Introduction
Lymphoblastic lymphoma (LBL) is a
lymphoproliferative disorder composed of immature, neoplastic
lymphocytes. T-cell LBL (T-LBL) is the second most common subtype
of non-Hodgkin lymphoma in children and adolescents, comprising
~85–90% of all LBLs (1,2). It is most frequent among adolescent
males, and is often characterized by rapid growth (1). T-LBL usually originates from lymph
nodes or extranodal sites, such as the skin, liver, spleen,
Waldeyer's ring, central nervous system and gonads. Approximately
50% of the patients present with a mediastinal mass (1,3). Up to
14% of extranodal non-Hodgkin lymphomas occur in the orbit
(4). The eye region may also be
secondarily affected by lymphomas arising in other extranodal
sites, and ~5% of all non-Hodgkin lymphomas affect the eye region
during the course of the disease (5).
Lymphoma is the most common orbital malignancy and
comprises ~10–20% of all orbital mass lesions. The majority of
these cases are of B-cell lineage and the most common subtype is
extranodal marginal B-cell lymphoma (5,6). To the
best of our knowledge, this is the first reported case of primary
T-LBL developing in the orbit of a 22-year-old man. The aim of this
study was to present a detailed description of the clinical,
histopathological, and genomic characteristics of this rare
neoplasm.
Case report
Clinical history
An otherwise healthy 22-year-old man sought medical
attention due to left-sided headache and swelling around the left
eye for the past 3 weeks. Initially, the patient was administered
antibiotics (Primcillin®) by a general practitioner, due
to a presumed sinusitis. The treatment was terminated as the
symptoms worsened and the patient was subsequently referred to the
Department of Ophthalmology at Rigshospitalet (Copenhagen,
Denmark). The patient complained of a history of pain in the left
eye, as well as fever and episodes of vomiting over the last few
days, without other signs or history of systemic disease. Clinical
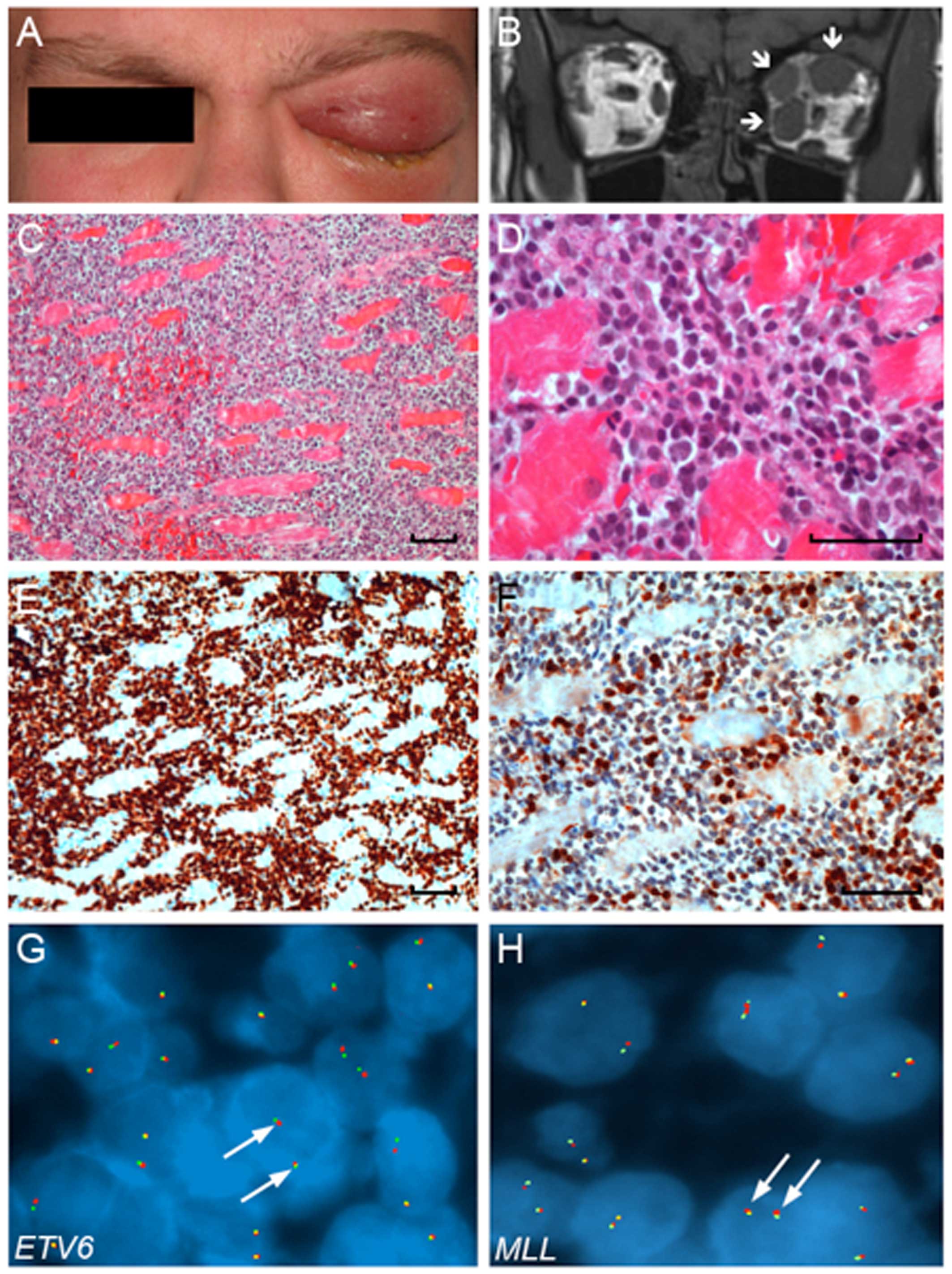
examination revealed left-sided exophthalmus (5 mm), periorbital
edema, chemosis, and reduced motility of the left eye (Fig. 1A). The visual acuity was 3/36 in the
left eye, with an ipsilateral positive relative afferent pupillary
defect. Ophthalmoscopy revealed edema of the optic nerve head. A
magnetic resonance imaging scan of the orbit revealed thickening of
the medial and superior rectus muscle, as well as of the superior
oblique muscle of the left eye (Fig.
