Introduction
Oral cancer is a subtype of head and neck cancer
(HNSCC), which is within the top-10 ranking incidences of cancers
worldwide (1). According to the
World Health Organization, the predicted mortality rate from HNSCC
could rise up to 595,000 in 2030 worldwide, which might claim
approximately 324,000 lives in South East Asia alone (2). Oral cancer arises on the lip or oral
cavity, and is traditionally defined as an oral squamous cell
carcinoma (OSCC), because 90% of oral cancers histologically
originate in the squamous cells (3,4).
The standard treatment for advanced or recurrent
OSCC is surgery in combination with chemoradiotherapy (CRT) or
bioradiotherapy (BRT), because these therapies can improve survival
rates of patients where surgical operation only cannot provide the
desired results (5). However,
radiotherapy (RT) or intensified CRT can cause acute mucositis of
the oral cavity, pharynx, and larynx in HNSCC patients, which poses
a significant clinical problem (6,7). After
the initiation of chemotherapy (CT) and/or RT, mucositis appears
within 3–10 days in labial and buccal mucosa, the tongue, the floor
of the mouth and soft palate (8).
This often results in acute oral pain, hampered nutritional intake,
and a compromised quality of life of the patients (9). Moreover, CRT-induced oral mucositis
causes interruptions to RT or concurrent chemotherapy in oral
cancer patients, which can prolong the hospitalization period and
negatively influences patients' outcome (10–13).
Current therapies against CRT-induced mucositis have shown very
limited efficacy in cancer patients (14–17).
Although there is no definitive treatment for preventing or curing
oral mucositis, provision of adequate nutritional support,
neutrophil recovery, and palliative care have shown good results
for the clinical improvement of the patients (18).
Previously, we carried out a retrospective study on
the efficacy of an amino-acid-rich elemental diet (ED),
Elental® (EA Pharma Co., Ltd., Tokyo, Japan), for the
treatment of mucositis in OSCC patients who had received CRT
(5). Elental® is Japan's
first ED that has been popular in Japan, as it is reported to
improve the nutritional status in patients and the elderly
(19). It has a well-blended mixture
of several amino acids, carbohydrates, vitamins, minerals, and
minimal fat and is easily digested and absorbed through the
digestive tract without digestive juice secretion (19–21). It
is a well-known fact that cancer can produce a state of glutamine
deficiency and malnutrition in patients (22,23).
Elental® is a good source of L-glutamine (2,415 mg
L-glutamine/100 g Elental®) and is supposed to help in
reducing the severity of CT- or RT-induced mucositis (19,20,24,25).
This ED has been reported to be effective in the treatment of a
number of diseases; namely, acute Crohn's disease by reducing the
mucosal proinflammatory cytokine production, and stomatitis induced
by chemotherapy in colorectal cancer and esophageal cancer patients
(20,21,26–29).
Moreover, according to one report, Elental® could
counteract sarcopenia progression during CRT therapy in esophageal
cancer patients and preserve lean body mass (30). Therefore, Elental® could
be effective in the treatment of mucositis in OSCC patients while
improving the nutritional status of the patients. Our retrospective
study showed that Elental® is useful against oral
mucositis in OSSC patients receiving CRT and is associated with an
improved completion rate of CRT treatment in those patients
(5).
This time, we carried out a prospective study to
confirm the efficacy of Elental® against oral mucositis
in OSCC patients and compared our new findings with those of our
retrospective study. The purpose of this study was to clarify,
prospectively, whether this amino-acid-rich ED is capable of
minimizing or preventing RT- or CRT-induced mucositis in patients
with OSCC.
Patients and methods
Patients
Fifty patients with OSCC who were scheduled for
60–70 Gy (mean 62.9 Gy) of RT with or without concurrent CRT at the
Yamaguchi University Hospital of Japan from January 2015 to
November 2017 participated in this study and were examined
prospectively. All patients enrolled in this study were over 20
years old and had an Eastern Cooperative Oncologic Group (ECOG)
performance status of 0–2. Moreover, patients who had dental
caries, periodontal disease, not fitting dentures and more
problems, were treated first before the administration of RT or
CRT, and Elental® treatment. In some cases, tooth
extraction was necessary. In that case, RT or CRT treatment and
Elental® treatment was started after 2 weeks of tooth
extraction. All participants gave written informed consent before
entering the study. This prospective study was approved by the
Institutional Review Board (IRB) of the ethics committee of the
Yamaguchi University Hospital (Ref. H26-120). This study was a
randomized open study (no one was blinded).
Administration of chemoradiotherapy
and Elental®
All patients received conventional fractioned
radiation (2 Gy per day for 5 days per week) to the oral cavity
with or without concurrent chemotherapeutic agents. Concurrent
chemotherapies were S-1 (65 mg/m2/day, two-week
administration and one-week rest, twice during RT), UFT (300–400
mg/day during RT), cisplatin (CDDP, 100 mg/m2,
intravenous infusion triweekly, three times during RT), Docetaxel
(DOC) plus CDDP (DOC 5 mg/m2 on day 1 plus CDDP 15
mg/m2 on days 1–5, superselective intra-arterial
infusions, 6–7 times during RT), Cetuximab (400 mg/m2 on
day 1, 250 mg/m2 on days 8, 15, 22, 29, 36, or 43 during
RT), or none (no chemotherapy, RT alone). The total treatment
period of RT or CRT was for ~6–7 weeks.
