC35D3 | Solute carrier
family 35, member D3 | −1.638228 | −2.0054185 | −1.821823 |
| ANO1-AS2 | ANO1 antisense RNA
2 (head to head) | −1.763001 | −1.8544457 | −1.808723 |
| C22orf23 | Chromosome 22 open
reading frame 23 | −1.741993 | −1.8479792 | −1.794986 |
| TCTN3 | Tectonic family
member 3 | −1.880425 | −1.6976513 | −1.789038 |
| FAM198A | Family with
sequence similarity 198, member A | −1.940336 | −1.6032256 | −1.771781 |
| TG | Thyroglobulin | −1.719962 | −1.8222181 | −1.771090 |
| RASGEF1A | RasGEF domain
family, member 1A | −1.865154 | −1.6762245 | −1.770689 |
| KATNAL2 | Katanin p60 subunit
A-like 2 | −1.982902 | −1.5259477 | −1.754425 |
| KLHL14 | Kelch-like family
member 14 | −1.575037 | −1.9020982 | −1.738568 |
| NANOS1 | Nanos homolog 1
(Drosophila) | −1.785024 | −1.6778822 | −1.731453 |
| BTBD16 | BTB (POZ) domain
containing 16 | −1.769118 | −1.6703396 | −1.719729 |
| APOL4 | Apolipoprotein L,
4 | −1.542360 | −1.8804932 | −1.711427 |
| ZNF385C | Zinc finger protein
385C | −1.792121 | −1.6219425 | −1.707032 |
| ABO | ABO blood
group | −1.624970 | −1.7806495 | −1.702810 |
| CD200R1 | CD200 receptor
1 | −1.778945 | −1.5290467 | −1.653996 |
| VWA3A | Von Willebrand
factor A domain containing 3A | −1.527752 | −1.7406074 | −1.634180 |
| CYP19A1 | Cytochrome P450,
family 19, subfamily A, polypeptide 1 | −1.572258 | −1.6706244 | −1.621441 |
| ZNF880 | Zinc finger protein
880 | −1.604488 | −1.6321578 | −1.618323 |
| NKX6-2 | NK6 homeobox 2 | −1.597461 | −1.6252115 | −1.611336 |
| GPR157 | G protein-coupled
receptor 157 | −1.583724 | −1.6133837 | −1.598554 |
| ST3GAL5-AS1 | ST3GAL5 antisense
RNA 1 (head to head) | −1.526179 | −1.5542628 | −1.540221 |
Discussion
Accumulating evidence has shown that aberrant
expression of miRNAs disrupts the well-ordered RNA networks in
cancer cells and is involved in the pathogenesis of human cancers
(22). Based on the miRNA
expression signatures of human cancers, we have sequentially
identified antitumor miRNAs that regulate novel cancer networks
(16,23–26).
Analyses of our miRNA signature of HNSCC by RNA sequencing showed
that several passenger strands of miRNAs were significantly
downregulated in cancer tissues (8). Our recent study demonstrated that
both strands of pre-miR-150 (miR-150-5p, guide
strand; and miR-150-3p, passenger strand) had antitumor
functions and that these miRNAs cooperatively regulated oncogenic
ITGA3, ITGA6 and TNC in HNSCC cells (8). Our other studies showed that the
passenger strand of miR-150 acted as an anti-tumor miRNA in
several types of cancers, such as esophageal cancer and prostate
cancer (9,27). These findings suggested that miRNA
passenger strands also contribute substantially to cancer
pathogenesis and that identification of RNA networks mediated by
miRNA passenger strands may provide novel insights into the
pathogenesis of HNSCC.
Based on our miRNA signature of HNSCC, we focused
on the passenger strand miR-145-3p in this study. Similarly,
miR-145-5p, the guide strand of miR-145, was significantly
reduced in this signature. Downregulation of miR-145-5p is
frequently observed in many types of cancer, and prior studies have
confirmed the antitumor function of miR-145-5p by
demonstration of its effects on several types of oncogenes in
cancer cells (10,11). Several studies have shown that
downregulation of miR-145-5p is caused by hypermethylation
of the promoter region of pre-miR-145 in prostate cancer
(28). Importantly, the tumor
suppressor p53 has been shown to directly bind p53-response
elements in the promoter region of pre-miR-145 and to
control the expression of miR-145-5p (29). p53 mutations are found in >50%
of patients with HNSCC (30).
Thus, downregulation of miR-145-5p and miR-145-3p may
be dependent on p53 inactivation in cancer cells.