1B). The right eye was unaffected. The blood profile revealed
leukocytosis 12.5×109/l (ref. interval
3.5–8.8×109/l) with neutrophilia 8.6×109/l
(ref. interval 1.6–5.9×109/l) and monocytosis
1.4×199/l (ref. interval 0.20–0.76×109/l).
Hemoglobin and platelet count were within normal limits. Treatment
with Prednisolon® was ineffective and 2 weeks later the
patient experienced total loss of vision (no light perception) and
complete loss of eye movements on the left side.
A positron emission tomography-computed tomography
(PET-CT) scan also confirmed activity in an enlarged subclavicular
lymph node. Biopsies of the orbital lesion and the enlarged
subclavicular lymph node were performed, together with a bone
marrow aspirate.
The patient started treatment according to the
high-risk NOPHO protocol (7).
High-dose chemotherapy, comprising cyclophosphamide, etoposide,
intrathecal methotrexate, and cytarabine, led to reduced swelling
of the left eye and left side of the face. The patient developed
neutropenia and anemia, was hospitalized due to febrile
neutropenia, and received several blood transfusions.
After the first three cycles of chemotherapy, a
PET-CT scan revealed metabolic remission of the left orbit and eye,
as well as of the subclavicular lymph node. The patient
subsequently received three additional cycles of chemotherapy and
two cycles of radiotherapy (2 Gy) followed by myeloablative
allogenic bone marrow transplantation from an unrelated donor. At
35 months after diagnosis, the patient remained in complete first
remission, but with no light perception in his left eye.
The study adheres to the tenets of the Declaration
of Helsinki, and has been approved by the Local Scientific Ethics
Committee (journal no. H-4-2013-003) and the Danish Data Protection
Agency (journal no. 2012-41-0747). Written informed consent was
obtained from the patient.
Histopathological and
immunohistochemical findings
Microscopic examination of the orbital and
subclavicular lymph node biopsy revealed infiltration by
medium-sized neoplastic lymphoid cells, with a high
nuclear-cytoplasmic ratio and a high mitotic index (Fig. 1C and D). Immunohistochemistry
revealed positivity for CD2, CD3, CD99, Tia-1, and GranzymB
(Fig. 1E and F) and variable
positivity for CD4. Immunostaining for CD5, CD7, CD8, CD10, CD20,
CD30, CD34, CD56, CD79α, ALK1, TdT, and CD1α was negative. Based on
the clinical and histopathological findings, a diagnosis of T-LBL
was established.
