Introduction
The incidence of oral cancer, particularly squamous
cell carcinomas (OSCC), is high in Southern China and there is also
a rising trend worldwide (1,2). Surgery serves an important role in the
treatment of OSCC (3). Oral defects
following oncological surgery may affect facial appearance and oral
function; therefore, reconstruction is an indispensable part of
oral cancer surgery (4).
Reconstructive methods used include using free flaps, prosthesis or
implantable materials. However, regardless of the method used, an
accurate evaluation of oral tissue defects is necessary.
Anatomically, the oral cavity comprises the upper, lateral and
lower walls. The upper wall is mainly bony structures, including
the hard palate and superior alveolus. A maxillectomy defect is a
defect associated with the upper wall of the oral cavity, which has
been well evaluated in the literature (5–8). However,
the lateral and lower wall of the oral cavity contains not only
mandibular structures, but also soft tissues, including the mobile
tongue, the floor of the mouth and the cheeks. Therefore, due to
the complexed anatomy and the existence of varied and compound
postoperative defects, there is no effective method for the
evaluation of oral defects occurring in the lower region of the
oral cavity. The establishment of a classification system for
postoperative oral defects could therefore aid clinical evaluation
and guide oral reconstruction.
There have been few previous studies regarding the
clinical evaluation of head and neck defects. For bony defects,
there have been several studies delineating the classification for
maxillectomy defects that defined the range of a maxillary defect
in horizontal and vertical dimensions (5–8). Schultz
et al (9) and Boyd et
al (10) also established a
classification system for mandibular defects (11). For oropharyngeal soft tissue defects
following a transoral robotic surgery procedure, de Almeida et
al (12) classified them into 4
types and provided the algorithm for the reconstruction. Brown's
classification covers the evaluation for the upper wall of the oral
cavity (5,6), however, there remains no effective
classification system for compound oral defects occurring in the
lower region of the oral cavity.
The present study reviewed a group of cases with
oral defects occurring in the lower region of the oral cavity and
proposed a classification system for the clinical evaluation and
oral reconstruction.
Materials and methods
Patients
Clinical data of 145 patients with oral tumors
treated in the Department Of Head And Neck Surgery of Cancer Center
of Sun Yat-Sen University (Guangdong, China) between January 2010
and December 2013 were reviewed. Inclusion criteria for the present
study were as follows: All patients were pathologically diagnosed
as having primary oral tumors, mainly SCC; all patients received
radical curative surgery; all postoperative oral defects received
primary reconstruction; and the clinical data were complete. Oral
cancer arising from the lip, hard palate and upper gum,
oropharyngeal cancer and cheek skin cancer were excluded from the
present study. The site and extent of the tumor were evaluated by
clinical examination and computed tomography/magnetic resonance
imaging. The postoperative oral defects were evaluated using
preoperative imaging and surgical records. The detailed data for
the 145 patients with oral tumors are summarized in Table I.
 | Table I.Clinical features of 145 patients with
oral tumors. |
Table I.
Clinical features of 145 patients with
oral tumors.
