Introduction
Inflammatory bowel disease (IBD) consists of two
types: Crohn's disease (CD) and ulcerative colitis (UC). Crohn's
disease is a chronic immune-mediated inflammatory disorder of the
gastrointestinal tract. It is widely accepted that the key
mechanism underlying the development of CD is a dysregulated immune
response to commensal flora in a genetically susceptible host
(1,2). However, the precise etiology remains
to be determined, which leads to limitations of corresponding
therapeutic methods.
Diet has been hypothesized to serve an important
role in the pathogenesis and clinical course of CD, as the
incidence of CD has risen markedly in countries where a western
diet culture prevails (3,4). In particular, dietary fat may
influence intestinal inflammation and regulate mucosal immunity,
which is considered to be a core etiological factor and may be a
promising therapeutic target for CD.
The fatty acids that are consumed as part of our
daily diets are broadly classified as cis- and trans-fatty acids
(TFAs). N-3 polyunsaturated fatty acids (PUFAs) belong to the group
of cis PUFAs, and include docosahexaenoic acid and eicosapentaenoic
acid. Numerous previous studies have demonstrated that TFA
consumption was a risk factor, while n-3 PUFA consumption was a
protective factor, for various diseases associated with systemic or
localized inflammation, including diabetes and coronary heart
disease (5,6). According to a retrospective study by
Nanthakrishnan et al (5),
which followed 170,805 women with IBD for 26 years, a high intake
of dietary long-chain n-3 PUFA may be associated with a reduced
risk of UC. By contrast, a high intake of TFA was reported be
associated with an increased risk of UC (7). Dietary n-3 PUFA competitively
inhibits the formation of proinflammatory prostaglandins and
leukotrienes via the arachidonic acid pathway (8), and inhibits angiogenesis and adaptive
immune responses (9,10). However, the effect of dietary n-3
PUFA on CD and the underlying mechanisms remain unclear.
Peroxisome proliferator-activated receptor (PPAR)-γ
is a member of the nuclear receptor superfamily of ligand-dependent
transcription factors, and is primarily expressed in adipose tissue
and the colonic epithelium (11).
Recently, its role in intestinal inflammation has emerged. Su et
al (12) reported that the use
of the PPAR-γ synthetic agonist thiazolidinedione (TZD) markedly
decreased colonic inflammation in mice with 2,4,6-trinitrobenzene
sulfonic acid (TNBS)-induced colitis. These results were
subsequently confirmed in different animal models with various
models of colitis induced by chemical compounds (13), ischaemia (14) or genetically modified animals
(15,16). PPAR-γ interferes with inflammatory
pathways by antagonizing the activity of various transcription
factors, such as nuclear factor of activated T-cells (NFAT). It is
established that NFAT has an important function in the
transcriptional induction of various proinflammatory genes,
including interleukin (IL)-4 and IL-2, during T-cell activation
(17,18). Thus, PPAR-γ may serve a key role in
the regulation of adipocyte differentiation and inflammation. In
addition, fatty acids and their metabolites are able to modulate
PPAR-γ. The precise association between dietary fatty acids and the
PPAR-γ/NFAT pathway remains unknown.
In the current study, the primary objectives were to
investigate the effect of long-term administration of an n-3
PUFA-enriched diet on CD rat models induced by TNBS, and to
characterize the association between n-3 PUFA and PPAR-γ/NFAT in
intestinal inflammation. In addition, the current study aimed to
determine whether n-3 PUFA may exhibit a potent anti-inflammatory
effect in experimental CD rats, and whether the protective effect
of this fatty acid involves the activation of PPAR-γ and inhibition
of NFAT.
Materials and methods
Ethics statement
The experiments in the present study were performed
in conformity with the National Institutes of Health (NIH)
(19) and Animal Research:
Reporting in vivo Experiments (ARRIVE) guidelines (20), and were approved by the Animal Care
and Use Committee of Sun Yat-Sen University (approval no.
20161652118). All surgical and experimental procedures were
performed according to the guidelines for the care and use of
animals approved by the Sun Yat-Sen University and were in
accordance with the code of Ethics of the EU Directive 2010/63/EU
for animal experiments. All efforts were made to minimize animal
suffering and to reduce the number of animals used.
Animals
Specific pathogen free male Sprague-Dawley rats
(n=90; 6-week-old; weight, 180–220 g) were obtained from the Animal
Experiment Center of the Sun Yat-Sen University. Animals were
maintained in controlled environment conditions with temperature
(23±1°C), humidity (60±10%) and a 12-h light/dark cycle. They were
housed in 15 cages each containing 6 rats and were allowed standard
rat chow and water ad libitum.
Experimental design
Following 1-week acclimation, 90 Sprague-Dawley rats
were randomly divided into the following 6 groups (n=15): Control,
intragastric administration of distilled water; PUFA control,
intragastric administration of n-3 PUFA (20 mg/kg/day); TFA
control, intragastric administration of TFA (13 mg/kg/day); model,
intragastric administration of distilled water + rectal TNBS
administration; PUFA model, intragastric administration of n-3 PUFA
(20 mg/kg/day) + rectal TNBS administration; and TFA model,
intragastric administration of TFA (13 mg/kg/day) + rectal TNBS
administration. CD rat models were induced by rectal administration
of TNBS (Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) in ethanol
(100 mg/kg TNBS + 50% ethanol; 0.25 ml) four times, with a 10 day
interval between each TNBS treatment (days 0, 10, 20 and 30)
(21). Rats in the control, PUFA
and TFA control groups were given rectal administration of 0.9%
NaCl solution instead of TNBS. TFA (13 mg/kg/day; Medicience Ltd.,
Yangzhou, China) and n-3 PUFA (20 mg/kg/day; Medicience Ltd.) were
administered intragastrically every day from day 0 to day 60. We
calculated the most suitable dose for rats according to the
following formula: Dose for rats = (X mg/kg × 70 kg × 0.018)/0.2 kg
= 6.3 X mg/kg. X denotes the effective dose for humans, 70 kg is
the standard weight for humans, 0.018 is the ratio of the
equivalent dose between humans and rats based on body surface area
and 0.2 kg is the standard weight of a rat (22).
Necropsy
At the end of the experiment (day 60), rats were
anesthetized and blood samples (2 ml from each rat) were collected
into tubes by cardiac puncture. Colon tissue specimens were
subjected to macroscopic assessment prior to being cut and fixed in
10% formaldehyde saline solution at room temperature for 24 h
before histological analysis. Serum was collected following blood
centrifugation at 550 × g for 10 min at 4°C. Tissue and serum
samples were snap frozen in liquid nitrogen and stored at −80°C
until further analysis.
