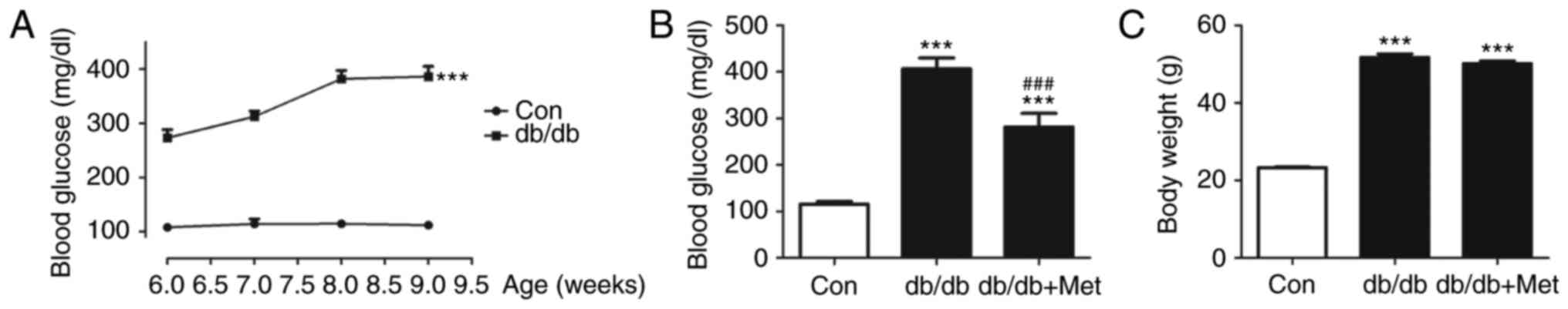
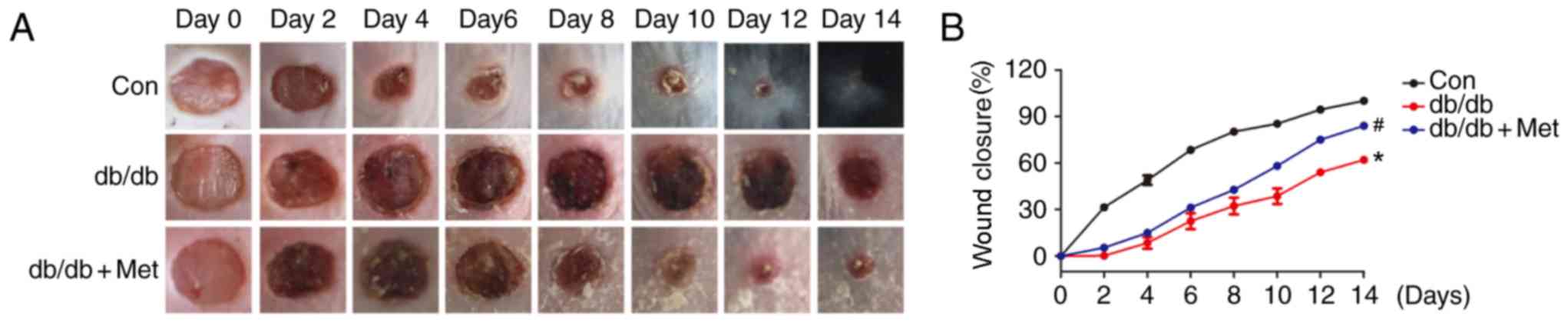
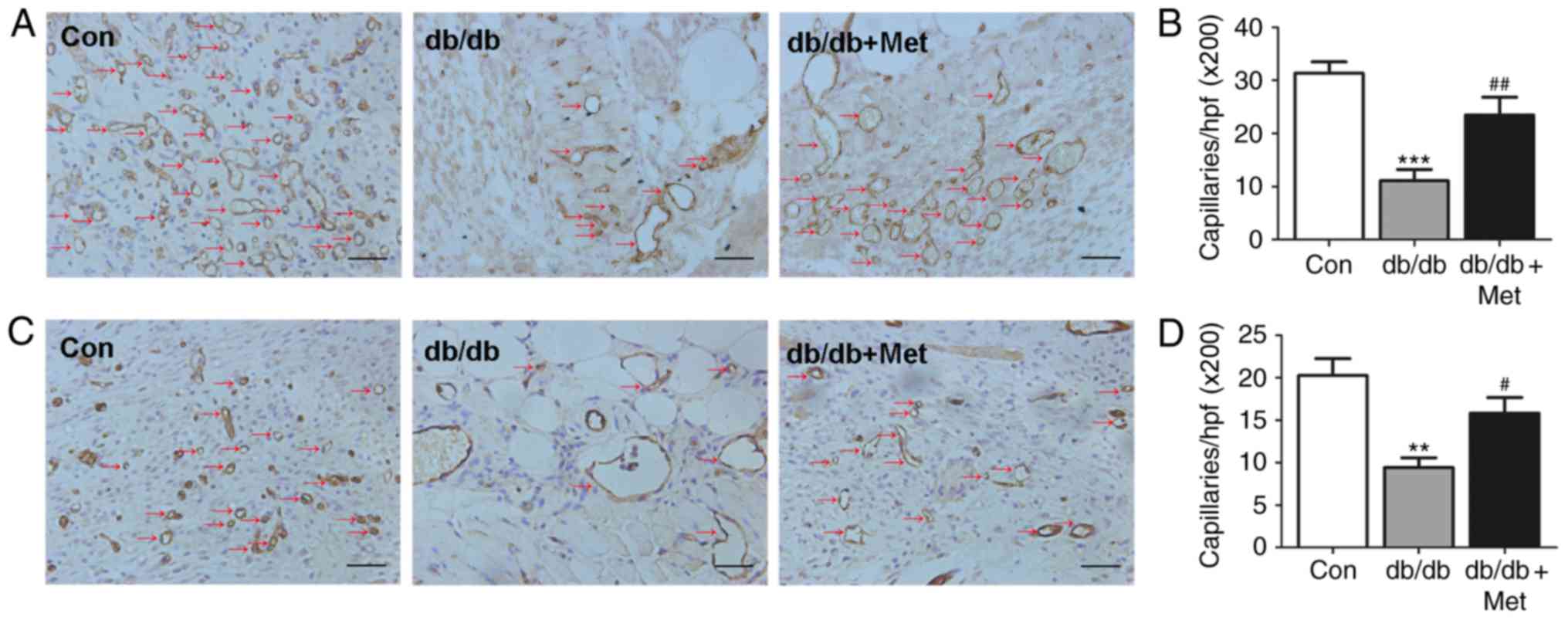
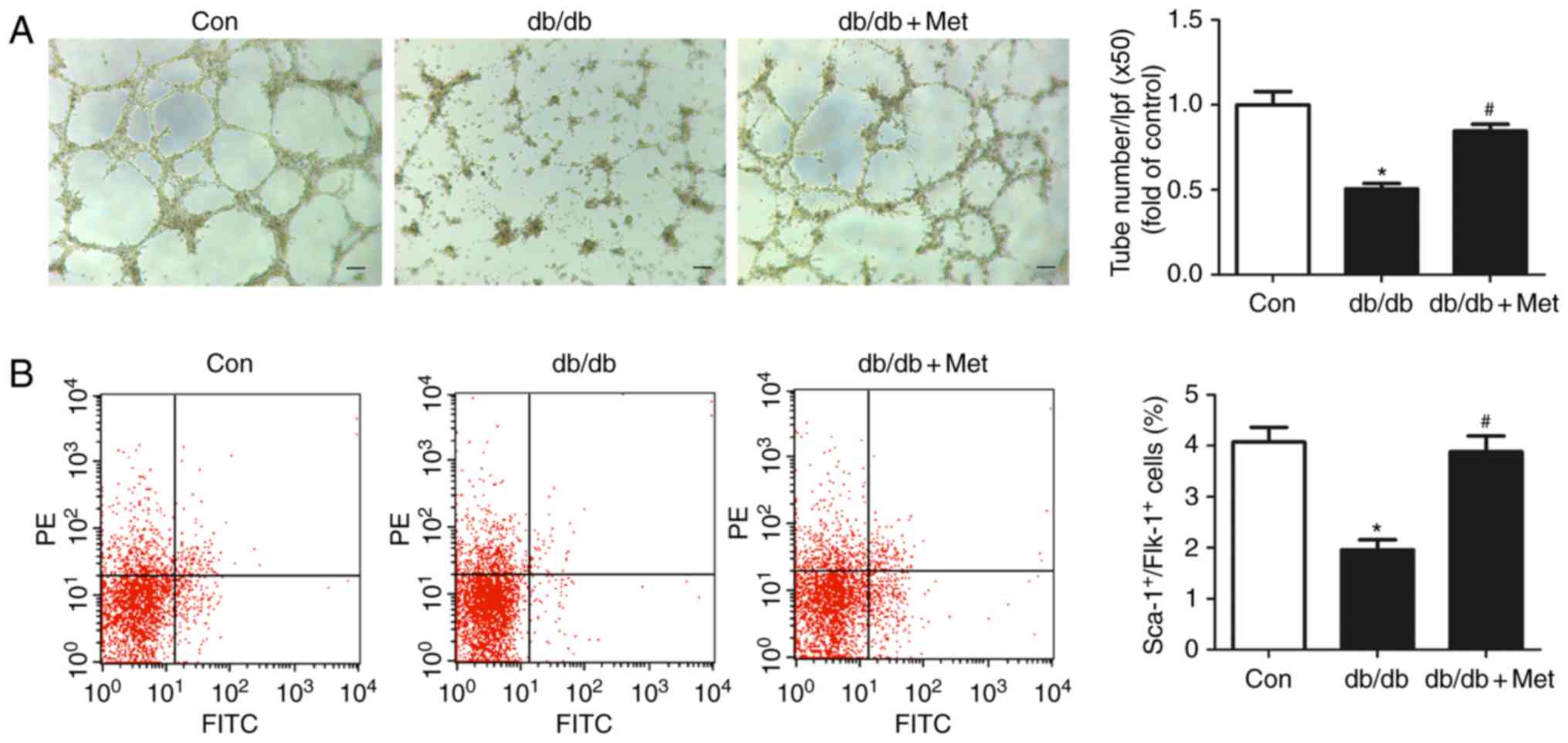