| Clinical
features | Patients, n | % |
|---|
| Sex |
|
|
| Male | 101 | 69.66 |
|
Female | 44 | 30.34 |
| Age, years |
|
|
|
<40 | 32 | 22.07 |
|
>40 | 113 | 77.93 |
| Primary sites |
|
|
|
Tongue | 69 | 47.59 |
| Floor of
mouth | 26 | 17.93 |
|
Mandible | 10 | 6.90 |
|
Gingiva | 24 | 16.55 |
| Buccal
mucosa | 16 | 11.03 |
| Pathology |
|
|
| Squamous
cell carcinoma | 125 | 86.21 |
|
Others | 20 | 13.79 |
| Ta |
|
|
| 1 | 8 | 6.40 |
| 2 | 34 | 27.20 |
| 3 | 51 | 40.80 |
| 4 | 32 | 25.60 |
| Na |
|
|
| 0 | 50 | 40.00 |
| 1 | 38 | 30.40 |
| 2 | 34 | 27.20 |
| 3 | 3 | 2.40 |
| Treatment |
|
|
| Surgery
alone | 76 | 52.41 |
|
Surgery+RT | 22 | 15.17 |
|
ICT+surgery | 23 | 15.86 |
| Other
therapies with surgery | 24 | 16.55 |
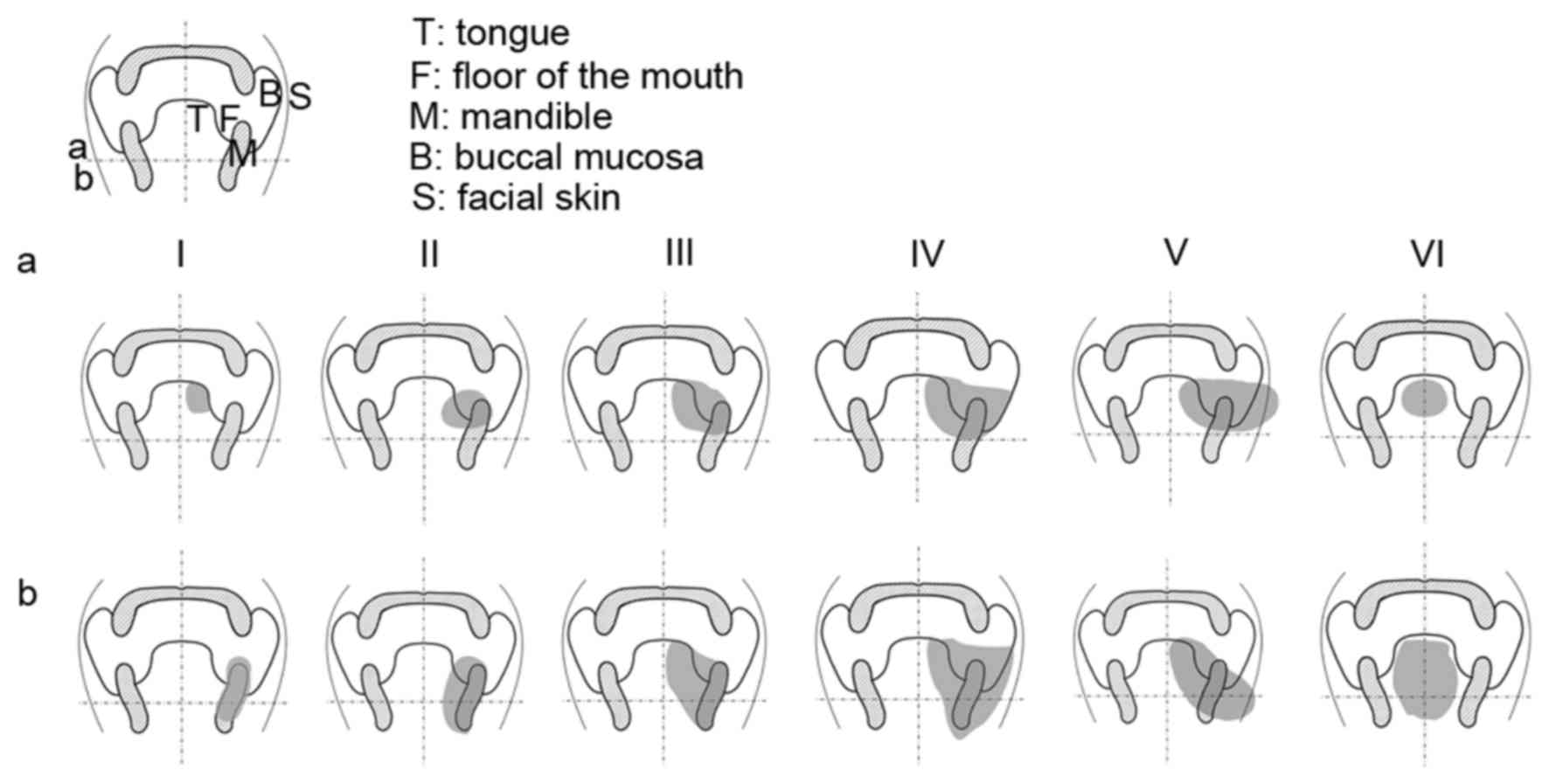
Classification system
The present study established a classification
system for the evaluation of postoperative oral defects based on
two dimensions. The extent of the oral defect from inside out was
evaluated in the horizontal dimension. The extent of the depth of
the oral defect was evaluated in the vertical dimension. Two
straight lines were then introduced into this classification
system. The midline vertically divided the oral cavity into 2
symmetrical parts, and the horizontal line connecting midpoints of
the bilateral mandible in the coronary section indicated the level
of mandibular nerve divided the oral cavity into 2 regions in terms
of depth. The 5 anatomical subsites of the oral cavity were
horizontally defined as the ipsilateral hemi-tongue (T), the floor
of the mouth (F), the mandible (M), the buccal mucosa (B) and the
cheek skin (S). The depth of the oral defects defined by the
horizontal line included shallow (a) and deep (b) types. This
classification system is represented in Fig. 1.
