Introduction
Subcutaneous effusion and skin flap necrosis have
been the most commonly reported complications at the incision area
of modified radical mastectomy for breast cancer. Furthermore, the
dissection of a partial latissimus dorsi muscle flap to clear the
axillary lymph nodes may be associated with unsatisfactory
aesthetic result and shape due to postoperative atrophy of the
latissimus dorsi muscle (1,2). The aim of the present study was to
evaluate the flap recovery, postoperative drainage fluid volume and
time to drainage tube removal following latissimus dorsi
restoration during modified radical mastectomy for breast cancer.
The usefulness of this surgical method was investigated with
respect to preoperative and postoperative complications, and the
results are reported below.
Patients and methods
General characteristics
The patients who were included in our study were
selected based on the following criteria: i) All the breast cancer
patients had a single lesion; ii) the patients had no disorders of
vital organs, such as the heart, brain, liver or kidney; and iii)
the patients did not suffer from any additional conditions, such as
diabetes, blood coagulation disorders or psychological
problems.
Based on the abovementioned criteria, a total of 365
primary breast cancer patients who were treated at the Fourth
Hospital of Hebei Medical University (Shijiazhuang, China) between
April 2014 and April 2015, were enrolled in the present study and
were randomly divided into two groups. The 185 patients in the
experimental group received modified radical mastectomy combined
with intraoperative latissimus dorsi restoration. The age of these
patients ranged between 25 and 76 years (mean ± standard deviation,
53.34±4.39 years). In terms of TNM staging classification, 37 cases
had stage I, 113 had stage II and 35 cases had stage IIIA disease.
The control group included 180 patients who received modified
radical mastectomy alone. These patients were aged 26–77 years
(54.71±5.26 years). As regards TNM stage, 38 control patients had
stage I, 112 had stage II and 30 had stage IIIA disease. There were
no significant differences between the two groups regarding age or
TNM stage (P>0.05). The clinical data are summarized in Table I. Following admission, all the
patients or their family members signed an informed consent and
operation agreement. The study protocol was sanctioned and
supervised by the Medical Ethics Committee of the Fourth Hospital
of Hebei Medical University.
 | Table I.Comparison of the drainage fluid
amount (ml) between the two groups. |
Table I.
Comparison of the drainage fluid
amount (ml) between the two groups.
|
| Days |
|---|
|
|
|
|---|
| Groups | First | Second | Third | Fourth |
|---|
| Experimental | 135.97±17.37 | 81.73±10.03 | 52.22±14.25 | 14.69±6.91 |
| Control | 173.94±16.11 | 98.33±11.36 | 69.02±11.88 | 30.91±8.12 |
| t | 4.9385 | 2.6641 | 2.9784 | 3.8046 |
| P-value | <0.05 | <0.05 | <0.05 | <0.05 |
All the patients underwent a modified radical
mastectomy, and the procedures were performed by an experienced
surgical team. The incision was performed layer by layer and the
breast flap was dissected. The tissue 0.4 cm below the subcutaneous
adipose layer was preserved and the blood supply by capillary
networks was protected. During surgery, the epidermis overlying the
tumor, breast tissue and pectoralis major fascia were resected. An
incision was made in the armpit parallel to the anterior axillary
fold, and the axillary lymph nodes were dissected. The
thoracodorsal vascular pedicle of the latissimus dorsi was
protected (3), as shown in Fig. 1. Following modified radical
mastectomy, the patients in the experimental group received
latissimus dorsi restoration. The leading edge of the latissimus
dorsi was sutured with 3–5 stitches along the original attachment
to be restored and fixed on the serratus anterior muscle; the
original point of attachment of the latissimus dorsi to the
serratus anterior is shown in Fig.
2.
Observational index
The surgical incisions were inspected daily after
surgery. During these inspections, the patients were examined for
presence of flap necrosis or subcutaneous effusion, along with the
measurement of volume of drainage fluid, every day postoperatively.
If there was no subcutaneous effusion, the drainage tube contained
clear plasma-like liquid, and the volume of drainage fluid was
<15 ml, a decision was made to remove the drainage tubes. The
changes in the shape of the latissimus dorsi were next compared and
the clinical effect and breast appearance were assessed at 1 week,
1 month, 3 months and 6 months postoperatively.