The patients were assigned randomly to
two groups
We prescribed Elental® (1 bottle/day) for
25 OSCC patients (Elental® group). In brief, one bottle
of Elental® (80 g, 300 kcal) powder was dissolved in 300
ml water (final concentration: 1 kcal/ml), and the patients were
asked to swish it around their mouths and swallow it orally once a
day during the (chemo) radiation period. In addition, patients in
both groups used a similar regimen of oral brushing, gargling with
4% azulene sodium sulfonate plus 4% lidocaine, and using NSAIDs
and/or opioids based on each patient's pain level when they
experienced severe oral pain with mucositis. We recorded data on a
daily basis and compared the data from 25 patients who received
Elental® (Elental® group) with those from 25
patients who did not receive Elental® (the control
group) during this prospective study period. The median follow up
period was 23 (8–37) months. The endpoint of the study was
determined by evaluating the changes of oral mucositis in patients
including the size, redness, and pain level of the affected area
that received RT or CRT, and by evaluating the CRP level in
patients. We finished our prospective study in March 2018.
Randomization, assessment of oral
mucositis, and completion of (chemo) radiation treatments
This study was a randomized open study (no one was
blinded). The degree of oral mucositis was graded according to the
Common Terminology Criteria for Adverse Events (CTCAE) version 4.0
(National Cancer Institute CTCAE v4.0). The CTCAE v4.0 grades for
oral mucositis are defined as follows: Grade 0, no mucositis; grade
1, asymptomatic or mild symptoms, and intervention not indicated;
grade 2, moderate pain not interfering with oral intake, and a
modified diet indicated; grade 3, severe pain interfering with oral
intake; grade 4, life-threatening consequences and urgent
intervention indicated; grade 5, death. Only patients without any
oral disease or mucositis (grade 3 or 4) were included in the
study. Briefly, all patients included in the study did not have
oral mucositis (grade 3 or 4) or any other type of oral disease
when we started RT/CRT and Elental® treatment. Eighteen
patients were grade 0, 6 patients were grade 1, and one patient was
grade 2 in Elental® group. On the other hand, 20
patients were grade 0, 4 patients were grade 1, and 1 patient was
grade 2 in control group. There was no big difference between both
groups.
Resident physicians and radiologists collected and
documented various data of patients including the severity of
mucositis, nutritional status, and efficacy of RT/CRT treatment.
Oral mucositis grade was assessed by independent physicians who
compared their findings with patients' personal assessment of the
mouth and throat soreness, pain level, and the activity score
recorded by the patients on a daily basis.
Moreover, incidence rates of grade 3 or 4 oral
mucositis and completion rates of scheduled (chemo) radiation
treatments by regimen were also evaluated. Treatment completion
included patients who underwent all scheduled chemotherapy and
>60 Gy of radiation without interruption. Furthermore,
nutritional status before and after (chemo) radiation was
investigated in terms of body weight and levels of total protein
and C-reactive protein (CRP) in blood serum. CRP level was checked
once per week during the 6-week treatment period. During the
follow-up period, CRP level was examined once per week in the case
of inpatients or once per month in the case of outpatients.
Statistical methods
We performed univariate and multivariate analyses to
identify the clinicopathological and therapeutic factors involved
in alleviation of oral mucositis during the (chemo) radiation
period. P<0.05 was considered to indicate a statistically
significant difference. Statistical analyses including Chi-square
for independence tests, Mann-Whitney U test, unpaired Student's
t-test, and multivariate logistic regression analysis were
performed using StatView software (version 5.0 J, SAS Institute,
Inc., Cary, NC, USA).
Results
Patient characteristics
Table I summarizes
the clinicopathological characteristics of all patients who
participated in this study. Most of patients in this study had
advanced stage OSCC (Stage III/IV), an ECOG performance status of 0
or 1, and tongue or gingival cancer. There were 35 males and 15
females with an average age of 68.3 years and range 40–92 years
(Table I). Among the 50 patients, 26
developed oral mucositis grade 1 or 2, and 24 patients developed
grade 3 or 4 oral mucositis after receiving RT or CRT (Table II). RT only was received by 11
patients, S-1 + RT by 8 patients, UFT + RT by 5 patients, CDDP + RT
by 20 patients, DOC + CDDP + RT by 2 patients, and Cetuximab + RT
by 4 patients. Half of the patients (25 patients,
Elental® group) received Elental® daily
(daily dose 80 g or 300 kcal/day), and half did not (control
group), while receiving RT or CRT (Table II). In addition, 6 patients in the
control group received a 4% azulene sodium sulfonate plus 4%
lidocaine gargle, 7 patients received NSAIDs only, and 12 patients
received NSAIDs plus opioids when they experienced severe oral pain
with mucositis. In the case of the Elental® group, 6
patients received a 4% azulene sodium sulfonate plus 4% lidocaine
gargle, 8 patients received NSAIDs only, 5 patients received NSAIDs
plus opioids, and 6 patients did not receive any of the above
mentioned treatments. The number of patients who needed NASIDs
and/or opioids was lower in the Elental® group than in
the control group.
 | Table I.Clinicopathological characteristics
of patients. |
Table I.
Clinicopathological characteristics
of patients.
|
Characteristics | No. of patients
(%) |
|---|
| Sex |
|
Male | 35 (70.0) |
|
Female | 15 (30.0) |
| EOCG performance
status |
| 0 | 25 (50.0) |
| 1 | 19 (38.0) |
| 2 | 6 (12.0) |
| Primary tumor
location |
|
Tongue | 24 (48.0) |
|
Gingiva | 22 (44.0) |
| Oral
floor | 2 (4.0) |
|
Lip | 1 (2.0) |
| Buccal
mucosa | 1 (2.0) |
| Stage |
| I | 0 (0) |
| II | 3 (6.0) |
|
III | 14 (28.0) |
| IV | 33 (66.0) |
 | Table II.Association of the grade of oral
mucositis with clinicopathological and therapeutic parameters. |
Table II.