Expression levels of passenger strand of
miR-145-3p was lower than miR-145-5p as a guide
strand miRNA in HNSCC clinical specimens and cell lines. Our
previous studies of bladder, lung, and prostate cancers showed that
expression levels of miR-145-3p was lower than
miR-145-5p in each cancer (10,11,31).
The results of the present data of HNSCC was similar to our
previous data. Explanation is incomplete as to in what kind of
molecular mechanisms the expression of the two miRNAs differ. This
problem is an important issue in miRNA biosynthesis.
Our functional assays showed that miR-145-3p
had antitumor functions similar to miR-145-5p in HNSCC
cells. We have also demonstrated miR-145-3p is downregulated
in cancer tissues and acts as an antitumor miRNA in bladder, lung,
and prostate cancers by targeting several oncogenic genes (10,11,31).
Previous studies demonstrated that several oncogenic genes were
regulated by miR-145-5p in several types of cancers
(32–34). There are few studies for target
genes by miR-145-3p regulation in cancer cells, including
HNSCC cells. Thus, we evaluated miR-145-3p regulatory
oncogenic networks in HNSCC cells; a total of 14 putative targets
of miR-145-3p in HNSCC cells were identified in this study.
Among these candidates, MYO1B, C16orf74, SP9
and RBP1 were found to be associated with poor prognosis in
patients with HNSCC by TCGA data analyses.
In this study, we focused on MYO1B because
high expression of MYO1B was strongly associated with poor
prognosis in patients with HNSCC. Myosins are actin-associated
molecular motor proteins that regulate membrane tension, anchor
membrane proteins and organelles, and transport inter-cellular
vesicles (35,36). We have demonstrated that antitumor
miRNAs inhibited cancer cell migration and invasion through
targeting several actin-binding proteins and actin-associate
proteins, e.g., FSCN1, LASP1, ARPC5 and ANLN (37–40).
Overexpression of these proteins has been detected in cancer
tissues and has been shown to contribute to cancer cell
aggressiveness.
Our present data of restoration of
miR-145-5p or miR-145-3p showed the inhibition of
cancer cell proliferation. However, inhibition of cell
proliferation was weak by knockdown of MYO1B in HNSCC cells.
These data suggest that miR-145-5p or miR-145-3p
inhibite cell proliferation genes and pathways which do not rely on
MYO1B in HNSCC cells.
MYO1B belongs to a member of the
membrane-associated class I myosin family and functions as a linker
between membranes and the actin cytoskeleton in several cellular
processes (41). Previous studies
have demonstrated other functions of MYO1B. For example, MYO1B is
localized in the endocytotic compartment and has pivotal roles in
endocytosis (42). MYO1B couples
with the actin assembly to organelles and controls membrane
remodeling at the trans-Golgi network (43). In cancer cells, MYO1B is
highly expressed in PC-3 metastatic prostate cancer cells, and
knockdown of MYO1B affects the cytoskeleton and cell
migration (44). Another study
showed that knockdown of MYO1B significantly inhibits
migratory and invasive abilities of HNSCC cells in vitro and
in vivo (35). Our present
data confirmed these findings and suggested that MYO1B may be an
effective target for the treatment of HNSCC.
To identify MYO1B-mediated HNSCC pathways, we
performed genome-wide gene expression analyses using
si-MYO1B transfectants. A total of 54 genes were found to be
mediated by MYO1B in HNSCC. Among them, 5 genes (ANXA10,
TRIM9, TCTN3, BTBD16 and CYP19A1) were
significantly associated with poor prognosis in patients with HNSCC
by TCGA database analyses. Annexin family proteins are
calcium-dependent phospholipid-binding proteins that regulate cell
growth and signal transduction (45). Overexpression of ANXA10 has
been reported in oral squamous cell carcinoma, and expression of
ANXA10 promotes cancer cell proliferation through regulating
mitogen-activated protein kinase signaling pathways (46). Exploration of novel
MYO1B-mediated pathways may improve our understanding of the
aggressiveness of this disease.
In conclusion, downregulation of miR-145-3p
was observed in HNSCC clinical specimens, and this passenger strand
acted as an antitumor miRNA through targeting MYO1B in HNSCC
cells. MYO1B was highly expressed in HNSCC clinical
specimens and was found to promote cancer aggressiveness in
functional assays. Elucidation of the pathways mediated by the
miR-145-3p/MYO1B axis is expected to contribute to
further analyses of oncogenesis mechanisms and treatment strategies
in HNSCC.
Acknowledgments
This study was supported by JSPS KAKENHI (grant
nos. 17K16893, 16K20229, 15K10801, 16K11224 and 17K11375).
