Introduction
Adenocarcinoma of the pancreas is an aggressive
malignancy with poor prognosis. Pancreatic neuroendocrine tumours
(PNET), comprising ~3% of primary pancreatic neoplasms (1), are more heterogeneous in their
histological character and outcome. Although partner and localizer
of breast cancer 2, early onset (PALB2) mutations have been
associated with pancreatic adenocarcinomas (2), pancreatic adenocarcinoma coexisting with
PNET is an uncommon finding. This is the presentation of a case
report of synchronous diagnosis of these two malignancies in the
setting of a PALB2 mutation.
Case report
A 73-year-old woman was admitted to the Royal North
Shore Hospital in late 2011 in late 2011 for medical review of
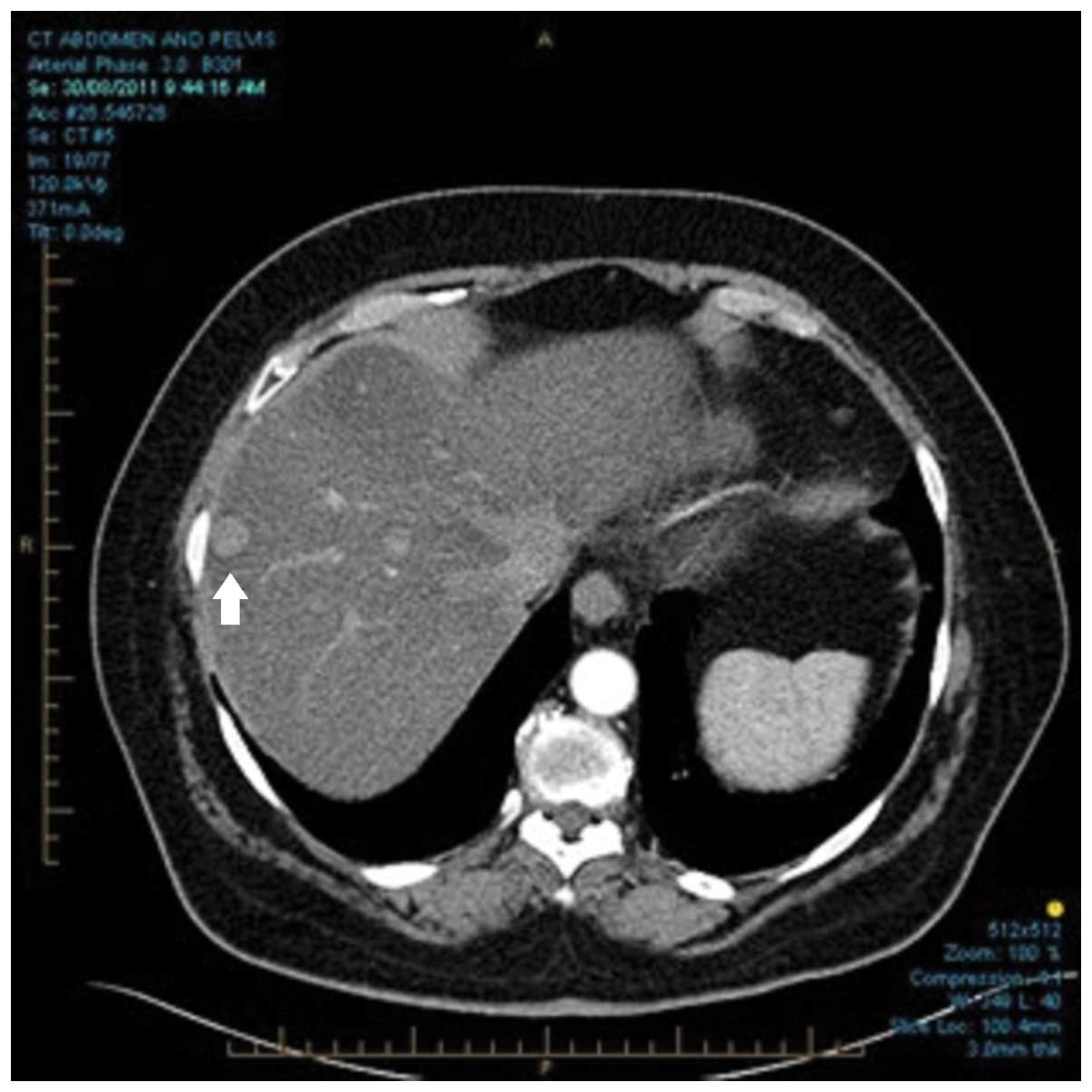
abdominal discomfort. Computed tomography (CT) of the abdomen and
pelvis revealed masses in the pancreatic head and terminal ileum,
as well as two enhancing lesions in the liver (Fig. 1). The patient underwent the Whipple's
procedure, ileal resection and biopsy of the liver lesions. The
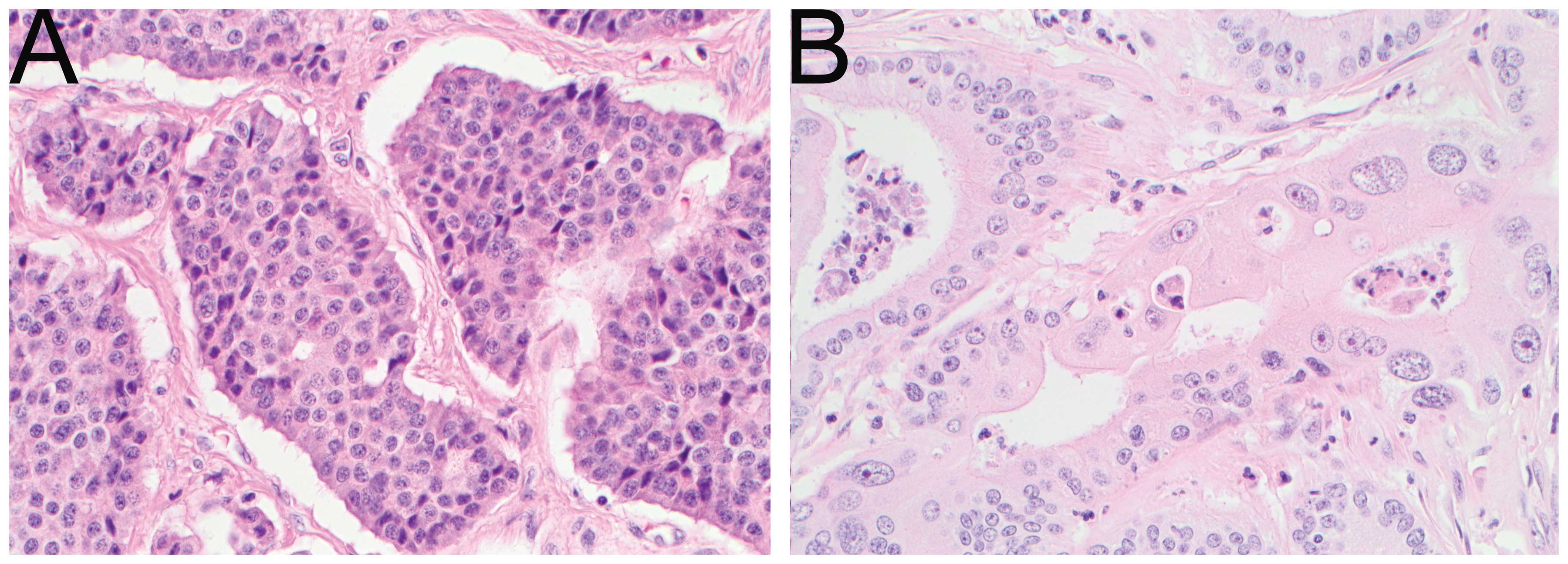
histological examination demonstrated a 40-mm moderately
differentiated pancreatic head adenocarcinoma (Fig. 2) arising from an intraductal papillary
mucinous neoplasm, with clear margins and no nodal involvement. A
low-grade neuroendocrine tumour [Ki-67<0.5%, 1 mitosis/10
high-power fields, p4(T3N1M1); Fig.
2] was also present in the terminal ileum and aforementioned
liver lesions. The patient's postoperative course was complicated
by diabetes mellitus and steatorrhoea, which were well controlled
with use of insulin and pancreatic enzyme supplements,
respectively.
The past history of the patient notably included
early left-sided breast cancer in 2007, treated by wide local
excision, adjuvant radiotherapy and adjuvant tamoxifen followed by
letrozole. The patient had also previously undergone laminectomy
for back pain, right total knee replacement, hysterectomy and
cholecystectomy. The patient had never smoked and did not drink
alcohol, but her family history was notable, as her mother and
sister had succumbed to pancreatic cancer and her father had
succumbed to hepatocellular carcinoma.