Association of the grade of oral
mucositis with clinicopathological and therapeutic parameters.
|
| Mucositis, oral by
CTCAE v4.0 (n=50) |
|
|---|
|
|
|
|
|---|
| Parameters | Grade 1 or 2 | Grade 3 or 4 | P-value |
|---|
| Sex |
|
| 0.656 |
|
Male | 21 | 14 |
|
|
Female | 5 | 10 |
|
| Age, years |
|
| 0.122 |
|
≤65 | 15 | 5 |
|
|
>65 | 16 | 14 |
|
| EOCG performance
status |
|
| 0.087 |
| 0 | 19 | 6 |
|
| 1 | 10 | 9 |
|
| 2 | 2 | 4 |
|
| Primary tumor
location |
|
| 0.834 |
|
Tongue | 16 | 8 |
|
|
Gingiva | 12 | 10 |
|
| Oral
floor | 1 | 1 |
|
|
Lip | 1 | 0 |
|
| Buccal
mucosa | 1 | 0 |
|
| Stage |
|
| 0.296 |
| II | 2 | 1 |
|
|
III | 11 | 3 |
|
| IV | 18 | 15 |
|
| Total radiation
dose, Gy |
|
| 0.864 |
|
≤60 | 13 | 7 |
|
|
>60 | 17 | 12 |
|
| Combined
chemotherapy |
|
| 0.075 |
|
None | 7 | 4 |
|
|
S-1 | 5 | 3 |
|
|
UFT | 2 | 3 |
|
|
CDDP | 17 | 3 |
|
| DOC +
CDDP | 1 | 1 |
|
|
Cetuximab | 1 | 3 |
|
| Elental®
administration |
|
| 0.0002a |
| Yes
(Elental®) | 22 | 3 |
|
| No
(Control) | 9 | 16 |
|
Assessment of oral mucositis and CRT
completion status after Elental® administration
We identified significant clinicopathological and
therapeutic factors associated with differences in the CTCAE v4.0
oral mucositis severity grade by univariate analysis. Our data
showed that Elental® administration (P=0.0002) was
significantly associated with the degree of oral mucositis
(Table II). The subsequent
multivariate logistic regression analysis included factors with
P-values of <0.05 from the univariate analysis, and the results
suggested that Elental® administration (P=0.0006) was a
significant factor affecting the grade of oral mucositis during RT
or CRT (i.e., patients in the Elental® group mostly
suffered from a lower grade of mucositis (grade 1 or 2) than the
control group (Table III).
 | Table III.Multivariate logistic regression for
grade 1 or 2 oral mucositis by CTCAE version 4.0. |
Table III.
Multivariate logistic regression for
grade 1 or 2 oral mucositis by CTCAE version 4.0.
| Variables | P-value | Odds ratio | 95% CI |
|---|
| Elental®
(vs. control) | 0.0006a | 13.037 | 3.037–55.961 |
| RT + S-1 (vs. RT
alone) | 0.2660 | 0.343 | 0.052–2.261 |
| RT + UFT (vs. RT
alone) | 0.3835 | 0.381 | 0.043–3.338 |
| RT + CDDP (vs. RT
alone) | 0.1848 | 3.238 | 0.570–18.388 |
| RT + CDDP+DOC (vs.
RT alone) | 0.7175 | 0.571 | 0.028–11.852 |
| RT + Cetuximab (vs.
RT alone) | 0.2069 | 0.190 | 0.015–2.502 |
Table IV compares
the characteristics of patients with Elental®
administration in addition to the basic supportive care for oral
mucositis (Elental® group) and patients who had only
supportive care (control group). Table
IV shows that the completion of treatment regimen was
significantly different between the Elental® group and
the control group (P=0.037). Therefore, we compared the two groups
based on CTCAE v4.0 oral mucositis grade and rates of completion of
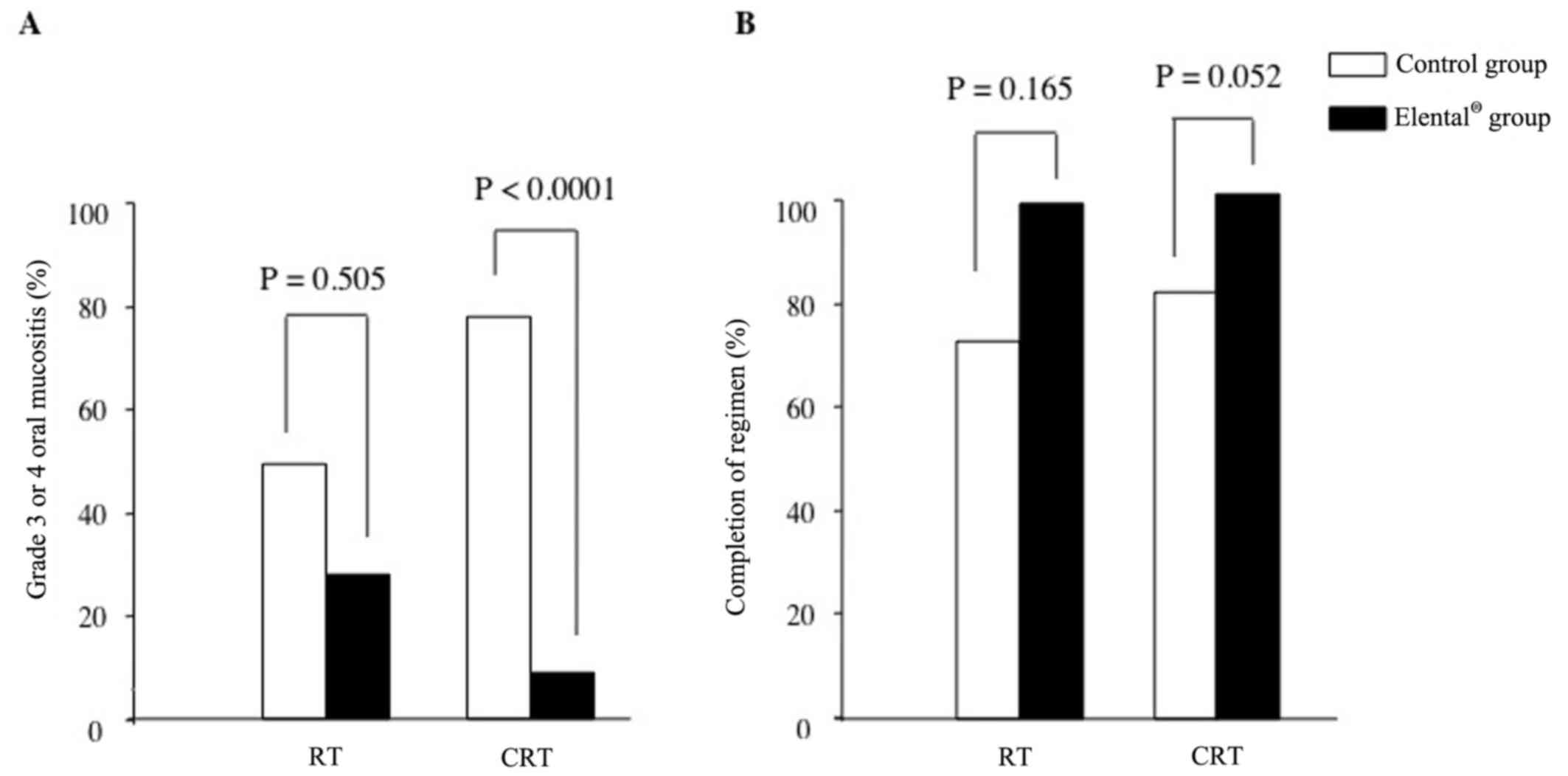