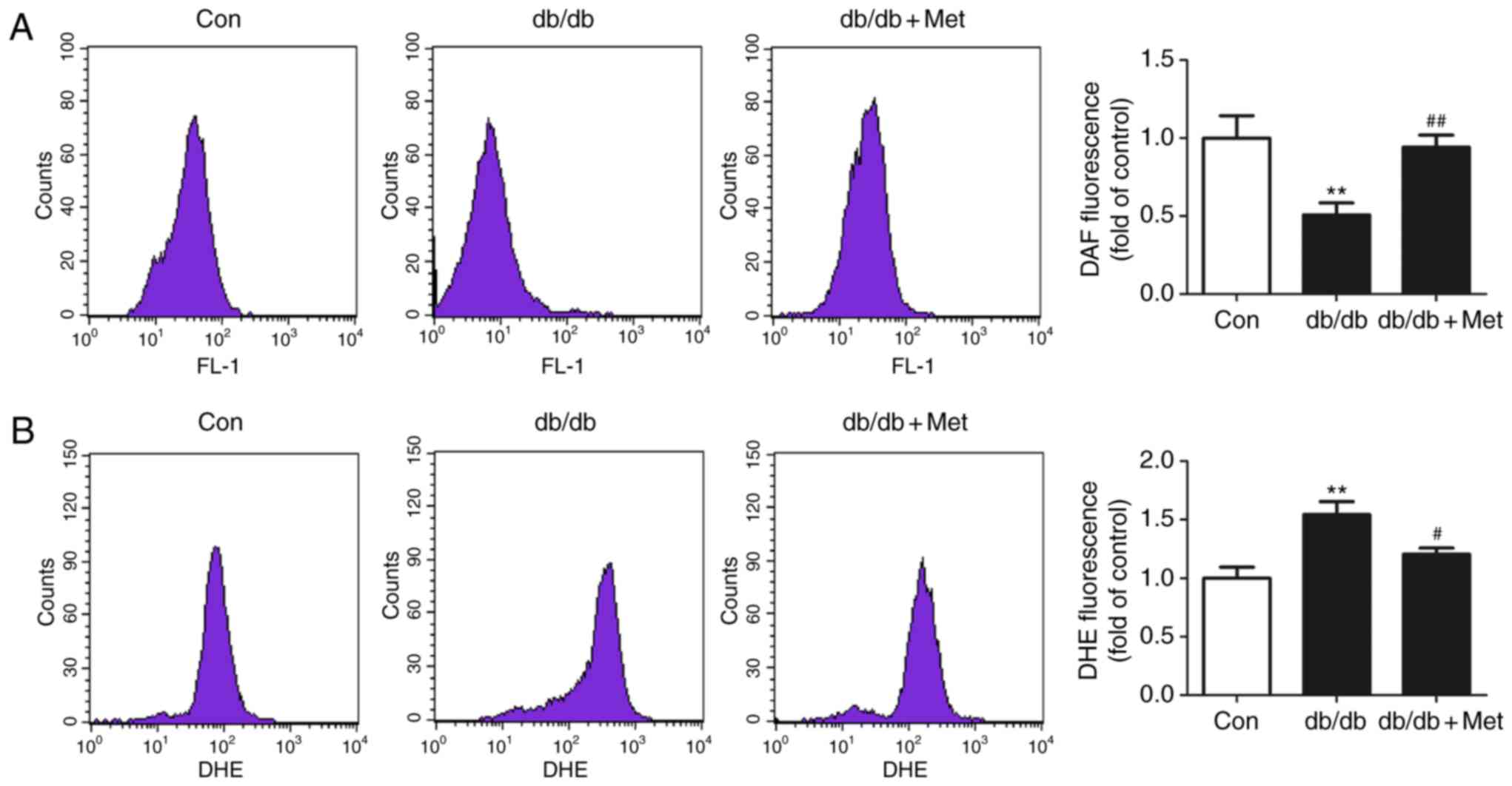
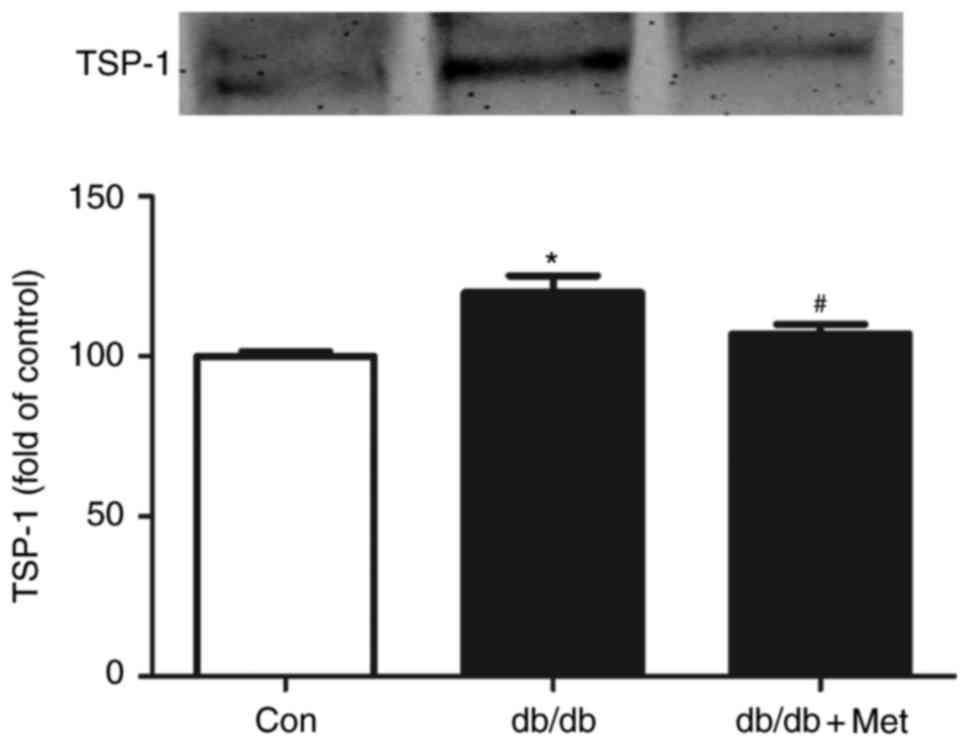
|
1
|
Chan JC, Cho NH, Tajima N and Shaw J:
Diabetes in the Western Pacific Region-past, present and future.
Diabetes Res Clin Pract. 103:244–255. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Thandavarayan RA, Garikipati VN,
Joladarashi D, Babu S Suresh, Jeyabal P, Verma SK, Mackie AR, Khan
M, Arumugam S, Watanabe K, et al: Sirtuin-6 deficiency exacerbates
diabetes-induced impairment of wound healing. Exp Dermatol.