Blood fat and inflammatory marker
detection
Blood samples were collected in tubes at day 60 by
cardiac puncture. Serum content of total cholesterol (TC),
triglycerides (TGs), erythrocyte sedimentation rate (ESR) and
high-sensitivity C-reactive protein (hs-CRP) was determined using
the Olympus AU400 Clinical Chemistry analyzer (Olympus Corporation,
Tokyo, Japan).
Disease activity index (DAI) of
rats
Rats were examined daily by a blinded investigator
to determine the DAI, which was achieved by scoring body weight
loss, stool consistency and bleeding, as described previously
(23,24), and scoring is described in Table I.
 | Table I.Criteria for scoring disease activity
index. |
Table I.
Criteria for scoring disease activity
index.
| Score | Weight loss, % | Stool
consistency | Occult blood or
gross bleeding |
|---|
| 0 | 0 | Normal | Negative |
| 1 | 1–5 | Loose stool | Negative |
| 2 |
5–10 | Loose stool | Hemoccult
positive |
| 3 | 10–15 | Diarrhea | Hemoccult
positive |
| 4 | >15 | Diarrhea | Gross bleeding |
Colon macroscopic damage index (CMDI)
of rats
The CMDI was the criteria for assessing macroscopic
damage. The following numerical rating scores were employed: 0, no
ulcer or inflammation; 1, local hyperemia without ulcers; 2,
ulceration without hyperemia; 3, ulceration and inflammation at one
site only; 4, two or more sites of ulceration and inflammation
extending >1 cm; and 5, ulceration extending >2 cm (24,25).
Tissue damage index (TDI) of rats
Formaldehyde-fixed colon specimens were obtained and
cut into 4 µm thick sections. The sections were then stained with
0.5% hematoxylin for 5 min, followed by 0.5% eosin for 1 min at
room temperature. Hematoxylin and eosin staining was performed via
a light microscope at ×100 magnification. An expert pathologist
evaluated the stained samples in a blind fashion and evaluated the
TDI according to previously described criteria (26), which involves scores from 0–4 (0,
no signs of inflammation; 1, low level of leukocyte infiltration;
2, moderate level of leukocyte infiltration; 3, high level of
leukocyte infiltration, high vascular density and thickening of the
colon wall; 4, in addition to points mentioned for score 3, the
lesions exceed 50% of the specimens; 5, transmural infiltration,
loss of goblet cells, high vascular density and thickening of the
colon wall; and 6, in addition to points mentioned for score 5, the
lesions exceed 50% of the specimens) (24).
ELISA analysis
The serum levels of cytokines associated with local
mucosal immunity, including IL-6, IL-12, tumor necrosis factor
(TNF)-α and interferon (IFN)-γ, were measured using commercial
ELISA kits (R&D Systems, Inc., Minneapolis, MN, USA) according
to the manufacturer's protocols. A standard curve was generated
with each assay, with the limit of detection for IL-6, IL-12, TNF-α
and IFN-γ being 12.0, 5.5, 20.0 and 15.4 pg/ml, respectively. Each
sample was performed in triplicate.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR) analysis
All the rats in each group were sampled for RT-qPCR
analysis. Total RNA was extracted from colon tissue using TRIzol
(Invitrogen, Thermo Fisher Scientific, Inc., Waltham, MA, USA),
according to manufacturer protocol. The extracted RNA was diluted
to a concentration of 1 µg/µl, and aliquots were stored at −80°C.
RT-qPCR was performed in triplicate for each specimen using
SYBR-Green PCR Master mix (Applied Biosystems; Thermo Fisher
Scientific, Inc.). Total RNA (500 ng) was used for cDNA synthesis
and 1 µl each reverse transcription product was added to a 9 µl
MasterMix reaction containing buffer, SYBR hydrolysis probes, Hot
Start Taq polymerase and dNTPs. The reaction consisted of 0.2 µl 25
mM dNTPs, 0.5 µl 25 µM corresponding primers and 9.3 µl
ddH2O for PCR amplification. qPCR cycling conditions
were as follows: 10 min pre-denaturation at 95°C followed by 35 or
38 cycles of 10 sec denaturation at 95°C and 60 sec annealing at
52°C. Sequences of the primers used and each number of cycles are
listed in Table II. Each
experiment was performed in triplicate. The relative levels of the
target mRNAs were normalized to the corresponding levels of GAPDH
mRNA in the same cDNA sample using a standard curve method
recommended in the LightCycler Software version 3.5 (Roche
Molecular Diagnostics, Pleasanton, CA, USA).
 | Table II.Primers and product size for each
target gene. |
Table II.
Primers and product size for each
target gene.
|
| Primer
sequence |
|
|
|
|---|
|
|
|
|
|
|
|---|
| Gene | Forward | Reverse | Length, bp | Number of
cycles | Annealing
temperature, °C |
|---|
| PPAR-γ |
5′-ATTACGATAGCCGTTCC-3′ |
5′-TAATGCTATCGGCAATT-3′ | 190 | 35 | 52 |
| NFAT |
5′-GTACCGTACATGCGTTCA-3′ |
5′-CATGGCATGTACGCAAGT-3′ | 201 | 35 | 52 |
| IL-2 |
5′-CGTATCATGCACTGCATA-3′ |
5′-GCATAGTACGTGACGTAT-3′ | 186 | 38 | 52 |
| IL-4 |
5′-ATCCGATAACGTACCGTAC-3′ |
5′-TAGGCTATTGCATGGCATG-3′ | 173 | 35 | 52 |
| GAPDH |
5′-GAATCTGGTGGCTGTGGA-3′ |
5′-CCCTGAAAGGCTTGGTCT-3′ | 202 | 35 | 52 |
Statistical analysis
Data are presented as the mean ± standard deviation.
Homoscedasticity tests and normal distribution were performed, and
data were subsequently analyzed by one-way analysis of variance
followed by the Bonferroni multiple comparisons test. P<0.05 was
considered to indicate a statistically significant difference. All
statistical analyses were performed using SPSS version 13.0 (SPSS,
Inc., Chicago, IL, USA).
Results
Serum fat and inflammatory
indices
The results in Table
III demonstrated that TC in the TFA model group was
significantly higher compared with other groups (P<0.05), which
indicates that a TFA-enriched diet may disturb fat metabolism.