Statistical methods
All the data were analyzed using statistical
analysis software, SPSS 19.0 (SPSS Inc., Chicago, IL, USA). The
quantitative data (mean ± standard deviation) were analyzed using
the t-test and the percentages were calculated using the
χ2 test. P-values <0.05 were considered to indicate
statistically significant differences.
Results
Comparison of drainage fluid
volume
The comparison of the amount of drainage fluid
between the two groups after surgery revealed that the experimental
group patients exhibited significantly lower volumes of drainage
fluid compared with the control group at 1, 2, 3 and 4 days
postoperatively (P<0.05; Table
I).
Comparison of time to drainage tube
removal
The mean time to drainage tube removal in the
experimental group was 4.61±1.78 days, compared with 6.21±2.43 days
in the control group; the difference was statistically significant
(P<0.05).
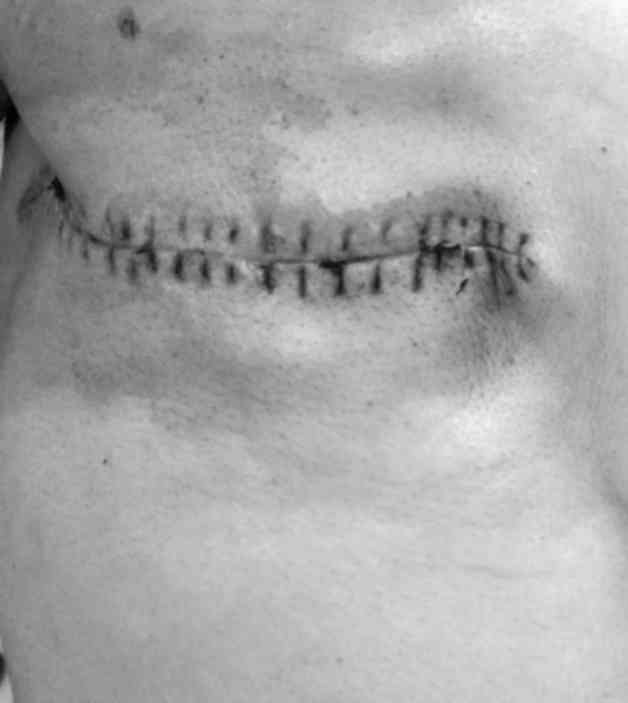
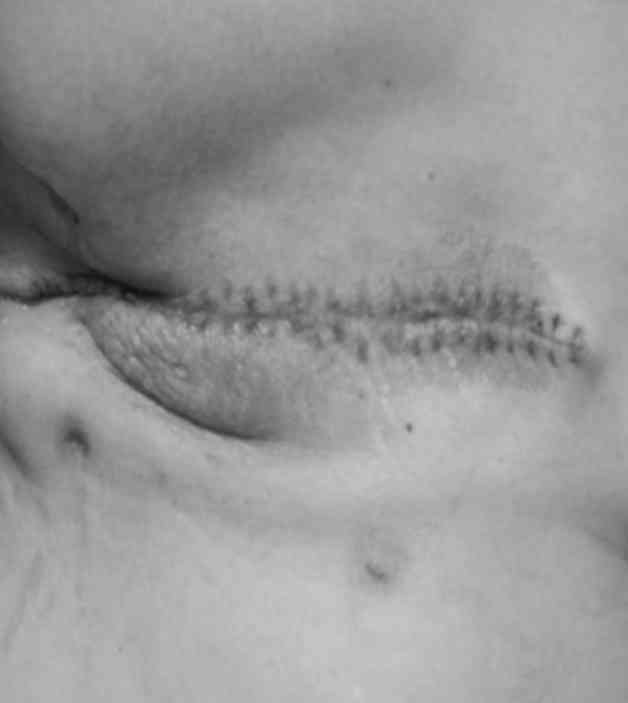
Cosmetic outcome
In addition, the patients who received
intraoperative latissimus dorsi restoration exhibited a better
shape compared with those who did not undergo restoration (Figs. 3 and 4).
Comparison of flap skin ischemia and
necrosis
Flap skin ischemia and necrosis were observed in 5
cases (5/185, 2.70%) in the experimental group and 17 cases
(17/180, 9.44%) in the control group (P<0.05). Furthermore, 3
cases (3/112, 2.68%) in the experimental group exhibited necrosis
with a transverse incision, while 5 such cases (5/111, 4.5%) were
observed in the control group (P=0.499). However, 2 cases (2/73,
2.74%) in the experimental group exhibited necrosis with a vertical
incision, whereas 12 such cases (12/69, 17.39%) were observed in
the control group (P<0.05).