Genomic profile
Conventional cytogenetic analysis of the bone marrow
aspirate revealed a normal karyotype, without apparent chromosome
rearrangements. Fluorescence in situ hybridization (FISH)
analyses of bone marrow cells with probes for gene fusions
involving MLL (KMT2A), TCF3-PBX1,
BCR-ABL, and ETV6-RUNX1 (TEL-AML1), were also
negative.
Array-based comparative genomic hybridization
(arrayCGH) analysis of genomic DNA isolated from the orbital lesion
(containing >70% tumor cells) was performed with the Human
Genome CGH Microarray 244K oligonucleotide array (G4411B; Agilent
Technologies, Palo Alto, CA, USA). Data analysis was performed with
Nexus Copy Number software v.7.5 (BioDiscovery Inc., El Segundo,
CA, USA). Regions partially or completely covered by a previously
reported copy number variation were excluded from the analysis
(8,9). ArrayCGH analysis of the orbital T-LBL
revealed an apparently normal genomic profile. No major copy number
alterations were detected and there was no evidence of homozygous
deletions or gene amplifications.
To identify possible rearrangements of the
ETV6 and MLL (KMT2A) genes, we also performed
FISH analysis of formalin-fixed, paraffin-embedded sections from
the orbital lesion. The fluorescence signals were digitized,
processed and analyzed with the Isis FISH imaging system v.5.5
(MetaSystems GmbH, Altlussheim, Germany) and at least 50 nuclei
were scored for each probe. Using dual-color FISH break-apart
probes (Kreatech Diagnostics, Amsterdam, The Netherlands) we did
not detect any evidence of rearrangements or copy number
gain/amplifications of the ETV6 or MLL loci (Fig. 1G and H).
Since NOTCH1 mutations in LBLs and leukemias
are located predominantly within the heterodimerization domain and
the C-terminal PEST domain, we sequenced exons 26–27, and 34 of the
NOTCH1 gene (10). However,
no NOTCH1 mutations were detected in any of the fragments
sequenced (>70% neoplastic cells in the orbital biopsy from
which the DNA was isolated).
Discussion
The present study presented a unique case of primary
T-LBL involving the extraocular muscles of the left orbit of a
22-year-old previously healthy man. LBLs of the orbit are very
rare, with only a few cases of B-LBL reported in the literature
(5,6,11). To
the best of our knowledge, this is the first case of T-LBL
involving the orbit. Notably, a similar case of T-LBL, but
affecting Tenon's capsule (fascia bulbi), was recently described in
a 30-year-old man (4).
Intramuscular, primary T-LBLs are exceedingly uncommon, with only
one previously reported case of a 36-year-old male patient with
intramuscular T-LBL involving the anterior, lateral, and deep
posterior muscle compartments of the left leg (12). The symptoms of intramuscular LBLs are
insidious and may include arthralgia, muscle swelling, and/or
neuropathy (12). Our patient
presented with proptosis, periorbital edema, pain, ptosis,
restricted eye movements, and reduced visual acuity, which are
symptoms typical of orbital lymphomas (3).
T-LBL and T-cell acute lymphoblastic leukemia
(T-ALL) are considered to be the same disease, differing only by
the extent of bone marrow infiltration (1,6). Our
patient exhibited no involvement of the bone marrow or peripheral
blood. Lymphomas of the orbit are treated with moderate-dose
orbital radiotherapy (3) and/or
multiagent chemotherapy. In the Nordic countries, T-LBL and B-LBL
are treated according to the NOPHO protocol, which is the same
protocol as that used for lymphoblastic leukemias. Accordingly, our
patient was also treated with the high-risk NOPHO protocol; he
responded well and remains in complete first remission 35 months
after the initiation of therapy. Using multiagent chemotherapy, the
event-free survival rates for children and adolescents with T-LBL
and B-LBL is 75–85% (2). By
contrast, patients with relapsed disease have a poor outcome.
Accurate diagnosis and prompt initiation of treatment according to
high-risk protocols are therefore imperative for such patients.
To further characterize the present case of T-LBL, a
high-resolution arrayCGH analysis was performed. The tumor had a
stable genome, with no major detectable copy number alterations.
Sequence analysis of frequently mutated regions of the
NOTCH1 gene revealed no mutations. However, we cannot rule
out the possibility of mutations located outside the hotspot
regions sequenced. We were also unable to detect any rearrangements
of the MLL and ETV6 genes. Further studies using
whole exome and RNA sequencing may reveal novel oncogenic driver
mutations in cases with a normal genomic profile. Epigenetic
changes may also contribute to neoplastic transformation in such
cases.