However, no differences were observed in the TG content between all
groups. Inflammatory indices, including ESR and hs-CRP, were
significantly increased in all three model groups compared with the
control groups, but the n-3 PUFA model group exhibited
significantly reduced levels compared with the model and TFA model
groups (P<0.05; Table
III).
 | Table III.Serum fat and inflammatory index in
rats in each group. |
Table III.
Serum fat and inflammatory index in
rats in each group.
| Group | Triglyceride,
mmol/l | Total cholesterol,
mmol/l | Erythrocyte
sedimentation rate, mm/h | High-sensitivity
C-reactive protein, mg/l |
|---|
| Control | 1.21±0.22 |
1.72±0.42a |
12.00±4.32a–c |
2.11±0.28a–c |
| PUFA control | 1.32±0.35 |
1.69±0.61a |
13.41±4.36a–c |
2.78±0.34a–c |
| TFA control | 1.37±0.28 |
2.14±0.42a | 15.67±4.84
a–c |
2.65±0.27a–c |
| Model | 1.17±0.26 |
2.01±0.53a |
23.07±4.90b |
9.05±2.50b |
| PUFA model | 1.39±0.33 |
1.97±0.45a |
19.34±5.07a,c |
6.15±1.75a,c |
| TFA model | 1.37±0.30 | 3.01±0.55 |
24.05±4.82b |
8.98±2.63b |
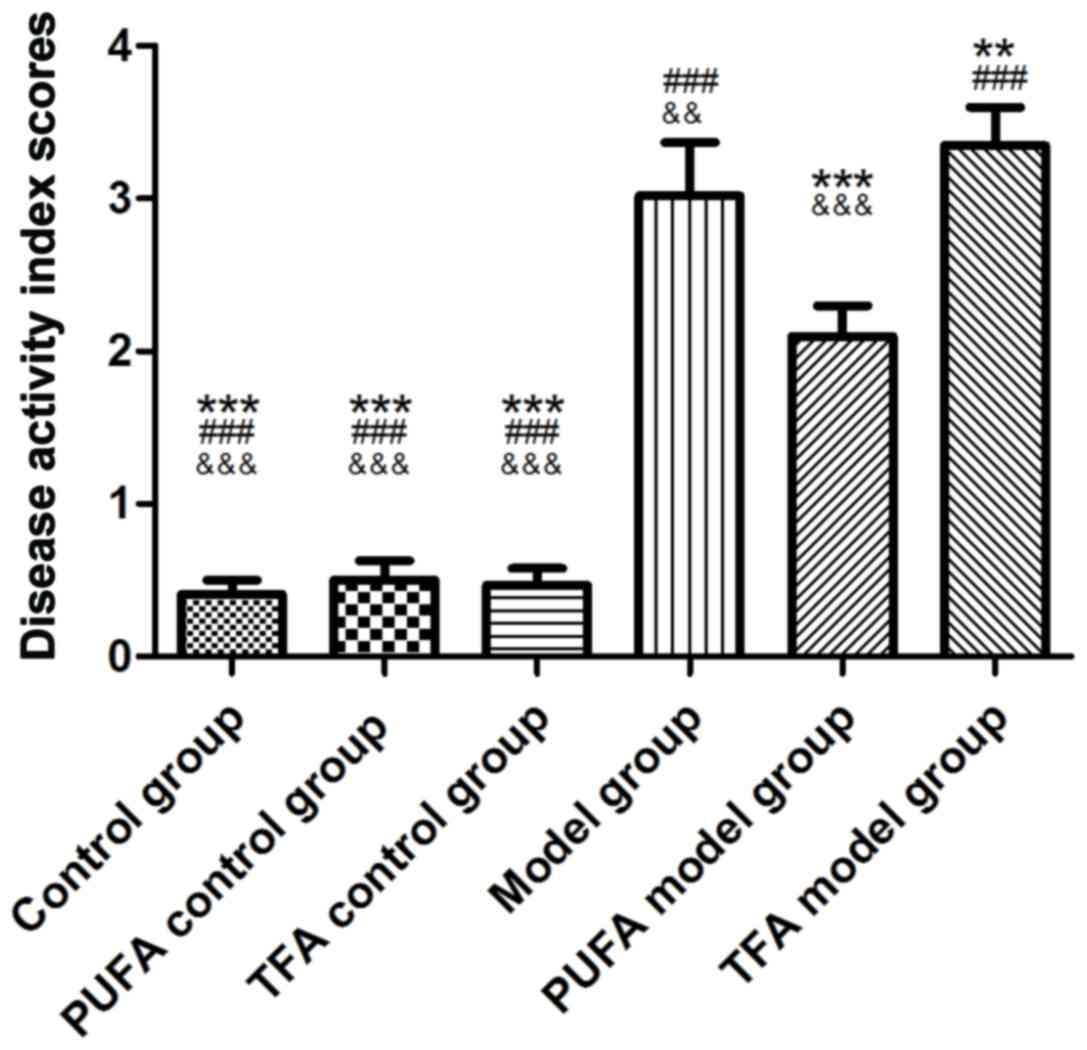
DAI scores of rats
DAI scores in the model, PUFA model and TFA model
groups were significantly higher compared with those in control,
PUFA control and TFA control groups (P<0.001 vs. all control
groups; Fig. 1). No differences
were observed in the DAI scores among the three control groups,
which indicate that diet intervention did not have effects in
healthy rats (Fig. 1). However, a
significant decrease in DAI scores was observed in the PUFA model
group (P<0.001 vs. model group) and an increase in the TFA model
group (P<0.01 vs. model group; Fig.
1).