References
|
1
|
Jemal A, Siegel R, Xu J and Ward E: Cancer
statistics, 2010. CA Cancer J Clin. 60:277–300. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Jou A and Hess J: Epidemiology and
molecular biology of head and neck cancer. Oncol Res Treat.
40:328–332. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Sawicki M, Szudy A, Szczyrek M, Krawczyk P
and Klatka J: Molecularly targeted therapies in head and neck
cancers. Otolaryngol Pol. 66:307–312. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bartel DP: MicroRNAs: Genomics,
biogenesis, mechanism, and function. Cell. 116:281–297. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Filipowicz W, Bhattacharyya SN and
Sonenberg N: Mechanisms of post-transcriptional regulation by
microRNAs: Are the answers in sight? Nat Rev Genet. 9:102–114.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Adams BD, Kasinski AL and Slack FJ:
Aberrant regulation and function of microRNAs in cancer. Curr Biol.
24:R762–R776. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Mah SM, Buske C, Humphries RK and
Kuchenbauer F: miRNA*: A passenger stranded in RNA-induced
silencing complex? Crit Rev Eukaryot Gene Expr. 20:141–148. 2010.
View Article : Google Scholar
|
|
8
|
Koshizuka K, Nohata N, Hanazawa T, Kikkawa
N, Arai T, Okato A, Fukumoto I, Katada K, Okamoto Y and Seki N:
Deep sequencing-based microRNA expression signatures in head and
neck squamous cell carcinoma: Dual strands of pre-miR-150 as
antitumor miRNAs. Oncotarget. 8:30288–30304. 2017.PubMed/NCBI
|
|
9
|
Okato A, Arai T, Kojima S, Koshizuka K,
Osako Y, Idichi T, Kurozumi A, Goto Y, Kato M, Naya Y, et al: Dual
strands of pre-miR-150 (miR-150-5p and miR-150-3p) act as antitumor
miRNAs targeting SPOCK1 in naïve and castration-resistant prostate
cancer. Int J Oncol. 51:245–256. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Mataki H, Seki N, Mizuno K, Nohata N,
Kamikawaji K, Kumamoto T, Koshizuka K, Goto Y and Inoue H:
Dual-strand tumor-suppressor microRNA-145 (miR-145-5p and
miR-145-3p) coordinately targeted MTDH in lung squamous cell
carcinoma. Oncotarget. 7:72084–72098. 2016.PubMed/NCBI
|
|
11
|
Matsushita R, Yoshino H, Enokida H, Goto
Y, Miyamoto K, Yonemori M, Inoguchi S, Nakagawa M and Seki N:
Regulation of UHRF1 by dual-strand tumor-suppressor microRNA-145
(miR-145-5p and miR-145-3p): Inhibition of bladder cancer cell
aggressiveness. Oncotarget. 7:28460–28487. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Matsushita R, Seki N, Chiyomaru T,
Inoguchi S, Ishihara T, Goto Y, Nishikawa R, Mataki H, Tatarano S,
Itesako T, et al: Tumour-suppressive microRNA-144-5p directly
targets CCNE1/2 as potential prognostic markers in bladder cancer.
Br J Cancer. 113:282–289. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yonemori M, Seki N, Yoshino H, Matsushita
R, Miyamoto ZK, Nakagawa M and Enokida H: Dual tumor-suppressors
miR-139-5p and miR-139-3p targeting matrix metalloprotease 11 in
bladder cancer. Cancer Sci. 107:1233–1242. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Karatas OF, Yuceturk B, Suer I, Yilmaz M,
Cansiz H, Solak M, Ittmann M and Ozen M: Role of miR-145 in human
laryngeal squamous cell carcinoma. Head Neck. 38:260–266. 2016.
View Article : Google Scholar
|
|
15
|
Huang SH and O'Sullivan B: Overview of the
8th edition TNM classification for head and neck cancer. Curr Treat
Options Oncol. 18:402017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Fukumoto I, Hanazawa T, Kinoshita T,
Kikkawa N, Koshizuka K, Goto Y, Nishikawa R, Chiyomaru T, Enokida
H, Nakagawa M, et al: MicroRNA expression signature of oral
squamous cell carcinoma: Functional role of microRNA-26a/b in the
modulation of novel cancer pathways. Br J Cancer. 112:891–900.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Koshizuka K, Hanazawa T, Fukumoto I,
Kikkawa N, Matsushita R, Mataki H, Mizuno K, Okamoto Y and Seki N:
Dual-receptor (EGFR and c-MET) inhibition by tumor- suppressive
miR-1 and miR-206 in head and neck squamous cell carcinoma. J Hum
Genet. 62:113–121. 2017. View Article : Google Scholar
|
|
18
|
Nohata N, Sone Y, Hanazawa T, Fuse M,
Kikkawa N, Yoshino H, Chiyomaru T, Kawakami K, Enokida H, Nakagawa
M, et al: miR-1 as a tumor suppressive microRNA targeting TAGLN2 in
head and neck squamous cell carcinoma. Oncotarget. 2:29–42. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Goto Y, Kojima S, Nishikawa R, Enokida H,
Chiyomaru T, Kinoshita T, Nakagawa M, Naya Y, Ichikawa T and Seki
N: The microRNA-23b/27b/24-1 cluster is a disease progression
marker and tumor suppressor in prostate cancer. Oncotarget.
