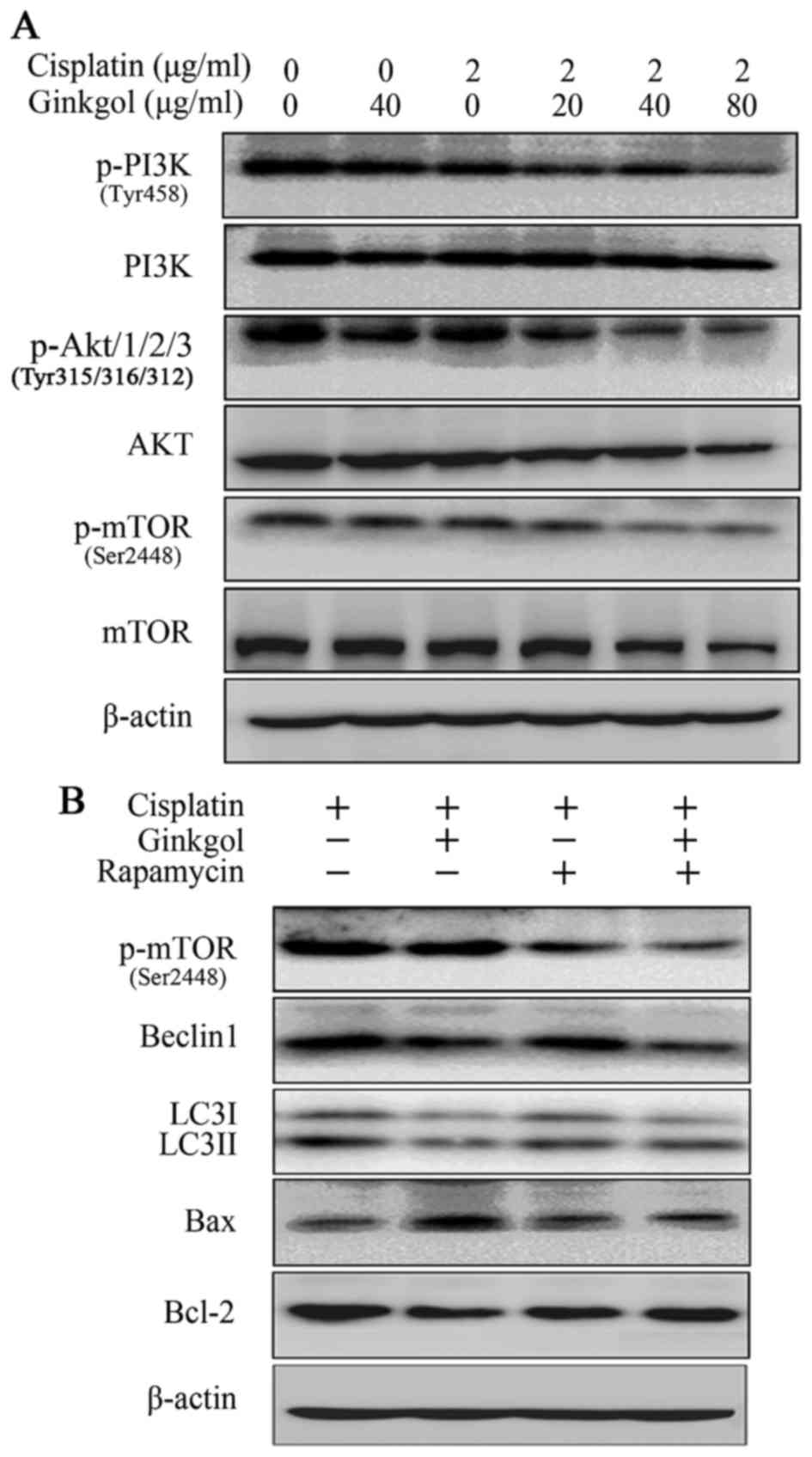
|
1
|
Farazi PA and Depinho RA: Hepatocellular
carcinoma pathogenesis: From genes to environment. Nat Rev Cancer.
6:674–687. 2006. View
Article : Google Scholar : PubMed/NCBI
|
|
2
|
Khaderi S, Guiteau J, Cotton RT, O'Mahony
C, Rana A and Goss JA: Role of liver transplantation in the
management of hepatoblastoma in the pediatric population. World J
Transplant. 4:294–298. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Abdul HA, Lai ML and Phaik LC: Tissue
microarray immunohistochemical profiles of p53 and pRB in
hepatocellular carcinoma and hepatoblastoma. Asian Pac J Cancer
Prev. 15:3959–3963. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
López-Terrada D, Cheung SW, Finegold MJ
and Knowles BB: Hep G2 is a hepatoblastoma-derived cell line. Hum
Pathol. 40:1512–1517. 2009. View Article : Google Scholar
|
|
5
|
Pang RTK, Poon TCW, Wong N, Lai PSB, Wong
NLY, Chan CML, Yu JWS, Chan ATC and Sung JJY: Comparison of protein
expression patterns between hepatocellular carcinoma cell lines and
a hepatoblastoma cell line. Clin Proteomics. 1:313–331. 2004.