RT or CRT therapy by regimen. For RT alone, 50.0% of patients in
the control group had grade 3 or 4 mucositis vs. 28.6% in the
Elental® group (P=0.505). For CRT, the rates of grade 3
or 4 mucositis were 77.8% vs. 4.76%, respectively, in the control
group and the Elental® group (P<0.0001; Fig. 1A). Elental® showed a
statistically significant difference in reducing the severity of
oral mucositis in CRT cases, though the statistical significance
was not observed in cases of RT alone possibly because of the small
number of cases we investigated by regimen. The rates of completion
by group (control vs. Elental®) were 75.0 and 100%,
respectively, in RT alone (P=0.165), and 83.3 and 100% in CRT
(P=0.052; Fig. 1B). Our data
suggested that, although there was no statistically significant
difference observed between the Elental® and control
groups, Elental® still helped to reduce the
interruptions of treatment regimen in RT and CRT cases (Fig. 1B). Here, treatment interruptions
implicated the cessation of combined chemotherapy. In addition, we
had to discontinue radiation treatment in four patients who
belonged to the control group. The treatment interruptions happened
due to oral mucositis-related severe pain and bleeding. Our data
showed that Elental® was helpful in decreasing the
severity of CRT-induced oral mucositis and in improving the
completion rates of RT or CRT therapy regardless of the
regimen.
 | Table IV.Clinicopathological and therapeutic
characteristics of Elental® treatment group vs. control
group. |
Table IV.
Clinicopathological and therapeutic
characteristics of Elental® treatment group vs. control
group.
|
| No. of patients
(n=50) |
|
|---|
|
|
|
|
|---|
|
Characteristics | Elental®
(n=25) | Control (n=25) | P-value |
|---|
| Sex |
|
| 0.758 |
|
Male | 18 | 17 |
|
|
Female | 7 | 8 |
|
| Mean age, range,
years | 65.8 (40–92) | 70.8 (40–91) |
|
| EOCG performance
status |
|
| 0.215 |
| 0 | 14 | 11 |
|
| 1 | 10 | 9 |
|
| 2 | 1 | 5 |
|
| Primary tumor
location |
|
| 0.526 |
|
Tongue | 13 | 11 |
|
|
Gingiva | 11 | 11 |
|
| Oral
floor | 0 | 2 |
|
|
Lip | 1 | 0 |
|
| Buccal
mucosa | 0 | 1 |
|
| Stage |
|
| 0.723 |
| II | 1 | 2 |
|
|
III | 8 | 6 |
|
| IV | 16 | 17 |
|
| Total radiation
dose, Gy |
|
| 0.564 |
|
≤60 | 11 | 11 |
|
|
>60 | 14 | 14 |
|
| Completion of
regimen |
|
| 0.037a |
|
Completion | 25 | 21 |
|
|
Interruption | 0 | 4 |
|
| Combined
chemotherapy |
|
| 0.0573 |
|
None | 4 | 7 |
|
|
S-1 | 3 | 7 |
|
|
UFT | 2 | 3 |
|
|
CDDP | 14 | 6 |
|
| DOC +
CDDP | 2 | 0 |
|
|
Cetuximab | 0 | 4 |
|
Assessment of nutritional and
inflammatory status after Elental® administration
We evaluated the nutritional and inflammatory status
of patients in both groups before and after CRT and compared the
changes in pre- and post-CRT nutritional or inflammatory status for
each group. We retrospectively evaluated body weight, serum total
protein, and CRP in blood, which is a marker for inflammation.