24:773–778. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Papanas N, Demetzos C, Pippa N, Maltezos E
and Tentolouris N: Efficacy of a new heparan sulfate mimetic
dressing in the healing of foot and lower extremity ulcerations in
type 2 diabetes: A case series. Int J Low Extrem Wounds. 15:63–67.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zgheib C and Liechty KW: Shedding light on
miR-26a: Another key regulator of angiogenesis in diabetic wound
healing. J Mol Cell Cardiol. 92:203–205. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Li DW, Liu ZQ, Wei J, Liu Y and Hu LS:
Contribution of endothelial progenitor cells to neovascularization
(Review). Int J Mol Med. 30:1000–1006. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Fadini GP, Sartore S, Albiero M, Baesso I,
Murphy E, Menegolo M, Grego F, de Kreutzenberg S Vigili, Tiengo A,
Agostini C and Avogaro A: Number and function of endothelial
progenitor cells as a marker of severity for diabetic vasculopathy.
Arterioscler Thromb Vasc Biol. 26:2140–2146. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kovacic JC, Moore J, Herbert A, Ma D,
Boehm M and Graham RM: Endothelial progenitor cells, angioblasts,
and angiogenesis-old terms reconsidered from a current perspective.
Trends Cardiovasc Med. 18:45–51. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Liao YF, Chen LL, Zeng TS, Li YM, Fan Yu,
Hu LJ and Ling Yue: Number of circulating endothelial progenitor
cells as a marker of vascular endothelial function for type 2
diabetes. Vasc Med. 15:279–285. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Tepper OM: Human endothelial progenitor
cells from type II diabetics exhibit impaired proliferation,
adhesion, and incorporation into vascular structures. Circulation.
106:2781–2786. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wang CH, Ting MK, Verma S, Kuo LT, Yang
NI, Hsieh IC, Wang SY, Hung A and Cherng WJ: Pioglitazone increases
the numbers and improves the functional capacity of endothelial
progenitor cells in patients with diabetes mellitus. Am Heart J.
152:1051.e1–8. 2006. View Article : Google Scholar
|
|
11
|
Fadini GP, Miorin M, Facco M, Bonamico S,
Baesso I, Grego F, Menegolo M, de Kreutzenberg SV, Tiengo A,
Agostini C and Avogaro A: Circulating endothelial progenitor cells
are reduced in peripheral vascular complications of type 2 diabetes
mellitus. J Am Coll Cardiol. 45:1449–1457. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Nakamura K, Oe H, Kihara H, Shimada K,
Fukuda S, Watanabe K, Takagi T, Yunoki K, Miyoshi T, Hirata K, et
al: DPP-4 inhibitor and alpha-glucosidase inhibitor equally improve
endothelial function in patients with type 2 diabetes: EDGE study.
Cardiovasc Diabetol. 13:1102014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yue WS, Lau KK, Siu CW, Wang M, Yan GH,
Yiu KH and Tse HF: Impact of glycemic control on circulating
endothelial progenitor cells and arterial stiffness in patients
with type 2 diabetes mellitus. Cardiovasc Diabetol. 10:1132011.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Badr G, Hozzein WN, Badr BM, Al Ghamdi A,
Eldien HM Saad and Garraud O: Bee venom accelerates wound healing
in diabetic mice by suppressing activating transcription factor-3
(ATF-3) and inducible nitric oxide synthase (iNOS)-mediated
oxidative stress and recruiting bone marrow-derived endothelial
progenitor cells. J Cell Physiol. 231:2159–2171. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Gallagher KA, Goldstein LJ, Thom SR and
Velazquez OC: Hyperbaric oxygen and bone marrow-derived endothelial
progenitor cells in diabetic wound healing. Vascular. 14:328–337.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Gallagher KA, Liu ZJ, Xiao M, Chen H,
Goldstein LJ, Buerk DG, Nedeau A, Thom SR and Velazquez OC:
Diabetic impairments in NO-mediated endothelial progenitor cell
mobilization and homing are reversed by hyperoxia and SDF-1 alpha.
J Clin Invest. 117:1249–1259. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Hattori Y, Hattori K and Hayashi T:
Pleiotropic benefits of metformin: Macrophage targeting its
anti-inflammatory mechanisms. Diabetes. 64:1907–1909. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Li DJ, Huang F, Lu WJ, Jiang GJ, Deng YP
and Shen FM: Metformin promotes irisin release from murine skeletal
muscle independently of AMP-activated protein kinase activation.
