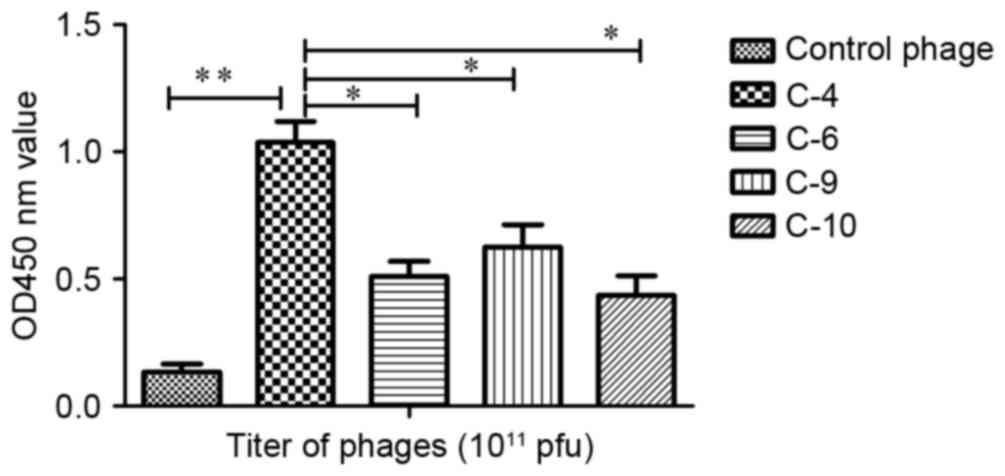
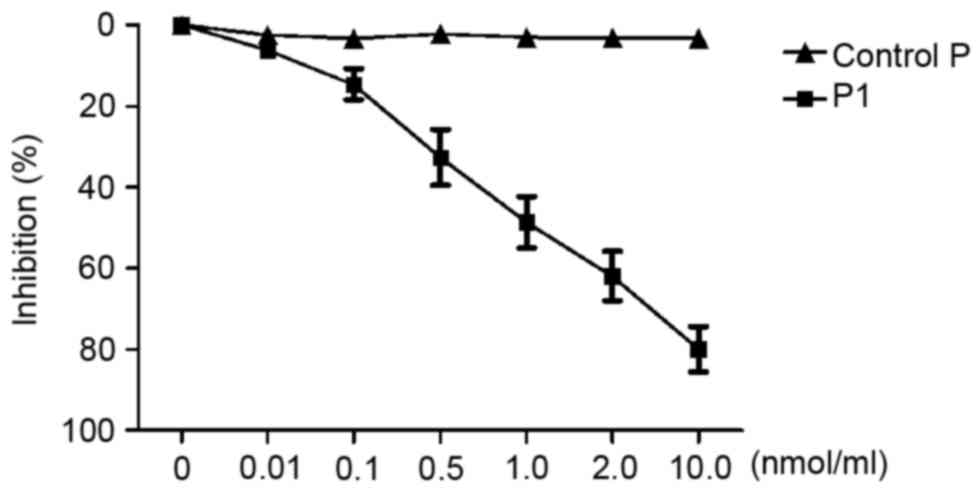
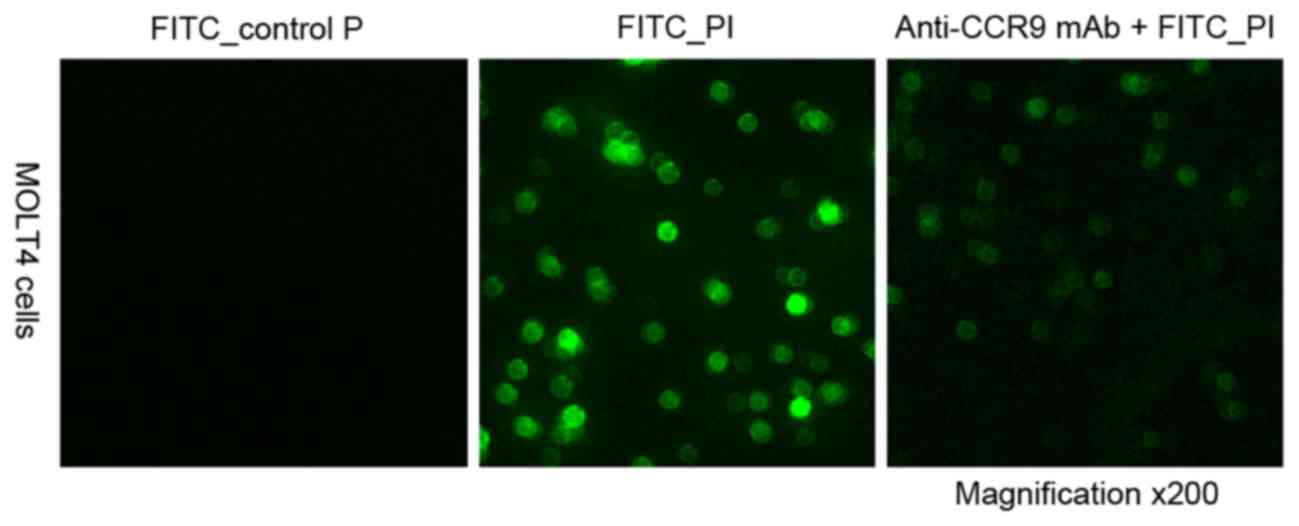