Table V shows that there was a
significant difference in CRP values between the
Elental® group and the control group: A lower CRP level
in serum was maintained in the Elental® group than in
the control group. CRP level showed the most significant difference
in the 4th to 6th week of the treatment period. However, we could
not detect any significant difference in body weight or serum total
protein (Table V). Also, the number
of cases that required nutrition management by tube feeding was two
in the Elental® group and 10 in the control group. Two
cases in the control group underwent parenteral nutrition, whereas
no cases received parenteral nutrition in the Elental®
group.
 | Table V.Body weight and serum level of total
protein and CRP before and after chemoradiation and their changes
during the study. |
Table V.
Body weight and serum level of total
protein and CRP before and after chemoradiation and their changes
during the study.
|
| Before
treatment |
| After
treatment |
| Change |
|
|---|
|
|
|
|
|
|
|
|
|---|
| Variable | Mean ± SD | P-value | Mean ± SD | P-value | Mean ± SD | P-value |
|---|
| Body weight,
kg |
| 0.839 |
| 0.801 |
| 0.614 |
|
Elental® | 49.8±12.2 |
| 48.8±12.3 |
| −2.94±3.37 |
|
| Control | 50.5±10.8 |
| 46.9±9.82 |
| −2.80±2.58 |
|
| TP, g/dl |
| 0.460 |
| 0.059 |
| 0.146 |
|
Elental® | 6.48±0.43 |
| 5.76±0.49 |
| −0.72±0.42 |
|
| Control | 6.56±0.49 |
| 5.96±0.54 |
| −0.57±0.51 |
|
| CRP, g/dl |
| 0.712 |
| 0.004 |
| 0.001a |
|
Elental® | 0.47±0.87 |
| 1.44±1.37 |
| 0.98±0.98 |
|
| Control | 0.56±0.98 |
| 4.54±4.66 |
| 3.97±3.91 |
|
Discussion
In this prospective study, we demonstrated that the
ED Elental® was effective for the treatment of RT or
CRT-induced oral mucositis in OSCC patients. Here, BRT cases
(Cetuximab + RT) were included in the CRT group because we had only
4 patients receiving BRT. Our data also showed that
Elental® administration improved the completion rates of
RT or CRT in OSCC patients. However, CRT completion status may not
be a good indicator of Elental® efficiency because many
parameters can affect CRT completion. There are some real
limitations to gather enough participants by single-center study.
Further multicenter study must be needed to clarify a good
indicator of Elental® efficiency. We are preparing for
prospective multicenter clinical trials to confirm the benefits
indicated from this prospective single-center study. Basic
investigations are also necessary to clarify the mechanism of
action of elemental diet for (chemo) radiation-induced mucositis in
OSCC.
Many HNSCC patients suffer from mucositis,
swallowing disorders, and distortion of taste and smell soon after
receiving CRT, followed by dysphagia, xerostomia, acute pain,
trismus, osteoradionecrosis, and several dental diseases (13). Severe oral mucositis can cause
unplanned breaks and delays in RT and CRT treatment, which can lead
to a poor outcome for patients (31,32).
These CRT side effects can also negatively affect the patient's
ability to eat and drink, which can result in malnutrition,
dehydration, and weight loss (33,34).
Therefore, patients might require additional nutritional
supplements. There are many nutritional supplements with
L-glutamine that are prescribed for cancer patients, because
L-glutamine can encourage protein synthesis and enterocyte
proliferation and is reported to be useful in reducing inflammation
(35,36). Moreover, in vivo animal
studies have demonstrated the safety of glutamine supplements and
its ameliorating effect against cytotoxicity-induced mucositis
(24,25,37).
Elental®, with a high L-glutamine
content, has been used in Japan for four decades, and its safety
has been well established (5,19,20).
It costs <US$4.00 per day and has been approved and covered by
public insurance as a prescription medication for the treatment of
malnutrition in Japan (19). It has
almost the same formula as VIVONEX® T.E.N. (Nestlé,
Vevey, Switzerland) prescribed in many Western countries (38). Elental® has been reported
to be effective against various gastrointestinal disorders, such as
inflammatory bowel disease or Crohn's disease (26,27,39,40).
Several authors have reported the benefits of Elental®
against oral mucositis during CT and/or RT in patients with
esophageal, colorectal, and oral cancer while preserving the lean
body mass of patients (5,28–30).
Compared to other treatment options available for mucositis in
cancer patients, Elental® could be an attractive agent
because it is neither costly nor a growth factor such as
palifermin, and no side effects of Elental® have been
reported thus far (5,14,29,39,41).
However, only a few published reports are available on the efficacy
of Elental® for the treatment of RT and/or CT-induced
oral mucositis (5,37).
The preventive and healing effects of
Elental® against oral mucositis have been assessed in
several clinical trials, but very few were carried out
prospectively or with randomization (23,28,42).
