Spandidos Publications style
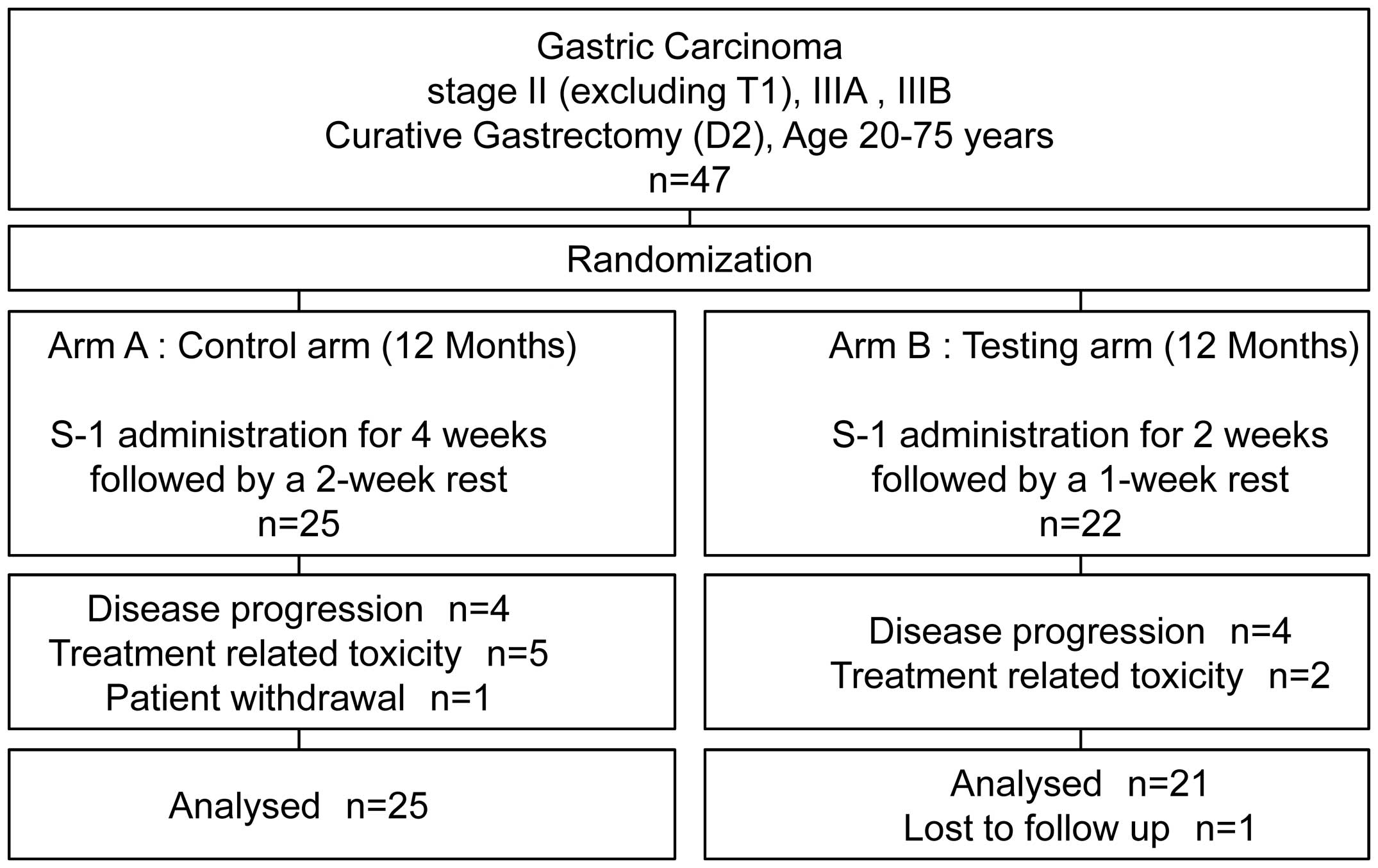
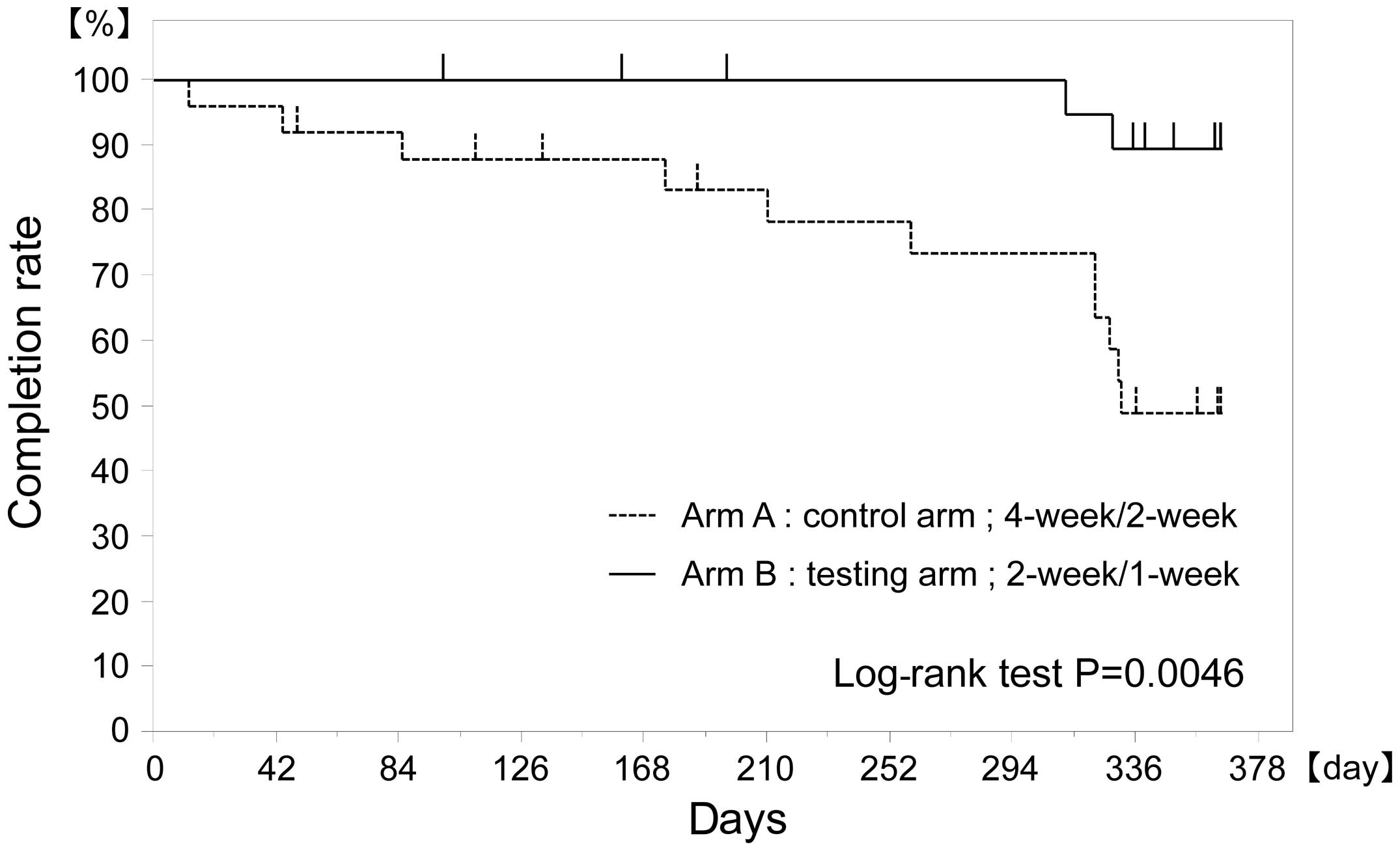
Yamatsuji T, Fujiwara Y, Matsumoto H, Hato S, Namikawa T, Hanazaki K, Takaoka M, Hayashi J, Shigemitsu K, Yoshida K, Yoshida K, et al: Feasibility of oral administration of S-1 as adjuvant chemotherapy in gastric cancer: 4-week S‑1 administration followed by 2-week rest vs. 2-week administration followed by 1-week rest. Mol Clin Oncol 3: 527-532, 2015.
APA
Yamatsuji, T., Fujiwara, Y., Matsumoto, H., Hato, S., Namikawa, T., Hanazaki, K. ... Naomoto, Y. (2015). Feasibility of oral administration of S-1 as adjuvant chemotherapy in gastric cancer: 4-week S‑1 administration followed by 2-week rest vs. 2-week administration followed by 1-week rest. Molecular and Clinical Oncology, 3, 527-532. https://doi.org/10.3892/mco.2015.500
MLA
