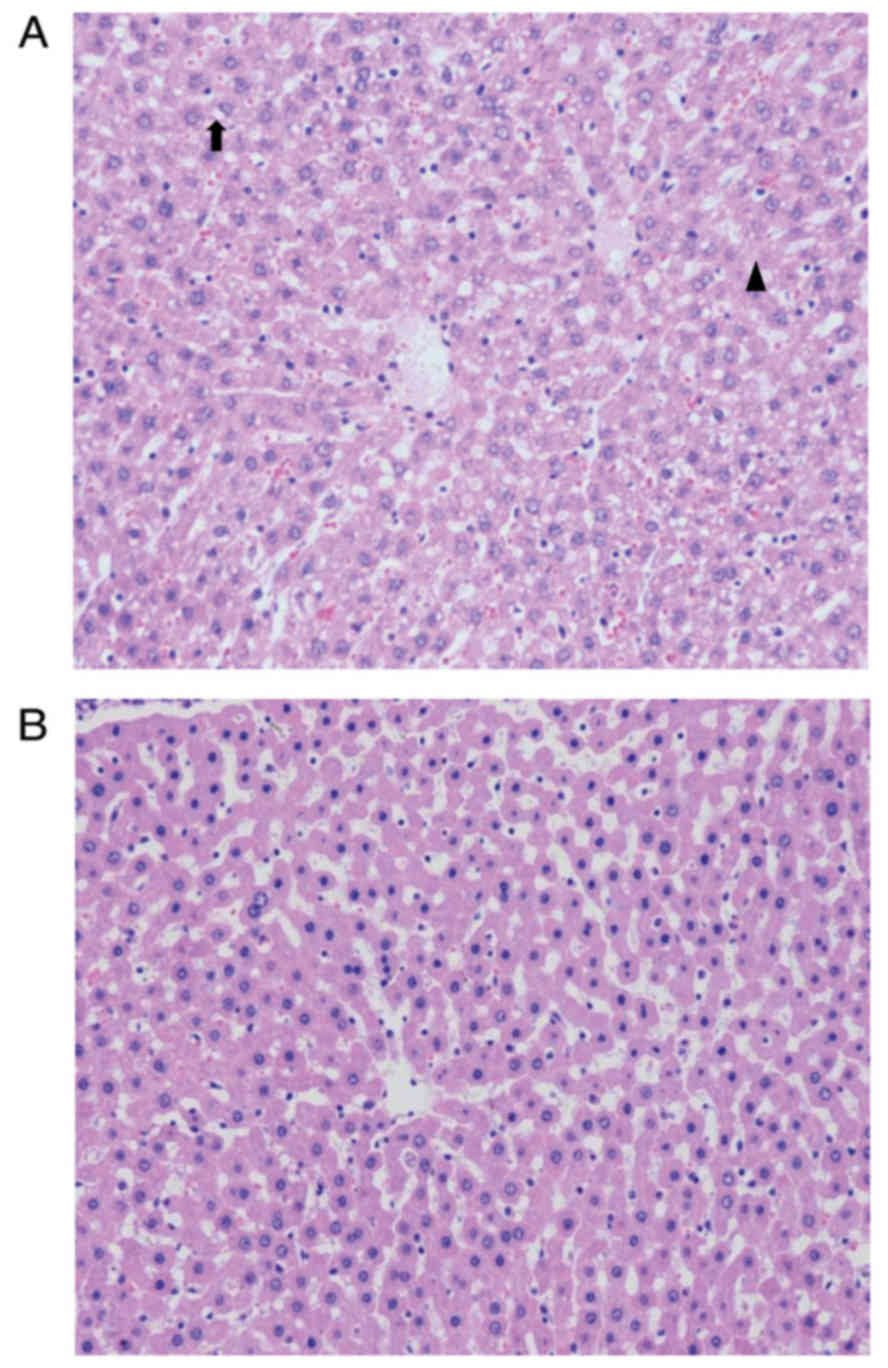
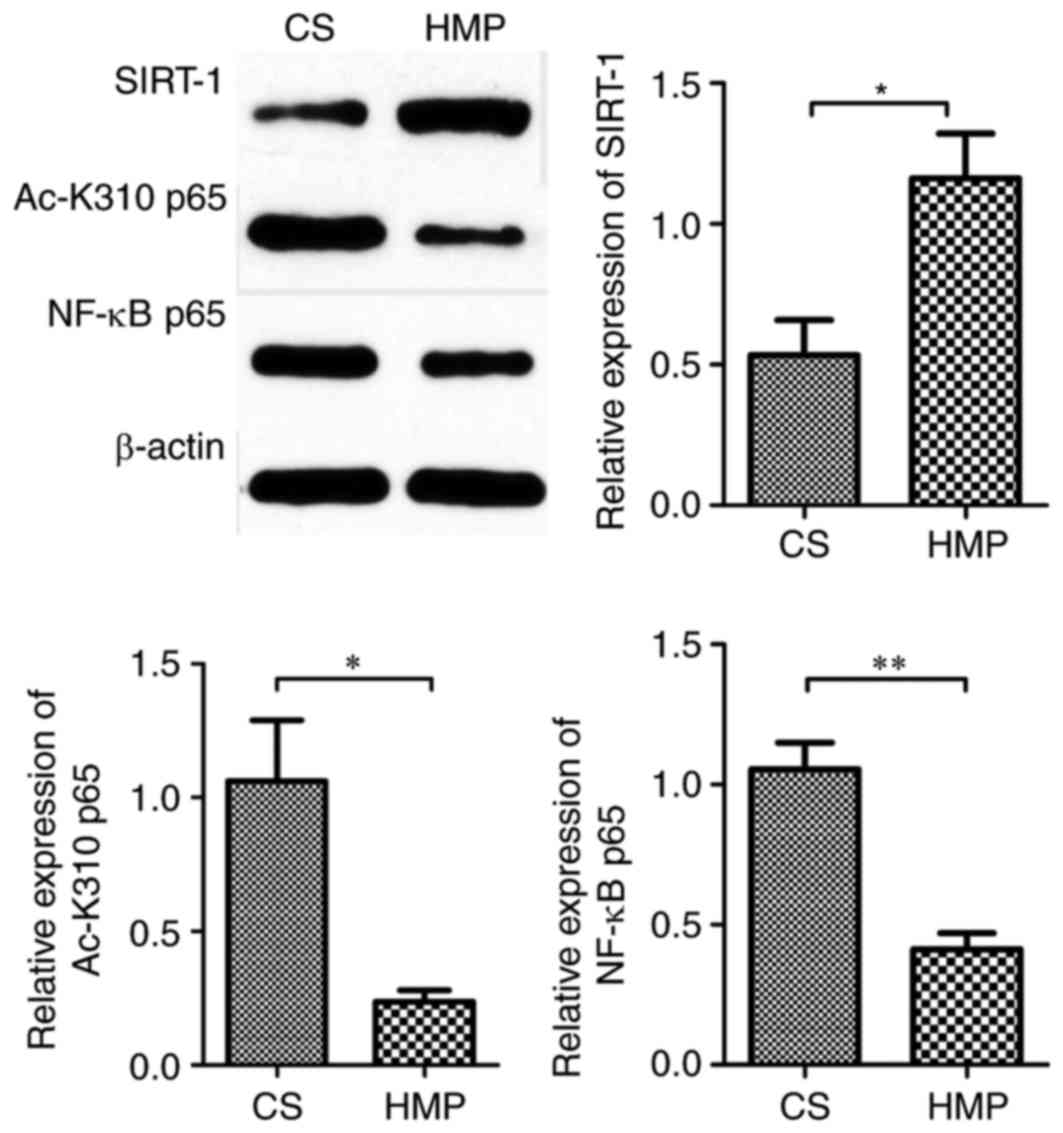
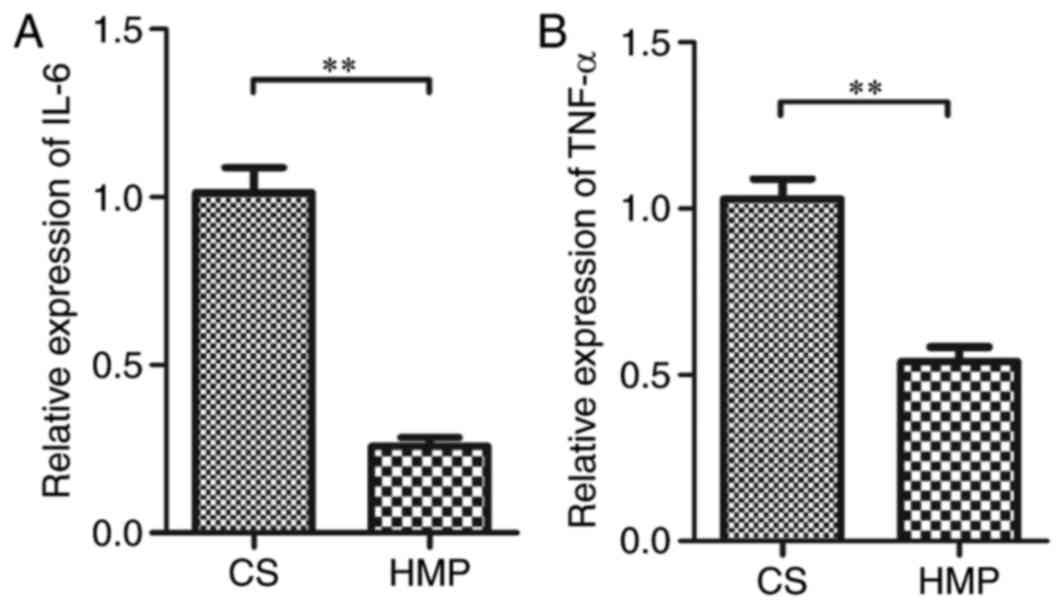
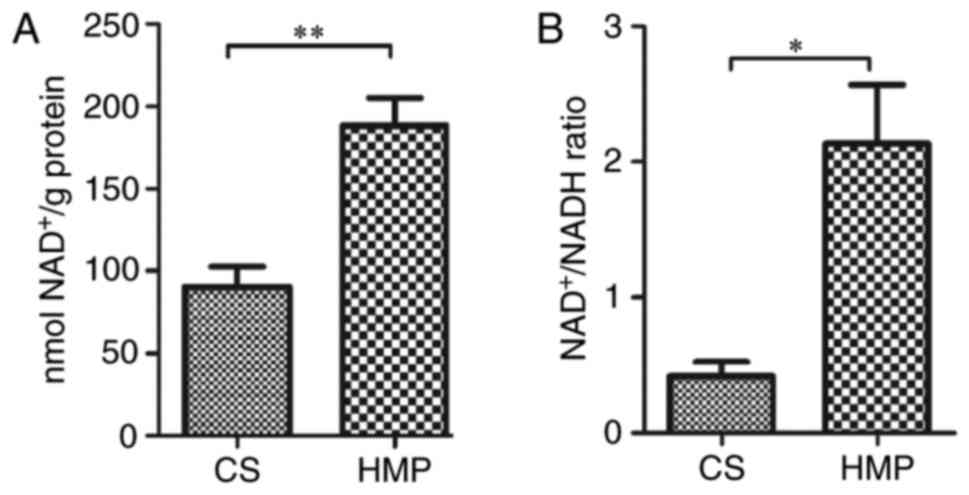
|
1
|
Blok JJ, Detry O, Putter H, Rogiers X,
Porte RJ, van Hoek B, Pirenne J, Metselaar HJ, Lerut JP, Ysebaert
DK, et al: Longterm results of liver transplantation from donation
after circulatory death. Liver Transpl. 22:1107–1114. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Perico N, Cattaneo D, Sayegh MH and
Remuzzi G: Delayed graft function in kidney transplantation.