Previously, we carried out a retrospective study that showed the
effectiveness of Elental® against oral mucositis in OSSC
patients receiving CRT (5). Our
present study was conducted prospectively on OSCC patients with
CRT-induced mucositis. We observed similar data as our previous
retrospective study, which confirms the ameliorating effect of
Elental® against oral mucositis. In this study, we
observed a significant association (P=0.0002) between
Elental® administration and the degree of oral mucositis
by univariate analysis. Our multivariate logistic regression
analysis also showed that patients in the Elental® group
mostly suffered from a lower grade of mucositis (grade 1 or 2) than
the control group (P=0.0002). Moreover, OSCC patients who received
Elental® showed an improved completion rate of RT or CRT
compared to the control group. We did not detect any adverse
effects in relation to the clinical use of Elental® in
this study (data not shown).
We assessed the CRP level in the blood of patients
because a high level of CRP is considered as a marker of
inflammation. Our data showed that administration of
Elental® was associated with suppressed expression of
CRP. However, because CRP is increased by various factors, its
relation to mucositis grade is still unclear. According to our
in vitro data, Elental® treatment could
successfully downregulate the expression of inflammatory cytokines
in the immortalized human keratinocyte cell line HaCaT (43). Therefore, we assumed that
Elental® might suppress CRP expression via the
downregulation of inflammatory cytokines. We also showed previously
that Elental® can treat mucositis and dermatitis by
accelerating mucosal and skin recovery through FGF2 induction and
reepithelization in vivo (37). CRT- or RT-induced mucositis occurs
through a sequence of stepwise events, namely, direct DNA damage
leading to cell damage, followed by the activation of several
transcription factors including nuclear factor-κB (NF-κB), Wnt, and
p53, and their molecular pathways (43). Therefore, it is important to
understand whether these factors and molecular pathways are
affected by Elental® treatment or not. There could be
other factors that contribute to the development and degree of oral
mucosistis, and consumption of Elental® alone might not
be able to reduce the severity of oral mucositis induced by more RT
or CRT regimens that are more intensive than those administered in
our current study. In order to clarify the mechanism underlying the
efficacy of Elental® against oral mucositis in OSCC,
further investigations are necessary.
Our study did not show some of the desired results
expected by nutritional supplementation. According to several
reports, Elental® nutrition therapy has supportive
effects in patients with Crohn's disease and esophageal cancer,
including nutritional status improvement and average body mass
index preservation (26,27,30).
However, we did not find any association between
Elental® administration and the improvement of
nutritional status (total protein) or body weight of OSCC patients
in this study. Therefore, the effect of Elental® on the
maintenance of nutritional status of patients should be clarified
by future, randomized clinical trials. Moreover, only 2 patients in
the Elental® group required tube feeding as a nutrition
therapy, whereas it was 10 patients in the control group. In
addition, the control group received only central venous
alimentation. Additional clinical data are needed to clarify the
reason behind the marked decreases of oral intake that were seen in
the control group patients.
Our present prospective clinical trial demonstrated
that Elental® is beneficial in the treatment of oral
mucositis for OSCC patients receiving CRT or BRT and can also
improve the chemo (radiation) treatment completion rate. Thus, it
confirmed the findings of our previous retrospective study on the
usefulness of Elental® against oral mucositis in OSSC
patients. These findings warrant the initiation of future
prospective studies with a larger group of patients to confirm
further the efficacy of Elental® for RT- or CRT-induced
oral mucositis.
Acknowledgements
Not applicable.
Funding
This study was supported in part by a Grant-in-Aid
from the Japanese Ministry of Education, Science, and Culture
(grant no. 15K11292). This study was also supported by EA Pharma
Co., Ltd., Tokyo, Japan.
Availability of data and materials
The datasets used during the present study are
available from the corresponding author upon reasonable
request.
Authors' contributions
KH assisted in the study design, performed the study
and data analysis, and wrote the manuscript. HaM carried out the
prospective study and collected data from patients. TF carried out
data evaluation and assisted in manuscript writing. HaM, YK, HU,
DH, KU, SP, SC, HH, ST, MO, HiM, JM, HK, SA, and KS helped to carry
out the prospective study and helped in data collection. KM helped
with data collection and analysis, and edited and revised the
manuscript. All the authors read and approved the final version of
the manuscript.
Ethics approval and consent to
participate
This prospective study was approved by the
Institutional Review Board (IRB) of the Ethics Committee of the
Yamaguchi University Hospital (Ref. H26-120). All procedures
performed in the studies involving human participants were in
accordance with the standards of the ethics committee of the
Yamaguchi University Hospital. Written informed consent was
obtained from all individual who participated in this study.
Clinical trial registration no. H26-120, name of registry: Efficacy
of enteral nutrition on chemoradiotherapy against oral cancer, date
of registration: January 23, 2015.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviation
Abbreviations:
|
OSCC
|
oral squamous cell carcinoma
|
References
|
1
|
Rivera C: Essentials of oral cancer. Int J
Clin Exp Pathol. 8:11884–11894. 2015.PubMed/NCBI
|
|
2
|
Mehanna H, Paleri V, West CM and Nutting
C: Head and neck cancer-part 1: Epidemiology, presentation and
preservation. Clin Otolaryngol. 36:65–68. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Dhanuthai K, Rojanawatsirivej S, Thosaporn
W, Kintarak S, Subarnbhesaj A, Darling M, Kryshtalskyj E, Chiang
CP, Shin HI, Choi SY, et al: Oral cancer: A multicenter study. Med
Oral Patol Oral Cir Bucal. 23:e23–e29. 2018.PubMed/NCBI
|
|
4
|
Lingen MW, Kalmar JR, Karrison T and
Speight PM: Critical evaluation of diagnostic aids for the
detection of oral cancer. Oral Oncol. 44:10–22. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Harada K, Ferdous T, Horinaga D, Uchida K,
Mano T, Mishima K, Park S, Hanazawa H, Takahashi S, Okita A, et al:
Efficacy of elemental diet on prevention for
chemoradiotherapy-induced oral mucositis in patients with oral
squamous cell carcinoma. Support Care Cancer. 24:953–959. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Sonis ST: Oral mucositis. Anticancer
Drugs. 22:607–612. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Moslemi D, Nokhandani AM, Otaghsaraei MT,
Moghadamnia Y, Kazemi S and Moghadamnia AA: Management of
chemo/radiation-induced oral mucositis in patients with head and
neck cancer: A review of the current literature. Radiother Oncol.