Discussion
Modified radical mastectomy is currently the
preferred surgical method for the treatment of breast cancer
(4). The resection of the mammary
gland and axillary fat tissue usually results in the creation of a
large space between the breast flap and the chest wall and axilla
(5). The main causes of
postoperative suboptimal healing are as follows: Inability to
achieve complete intraoperative hemostasis, unligated
macro-lymphatics, postoperative fat liquefaction, effusion
underneath the flap and its insufficient drainage (6). Currently, the electrotome is commonly
used to perform modified radical mastectomy for breast cancer, to
isolate the flap and remove the axillary lymph nodes; however, it
leads to bleeding and lymphatic fistula during and after surgery,
and increases the drainage fluid volume, thus requiring a longer
recovery time (12.5±3.6 days). Alternatively, the utilization of
the ultrasound knife to remove the axillary lymph nodes may shorten
the recovery time, although it remains relatively long (7.1±2.3
days) (7). During intraoperative
clearing of the axillary lymph nodes, the anterior border of the
latissimus dorsi must be resected and isolated along the serratus
anterior muscle. If the restoration is not performed, the anterior
border of the latissimus dorsi must be repositioned at 3–5 cm
posteriorly, and the contracture in this site may lead to an
unsatisfactory aesthetic result and appearance, increasing patient
discomfort and adversely affecting the cosmetic outcome (8). Intraoperative latissimus dorsi
restoration may recover the original anterior border of the
latissimus dorsi, close the residual cavity in the axillary region,
and accelerate the healing of the axillary lymphatics, reduce the
amount of the effusion and shorten postoperative recovery time.
In addition, part of the skin overlying the tumors
must also be resected in modified radical mastectomy for breast
cancer. Generally, the transverse fusiform incision in the chest
wall may mitigate the flap tension, which occurs due to
longitudinal or other surgical incisions performed according to the
locations of the breast lesion(s). The high flap tension may lead
to the non-adhesion of the flap in the sagging area of the chest
wall, particularly the axillary region, and finally induce skin
flap edema and skin flap necrosis (9,10).
Several factors may lead to skin flap necrosis postoperatively,
such as the location of the incision, the thickness of the free
flap, the flap tension, subcutaneous effusion, electric knife burns
and infections (11–13). Ming et al (14) performed a comparison and observed
that the skin flap necrosis rate due to transverse fusiform
incision (13.5%) postoperatively was significantly lower compared
with that with the longitudinal incision method (28.9%) in modified
radical mastectomy for breast cancer. Therefore, it appears that by
simply minimizing the skin flap tension, the patients recover
faster. Thus, with intraoperative latissimus dorsi restoration, the
high tension of the skin flap was reduced, subsequently resulting
in relaxation of bilateral skin flap involution and lowering the
incidence of skin flap necrosis caused by reduced blood supply.
Therefore, latissimus dorsi recovery may reduce drainage fluid
seepage and the risk of ischemia and necrosis of the skin flap,
particularly in cases where the vertical incision is applied.
During latissimus dorsi resection for removing the
axillary lymph nodes, patients usually feel uncomfortable when
lying on the operated side postoperatively, due to the retraction
of the anterior border of the latissimus dorsi, which appears to
assume an uneven shape and is sensitive to touch. Moreover, the
edges of the scar may be pulled apart during postoperative
functional exercises, and the functional recovery of the affected
limb is slow (15). In the
experimental group, intraoperative latissimus dorsi restoration was
performed following modified radical mastectomy for breast cancer.
The flap tension in the experimental group patients was lower
compared with that of patients in the control group. Consistent
with this observation, high flap tension was also observed in
patients who received a longitudinal incision, and this high
tension may adversely affect the blood supply of the flap. Patients
who received intraoperative latissimus dorsi restoration and
removed the pressure dressing postoperatively, were more satisfied
with the cosmetic outcome compared with those who did not receive
this restoration. Furthermore, the breast appearance was assessed
at 6 months postoperatively, and patients in the experimental group
were satisfied with the cosmetic result. Moreover, there was no
shrinkage of the latissimus dorsi or foreign body sensation upon
touching. There was a definitive improvement in the patient's
quality of life. The additional comparison of the clinical effect
and breast appearance between the two groups at 6 months
postoperatively led to the observation that the experimental group
patients exhibited better clinical efficacy and superior cosmetic
effect. However, latissimus dorsi restoration may cause adhesion
between the latissimus dorsi and serratus anterior muscle and delay
breast reconstruction.