Lancet. 364:1814–1827. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Brasile L, Stubenitsky BM, Booster MH,
Arenada D, Haisch C and Kootstra G: Hypothermia a limiting factor
in using warm ischemically damaged kidneys. Am J Transplant.
1:316–320. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Henry SD, Nachber E, Tulipan J, Stone J,
Bae C, Reznik L, Kato T, Samstein B, Emond JC and Guarrera JV:
Hypothermic machine preservation reduces molecular markers of
ischemia/reperfusion injury in human liver transplantation. Am J
Transplant. 12:2477–2486. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Carnevale ME, Balaban CL, Guibert EE,
Bottai H and Rodriguez JV: Hypothermic machine perfusion versus
cold storage in the rescuing of livers from non-heart-beating donor
rats. Artif Organs. 37:985–991. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Guarrera JV, Henry SD, Samstein B,
Odeh-Ramadan R, Kinkhabwala M, Goldstein MJ, Ratner LE, Renz JF,
Lee HT, Brown RS Jr and Emond JC: Hypothermic machine preservation
in human liver transplantation: The first clinical series. Am J
Transplant. 10:372–381. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Taylor MJ and Baicu SC: Current state of
hypothermic machine perfusion preservation of organs: The clinical
perspective. Cryobiology. 60(3 Suppl): S20–S35. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Arnaudo AM and Garcia BA: Proteomic
characterization of novel histone post-translational modifications.
Epigenetics Chromatin. 6:242013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Chen LF and Greene WC: Assessing
acetylation of NF-kappaB. Methods. 36:368–375. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Huang B, Yang XD, Lamb A and Chen LF:
Posttranslational modifications of NFkappaB: Another layer of
regulation for NF-kappaB signaling pathway. Cell Signal.
22:1282–1290. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Chen LF, Mu Y and Greene WC: Acetylation
of RelA at discrete sites regulates distinct nuclear functions of
NF-kappaB. EMBO J. 21:6539–6548. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Sun T, Luo J, Jia M, Li H, Li K and Fu Z:
Small interfering RNA-mediated knockdown of NF-κBp65 attenuates
neuropathic pain following peripheral nerve injury in rats. Eur J
Pharmacol. 682:79–85. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ramachandran S, Liaw JM, Jia J, Glasgow
SC, Liu W, Csontos K, Upadhya GA, Mohanakumar T and Chapman WC:
Ischemia-reperfusion injury in rat steatotic liver is dependent on
NFκB P65 activation. Transpl Immunol. 26:201–206. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Andrade-Silva AR, Ramalho FS, Ramalho LN,
Saavedra-Lopes M, Jordão AA Jr, Vanucchi H, Piccinato CE and
Zucoloto S: Effect of NFkappaB inhibition by CAPE on skeletal
muscle ischemia-reperfusion injury. J Surg Res. 153:254–262. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Suetsugu H, Iimuro Y, Uehara T, Nishio T,
Harada N, Yoshida M, Hatano E, Son G, Fujimoto J and Yamaoka Y:
Nuclear factor {kappa}B inactivation in the rat liver ameliorates
short term total warm ischaemia/reperfusion injury. Gut.
54:835–842. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhang C, Li X and Liu Q: Sorbitol
dehydrogenase inhibitor protects the liver from
ischemia/reperfusion-induced injury via elevated glycolytic flux
and enhanced sirtuin 1 activity. Mol Med Rep. 11:283–288. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yan H, Jihong Y, Feng Z, Xiaomei X,
Xiaohan Z, Guangzhi W, Zhenhai M, Dongyan G, Xiaochi M, Qing F, et
al: Sirtuin 1-mediated inhibition of p66shc expression alleviates
liver ischemia/reperfusion injury. Crit Care Med. 42:e373–e381.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Pantazi E, Zaouali MA, Bejaoui M, Serafin
A, Folch-Puy E, Petegnief V, De Vera N, Ben Abdennebi H, Rimola A
and Roselló-Catafau J: Silent information regulator 1 protects the
liver against ischemia-reperfusion injury: Implications in
steatotic liver ischemic preconditioning. Transpl Int. 27:493–503.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Yeung F, Hoberg JE, Ramsey CS, Keller MD,
Jones DR, Frye RA and Mayo MW: Modulation of NF-kappaB-dependent
transcription and cell survival by the SIRT1 deacetylase. EMBO J.