Acta Physiol (Oxf). 213:711–721. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Chen LL, Liao YF, Zeng TS, Yu F, Li HQ and
Feng Y: Effects of metformin plus gliclazide compared with
metformin alone on circulating endothelial progenitor cell in type
2 diabetic patients. Endocrine. 38:266–275. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Varma V, Yao-Borengasser A, Bodles AM,
Rasouli N, Phanavanh B, Nolen GT, Kern EM, Nagarajan R, Spencer HJ
III, Lee MJ, et al: Thrombospondin-1 is an adipokine associated
with obesity, adipose inflammation, and insulin resistance.
Diabetes. 57:432–439. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Dabir P, Marinic TE, Krukovets I and
Stenina OI: Aryl hydrocarbon receptor is activated by glucose and
regulates the thrombospondin-1 gene promoter in endothelial cells.
Circ Res. 102:1558–1565. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Stenina OI, Krukovets I, Wang K, Zhou Z,
Forudi F, Penn MS, Topol EJ and Plow EF: Increased expression of
thrombospondin-1 in vessel wall of diabetic Zucker rat.
Circulation. 107:3209–3215. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Tie L, Chen LY, Chen DD, Xie HH, Channon
KM and Chen AF: GTP cyclohydrolase I prevents diabetic-impaired
endothelial progenitor cells and wound healing by suppressing
oxidative stress/thrombospondin-1. Am J Physiol Endocrinol Metab.
306:E1120–E1131. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Tan BK, Adya R, Chen J, Farhatullah S,
Heutling D, Mitchell D, Lehnert H and Randeva HS: Metformin
decreases angiogenesis via NF-kappaB and Erk1/2/Erk5 pathways by
increasing the antiangiogenic thrombospondin-1. Cardiovasc Res.
83:566–574. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Xie HH, Zhou S, Chen DD, Channon KM, Su DF
and Chen AF: GTP cyclohydrolase I/BH4 pathway protects EPCs via
suppressing oxidative stress and thrombospondin-1 in salt-sensitive
hypertension. Hypertension. 56:1137–1144. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Li ZP, Xin RJ, Yang H, Jiang GJ, Deng YP,
Li DJ and Shen FM: Diazoxide accelerates wound healing by improving
EPC function. Front Biosci (Landmark Ed). 21:1039–1051. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Cai J, Lu S, Yao Z, Deng YP, Zhang LD, Yu
JW, Ren GF, Shen FM and Jiang GJ: Glibenclamide attenuates
myocardial injury by lipopolysaccharides in streptozotocin-induced
diabetic mice. Cardiovasc Diabetol. 13:1062014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Marrotte EJ, Chen DD, Hakim JS and Chen
AF: Manganese superoxide dismutase expression in endothelial
progenitor cells accelerates wound healing in diabetic mice. J Clin
Invest. 120:4207–4219. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Chen JK, Deng YP, Jiang GJ, Liu YZ, Zhao T
and Shen FM: Establishment of tube formation assay of bone
marrow-derived endothelial progenitor cells. CNS Neurosci Ther.
19:533–535. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Lee MH, Choi EN, Jeon YJ and Jung SC:
Possible role of transforming growth factor-β1 and vascular
endothelial growth factor in Fabry disease nephropathy. Int J Mol
Med. 30:1275–1280. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Aoyama H, Daitoku H and Fukamizu A:
Nutrient control of phosphorylation and translocation of Foxo1 in
C57BL/6 and db/db mice. Int J Mol Med. 18:433–439. 2006.PubMed/NCBI
|
|
32
|
Bao Q, Shen X, Qian L, Gong C, Nie M and
Dong Y: Anti-diabetic activities of catalpol in db/db mice. Korean
J Physiol Pharmacol. 20:153–160. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Tamura Y, Murayama T, Minami M, Yokode M
and Arai H: Differential effect of statins on diabetic nephropathy
in db/db mice. Int J Mol Med. 28:683–687. 2011.PubMed/NCBI
|
|
34
|
Galiano RD, Tepper OM, Pelo CR, Bhatt KA,
Callaghan M, Bastidas N, Bunting S, Steinmetz HG and Gurtner GC:
Topical vascular endothelial growth factor accelerates diabetic
wound healing through increased angiogenesis and by mobilizing and
recruiting bone marrow-derived cells. Am J Pathol. 164:1935–1947.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Galeano M, Altavilla D, Cucinotta D, Russo
GT, Calò M, Bitto A, Marini H, Marini R, Adamo EB, Seminara P, et
al: Recombinant human erythropoietin stimulates angiogenesis and
wound healing in the genetically diabetic mouse. Diabetes.