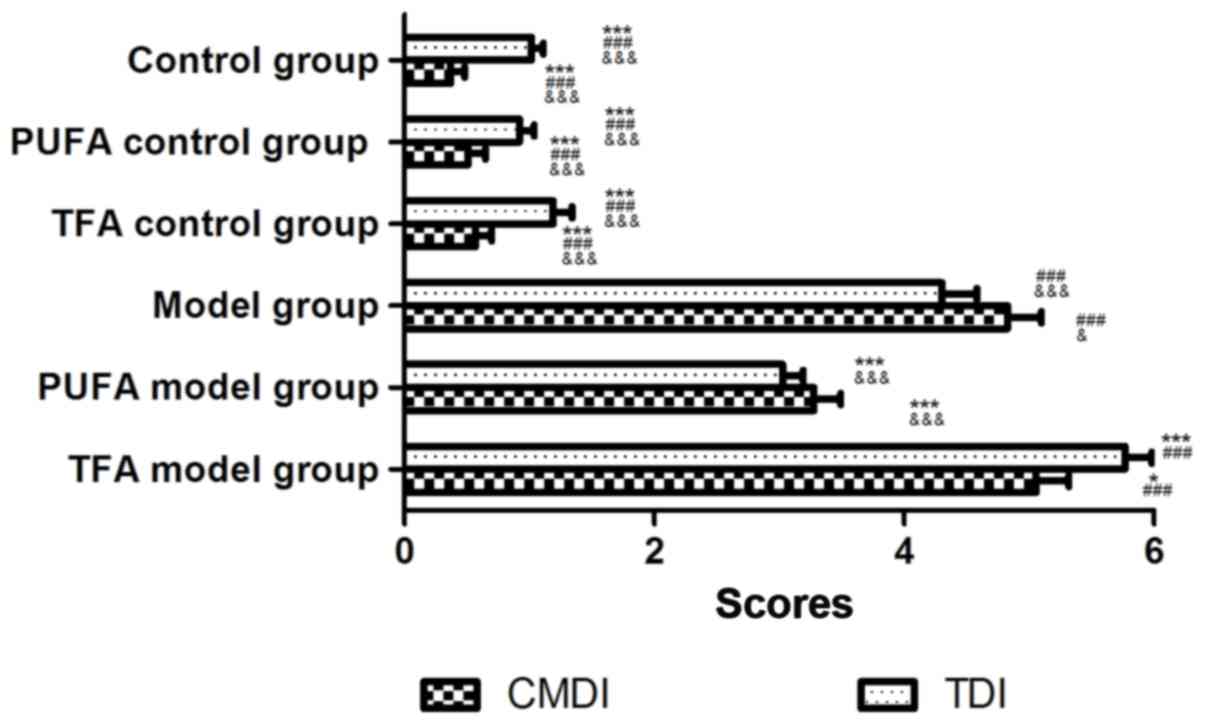
CMDI and TDI
CMDI and TDI indices in the model group were
significantly higher compared with the three control groups
(P<0.001). A high intake of n-3 PUFA significantly lowered both
scores, exerting a protective effect on rats with CD (P<0.01 for
CMDI and P<0.001 for TDI). A TFA-enriched diet significantly
increased TDI scores (P<0.001) and CMDI scores were increased
(without statistical significance; P>0.05) compared with the
model group (Fig. 2).
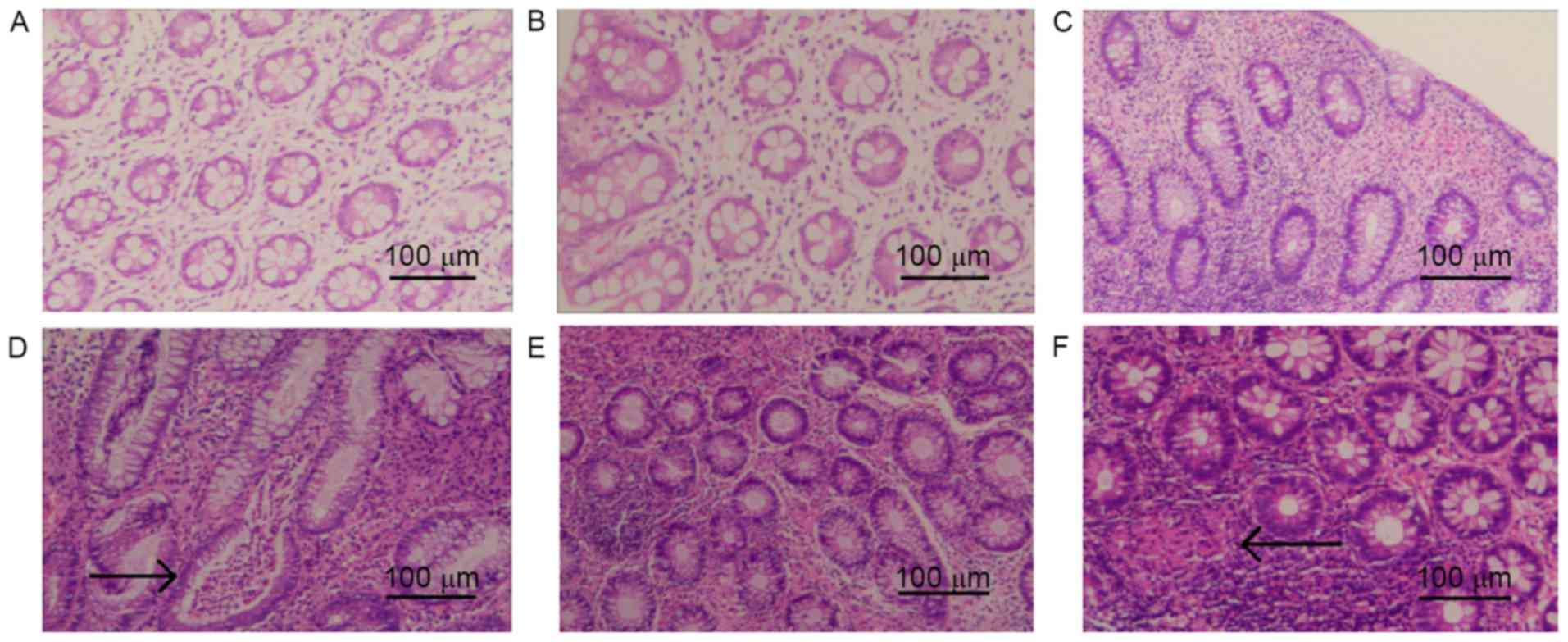
Pathological changes in rat colon
tissue
Colon tissue in TNBS-induced CD model rats was
characterized by infiltration of the lamina propria with
inflammatory cells and crypt abscesses (Fig. 3). Following intervention with n-3
PUFA treatment, a marked improvement in the level inflammation was
observed (Fig. 3). However, TFA
intervention appeared to aggravate inflammation with epithelioid
granulomas, which are typical pathological changes of CD (Fig. 3).