According to the missing numbers of continuous oral
anatomical subsites, oral defects in the horizontal dimension could
be classified into 6 types (I–VI). According to the depth, oral
defects in the vertical dimension could be classified into 2 types
(a and b). The detailed definitions are shown in Table II.
 | Table II.Definitions for the classification
system of oral defects. |
Table II.
Definitions for the classification
system of oral defects.
| A, Horizontal |
|---|
|
|---|
| Types | Subsites | Definition |
|---|
| I |
| Defects with only 1
ipsilateral subsite involved |
|
| T | Tongue |
|
| F | Floor of mouth |
|
| M | Mandible |
|
| B | Buccal mucosa |
| II |
| Defects with 2
ipsilateral continuous subsites involved |
|
| TF | Tongue and floor of
mouth |
|
| FM | Floor of mouth and
mandible |
|
| MB | Mandible and buccal
mucosa |
|
| BS | Buccal mucosa and
facial skin |
| III |
| Defects with 3
ipsilateral continuous subsites involved |
|
| TFM | Tongue, floor of
mouth and mandible |
|
| FMB | Floor of mouth,
mandible and buccal mucosa |
|
| MBS | Mandible, buccal
mucosa and facial skin |
| IV |
| Defects with 4
ipsilateral continuous subsites involved |
|
| TFMB | Tongue, floor of
mouth, mandible and buccal mucosa |
|
| FMBS | Floor of mouth,
mandible, buccal mucosa and facial skin |
| V | TFMBS | Defects with all of
the 5 ipsilateral subsites involved |
| VI |
| Defects with
bilateral subsites involved |
|
| B,
Vertical |
|
| a |
| Defects above the
horizontal line |
| b |
| Defects across the
horizontal line to include deep floor of mouth and whole height of
mandible |
According to this classification system,
postoperative oral defects can be defined into 12 types (Fig. 1). Each type of tissue defect could be
expressed with an alphabetical sign; for example, IIIb (FMB)
indicating an IIIb oral defect with 3 continuous subsites (floor of
mouth, mandible and buccal mucosa) involved.
Results
A total of 145 patients with oral tumors received
curative radical surgery, and their postoperative oral defects were
evaluated using the proposed classification system. The results are
displayed in Table III. The
proportion of different types of oral defects was as follows: Type
I, 35.9%; type II, 21.4%; type III, 23.4%; type IV, 4.8%; type V,
2.1%; and type VI, 12.4%. Among them, 91 (62.8%) cases were type a
and 54 (37.2%) cases were type b.
 | Table III.Postoperative classification of
defects for 145 patients with oral tumors. |
Table III.