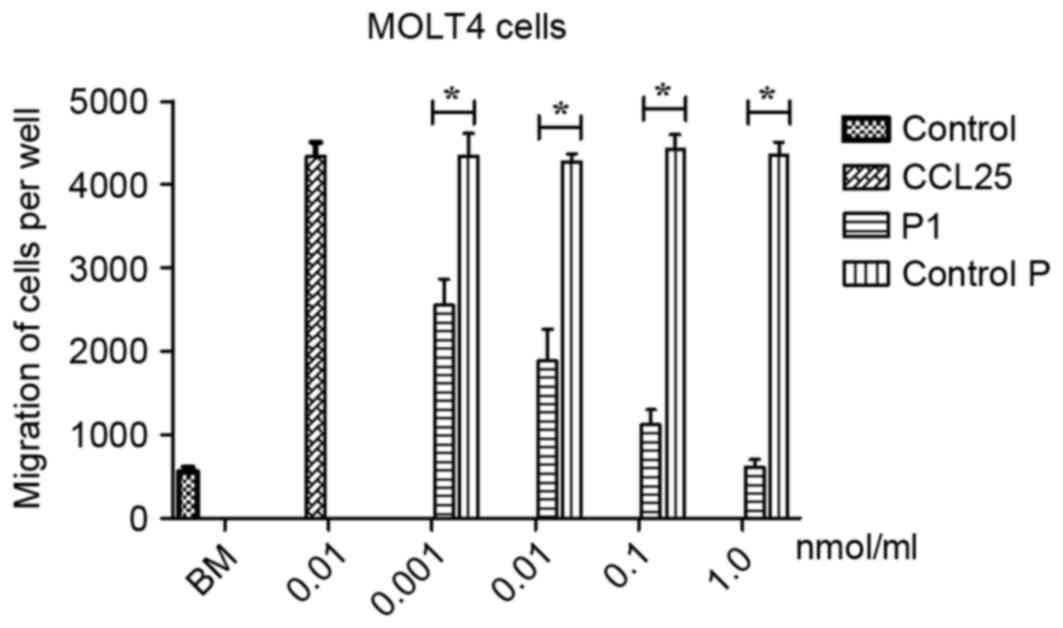
|
1
|
Zlotnik A and Yoshie O: Chemokines: A new
classification system and their role in immunity. Immunity.