A previous study of pediatric patients with T-LBL
has shown that mutations in NOTCH1 (involved in the
regulation of early T-cell development) are associated with a
favorable prognosis (2). Similar
observations have also been made in T-ALL (10). Conversely, patients with T-LBL and
loss of heterozygosity of 6q14-24 have a poor prognosis and an
increased risk of relapse (2).
T-LBL is an aggressive form of non-Hodgkin lymphoma
(6). It is therefore important to
consider T-LBL in the differential diagnosis of patients with
symptoms such as proptosis, reduced visual acuity and mobility of
the eye, periorbital edema, ptosis, fever, and pain. Prompt
diagnosis and treatment of these patients are imperative in order
to improve their prognosis and outcome.
Acknowledgements
This study was supported by the Synoptik-Fonden, the
Swedish Cancer Society, and BioCARE, a National Strategic Cancer
Research Program at the University of Gothenburg.
References
|
1
|
Jaffe ES, Harris NL, Stein H and Vardiman
JW: World Health Organization Classification of TumorsPathology and
Genetics Tumours of Haematopoietic and Lymphoid Tissues. IARC
Press; Lyon: pp. 111–117. 2001
|
|
2
|
Bonn BR, Rohde M, Zimmermann M, Krieger D,
Oschlies I, Niggli F, Wrobel G, Attarbaschi A, Escherich G, Klapper
W, et al: Incidence and prognostic relevance of genetic variations
in T-cell lymphoblastic lymphoma in childhood and adolescence.
Blood. 121:3153–3160. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Nutting CM, Jenkins CD, Norton AJ, Cree I,
Rose GE and Plowman PN: Primary orbital lymphoma. Hematol J.
3:14–16. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Wadhwani M, Verma D, Agarwal K, Prakash O
and Shukla S: T-cell lymphoblastic lymphoma of Tenon's capsule: An
unusual presentation. Int Ophtalmol. 34:639–642. 2014. View Article : Google Scholar
|
|
5
|
Ferry JA, Fung CY, Zukerberg L, Lucarelli
MJ, Hasserjian RP, Preffer FI and Harris NL: Lymphoma of the ocular
adnexa: A study of 353 cases. Am J Surg Pathol. 31:170–184. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Faridpooya K, Mulder MM, Merks JH, de Smet
MD, Pals ST and Saeed P: Precursor B-lymphoblastic lymphoma of the
orbit in a child: An unusual presentation of non-Hodgkin lymphoma.
Orbit. 25:153–157. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lund B, Åsberg A, Heyman M, Kanerva J,
Harila-Saari A, Hasle H, Söderhäll S, Jónsson ÓG, Lydersen S and
Schmiegelow K: Nordic Society of Paediatric Haematology and
Oncology: Risk factors for treatment related mortality in childhood
acute lymphoblastic leukemia. Pediatr Blood Cancer. 56:551–559.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
MacDonald JR, Ziman R, Yuen RK, Feuk L and
Scherer SW: The database of genomic variants: A curated collection
of structural variation in the human genome. Nucleic Acids Res.
42:(Database Issue). D986–D992. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Iafrate AJ, Feuk L, Rivera MN, Listewnik
ML, Donahoe PK, Qi Y, Scherer SW and Lee C: Detection of
large-scale variation in the human genome. Nat Genet. 36:949–951.
2004. View
Article : Google Scholar : PubMed/NCBI
|
|
10
|
Fogelstrand L, Staffas A, Wasslavik C,
Sjögren H, Söderhäll S, Frost BM, Forestier E, Degerman S,
Behrendtz M, Heldrup J, et al: Prognostic implications of mutations
in NOTCH1 and FBXW7 in childhood T-ALL treated according to the
NOPHO ALL-1992 and ALL-2000 protocols. Pediatr Blood Cancer.
61:424–430. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Shinkuma S, Natsuga K, Akiyama M, Saito A,
Saito W, Ota S, Kondo T, Abe R, Kodama K and Shimizu H: Precursor
B-cell lymphoblastic lymphoma presented with intraocular
involvement and unusual skin manifestations. Ann Hematol.
87:677–679. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Lim Z, Gupta S, Salisbury JR, Elias D,
Venkatram NK, Mufti GJ and Pagliuca A: T-cell lymphoblastic
lymphoma presenting as an intra-muscular mass. Br J Haematol.
132:5372006. View Article : Google Scholar : PubMed/NCBI
|