23:2369–2380. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Institute of laboratory animal resources
(US), . Committee on care, use of laboratory animals and national
institutes of health (US)Division of research resources: Guide for
the care and use of laboratory animals. 8th edition. National
academies press; Washington, DC: 2011
|
|
21
|
Pizarro MD, Rodriguez JV, Mamprin ME,
Fuller BJ, Mann BE, Motterlini R and Guibert EE: Protective effects
of a carbon monoxide-releasing molecule (CORM-3) during hepatic
cold preservation. Cryobiology. 58:248–255. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Balaban CL, Rodriguez JV and Guibert EE:
Delivery of the bioactive gas hydrogen sulfide during cold
preservation of rat liver: Effects on hepatic function in an ex
vivo model. Artif Organs. 35:508–515. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Schlegel A, Kron P, Graf R, Dutkowski P
and Clavien PA: Warm vs. cold perfusion techniques to rescue rodent
liver grafts. J Hepatol. 61:1267–1275. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kamada N and Calne RY: A surgical
experience with five hundred thirty liver transplants in the rat.
Surgery. 93:64–69. 1983.PubMed/NCBI
|
|
25
|
Minor T, Manekeller S, Sioutis M and
Dombrowski F: Endoplasmic and vascular surface activation during
organ preservation: Refining upon the benefits of machine
perfusion. Am J Transplant. 6:1355–1366. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Minor T and Manekeller S: Assessment of
hepatic integrity after ischemic preservation by isolated perfusion
in vitro: The role of albumin. Cryobiology. 54:188–195. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Khandoga A, Biberthaler P, Enders G and
Krombach F: 5-Aminoisoquinolinone, a novel inhibitor of
poly(adenosine disphosphate-ribose) polymerase, reduces
microvascular liver injury but not mortality rate after hepatic
ischemia-reperfusion. Crit Care Med. 32:472–477. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Lentsch AB, Kato A, Yoshidome H, McMasters
KM and Edwards MJ: Inflammatory mechanisms and therapeutic
strategies for warm hepatic ischemia/reperfusion injury.
Hepatology. 32:169–173. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Yamamoto T, Ono T, Ito T, Yamanoi A,
Maruyama I and Tanaka T: Hemoperfusion with a high-mobility group
box 1 adsorption column can prevent the occurrence of hepatic
ischemia-reperfusion injury in rats. Crit Care Med. 38:879–885.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Peralta C, Jiménez-Castro MB and
Gracia-Sancho J: Hepatic ischemia and reperfusion injury: Effects
on the liver sinusoidal milieu. J Hepatol. 59:1094–1106. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Jia JJ, Zhang J, Li JH, Chen XD, Jiang L,
Zhou YF, He N, Xie HY, Zhou L and Zheng SS: Influence of perfusate
on liver viability during hypothermic machine perfusion. World J
Gastroenterol. 21:8848–8857. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Vekemans K, Liu Q, Brassil J, Komuta M,
Pirenne J and Monbaliu D: Influence of flow and addition of oxygen
during porcine liver hypothermic machine perfusion. Transplant
Proc. 39:pp. 2647–2651. 2007; View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Pamukcu B, Lip GY and Shantsila E: The
nuclear factor-kappa B pathway in atherosclerosis: A potential
therapeutic target for atherothrombotic vascular disease. Thromb
Res. 128:117–123. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Lin SJ, Ford E, Haigis M, Liszt G and
Guarente L: Calorie restriction extends yeast life span by lowering
the level of NADH. Genes Dev. 18:12–16. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Vaziri H, Dessain SK, Ng Eaton E, Imai SI,
Frye RA, Pandita TK, Guarente L and Weinberg RA: hSIR2(SIRT1)
functions as an NAD-dependent p53 deacetylase. Cell. 107:149–159.
2001. View Article : Google Scholar : PubMed/NCBI
|