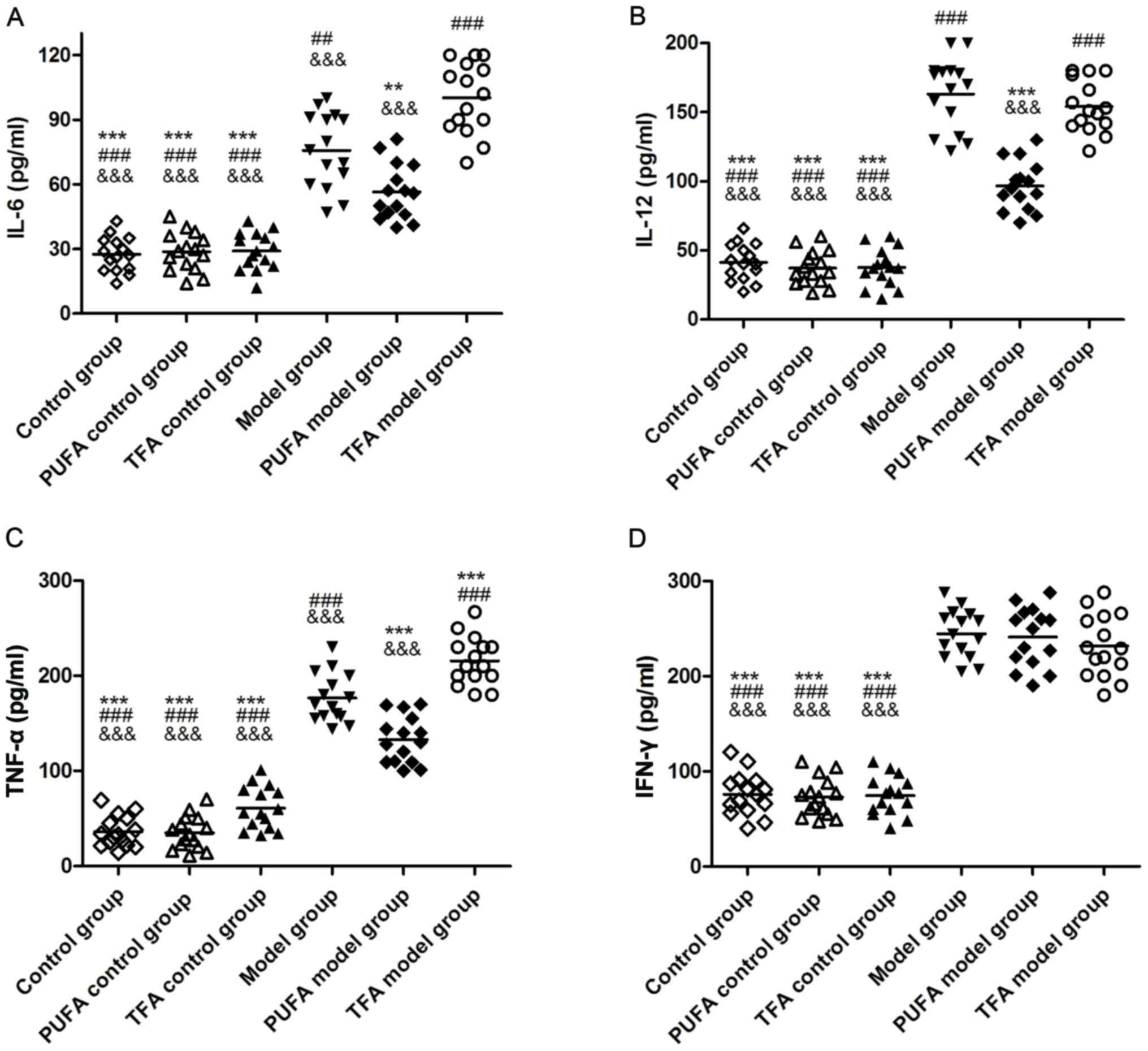
Serum levels of cytokines in each
group detected by ELISA
Marked increases in IL-6, IL-12, TNF-α and IFN-γ
serum levels were observed in the three model groups compared with
the three control groups (Fig. 4),
indicating that TNBS intervention successfully altered local
mucosal immunity. IL-6, IL-12 and TNF-α concentrations were
significantly decreased in the PUFA model group compared with the
model group (P<0.05; Fig. 4).
However, a TFA-enriched diet appeared to worsen local mucosal
immunity as levels of serum IL-6 and TNF-α were significantly
increased in the TFA model group compared with the model group
(P<0.05; Fig. 4).
Expression of PPAR-γ/NFAT-associated
genes detected by RT-qPCR
The mRNA expression of genes associated with the
PPAR-γ/NFAT pathway, including PPAR-γ, NFAT, IL-2 and IL-4, was
detected by RT-qPCR. A significant increase in the expression of
the proinflammatory cytokines IL-2 and IL-4 was observed in the
model and TFA model groups (P<0.05; Fig. 5) compared with the control group.
However, IL-2 and IL-4 expression was reduced in the n-3PUFA model
group, indicating a potential anti-inflammatory effect of n-3 PUFA
diet intervention (Fig. 5).
Furthermore, the expression of PPAR-γ was enhanced in the n-3 PUFA
intervention group compared with model and TFA model groups
(Fig. 5), which indicates that
PPAR-γ may be activated by an n-3 PUFA-enriched diet. In addition,
the expression of NFAT was reduced in the n-3 PUFA model group
compared with model and TFA model groups (Fig. 5), indicating that n-3 PUFA may
inhibit NFAT, potentially via the activation of PPAR-γ in
TNBS-induced CD rats.
