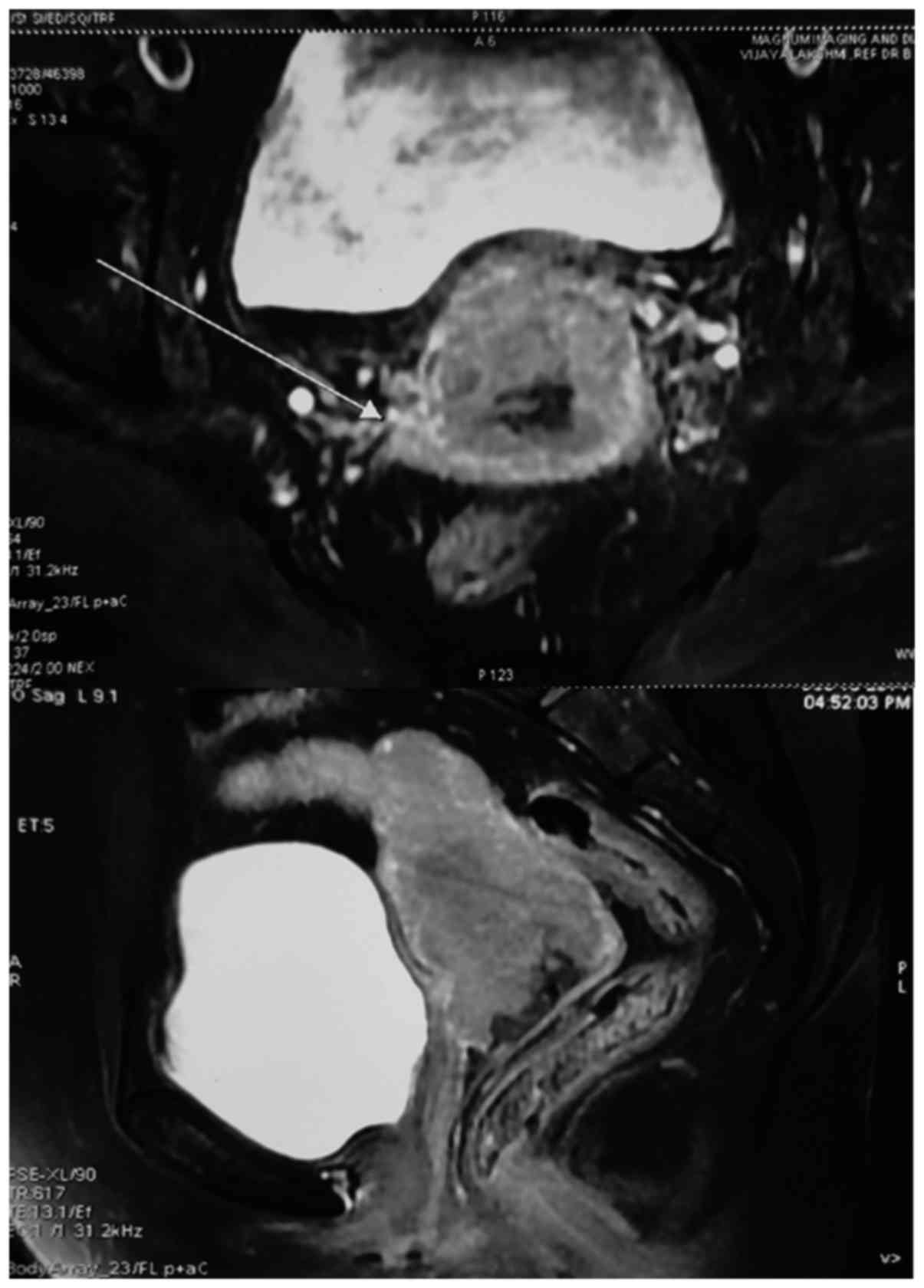
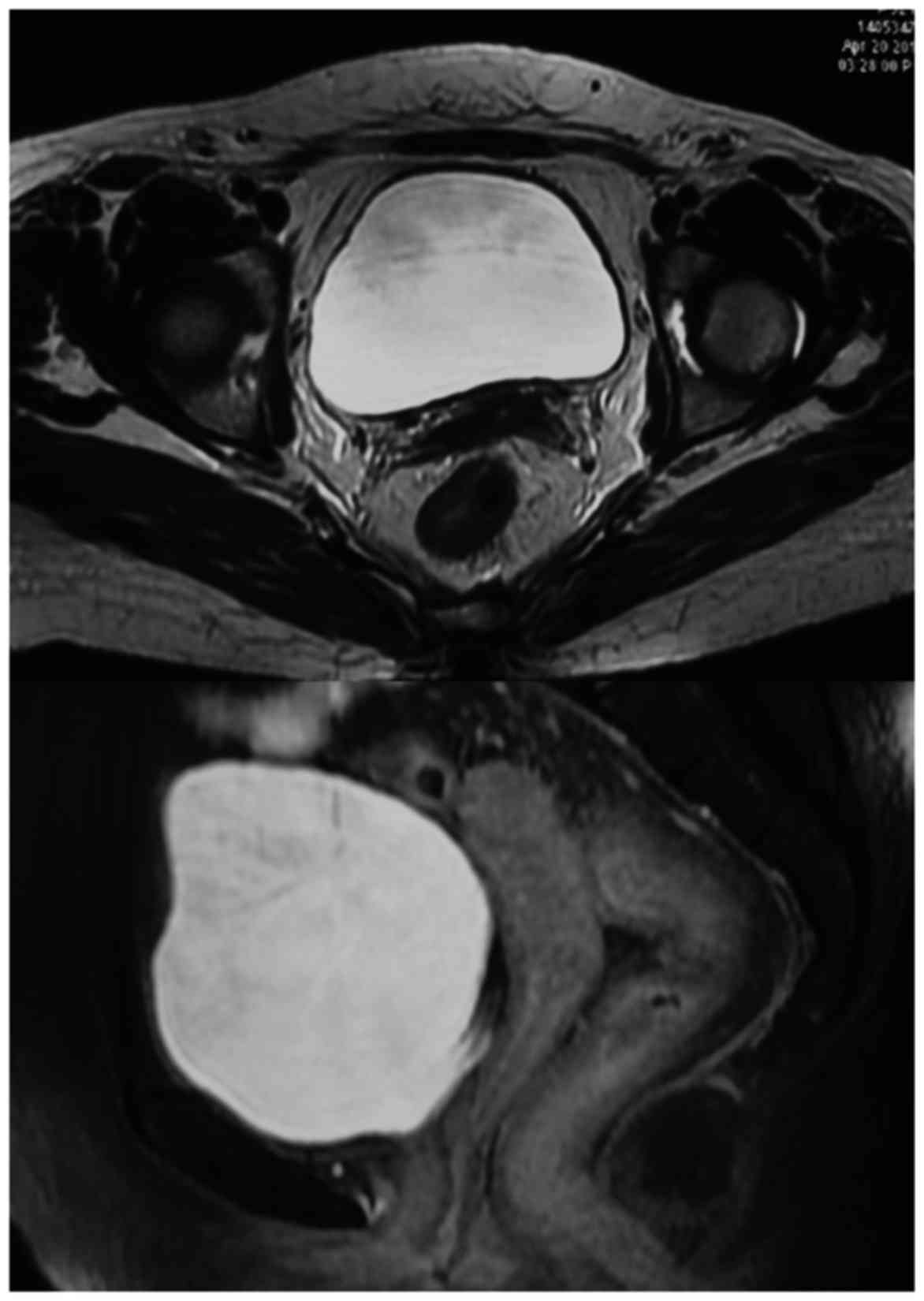
|
1
|
Asthana S, Chauhan S and Labani S: Breast
and cervical cancer risk in India: An update. Indian J Public
Health. 58:5–10. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Harjani RR, Janaki MG, Somashekhar M,
Ponni A, Alva RC, Koushik K, Kannan RA and Sathyamurthy A:
Feasibility of Concurrent Chemoradiation in Cervical Cancer
Patients From Rural Background. Clin Ovarian Other Gynecol Cancer.
7:29–32. 2014. View Article : Google Scholar
|
|
3
|
Nandakumar A, Ramnath T and Chaturvedi M:
The magnitude of cancer cervix in India. Indian J Med Res.