In conclusion, our study demonstrated that modified
radical mastectomy combined with intraoperative latissimus dorsi
restoration method in breast cancer patients may significantly
decrease drainage fluid effusion, shorten hospitalization time,
reduce intraoperative flap tension, and decrease the risk of skin
flap necrosis. The clinical effectiveness and patients'
satisfaction with the cosmetic outcome were also improved. Thus,
this method appears to be suitable for wider application in the
clinical setting.
References
|
1
|
Han Z, Zhou Y and Han K: Breast cancer
radical mastectomy preserving intercostobrachial nerve: Improve the
quality of life for postoperative patients. Chin J Clin Anat.
29:591–593. 2011.
|
|
2
|
Wang Y, Chen J and Chen B: Application of
extended latissimus dorsi flap in immediate breast reconstruction
after modified radical mastectomy. Chin J Bases Clin Gen Surg.
17:735–738. 2010.
|
|
3
|
Mo J and Tan S: Immediate breast
reconstruction with the latissimus dorsimuscle flap after modified
radical mastectomy for breast cancer. Pract J Cancer. 28:357–359.
2013.
|
|
4
|
Staradub VL and Morrow M: Modified radical
mastectomy with knife technique. Arch Surg. 137:105–110. 2002.(J).
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zhang H, Wang Y, Peng D, et al:
Nipple-areola complex sparing modified radical mastectomy for
breast cancer: Report of 159 cases. Chin J Gen Surg. 29:751–754.
2011.
|
|
6
|
Zhong T, Hofer SO, McCready DR, Jacks LM,
Cook FE and Baxter N: A comparison of surgical complications
between immediate breast reconstruction and mastectomy: The impact
on delivery of chemotherapy-an analysis of 391 procedures. Ann Surg
Oncol. 19:560–566. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Wang D, Tang J, Qin J, et al: Application
and evaluation of harmonic scalpel in breast cancer surgery
axillary lymph node dissection. Chin J Surg Oncol. 7:30–32.
2015.
|
|
8
|
Cao Z, Liu H and Chen J: Immediate breast
reconstruction in 24 patients after Nipple-sparing modified radical
mastectomy of breast cancer. Chin J Clin Oncol. 37:1042010.
|
|
9
|
Yan X, Hu Y and Cui R: Clinical analysis
of preserving intercostobrachial nerves in modified radical
mastectomy for breast cancer. Chin Arch Gen Surg. 9:37–39.
2015.(Electronic Edition).
|
|
10
|
Phillips BT, Lanier ST, Conkling N, Wang
ED, Dagum AB, Ganz JC, Khan SU and Bui DT: Intraoperative perfusion
techniques can accurately predict mastectomy skin flap necrosis in
breast reconstruction: Results of a prospective trial. Plast
Reconstr Surg. 129:778e–788e. 2012.(J). View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Bayram Y, Kulahci Y, Irgil C, Calikapan M
and Noyan N: Skin-reducing subcutaneous mastectomy using a dermal
barrier flap and immediate breast reconstruction with an implant: A
new surgical design for reconstruction of early-stage breast
cancer. Aesthetic Plast Surg. 34:71–77. 2010.(J). View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Garvey EM, Gray RJ, Wasif N, et al:
Neoadjuvant therapy and a closer look at postoperative
complication. Am J Surg. 206:894–899. 2013.(J). View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ribeiro GHFP, Kerr LM, Haikel RL, et al:
Modified radical mastectomy: Apilot clinical trail comparing the
conventional. Int J SurgeryLond Engl. 11:496–500. 2013.
|
|
14
|
Ming L, Liu H and Li J: An analysis of
transverse incisional and longitudinal incision in modified radical
mastectomy of breast cancer. Mod Oncol. 18:1765–1767. 2010.
|
|
15
|
Tan Y, Zhou E and He D: The clinical
significance of reserving intercostobrachial nerve in modified
radical mastectomy for breast cancer. Chin J Mod Operative Surg.
14:33–35. 2010.
|