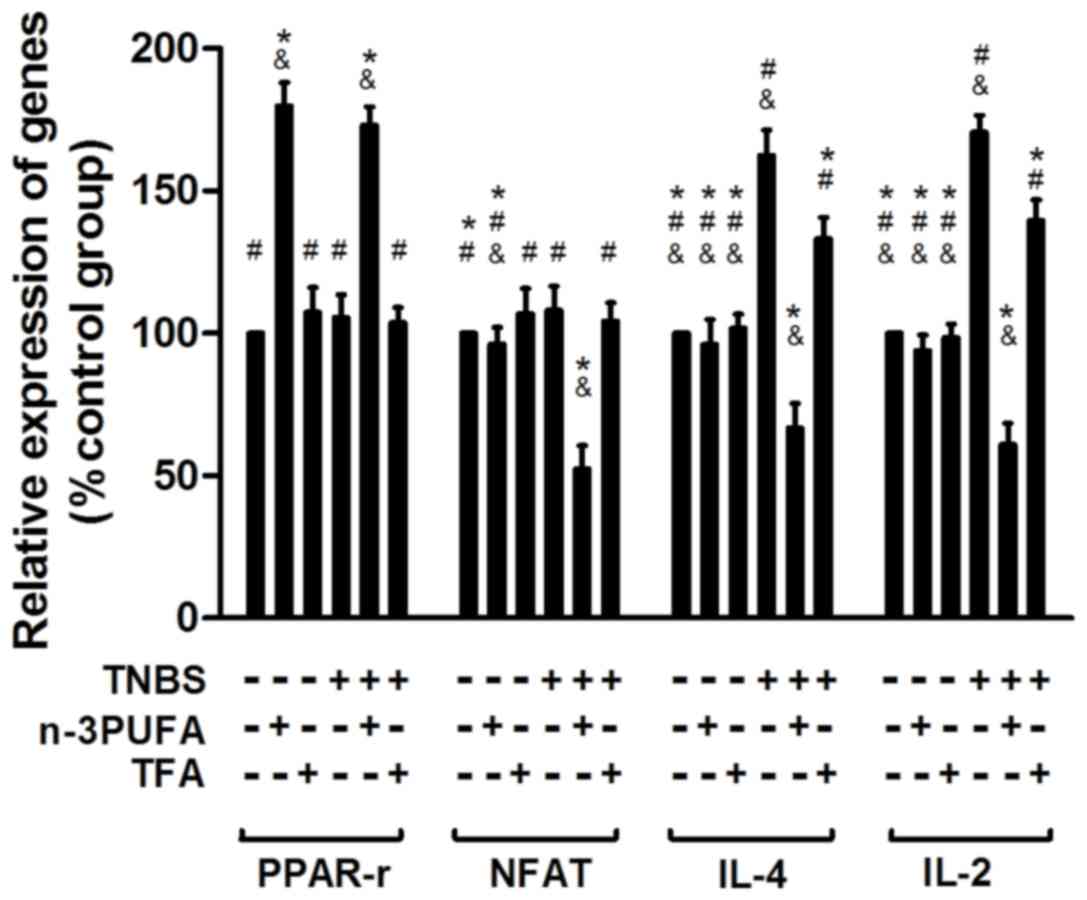 | Figure 5.Relative mRNA expression of genes
associated with the PPAR-γ/NFAT pathway in each group. The relative
mRNA expression of PPAR-γ, NFAT, IL-4 and IL-2 was analyzed in
colon samples taken from rats in each treatment group by reverse
transcription-quantitative polymerase chain reaction, and
expression was presented normalized to the control group. PPAR,
peroxisome proliferator-activated receptor; NFAT, nuclear factor of
activated T-cells; IL, interleukin; TNBS, 2,4,6-trinitrobenzene
sulfonic acid; PUFA, polyunsaturated fatty acid; TFA, trans fatty
acid. *P<0.05 vs. model group, #P<0.05 vs. PUFA
model group and &P<0.05 vs. TFA model group. |
Discussion
At present, no curative treatment is available for
CD. Traditional treatments, including aminosalicylates,
corticosteroids, immunomodulating drugs and biological agents, are
associated with limitations due to adverse side effects (27–29).
Therefore, the identification of alternative therapeutic strategies
that are effective with reduced side effects is necessary. Previous
studies have demonstrated the association between dietary fat and
IBD. Borniquel et al (30)
reported that dietary conjugated linoleic acid ameliorated colonic
inflammation induced by dextran sulfate sodium in mice. In
addition, another study demonstrated the efficacy of short chain
fatty acids in accelerating clinical disease in an
IL-10−/− mouse model (31).
Dietary n-3 PUFA is present in fish oil. It was
reported to ameliorate intestinal inflammation in a colitis rat
model. However, the underlying mechanisms were not determined
(32). Based on this, the present
study aimed to determine whether a similar protective effect of
dietary n-3 PUFA may be observed in CD. Thus, TNBS-induced CD rat
models were established, which were characterized as IL-12 driven
and resembled the alterations observed in patients with CD
(33). An n-3 PUFA diet was
employed as an intervention strategy. The present study
successfully established CD rat models with pathological changes
such as massive transmural infiltration of inflammatory cells,
typical epithelioid granulomas and crypt abscess formation. The
results demonstrated that dietary supplementation with n-3 PUFA
ameliorated the symptoms of experimental CD rats, with lower DAI,
CDI and CMDI scores and milder changes demonstrated by pathological
observation. In addition, the results indicated that the protective
effects of n-3 PUFA may involve activation of the PPAR-γ/NFAT
pathway, which ultimately led to improvements in mucosal
inflammation and amelioration of tissue damage. This study, to the
best of our knowledge, was the first to report the protective
effect of dietary n-3 PUFA on CD rats and to identify potential
underlying mechanisms.
The role of PPAR-γ in the etiology and treatment of
colitis is a subject of great interest. Thiazolidinediones, PPAR-γ
ligands, have been previously reported to decrease the severity of
UC in rats (34). Furthermore,
PPAR-γ+/− heterozygous mice exhibited increased
susceptibility to experimental-induced colitis, which further
indicated a role for PPAR-γ in the maintenance of gut homeostasis
(35,36). Despite these observations, the role
of PPAR-γ in experimental CD rats and its association with dietary
fatty acid intervention has not been previously reported. It is
established that n-3 PUFAs and their metabolites are endogenous
PPAR-γ ligands (37). The results
of the current study revealed that n-3 PUFA intake activated PPAR-γ
expression in healthy rats and TNBS-induced CD rats. Therefore, the
protective effects of n-3 PUFA may be attributed to the activation
and increased expression of PPAR-γ, which was consistent with a
previous study (30). PPAR-γ
usually exists as a heterodimer with retinoid X receptor, and has
been reported to be associated with PPAR response elements in the
regulation of NFAT, a transcription factor that binds to the IL-2
and IL-4 promoters and is necessary for gene transcription
(18,38). The present study demonstrated that
an n-3 PUFA-enriched diet inhibited the expression of NFAT in CD
rats. In addition, the gene expression of the proinflammatory
cytokines IL-2 and IL-4 was reduced, which may contribute to
ameliorated mucosal localized inflammation in n-3 PUFA intervention
CD rats. However, no difference in NFAT expression was observed
between healthy rats with or without n-3 PUFA, which indicates that
factors other than n-3 PUFA and PPAR-γ may be involved in the
modulation of NFAT expression, and further investigation is
required.