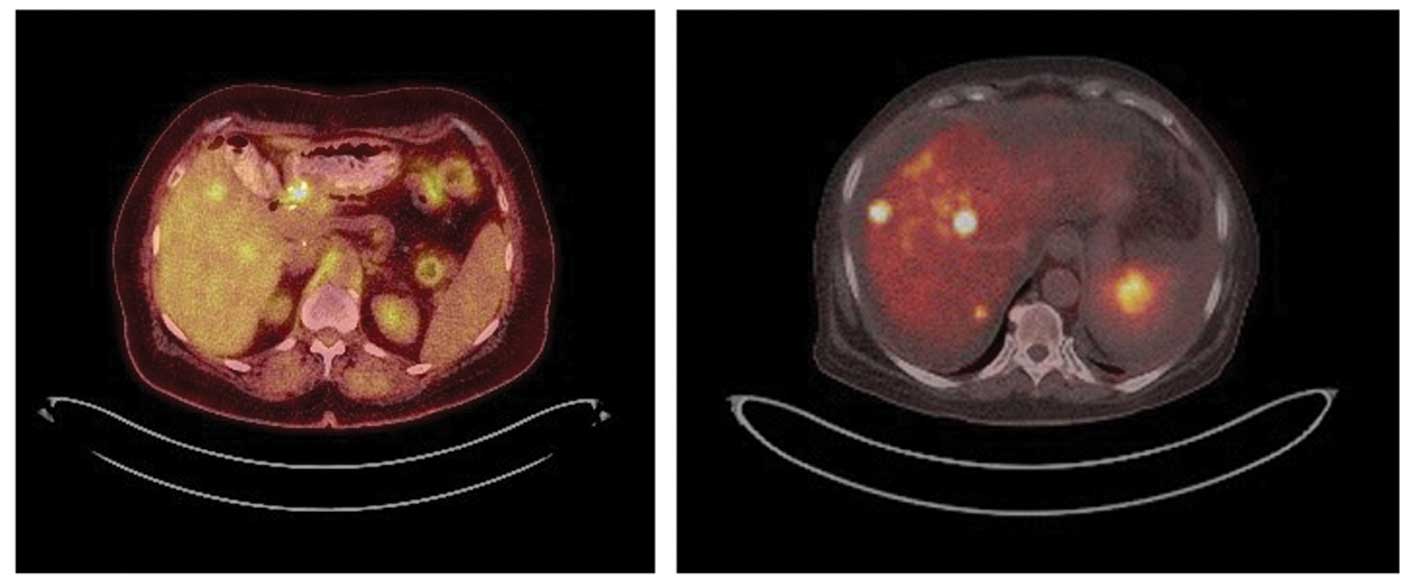
A 68Ga DOTATATE positron emission
tomography (PET) scan revealed uptake by multiple liver lesions and
para-aortic nodes (Fig. 3A). The
patient was commenced on long-acting octreotide at a dose of 30 mg
monthly and remained well over the following year; however, a
progress CT performed 15 months postoperatively revealed a lesion
at the surgical margin. PET scanning demonstrated
18F-fluorodeoxyglucose (FDG) but not 68Ga
avidity for this lesion, as well as two new hepatic lesions
(Fig. 3B). The serum CA 19.9 level
was 4,045 U/ml. Taken together, these findings suggested the
diagnosis of metastatic pancreatic cancer.
The patient was commenced on cisplatin + gemcitabine
chemotherapy in December, 2012 and tolerated treatment well, apart
from dose reductions due to myelosuppression. The treatment was
ceased after 5 cycles, as the patient was asymptomatic and the CA
19.9 levels had normalized.
New lesions were evident in the liver 2 months after
cessation of treatment, with 18F-FDG avidity confirmed
by PET and histological confirmation of metastatic pancreatic
adenocarcinoma via fine-needle biopsy. The patient was commenced on
gemcitabine and capecitabine chemotherapy and subsequently received
selective internal radiation therapy [90Y microspheres -
SIR-Spheres® (SIRTeX, Sydney, Australia)] to the liver.