12:121–127. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Nagakubo D, Jin Z, Hieshima K, Nakayama T,
Shirakawa AK, Tanaka Y, Hasegawa H, Hayashi T, Tsukasaki K, Yamada
Y, et al: Expression of CCR9 in HTLV-1+T cells and ATL cells
expressing tax. Int J Cancer. 120:1591–1597. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Heinrich EL, Arrington AK, Ko ME, Luu C,
Lee W, Lu J and Kim J: Paracrine activation of chemokine receptor
CCR9 enhances the invasiveness of pancreatic cancer cells. Cancer
Microenviron. 6:241–245. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Gupta P, Sharma PK, Mir H, Singh R, Singh
N, Kloecker GH, Lillard JW Jr and Singh S: CCR9/CCL25 expression in
non-small cell lung cancer correlates with aggressive disease and
mediates key steps of metastasis. Oncotarget. 5:10170–10179. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Sharma PK, Singh R, Novakovic KR, Eaton
JW, Grizzle WE and Singh S: CCR9 mediates PI3K/AKT-dependent
antiapoptotic signals in prostate cancer cells and inhibition of
CCR9-CCL25 interaction enhances the cytotoxic effects of etoposide.
Int J Cancer. 127:2020–2030. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Singh S, Singh UP, Stiles JK, Grizzle WE
and Lillard JW Jr: Expression and functional role of CCR9 in
prostate cancer cell migration and invasion. Clin Cancer Res.
10:8743–8750. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Chen HJ, Edwards R, Tucci S, Bu P, Milsom
J, Lee S, Edelmann W, Gumus ZH, Shen X and Lipkin S: Chemokine
25-induced signaling suppresses colon cancer invasion and
metastasis. J Clin Invest. 122:3184–3196. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Tu Z, Xiao R, Xiong J, Tembo KM, Deng X,
Xiong M, Liu P, Wang M and Zhang Q: CCR9 in cancer: Oncogenic role
and therapeutic targeting. J Hematol Oncol. 9:102016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Hu Y, Zhang L, Wu R, Han R, Jia Y, Jiang
Z, Cheng M, Gan J, Tao X and Zhang Q: Specific killing of CCR9
high-expressing acute T lymphocytic leukemia cells by CCL25 fused
with PE38 toxin. Leuk Res. 35:1254–1260. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Brasnjevic I, Steinbusch HW, Schmitz C and
Martinez-Martinez P: European NanoBioPharmaceutics research
initiative: Delivery of peptide and protein drugs over the
blood-brain barrier. Prog Neurobiol. 87:212–251. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Wang FY, Zhang TY, Luo JX, He GA, Gu QL
and Xiao F: Selection of CC chemokine receptor 5-binding peptide
from a phage display peptide library. Biosci Biotechnol Biochem.
70:2035–2041. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Skelton NJ, Chen YM, Dubree N, Quan C,
Jackson DY, Cochran A, Zobel K, Deshayes K, Baca M, Pisabarro MT,
et al: Structure-function analysis of a phage display-derived
peptide that binds to insulin-like growth factor binding protein 1.
Biochemistry. 40:8487–8498. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Tang B, Li Z, Huang D, Zheng L and Li Q:
Screening of a specific peptide binding to VPAC1 receptor from a
phage display peptide library. PLoS One. 8:e542642013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Houimel M and Mazzucchelli L:
Identification of biologically active peptides that inhibit binding
of human CXCL8 to its receptors from a random phage-epitope
library. J Leukoc Biol. 85:728–738. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Li B, Wang Z, Zhong Y, Lan J, Li X and Lin
H: CCR9-CCL25 interaction suppresses apoptosis of lung cancer cells
by activating the PI3K/Akt pathway. Med Oncol. 32:662015.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhang L, Yin G, Yan D, Wei Y, Ma C, Huang
Z, Liao X, Yao Y, Chen X and Hao B: In vitro screening of ovarian
tumor specific peptides from a phage display peptide library.
Biotechnol Lett. 33:1729–1735. 2011. View Article : Google Scholar : PubMed/NCBI
|