TFAs are steric isomers of cis unsaturated fatty
acids with distinct differences in biological effects. TFAs are
primarily present in dairy products and meats, and are generated
during the manufacturing process. Western-style diets usually
contain high levels of TFAs (39).
Recently, a number of reports have demonstrated that TFA intake is
a risk factor for diabetes mellitus, coronary heart disease and
hyperlipemia (40,41). The present study demonstrated that
TFA intake enhanced the risk of hypertriglyceridemia in CD rats,
which was consistent with the results of a previous study (42). However, no differences in blood fat
levels were observed in healthy rats despite the different diets,
which indicated an association between intestinal inflammation and
susceptibility to blood lipid disorders. Furthermore, the results
of the current study revealed that a TFA-enriched diet enhanced the
serum levels of IL-6 and TNF-α, exacerbated mucosal localized
inflammation determined by pathological observation and increased
the gene expression of IL-2 and IL-4. However, TFA exhibited no
effect on the gene expression of PPAR-γ and NFAT, indicating that
TFA may worsen inflammatory in CD rats via pathways other than
PPAR-γ/NFAT.
In conclusion, the present study demonstrated a
potent anti-inflammatory effect of dietary n-3 PUFA in experimental
CD rats, and the protective effect of this fatty acid may involve
the activation of PPAR-γ and inhibition of NFAT. Therefore, an n-3
PUFA-enriched diet, with few adverse side effects and established
mechanisms, may be a novel therapeutic approach for patients with
CD. However, further investigation is required. An improved
mechanistic understanding in combination with more comprehensive
data from well-designed clinical studies will provide the basis for
accelerating the development of nutrition-based therapies against
CD.
Acknowledgements
The present study was supported by grants from the
National Natural Science Foundation of China (grant no. 81370498)
and the Natural Science Foundation of Guangdong Province (grant no.
2011B031800097). The funders had no role in the study design, data
collection and analysis, decision to publish or preparation of the
manuscript. The authors thank Professor Yongfei Wu from the
Department of Neurology, Northwestern University Feinberg School of
Medicine (Chicago, IL, USA) for his assistance in improving the
language.
References
|
1
|
Abraham C and Cho JH: Inflammatory bowel
disease. N Engl J Med. 361:2066–2078. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Khor B, Gardet A and Xavier RJ: Genetics
and pathogenesis of inflammatory bowel disease. Nature.
474:307–317. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Asakura H, Suzuki K, Kitahora T and
Morizane T: Is there a link between food and intestinal microbes
and the occurrence of Crohn's disease and ulcerative colitis? J
Gastroenterol Hepatol. 23:1794–1801. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Thia KT, Loftus EV Jr, Sandborn WJ and
Yang SK: An update on the epidemiology of inflammatory bowel
disease in Asia. Am J Gastroenterol. 103:3167–3182. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Nakanishi S, Nagano C, Miyahara M and
Sawano F: Sex differences in the association between the
eicosapentaenoic acid/arachidonic acid ratio and the visceral fat
area among patients with type 2 diabetes. Intern Med. 55:1269–1274.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Borgeraas H, Hertel JK, Seifert R, Berge
RK, Bohov P, Ueland PM, Nygård O and Hjelmesæth J: Serum trans
fatty acids, asymmetric dimethylarginine and risk of acute
myocardial infarction and mortality in patients with suspected
coronary heart disease: A prospective cohort study. Lipids Health
Dis. 15:382016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ananthakrishnan AN, Khalili H, Konijeti
GG, Higuchi LM, de Silva P, Fuchs CS, Willett WC, Richter JM and
Chan AT: Long-term intake of dietary fat and risk of ulcerative
colitis and Crohn's disease. Gut. 63:776–784. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Marion-Letellier R, Savoye G, Beck PL,
Panaccione R and Ghosh S: Polyunsaturated fatty acids in
inflammatory bowel diseases: A reappraisal of effects and
therapeutic approaches. Inflamm Bowel Dis. 19:650–661. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yang Y, Lu N, Chen D, Meng L, Zheng Y and
Hui R: Effects of n-3 PUFA supplementation on plasma soluble
adhesion molecules: A meta-analysis of randomized controlled
trials. Am J Clin Nutr. 95:972–980. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Calder PC: Mechanisms of action of (n-3)
fatty acids. J Nutr. 142 Suppl:592S–599S. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Adachi M, Kurotani R, Morimura K, Shah Y,
Sanford M, Madison BB, Gumucio DL, Marin HE, Peters JM, Young HA
and Gonzalez FJ: Peroxisome proliferator activated receptor gamma
in colonic epithelial cells protects against experimental
inflammatory bowel disease. Gut. 55:1104–1113. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Su CG, Wen X, Bailey ST, Jiang W, Rangwala
SM, Keilbaugh SA, Flanigan A, Murthy S, Lazar MA and Wu GD: A novel
therapy for colitis utilizing PPAR-gamma ligands to inhibit the
epithelial inflammatory response. J Clin Invest. 104:383–389. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Bassaganya-Riera J, Viladomiu M, Pedragosa
M, De Simone C and Hontecillas R: Immunoregulatory mechanisms
underlying prevention of colitis-associated colorectal cancer by
probiotic bacteria. PLoS One. 7:e346762012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zeng C, Xiao JH, Chang MJ and Wang JL:
Beneficial effects of THSG on acetic acid-induced experimental
colitis: Involvement of upregulation of PPAR-γ and inhibition of
the Nf-Kb inflammatory pathway. Molecules. 16:8552–8568. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Hontecillas R, Horne WT, Climent M, Guri
AJ, Evans C, Zhang Y, Sobral BW and Bassaganya-Riera J:
Immunoregulatory mechanisms of macrophage PPAR-γ in mice with
experimental inflammatory bowel disease. Mucosal Immunol.
4:304–313. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Nakajima A, Wada K, Miki H, Kubota N,
Nakajima N, Terauchi Y, Ohnishi S, Saubermann LJ, Kadowaki T,
Blumberg RS, et al: Endogenous PPAR gamma mediates
anti-inflammatory activity in murine ischemia-reperfusion injury.