5:7748–7759. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Arai T, Okato A, Kojima S, Idichi T,
Koshizuka K, Kurozumi A, Kato M, Yamazaki K, Ishida Y, Naya Y, et
al: Regulation of spindle and kinetochore-associated protein 1 by
antitumor miR-10a-5p in renal cell carcinoma. Cancer Sci.
108:2088–2101. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kurozumi A, Goto Y, Matsushita R, Fukumoto
I, Kato M, Nishikawa R, Sakamoto S, Enokida H, Nakagawa M, Ichikawa
T, et al: Tumor-suppressive microRNA-223 inhibits cancer cell
migration and invasion by targeting ITGA3/ITGB1 signaling in
prostate cancer. Cancer Sci. 107:84–94. 2016. View Article : Google Scholar
|
|
22
|
Esquela-Kerscher A and Slack FJ: Oncomirs
- microRNAs with a role in cancer. Nat Rev Cancer. 6:259–269. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Nohata N, Hanazawa T, Kikkawa N, Sakurai
D, Fujimura L, Chiyomaru T, Kawakami K, Yoshino H, Enokida H,
Nakagawa M, et al: Tumour suppressive microRNA-874 regulates novel
cancer networks in maxillary sinus squamous cell carcinoma. Br J
Cancer. 105:833–841. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Fukumoto I, Kinoshita T, Hanazawa T,
Kikkawa N, Chiyomaru T, Enokida H, Yamamoto N, Goto Y, Nishikawa R,
Nakagawa M, et al: Identification of tumour suppressive
microRNA-451a in hypopharyngeal squamous cell carcinoma based on
microRNA expression signature. Br J Cancer. 111:386–394. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Goto Y, Kojima S, Nishikawa R, Kurozumi A,
Kato M, Enokida H, Matsushita R, Yamazaki K, Ishida Y, Nakagawa M,
et al: MicroRNA expression signature of castration-resistant
prostate cancer: The microRNA-221/222 cluster functions as a tumour
suppressor and disease progression marker. Br J Cancer.
113:1055–1065. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Goto Y, Kurozumi A, Nohata N, Kojima S,
Matsushita R, Yoshino H, Yamazaki K, Ishida Y, Ichikawa T, Naya Y,
et al: The microRNA signature of patients with sunitinib failure:
Regulation of UHRF1 pathways by microRNA-101 in renal cell
carcinoma. Oncotarget. 7:59070–59086. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Osako Y, Seki N, Koshizuka K, Okato A,
Idichi T, Arai T, Omoto I, Sasaki K, Uchikado Y, Kita Y, et al:
Regulation of SPOCK1 by dual strands of pre-miR-150 inhibit cancer
cell migration and invasion in esophageal squamous cell carcinoma.
J Hum Genet. Jun 29–2017.Epub ahead of print. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Xia W, Chen Q, Wang J, Mao Q, Dong G, Shi
R, Zheng Y, Xu L and Jiang F: DNA methylation mediated silencing of
microRNA-145 is a potential prognostic marker in patients with lung
adenocarcinoma. Sci Rep. 5:169012015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Yang P, Yang Y, An W, Xu J, Zhang G, Jie J
and Zhang Q: The long noncoding RNA-ROR promotes the resistance of
radiotherapy for human colorectal cancer cells by targeting the
p53/miR-145 pathway. J Gastroenterol Hepatol. 32:837–845. 2017.
View Article : Google Scholar
|
|
30
|
Wood NB, Kotelnikov V, Caldarelli DD,
Hutchinson J, Panje WR, Hegde P, Leurgans S, LaFollette S, Taylor
SG IV, Preisler HD, et al: Mutation of p53 in squamous cell cancer
of the head and neck: Relationship to tumor cell proliferation.
Laryngoscope. 107:827–833. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Goto Y, Kurozumi A, Arai T, Nohata N,
Kojima S, Okato A, Kato M, Yamazaki K, Ishida Y, Naya Y, et al:
Impact of novel miR-145-3p regulatory networks on survival in
patients with castration-resistant prostate cancer. Br J Cancer.