130:219–221. 2009.PubMed/NCBI
|
|
4
|
Bobdey S, Sathwara J, Jain A and
Balasubramaniam G: Burden of cervical cancer and role of screening
in India. Indian J Med Paediatr Oncol. 37:278–285. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Boussios S, Seraj E, Zarkavelis G,
Petrakis D, Kollas A, Kafantari A, Assi A, Tatsi K, Pavlidis N and
Pentheroudakis G: Management of patients with recurrent/advanced
cervical cancer beyond first line platinum regimens: Where do we
stand? A literature review. Crit Rev Oncol Hematol. 108:164–174.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Morris M, Eifel PJ, Lu J, Grigsby PW,
Levenback C, Stevens RE, Rotman M, Gershenson DM and Mutch DG:
Pelvic radiation with concurrent chemotherapy compared with pelvic
and para-aortic radiation for high-risk cervical cancer. N Engl J
Med. 340:1137–1143. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
National Cancer Institute, . NCI Issues
Clinical Announcement on Cervical Cancer: Chemotherapy Plus
Radiation Improves Survival. February 22;
|
|
8
|
Takekida S, Fujiwara K, Nagao S, Yamaguchi
S, Yoshida N, Kitada F, Kigawa J, Terakawa N, Nishimura R and Phase
II: Phase II study of combination chemotherapy with docetaxel and
carboplatin for locally advanced or recurrent cervical cancer. Int
J Gynecol Cancer. 20:1563–1568. 2010.PubMed/NCBI
|
|
9
|
Chen CC, Wang L, Lin JC and Jan JS: The
prognostic factors for locally advanced cervical cancer patients
treated by intensity-modulated radiation therapy with concurrent
chemotherapy. J Formos Med Assoc. 114:231–237. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Monk BJ, Alberts DS, Burger RA, Fanta PT,
Hallum AV III, Hatch KD and Salmon SE: In vitro phase II comparison
of the cytotoxicity of a novel platinum analog, nedaplatin (254-S),
with that of cisplatin and carboplatin against fresh, human
cervical cancers. Gynecol Oncol. 71:308–312. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Naik R and Khan MA: Doceaqualip in a
patient with prostate cancer who had an allergic reaction to
conventional docetaxel: A case report. Mol Clin Oncol. 6:341–343.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ahmad A, Sheikh S, Taran R, Srivastav SP,
Prasad K, Rajappa SJ, Kumar V, Gopichand M, Paithankar M, Sharma M,
et al: Therapeutic efficacy of a novel nanosomal docetaxel lipid
suspension compared with taxotere in locally advanced or metastatic
breast cancer patients. Clin Breast Cancer. 14:177–181. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ashraf M SR, Khan MA, Shah M, Bhat Y and
Wani ZA: Efficacy and safety of a novel nanosomal docetaxel lipid
suspension (NDLS) as an anti cancer agent a retrospective study.
Ann Oncol. 27(suppl_9): mdw579.008. 2016. View Article : Google Scholar
|
|
14
|
Kong TW, Chang S-J, Paek J, Yoo S-C, Yoon
J-H, Chang K-H, Chun M and Ryu H-S: Comparison of concurrent
chemoradiation therapy with weekly cisplatin versus monthly
fluorouracil plus cisplatin in FIGO stage IIB-IVA cervical cancer.
J Gynecol Oncol. 23:235–241. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Nam EJ, Lee M, Yim GW, Kim JH, Kim S, Kim
SW, Kim JW and Kim YT: Comparison of carboplatin- and
cisplatin-based concurrent chemoradiotherapy in locally advanced
cervical cancer patients with morbidity risks. Oncologist.
18:843–849. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Adams M, Kerby IJ, Rocker I, Evans A,
Johansen K and Franks CR; The Swons Gynaecological Cancer Group, :
A comparison of the toxicity and efficacy of cisplatin and
carboplatin in advanced ovarian cancer. Acta Oncol. 28:57–60. 1989.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Weiss GR, Green S, Hannigan EV, Boutselis
JG, Surwit EA, Wallace DL and Alberts DS: A phase II trial of
carboplatin for recurrent or metastatic squamous carcinoma of the
uterine cervix: A Southwest Oncology Group study. Gynecol Oncol.
39:332–336. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Schwab CL, English DP, Roque DM and Santin
AD: Taxanes: Their impact on gynecologic malignancy. Anticancer
Drugs. 25:522–535. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Rein DT, Kurbacher CM, Breidenbach M,
Schöndorf T, Schmidt T, König E, Göhring UJ, Blohmer JU and
Mallmann P: Weekly carboplatin and docetaxel for locally advanced
primary and recurrent cervical cancer: A phase I study. Gynecol
Oncol. 87:98–103. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Nagao S, Fujiwara K, Oda T, Ishikawa H,
Koike H, Tanaka H and Kohno I: Combination chemotherapy of
docetaxel and carboplatin in advanced or recurrent cervix cancer. A
pilot study. Gynecol Oncol. 96:805–809. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lyman GH, Dale DC, Legg JC, Abella E,
Morrow PK, Whittaker S and Crawford J: Assessing patients' risk of
febrile neutropenia: Is there a correlation between
physician-assessed risk and model-predicted risk? Cancer Med.
4:1153–1160. 2015. View
Article : Google Scholar : PubMed/NCBI
|