120:13–20. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Gutiérrez-Vargas R, Díaz-García ML,
Villasís-Keever MÁ, Portilla-Robertson J and Zapata-Tárres M:
Instruments to measure the quality of life in patients with oral
mucositis undergoing oncological treatment: A systematic review of
the literature. Bol Med Hosp Infant Mex. 73:457–466.
2016.PubMed/NCBI
|
|
9
|
Lalla RV, Sonis ST and Peterson DE:
Management of oral mucositis in patients who have cancer. Dent Clin
North Am. 52:61–77. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Bese NS, Hendry J and Jeremic B: Effects
of prolongation of overall treatment time due to unplanned
interruptions during radiotherapy of different tumor sites and
practical methods for compensation. Int J Radiat Oncol Biol Phys.
68:654–661. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Russo G, Haddad R, Posner M and Machtay M:
Radiation treatment breaks and ulcerative mucositis in head and
neck cancer. Oncologist. 13:886–898. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Sonis ST, Oster G, Fuchs H, Bellm L,
Bradford WZ, Edelsberg J, Hayden V, Eilers J, Epstein JB, LeVeque
FG, et al: Oral mucositis and the clinical and economic outcomes of
hematopoietic stem-cell transplantation. J Clin Oncol.
19:2201–2205. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Bressan V, Stevanin S, Bianchi M, Aleo G,
Bagnasco A and Sasso L: The effects of swallowing disorders,
dysgeusia, oral mucositis and xerostomia on nutritional status,
oral intake and weight loss in head and neck cancer patients: A
systematic review. Cancer Treat Rev. 45:105–119. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Henke M, Alfonsi M, Foa P, Giralt J,
Bardet E, Cerezo L, Salzwimmer M, Lizambri R, Emmerson L, Chen MG
and Berger D: Palifermin decreases severe oral mucositis of
patients undergoing postoperative radiochemotherapy for head and
neck cancer: A randomized, placebo-controlled trial. J Clin Oncol.
29:2815–2820. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Svanberg A, Ohrn K and Birgegard G: Oral
cryotherapy reduces mucositis and improves nutrition-a randomised
controlled trial. J Clin Nurs. 19:2146–2151. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Scully C, Epstein J and Sonis S: Oral
mucositis: A challenging complication of radiotherapy, chemotherapy
and radiochemotherapy. Part 2: Diagnosis and management of
mucositis. Head Neck. 26:77–84. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Cowen D, Tardieu C, Schubert M, Peterson
D, Resbeut M, Faucher C and Franquin JC: Low energy helium-neon
laser in the prevention of oral mucositis in patients undergoing
bone marrow transplant: Results of a double blind randomized trial.
Int J Radiat Oncol Biol Phys. 38:697–703. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Cheng KK, Molassiotis A, Chang AM, Wai WC
and Cheung SS: Evaluation of an oral care protocol intervention in
the prevention of chemotherapy-induced oral mucositis in paediatric
cancer patients. Eur J Cancer. 37:2056–2063. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Online EA Pharma Co. Ltd Products
Information, Elental®, . http://www.eapharma.co.jp/medicalexpert/product/elental/elental_information.htmlWebpage
in Japanese. August 19–2017
|
|
20
|
Ikeura T, Takaoka M, Uchida K, Miyoshi H
and Okazaki K: Beneficial effect of low-fat elemental diet therapy
on pain in chronic pancreatitis. Int J Chronic Dis.
2014:8620912014.PubMed/NCBI
|
|
21
|
Nakayama G, Morioka D, Murakami T,
Takakura H, Miura Y and Togo S: Chylous ascites occurring after low
anterior resection of the rectum successfully treated with an oral
fat-free elemental diet (Elental®). Clin J
Gastroenterol. 5:216–219. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Gaurav K, Goel RK, Shukla M and Pandey M:
Glutamine: A novel approach to chemotherapy-induced toxicity.
Indian J Med Paediatr Oncol. 33:13–20. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Okada T, Nakajima Y, Nishikage T,
Ryotokuji T, Miyawaki Y, Hoshino A, Tokairin Y, Kawada K, Nagai K
and Kawano T: A prospective study of nutritional supplementation
for preventing oral mucositis in cancer patients receiving
chemotherapy. Asia Pac J Clin Nutr. 26:42–48. 2017.PubMed/NCBI
|
|
24
|
Choi K, Lee SS, Oh SJ, Lim SY, Lim SY,
Jeon WK, Oh TY and Kim JW: The effect of oral glutamine on
5-fluorouracil/leucovorin-induced mucositis/stomatitis assessed by
intestinal permeability test. Clin Nutr. 26:57–62. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Savarese DM, Savy G, Vahdat L, Wischmeyer
PE and Corey B: Prevention of chemotherapy and radiation toxicity
with glutamine. Cancer Treat Rev. 29:501–513. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yamamoto T, Nakahigashi M, Umegae S,
Kitagawa T and Matsumoto K: Acute duodenal Crohn's disease
successfully managed with low-speed elemental diet infusion via
nasogastric tube: A case report. World J Gastroenterol. 12:649–651.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Yamamoto T, Nakahigashi M, Umegae S,
Kitagawa T and Matsumoto K: Impact of elemental diet on mucosal
inflammation in patients with active Crohn's disease: Cytokine
production and endoscopic and histological findings. Inflamm Bowel
Dis. 11:580–588. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ogata Y, Takeuchi M, Ishibashi N, Kibe S,
Takahashi K, Uchida S, Murakami N, Yahara T and Shirouzu K:
Efficacy of Elental on prevention for chemotherapy-induced oral
mucositis in colorectal cancer patients. Gan To Kagaku Ryoho.