53:2509–2517. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Zhang XN, Ma ZJ, Wang Y, Li YZ, Sun B, Guo
X, Pan CQ and Chen LM: The four-herb Chinese medicine formula
Tuo-Li-Xiao-Du-San accelerates cutaneous wound healing in
streptozotocin-induced diabetic rats through reducing inflammation
and increasing angiogenesis. J Diabetes Res. 2016:56391292016.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Caligiuri G, Groyer E, Khallou-Laschet J,
Al Haj Zen A, Sainz J, Urbain D, Gaston AT, Lemitre M, Nicoletti A
and Lafont A: Reduced immunoregulatory CD31+ T cells in the blood
of atherosclerotic mice with plaque thrombosis. Arterioscler Thromb
Vasc Biol. 25:1659–1664. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Tellechea A, Kafanas A, Leal EC,
Tecilazich F, Kuchibhotla S, Auster ME, Kontoes I, Paolino J,
Carvalho E, Nabzdyk LP and Veves A: Increased skin inflammation and
blood vessel density in human and experimental diabetes. Int J Low
Extrem Wounds. 12:4–11. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Desouza CV: Does drug therapy reverse
endothelial progenitor cell dysfunction in diabetes? J Diabetes
Complications. 27:519–525. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Lin JT, Chen HM, Chiu CH and Liang YJ:
AMP-activated protein kinase activators in diabetic ulcers: From
animal studies to Phase II drugs under investigation. Expert Opin
Investig Drugs. 23:1253–1265. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Ochoa-Gonzalez F, Cervantes-Villagrana AR,
Fernandez-Ruiz JC, Nava-Ramirez HS, Hernandez-Correa AC,
Enciso-Moreno JA and Castañeda-Delgado JE: Metformin induces cell
cycle arrest, reduced proliferation, wound healing impairment in
vivo and is associated to clinical outcomes in diabetic foot ulcer
patients. PLoS One. 11:e01509002016. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Mao L, Huang M, Chen SC, Li YN, Xia YP, He
QW, Wang MD, Huang Y, Zheng L and Hu B: Endogenous endothelial
progenitor cells participate in neovascularization via CXCR4/SDF-1
axis and improve outcome after stroke. CNS Neurosci Ther.
20:460–468. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Kim KA, Shin YJ, Kim JH, Lee H, Noh SY,
Jang SH and Bae ON: Dysfunction of endothelial progenitor cells
under diabetic conditions and its underlying mechanisms. Arch Pharm
Res. 35:223–234. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Ghosh S, Lakshmanan AP, Hwang MJ, Kubba H,
Mushannen A, Triggle CR and Ding H: Metformin improves endothelial
function in aortic tissue and microvascular endothelial cells
subjected to diabetic hyperglycaemic conditions. Biochem Pharmacol.
98:412–421. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Shi Y, He Z, Jia Z and Xu C: Inhibitory
effect of metformin combined with gemcitabine on pancreatic cancer
cells in vitro and in vivo. Mol Med Rep. 14:2921–2928. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Gao L, Li P, Zhang J, Hagiwara M, Shen B,
Bledsoe G, Chang E, Chao L and Chao J: Novel role of kallistatin in
vascular repair by promoting mobility, viability, and function of
endothelial progenitor cells. J Am Heart Assoc. 3:e0011942014.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Bhattacharyya S, Marinic TE, Krukovets I,
Hoppe G and Stenina OI: Cell type-specific post-transcriptional
regulation of production of the potent antiangiogenic and
proatherogenic protein thrombospondin-1 by high glucose. J Biol
Chem. 283:5699–5707. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Isenberg JS, Wink DA and Roberts DD:
Thrombospondin-1 antagonizes nitric oxide-stimulated vascular
smooth muscle cell responses. Cardiovasc Res. 71:785–793. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Ridnour LA, Isenberg JS, Espey MG, Thomas
DD, Roberts DD and Wink DA: Nitric oxide regulates angiogenesis
through a functional switch involving thrombospondin-1. Proc Natl
Acad Sci USA. 102:pp. 13147–13152. 2005; View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Xu L, Xun G, Yao Z, Liu Y, Qiu Y, Liu K,
Zhu D, Gu Q, Xu X and Ho PC: Effects of generated trans-arachidonic
acids on retinal capillary during nitrative stress in diabetic
rats. Ophthalmologica. 222:37–41. 2008. View Article : Google Scholar : PubMed/NCBI
|