Postoperative classification of
defects for 145 patients with oral tumors.
| Types | Subsites | Type a | Type b | Total, n (%) |
|---|
| I | T | 31 | – | 31 |
|
| F | 5 | – | 5 |
|
| M | 2 | 5 | 7 |
|
| B | 9 | – | 9 |
|
|
| 47 | 5 | 52 (35.9) |
| II | TF | 8 | 1 | 9 |
|
| FM | 10 | 7 | 17 |
|
| MB | 4 | – | 4 |
|
| BS | 1 | – | 1 |
|
|
| 23 | 8 | 31 (21.4) |
| III | TFM | 15 | 14 | 29 |
|
| FMB | – | 3 | 3 |
|
| MBS | 2 | – | 2 |
|
|
| 17 | 17 | 34 (23.4) |
| IV | TFMB | – | 3 | 3 |
|
| FMBS | 1 | 3 | 4 |
|
|
| 1 | 6 | 7 (4.8) |
| V |
| – | 3 | 3 (2.1) |
| VI |
| 3 | 15 | 18 (12.4) |
To further elucidate the clinical relevance, the
classifications in Table III could
be summarized into 4 groups (counting may overlap as some patients
were included in more than one category in order to highlight
facial skin and bone involvement): Ia-Va representing the
unilateral 1–5 subsites involving superficial oral defects without
mandibular continuity destruction (88 cases, 60.7%); Ib-Vb (+M)
representing the unilateral 1–5 subsites involving deep oral
defects with segmental mandibular continuity destruction (39 cases,
26.9%); I–V (+S) representing the unilateral through and through
oral defects with cheek skin involvement (7 cases, 4.8%); and VI
representing bilateral oral defects (18 cases, 12.4%).
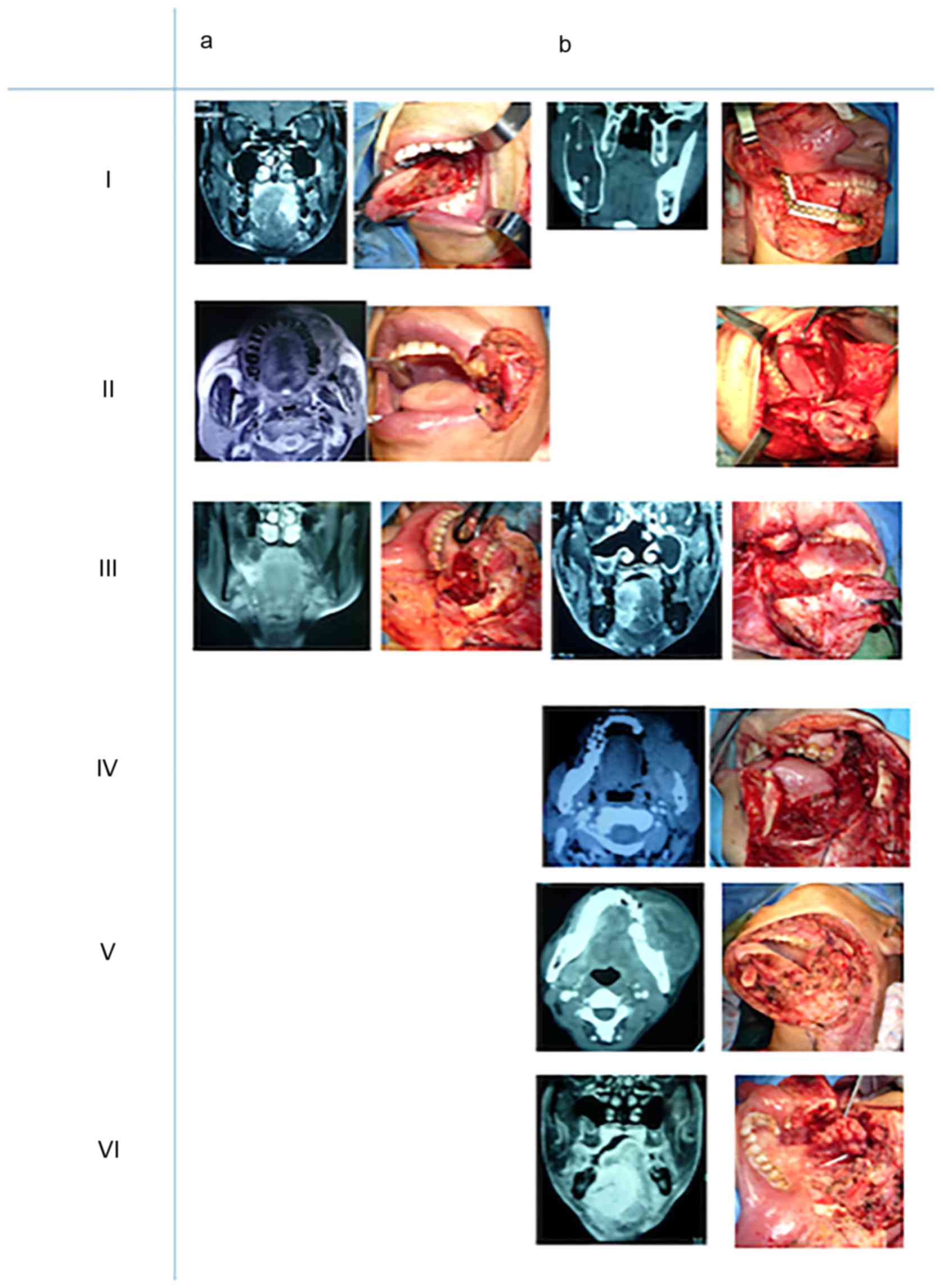
The different types of representative postoperative
oral defects are shown in representative in Fig. 2. All cases received primary surgical
reconstruction and Table IV
demonstrates the reconstructive methods used in this series of
patients. A total of 4 reconstructive methods were used in 145
cases: 101 cases (69.7%) with free flaps; 21 cases (14.5%) with
flaps combined with titanium plate reconstruction; 13 cases (9.0%)
with local pedicled flaps; and 10 cases (6.9%) with other methods.
Among the 101 cases which received free flap reconstruction: 79
cases (78.2%) used an anterolateral thigh flap; 5 cases (5.0%) used
a forearm flap; and 17 cases (16.8%) used a fibular flap. Among the
21 cases reconstructed by flaps combined with titanium plates, 16