Yamatsuji, T., Fujiwara, Y., Matsumoto, H., Hato, S., Namikawa, T., Hanazaki, K., Takaoka, M., Hayashi, J., Shigemitsu, K., Yoshida, K., Urakami, A., Uno, F., Nishizaki, M., Kagawa, S., Ninomiya, M., Fujiwara, T., Hirai, T., Nakamura, M., Haisa, M., Naomoto, Y."Feasibility of oral administration of S-1 as adjuvant chemotherapy in gastric cancer: 4-week S‑1 administration followed by 2-week rest vs. 2-week administration followed by 1-week rest". Molecular and Clinical Oncology 3.3 (2015): 527-532.
Chicago
Yamatsuji, T., Fujiwara, Y., Matsumoto, H., Hato, S., Namikawa, T., Hanazaki, K., Takaoka, M., Hayashi, J., Shigemitsu, K., Yoshida, K., Urakami, A., Uno, F., Nishizaki, M., Kagawa, S., Ninomiya, M., Fujiwara, T., Hirai, T., Nakamura, M., Haisa, M., Naomoto, Y."Feasibility of oral administration of S-1 as adjuvant chemotherapy in gastric cancer: 4-week S‑1 administration followed by 2-week rest vs. 2-week administration followed by 1-week rest". Molecular and Clinical Oncology 3, no. 3 (2015): 527-532. https://doi.org/10.3892/mco.2015.500