39:583–587. 2012.(In Japanese). PubMed/NCBI
|
|
29
|
Fukui T, Itoh Y, Orihara M, Yoshizawa K,
Takeda H, Kawada S and Yoshioka T: Elental prevented and reduced
oral mucositis during chemotherapy in patients esophageal cancer.
Gan To Kagaku Ryoho. 38:2597–2601. 2011.(In Japanese). PubMed/NCBI
|
|
30
|
Ishikawa T, Yasuda T, Doi T, Okayama T,
Sakamoto N, Gen Y, Dohi O, Yoshida N, Kamada K, Uchiyama K, et al:
The amino acid-rich elemental diet Elental® preserves
lean body mass during chemo- or chemoradiotherapy for esophageal
cancer. Oncol Rep. 36:1093–1100. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Bieri S, Bentzen SM, Huguenin P, Allal AS,
Cozzi L, Landmann C, Monney M and Bernier J: Early morbidity after
radiotherapy with or without chemotherapy in advanced head and neck
cancer. Experience from four nonrandomized studies. Strahlenther
Onkol. 179:390–395. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Cooper JS, Pajak TF, Forastiere AA, Jacobs
J, Campbell BH, Saxman SB, Kish JA, Kim HE, Cmelak AJ, Rotman M, et
al: Postoperative concurrent radiotherapy and chemotherapy for
high-risk squamous-cell carcinoma of the head and neck. N Engl J
Med. 350:1937–1944. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Van den Berg MG, Rasmussen-Conrad EL,
Gwasara GM, Krabbe PF, Naber AH and Merkx MA: A prospective study
on weight loss and energy intake in patients with head and neck
cancer, during diagnosis, treatment and revalidation. Clin Nutr.
25:765–772. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Ganzer H, Rothpletz-Puglia P, Byham-Gray
L, Murphy BA and Touger-Decker R: The eating experience in
long-term survivors of head and neck cancer: A mixed-methods study.
Support Care Cancer. 23:3257–3268. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Sornsuvit C, Komindr S, Chuncharunee S,
Wanikiat P, Archararit N and Santanirand P: Pilot study: Effects of
parenteral glutamine dipeptide supplementation on neutrophil
functions and prevention of chemotherapy-induced side-effects in
acute myeloid leukaemia patients. J Int Med Res. 36:1383–1391.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Maeda A, Ando H, Ura T, Komori A, Hasegawa
A, Taniguchi H, Kadowaki S, Muro K, Tajika M, Kobara M, et al:
Association between ABCG2 and SLCO1B1 polymorphisms and adverse
drug reactions to regorafenib: A preliminary study. Int J Clin
Pharmacol Ther. 55:409–415. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Harada K, Ferdous T, Kobayashi H and
Ueyama Y: Elemental diet accelerates the recovery from oral
mucositis and dermatitis induced by 5-Fluorouracil through the
induction of fibroblast growth factor 2. Integr Cancer Ther.
17:423–430. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Online Nestle Products Information.
VIVONEX® T.E.N. https://www.nestlehealthscience.us/brands/vivonex/vivonex-t-e-nMarch
1–2018
|
|
39
|
Yamamoto T, Shiraki M, Nakahigashi M,
Umegae S and Matsumoto K: Enteral nutrition to suppress
postoperative Crohn's disease recurrence: A five-year prospective
cohort study. Int J Colorectal Dis. 28:335–340. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Takagi S, Utsunomiya K, Kuriyama S,
Yokoyama H, Takahashi S, Iwabuchi M, Takahashi H, Takahashi S,
Kinouchi Y, Hiwatashi N, et al: Effectiveness of an ‘half elemental
diet’ as maintenance therapy for Crohn's disease: A
randomized-controlled trial. Aliment Pharmacol Ther. 24:1333–1340.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Bossi P, Locati LD and Licitra L:
Palifermin in prevention of head and neck cancer radiation-induced
mucositis: Not yet a definitive word on safety and efficacy
profile. J Clin Oncol. 30:565–567. 2012. View Article : Google Scholar
|
|
42
|
Ogata Y, Ishibashi N, Yamaguchi K, Uchida
S, Kamei H, Nakayama G, Hirakawa H, Tanigawa M and Akagi Y:
Preventive effects of amino-acid-rich elemental diet
Elental® on chemotherapy-induced oral mucositis in
patients with colorectal cancer: A prospective pilot study. Support
Care Cancer. 24:783–789. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Harada K, Ferdous T, Mizukami Y and
Mishima K: Elemental diet inhibits pro-inflammatory cytokine
production in keratinocytes through the suppression of NF-κB
activation. Oncol Rep. 40:361–368. 2018.PubMed/NCBI
|