cases used free anterolateral thigh flaps and 5 cases used
pectoralis major myocutaneous flaps. Other reconstructive methods
included adjacent tissue flaps (such as tongue flaps, forehead
flaps and strap muscle flaps) and a free skin graft.
 | Table IV.Reconstructive methods used for all
types of oral defects. |
Table IV.
Reconstructive methods used for all
types of oral defects.
|
| Free flap | Flap+titanium
plate |
|
|
|---|
|
|
|
|
|
|
|---|
| Types | ALT | FF | RF | ALT+T | PM+T | Local flap PM | Others |
|---|
| Ia | 32 | – | 4 | – | – | 1 | 9 |
| Ib | – | 6 | – | – | – | – | – |
| IIa | 16 | – | 1 | 2 | 1 | 1 | – |
| IIb | 2 | 4 | – | – | 2 | 2 | – |
| IIIa | 10 | – | – | 1 | – | 5 | 1 |
| IIIb | 4 | 2 | – | 7 | 1 | 3 | – |
| IVa | – | – | – | – | – | – | – |
| IVb | 1 | 2 | – | 3 | 1 | – | – |
| Va | – | – | – | – | – | – | – |
| Vb | 1 | – | – | 2 | – | – | – |
| VIa | 3 | – | – | – | – | – | – |
| VIb | 10 | 3 | – | 1 | – | 1 | – |
Discussion
An ideal reconstruction requires the accurate
evaluation of the tissue defects. An effective clinical
classification system should distinguish between simple and
complicated defects, clearly express the characteristics of the
defects, and be used easily and practically. The oral cavity is the
entrance of the digestive tract and can anatomically be abstracted
as a hollow tubular structure, delineated as a circle in Fig. 1. As defects of the upper jaw could be
included in maxillary defects, the evaluation of oral defects was
mainly concentrated on the lower half circle that was the basal and
lateral walls. Therefore, it was considered that the extent of the
oral defects could be sufficiently evaluated in two dimensions.
Horizontally, it was possible to measure the area of the defect
from inside out by the anatomical subsites involved and whether the
defect was through facial skin or not; vertically, the depth of the
oral defect was measured, and it could be determined whether the
mandibular continuity was destroyed or not. The current
classification system for the evaluation of postoperative oral
defects, except from the upper jaw, was based on the above
concepts.