117:409–420. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Pashaei E, Guzel E, Ozgurses ME, Demirel
G, Aydin N and Ozen M: A Meta-analysis: Identification of common
mir-145 target genes that have similar behavior in different GEO
datasets. PLoS One. 11:e01614912016. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Sachdeva M and Mo YY: miR-145-mediated
suppression of cell growth, invasion and metastasis. Am J Transl
Res. 2:170–180. 2010.PubMed/NCBI
|
|
34
|
Das AV and Pillai RM: Implications of miR
cluster 143/145 as universal anti-oncomiRs and their dysregulation
during tumorigenesis. Cancer Cell Int. 15:922015. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Ohmura G, Tsujikawa T, Yaguchi T, Kawamura
N, Mikami S, Sugiyama J, Nakamura K, Kobayashi A, Iwata T, Nakano
H, et al: Aberrant myosin 1b expression promotes cell migration and
lymph node metastasis of HNSCC. Mol Cancer Res. 13:721–731. 2015.
View Article : Google Scholar
|
|
36
|
Salas-Cortes L, Ye F, Tenza D, Wilhelm C,
Theos A, Louvard D, Raposo G and Coudrier E: Myosin Ib modulates
the morphology and the protein transport within multi-vesicular
sorting endosomes. J Cell Sci. 118:4823–4832. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Fuse M, Nohata N, Kojima S, Sakamoto S,
Chiyomaru T, Kawakami K, Enokida H, Nakagawa M, Naya Y, Ichikawa T,
et al: Restoration of miR-145 expression suppresses cell
proliferation, migration and invasion in prostate cancer by
targeting FSCN1. Int J Oncol. 38:1093–1101. 2011.PubMed/NCBI
|
|
38
|
Nishikawa R, Goto Y, Sakamoto S, Chiyomaru
T, Enokida H, Kojima S, Kinoshita T, Yamamoto N, Nakagawa M, Naya
Y, et al: Tumor-suppressive microRNA-218 inhibits cancer cell
migration and invasion via targeting of LASP1 in prostate cancer.
Cancer Sci. 105:802–811. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Kinoshita T, Nohata N, Watanabe-Takano H,
Yoshino H, Hidaka H, Fujimura L, Fuse M, Yamasaki T, Enokida H,
Nakagawa M, et al: Actin-related protein 2/3 complex subunit 5
(ARPC5) contributes to cell migration and invasion and is directly
regulated by tumor-suppressive microRNA-133a in head and neck
squamous cell carcinoma. Int J Oncol. 40:1770–1778. 2012.PubMed/NCBI
|
|
40
|
Idichi T, Seki N, Kurahara H, Yonemori K,
Osako Y, Arai T, Okato A, Kita Y, Arigami T, Mataki Y, et al:
Regulation of actin-binding protein ANLN by antitumor miR-217
inhibits cancer cell aggressiveness in pancreatic ductal
adenocarcinoma. Oncotarget. 8:53180–53193. 2017.PubMed/NCBI
|
|
41
|
Yamada A, Mamane A, Lee-Tin-Wah J, Di
Cicco A, Prévost C, Lévy D, Joanny JF, Coudrier E and Bassereau P:
Catch-bond behaviour facilitates membrane tubulation by
non-processive myosin 1b. Nat Commun. 5:36242014. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Komaba S and Coluccio LM: Localization of
myosin 1b to actin protrusions requires phosphoinositide binding. J
Biol Chem. 285:27686–27693. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Almeida CG, Yamada A, Tenza D, Louvard D,
Raposo G and Coudrier E: Myosin 1b promotes the formation of
post-Golgi carriers by regulating actin assembly and membrane
remodelling at the trans-Golgi network. Nat Cell Biol. 13:779–789.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Makowska KA, Hughes RE, White KJ, Wells CM
and Peckham M: Specific myosins control actin organization, cell
morphology, and migration in prostate cancer cells. Cell Rep.
13:2118–2125. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Qi H, Liu S, Guo C, Wang J, Greenaway FT
and Sun MZ: Role of annexin A6 in cancer. Oncol Lett. 10:1947–1952.
2015.PubMed/NCBI
|
|
46
|
Shimizu T, Kasamatsu A, Yamamoto A, Koike
K, Ishige S, Takatori H, Sakamoto Y, Ogawara K, Shiiba M, Tanzawa
H, et al: Annexin A10 in human oral cancer: Biomarker for tumoral
growth via G1/S transition by targeting MAPk signaling pathways.
PLoS One. 7:e455102012. View Article : Google Scholar : PubMed/NCBI
|