The patient had consented to having her pancreatic
tumour tissue analysed as part of the Australian Pancreatic Cancer
Genome Initiative (APGI), which is a prospective study
investigating broad-based sequencing of tumour and normal genomes
of patients with pancreatic adenocarcinoma (3). A mutation in the PALB2 gene (PALB2 g.23,
649, 207delACAA) in the tumour was detected at that time. Further
mitomycin C chemotherapy was planned; however, the patient was
admitted to the hospital with Escherichia coli sepsis,
delirium and gradual deterioration over the course of several weeks
and succumbed to the disease in June, 2014.
The patient and her husband provided informed
consent regarding the publication of the medical information.
Discussion
Pancreatic adenocarcinoma is the most common type of
exocrine pancreatic malignancy and is associated with poor outcome,
even following surgical resection. The 5-year survival rate from
metastatic pancreatic adenocarcinoma is currently <1% and the
median survival time is ~6–9 months, even with the recent advances
in chemotherapeutic regimens (4).
This has prompted the search for an improved understanding of
molecular pathogenesis in an attempt to identify targetable
mutations underlying the development of pancreatic cancer.
Multiple germline mutations have been implicated in
the pathogenesis of pancreatic cancer, such as adenomatous
polyposis coli, breast cancer 1, early onset (BRCA1), BRCA2, Lynch
syndrome mutations and PALB2. PALB2 mutations have been associated
with malignancies of the breast (5)
and pancreas, as well as non-malignant conditions, such as Fanconi
anaemia (6). In a series of 1,144
familial breast cancer patients (7),
3.4% of the patients harboured such a mutation. Although it is a
recognized pathogenic mutation in familial pancreatic cancer, PALB2
mutation frequency in a series of 96 familial pancreatic cancer
patients was only 3.1% (2). Mutations
in the domain of PALB2 which binds BRCA2 have been shown to affect
the double-stranded break repair mechanism (8), providing a rationale for
chemotherapeutic agents targeting this mechanism. Xenograft models
and case reports have suggested the efficacy of mitomycin C or
cisplatin in this setting (9).
The present case demonstrated the good clinical
response of this genotype to platinum combinations. Upon reviewing
other cases analysed by APGI, there are several examples of
exceptional responders to platinums in the DNA damage repair
pathway genotypes (10). Given the
patient's family history of pancreatic cancer and personal history
of breast cancer, the PALB2 mutation is almost certainly pathogenic
and genetic counselling for her family has been recommended.
This case highlights the need for ongoing research
into the molecular genomics of pancreatic cancer and continuous
refinement of the genetic testing system to optimize treatment
choices early on in diagnosis. The predictive nature of PALB2 for
response to DNA-damaging chemotherapeutic agents requires further
investigation.
References
|
1
|
Fesinmeyer MD, Austin MA, Li CI, et al:
Differences in survival by histologic type of pancreatic cancer.
Cancer Epidemiol Biomarkers Prev. 14:1766–1773. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Jones S, Hruban RH, Kamiyama M, et al:
Exomic sequencing identifies PALB2 as a pancreatic cancer
susceptibility gene. Science. 324:2172009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Johns AL, Miller DK, Simpson SH, et al:
Returning individual research results for genome sequences of
pancreatic cancer. Genome Med. 6:422014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Von Hoff DD, Ervin T, Arena FP, et al:
Increased survival in pancreatic cancer with nab-paclitaxel plus
gemcitabine. N Engl J Med. 369:1691–1703. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Antoniou AC, Casadei S, Heikkinen T, et
al: Breast-cancer risk in families with mutations in PALB2. N Engl
J Med. 371:497–506. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Southey MC, Teo ZL and Winship I: PALB2
and breast cancer: ready for clinical translation! Appl Clin Genet.
6:43–52. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Casadei S, Norquist BM, Walsh T, et al:
Contribution of inherited mutations in the BRCA2-interacting
protein PALB2 to familial breast cancer. Cancer Res. 71:2222–2229.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Xia B, Sheng Q, Nakanishi K, et al:
Control of BRCA2 cellular and clinical functions by a nuclear
partner, PALB2. Mol Cell. 22:719–729. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Villarroel MC, Rajeshkumar NV,
Garrido-Laguna I, et al: Personalizing cancer treatment in the age
of global genomic analyses: PALB2 gene mutations and the response
to DNA damaging agents in pancreatic cancer. Mol Cancer Ther.
10:3–8. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chang DK, Grimmond SM, Evans TRJ, et al:
Mining the genomes of exceptional responders. Nat Rev Cancer.
14:291–292. 2014. View
Article : Google Scholar : PubMed/NCBI
|