Oral defects following oncological surgery for
tumors are complicated. Therefore, two parameters were used to
summarize the complexity of oral defects in the current
classification system: The numbers of continuous oral anatomical
subsites involved in the defect and the depth of the oral defects.
The former could not only reflect the area of the defect, but also
clarify complicated defects; for example, a through and through
defect or a defect across the midline. The latter could express the
depth of the defect, particularly the mandibular continuity.
Therefore, the more anatomical subsites that were involved and the
deeper the defect was, the more complicated the defect was.
Accordingly, in the present classification system, higher level and
b-type defects represented more complicated defects. In the present
study, among the 145 cases with the oral defects, 28 cases (19.3%)
were classified as type IV–VI, which were difficult defects that
required more complicated reconstruction.
The postoperative oral defects observed in the
present study were varied and the classification system should
therefore be able to reflect the special types of oral defects. As
different reconstructive strategy may be required in certain oral
defects, the following 3 conditions should receive additional
attention: Through and through defects; composite defects combined
with mandibular discontinuity; and bilateral oral defects. In the
present classification system, the above 3 types of special oral
defects have been clearly indicated. Through and through oral
defects could be evaluated by cheek skin involvement (+S).
Composite oral defects with mandibular involvement could be
expressed as (+M), which could then be further classified into type
a (without mandibular continuity destruction) and type b (with
mandibular continuity destruction). Type VI could reflect the
bilateral oral defects.
The present proposed classification system for oral
defects is concise and should be easy to use clinically. The 2
parameters used in the classification system are familiar to
surgeons and could be easily evaluated on preoperative coronary
images of the oral cavity. In addition, it is easy to record types
of oral defects using this classification system. The present
classification could describe a through and through oral defect
with the involvement of 4 subsites (floor of mouth, mandible,
buccal mucosa and facial skin) and in combination with mandibular
continuity destruction simply by designating it as type IVb
(MBS).
In order to evaluate the depth of the oral defects
and determine whether mandibular continuity was destroyed, a
horizontal line was introduced into the present classification
system. Rectangular and segmental mandibulectomy were the two main
surgical procedures used in the treatment of OSCC with mandibular
invasion. This horizontal line could be used to distinguish the two
conditions, as anatomically, this line was almost on the same level
as the mandibular nerve, and by discriminating shallow from deep
oral defects this line may guide the choice of a different
reconstructive method.
There were no parameters to evaluate the extent of
oral defects in the sagittal dimension in the present proposed
classification system, as in the case of oral cancer with posterior
invasion of the tongue base or tonsil, the oropharyngeal defect
could be evaluated by de Almeida's system (12). In addition, the oral defects involving
the oropharynx would not significantly alter the method of
reconstruction.
The establishment of the present classification
system could aid the measurement of the extent of the oral defects,
distinguish special types of defects and guide the choice of
reconstruction. Previous studies on the classification of defects
of the upper jaw, mandible, mid-face and oropharynx provided an
algorithm for reconstruction (5–12). As an
example, the classification of upper jaw defects evaluated the
extent using only two dimensions, the advantage being that
different defects were easily identifiable in the clinical setting
(5,6).