Gastroenterology. 120:460–469. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Raman P, Kaplan BL, Thompson JT, Heuvel JP
Vanden and Kaminski NE: 15-Deoxy-delta12,14-prostaglandin
J2-glycerol ester, a putative metabolite of 2-arachidonyl glycerol,
activates peroxisome proliferator activated receptor gamma. Mol
Pharmacol. 80:201–209. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Chung SW, Kang BY and Kim TS: Inhibition
of interleukin-4 production in CD4+ T cells by
peroxisome proliferator-activated receptor-gamma (PPAR-gamma)
ligands: Involvement of physical association between PPAR-gamma and
the nuclear factor of activated T cells transcription factor. Mol
Pharmacol. 64:1169–1179. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
National Research Council, . Guide For The
Care And Use Of Laboratory Animals. No. 85-23. National Academies
Press; Washington, DC: 1996
|
|
20
|
Kilkenny C, Browne WJ, Cuthill IC, Emerson
M and Altman DG: Improving bioscience research reporting: The
ARRIVE guidelines for reporting animal research. PLoS Biol.
8:e10004122010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Mizoguchi A: Animal models of inflammatory
bowel disease. Prog Mol Biol Transl Sci. 105:263–320. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wei W: Pharmacokinetics and
bioavailability of drug metabolismExperimental Methodology of
Pharmacology. 6th edition. People's Medical Publishing Company;
Guangzhou: 2010, View Article : Google Scholar
|
|
23
|
Ito R, Shin-Ya M, Kishida T, Urano A,
Takada R, Sakagami J, Imanishi J, Kita M, Ueda Y, Iwakura Y, et al:
Interferon-gamma is causatively involved in experimental
inflammatory bowel disease in mice. Clin Exp Immunol. 146:330–338.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Yao JY, Lu Y, Zhi M, Li CJ, Hu PJ and Gao
X: Inhibition of the interleukin-23/interleukin-17 pathway by
anti-interleukin-23p19 monoclonal antibody attenuates
2,4,6-trinitrobenzene sulfonic acid-induced Crohn's disease in
rats. Mol Med Rep. 10:2105–2110. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Ding H, Gan HZ, Fan WJ, Cao LY, Xu JM and
Mei Q: Homocysteine promotes intestinal fibrosis in rats with
trinitrobenzene sulfonic acid-induced colitis. Dig Dis Sci.
60:375–381. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zingarelli B, Hake PW, Burroughs TJ,
Piraino G, O'Connor M and Denenberg A: Activator protein-1
signalling pathway and apoptosis are modulated by poly(ADP-ribose)
polymerase-1 in experimental colitis. Immunology. 113:509–517.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Deepak P and Loftus EV Jr: Ustekinumab in
treatment of Crohn's disease: Design, development, and potential
place in therapy. Drug Des Devel Ther. 10:3685–3698. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Bassaganya-Riera J, Reynolds K,
Martino-Catt S, Cui Y, Hennighausen L, Gonzalez F, Rohrer J,
Benninghoff AU and Hontecillas R: Activation of PPAR gamma and
delta by conjugated linoleic acid mediates protection from
experimental inflammatory bowel disease. Gastroenterology.
127:777–791. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Viladomiu M, Hontecillas R, Yuan L, Lu P
and Bassaganya-Riera J: Nutritional protective mechanisms against
gut inflammation. J Nutr Biochem. 24:929–939. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Borniquel S, Jädert C and Lundberg JO:
Dietary conjugated linoleic acid activates PPARγ and the intestinal
trefoil factor in SW480 cells and mice with dextran sulfate
sodium-induced colitis. J Nutr. 142:2135–2140. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Bassaganya-Riera J, DiGuardo M, Viladomiu
M, de Horna A, Sanchez S, Einerhand AW, Sanders L and Hontecillas
R: Soluble fibers and resistant starch ameliorate disease activity
in interleukin-10-deficient mice with inflammatory bowel disease. J
Nutr. 141:1318–1325. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Lian M, Luo W, Sui Y, Li Z and Hua J:
Dietary n-3 PUFA protects mice from con A induced liver injury by
modulating regulatory T cells and PPAR-γ expression. PLoS One.
10:e01327412015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Liu J, Zhou F, Chen Q, Kang A, Lu M, Liu
W, Zang X, Wang G and Zhang J: Chronic inflammation up-regulates
P-gp in peripheral mononuclear blood cells via the STAT3/Nf-κb
pathway in 2,4,6-trinitrobenzene sulfonic acid-induced colitis
mice. Sci Rep. 5:135582015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Lytle C, Tod TJ, Vo KT, Lee JW, Atkinson
RD and Straus DS: The peroxisome proliferator-activated receptor
gamma ligand rosiglitazone delays the onset of inflammatory bowel
disease in mice with interleukin 10 deficiency. Inflamm Bowel Dis.
11:231–243. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Speca S, Rousseaux C, Dubuquoy C, Rieder
F, Vetuschi A, Sferra R, Giusti I, Bertin B, Dubuquoy L, Gaudio E,
et al: Novel PPARγ modulator GED-0507-34 levo ameliorates
inflammation-driven intestinal fibrosis. Inflamm Bowel Dis.
22:279–292. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Kundu P, Ling TW, Korecka A, Li Y,
D'Arienzo R, Bunte RM, Berger T, Arulampalam V, Chambon P, Mak TW,
et al: Absence of intestinal PPARγ aggravates acute infectious
colitis in mice through a lipocalin-2-dependent pathway. PLoS
Pathog. 10:e10038872014. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Hwang D: Fatty acids and immune
responses-a new perspective in searching for clues to mechanism.
Annu Rev Nutr. 20:431–456. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Kim HB, Kumar A, Wang L, Liu GH, Keller
SR, Lawrence JC Jr, Finck BN and Harris TE: Lipin 1 represses
NFATc4 transcriptional activity in adipocytes to inhibit secretion
of inflammatory factors. Mol Cell Biol. 30:3126–3139. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Mozaffarian D, Katan MB, Ascherio A,
Stampfer MJ and Willett WC: Trans fatty acids and cardiovascular
disease. N Engl J Med. 354:1601–1613. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Micha R and Mozaffarian D: Trans fatty
acids: Effects on cardiometabolic health and implications for
policy. Prostaglandins Leukot Essent Fatty Acids. 79:147–152. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Ganguly R and Pierce GN: Trans fat
involvement in cardiovascular disease. Mol Nutr Food Res.
56:1090–1096. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Okada Y, Tsuzuki Y, Ueda T, Hozumi H, Sato
S, Hokari R, Kurihara C, Watanabe C, Tomita K, Komoto S, et al:
Trans fatty acids in diets act as a precipitating factor for gut
inflammation? J Gastroenterol Hepatol. 28(Suppl 4): S29–S32. 2013.
View Article : Google Scholar
|