For minor defects such as class 1a, 2a or 2b, an obturator may be
chosen in the reconstruction. Free flaps would be the better choice
for large defects like class 4a or 4b. We also saw the pattern in
the oral defects just as in the midfacial defects and organized
them into clinically actionable groups. The present classification
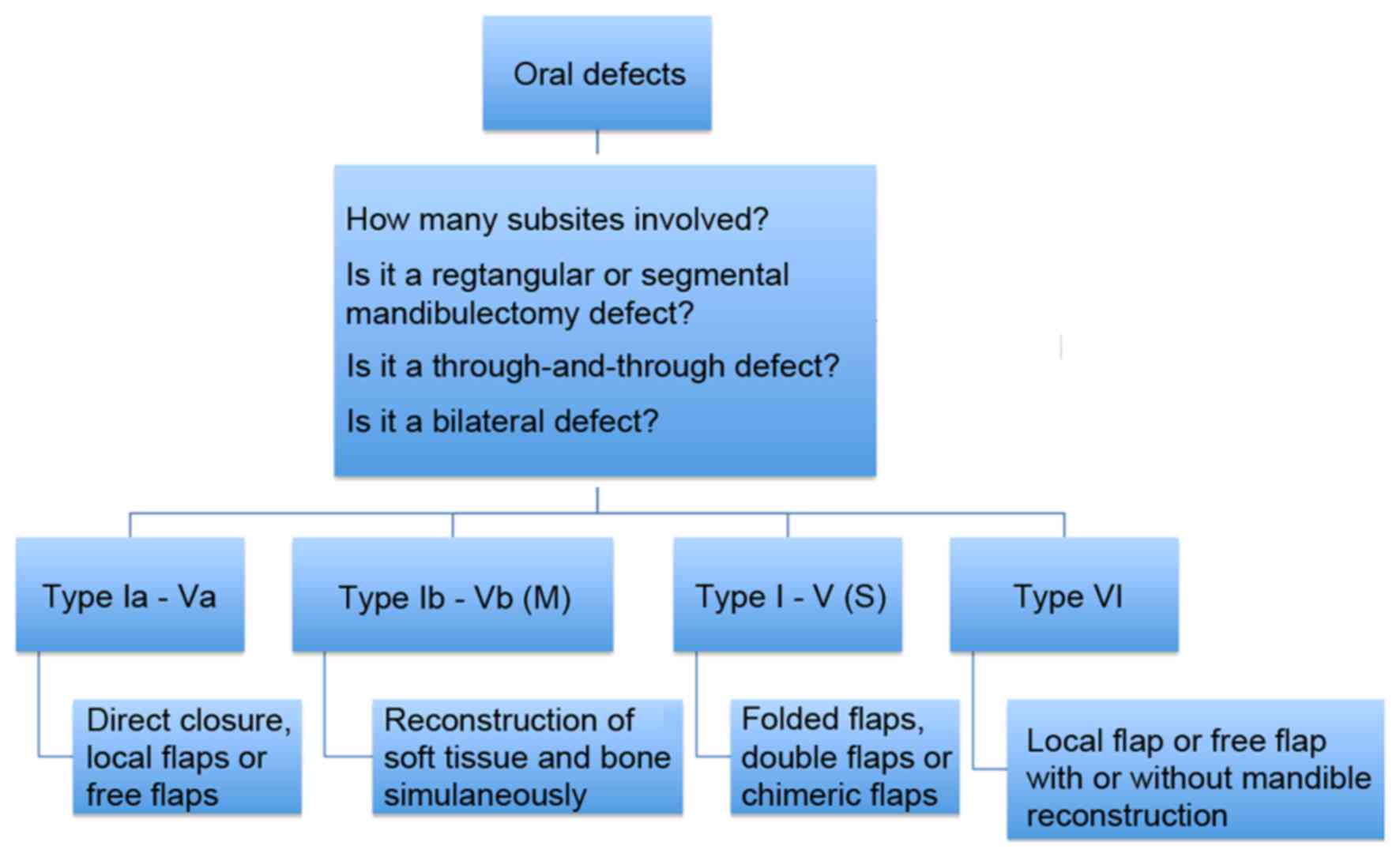
system could summarize the 4 common types of oral defects and guide
the choice of reconstructive methods (Fig. 3). For stage Ia-Va oral defects, due to
a shallow loss of tissue, the absence of destruction to mandibular
continuity and the complexity of the defects decided only by
numbers of involved anatomical subsites, reconstruction could be
completed by simple closure, local flaps or free flaps according to
the area of missing tissues. In this type of oral defect, as the
continuity of the mandible was preserved, it was relatively easy to
add dental implants. The restorability of oral function in these
types of defects should be good.
For type b (+M) defects with segmental
mandibulectomy, free fibula flaps or free flaps combined with
titanium plates should be considered for the reconstruction. The
size of the defect should be evaluated, as this may affect the
choice of the flaps and reconstructive plates. Occasionally,
three-dimensional computer-aided design was required to restore the
anatomy for certain bulky or wide compound oral defects. Dental
implants were feasible for these types of defects, however, the
restorability of the oral function depended not only on the size of
the defect itself, but also on the expected oncological result. For
the through and through defects with facial skin involvement,
folding flaps, double flaps or chimeric flaps may be the most
suitable reconstruction choice. The oral function could be restored
well for these types of defects. For bilateral oral defects, a
large block of composite tissue transfer was generally required to
reconstruct the total tongue, the floor of the mouth and a region
of the mandible. Due to a large volume of missing oral tissue, the
restorability of the oral function in this type of defect may be
compromised. If a total glossectomy was performed, the swallowing
function may also be affected.
There were numerous factors associated with the
evaluation of postoperative oral defects. The present
classification system was based on the coronary anatomy and
introduced 2 parameters, including the number of involved
anatomical subsites and the depth of the defect, to evaluate
mandibular continuity and distinguish special oral defects as
bilateral or through and through defects. This classification was
only a two-dimensional system, not including the sagittal
dimension, which influenced the complete evaluation of the oral
defects to a certain extent. In addition, it is necessary to
evaluate additional cases of oral defects using the proposed
classification system to further validate its efficacy and
practicability. Moreover, this classification could not provide an
evaluation for the size of the harvested flap. Another limitation
of the present classification system was that it could not define
the specific site of a mandibular defect when a compound defect
appeared, and it could not differentiate between anterior and
posterior mandibular defects that may influence the choice of
different reconstructive methods. Further modifications are
therefore required in order to improve the classification system.
Other associated factors, including adjuvant radiotherapy and the
systemic health of the patient, should also be considered in the
development of the classification system.
In summary, an accurate evaluation of oral defects
is necessary to guide the subsequent reconstruction. The present
retrospective study proposed a novel classification system for the
evaluation of oral defects, and confirmed that it was simple and
clinically practical. In the future, this classification system
could identify the common types of oral defects and assist in
guiding the reconstruction.
References
|
1
|
Saman DM: A review of the epidemiology of
oral and pharyngeal carcinoma: Update. Head Neck Oncol. 4:12012.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Chaturvedi AK, Anderson WF,
Lortet-Tieulent J, Curado MP, Ferlay J, Franceschi S, Rosenberg PS,
Bray F and Gillison ML: Worldwide trends in incidence rates for
oral cavity and oropharyngeal cancers. J Clin Oncol. 31:4550–4559.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ow TJ and Myers JN: Current management of
advanced resectable oral cavity squamous cell carcinoma. Clin Exp
Otorhinolaryngol. 4:1–10. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Wong CH and Wei FC: Microsurgical free
flap in head and neck reconstruction. Head Neck. 32:1236–1245.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Brown JS, Rogers SN, McNally DN and Boyle
M: A modified classification for the maxillectomy defect. Head
Neck. 22:17–26. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Brown JS and Shaw RJ: Reconstruction of
the maxilla and midface: Introducing a new classification. Lancet
Oncol. 11:1001–1008. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Cordeiro PG and Santamaria E: A
classification system and algorithm for reconstruction of
maxillectomy and midfacial defects. Plast Reconstr Surg.
105:2331–2348. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bidra AS, Jacob RF and Taylor TD:
Classification of maxillectomy defects: A systematic review and
criteria necessary for a universal description. J Prosthet Dent.
107:261–270. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Schultz BD, Sosin M, Nam A, Mohan R, Zhang
P, Khalifian S, Vranis N, Manson PN, Bojovic B and Rodriguez ED:
Classification of mandible defects and algorithm for microvascular
reconstruction. Plast Reconstr Surg. 135:743e–754e. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Boyd JB, Gullane PJ, Rotstein LE, Brown DH
and Irish JC: Classification of mandibular defects. Plast Reconstr
Surg. 92:1266–1275. 1993.PubMed/NCBI
|
|
11
|
Brown JS, Barry C, Ho M and Shaw R: A new
classification for mandibular defects after oncological resection.
Lancet Oncol. 17:e23–e30. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
de Almeida JR, Park RC, Villanueva NL,
Miles BA, Teng MS and Genden EM: Reconstructive algorithm and
classification system for transoral oropharyngeal defects. Head
Neck. 36:934–941. 2014. View Article : Google Scholar : PubMed/NCBI
|