View Article : Google Scholar
|
|
6
|
Rishi RR, Kimberlee KS, Ruth IH, Michael
SB, Max B and Lisa EH: Hepatoblastoma: A need for cell lines and
tissue banks to develop targeted drug therapies. Front Pediatr.
4:222016.PubMed/NCBI
|
|
7
|
Pateva IB, Egler RA and Stearns DS:
Hepatoblastoma in an 11-year-old: Case report and a review of the
literature. Medicine (Baltimore). 96:e58582017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Tomlinson GE and Kappler R: Genetics and
epigenetics of hepatoblastoma. Pediatr Blood Cancer. 59:785–792.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Boulikas T and Vougiouka M: Cisplatin and
platinum drugs at the molecular level (Review). Oncol Rep.
10:1663–1682. 2003.PubMed/NCBI
|
|
10
|
Amable L: Cisplatin resistance and
opportunities for precision medicine. Pharmacol Res. 106:27–36.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Muggia F: Platinum compounds 30 years
after the introduction of cisplatin: Implications for the treatment
of ovarian cancer. Gynecol Oncol. 112:275–281. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zhang HQ, Fang N, Liu XM, Xiong SP, Liao
YQ, Jin WJ, Song RF and Wan YY: Antitumor activity of chloroquine
in combination with Cisplatin in human gastric cancer xenografts.
Asian Pac J Cancer Prev. 16:3907–3912. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
García-Cano J, Ambroise G, Pascual-Serra
R, Carrión MC, Serrano-Oviedo L, Ortega-Muelas M, Cimas FJ, Sabater
S, Ruiz-Hidalgo MJ, Sanchez Perez I, et al: Exploiting the
potential of autophagy in cisplatin therapy: A new strategy to
overcome resistance. Oncotarget. 6:15551–15565. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Lu SZ and Harrison-Findik DD: Autophagy
and cancer. World J Biol Chem. 4:64–70. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Mizushima N and Komatsu M: Autophagy:
Renovation of cells and tissues. Cell. 147:728–741. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Mathew R, Karp CM, Beaudoin B, Vuong N,
Chen G, Chen HY, Bray K, Reddy A, Bhanot G, Gelinas C, et al:
Autophagy suppresses tumorigenesis through elimination of p62.
Cell. 137:1062–1075. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Liu M, Ma S, Liu M, Hou Y, Liang B, Su X
and Liu X: Synergistic killing of lung cancer cells by cisplatin
and radiation via autophagy and apoptosis. Oncol Lett. 7:1903–1910.
2014.PubMed/NCBI
|
|
18
|
Sakamoto A and Iwamoto Y: Current status
and perspectives regarding the treatment of osteo-sarcoma:
Chemotherapy. Rev Recent Clin Trials. 3:228–231. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Choi AM, Ryter SW and Levine B: Autophagy
in human health and disease. N Engl J Med. 368:651–662. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Chen J, Wang Q, Yin FQ, Zhang W, Yan LH
and Li L: MTRR silencing inhibits growth and cisplatin resistance
of ovarian carcinoma via inducing apoptosis and reducing autophagy.
Am J Transl Res. 7:1510–1527. 2015.PubMed/NCBI
|
|
21
|
Vanhaesebroeck B, Stephens L and Hawkins
P: PI3K signalling: The path to discovery and understanding. Nat
Rev Mol Cell Biol. 13:195–203. 2012. View
Article : Google Scholar : PubMed/NCBI
|
|
22
|
Tsai JP, Lee CH, Ying TH, Lin CL, Lin CL,
Hsueh JT and Hsieh YH: Licochalcone A induces autophagy through
PI3K/Akt/mTOR inactivation and autophagy suppression enhances
Licochalcone A-induced apoptosis of human cervical cancer cells.
Oncotarget. 6:28851–28866. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Laplante M and Sabatini DM: mTOR signaling
in growth control and disease. Cell. 149:274–293. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Nash KM and Shah ZA: Current perspectives
on the beneficial role of ginkgo biloba in neurological and
cerebrovascular disorders. Integr Med InsIghts. 10:1–9. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Chen Q, Yang GW and An LG: Apoptosis of
hepatoma cells SMMC-7721 induced by ginkgo biloba seed
polysaccharide. World J Gastroenterol. 8:832–836. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yang XM, Wang YF, Li YY and Ma HL: Thermal
stability of ginkgolic acids from ginkgo biloba and the effects of
ginkgol C17:1 on the apoptosis and migration of SMMMC7721 cells.
Fitoterapia. 98:66–76. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Ma X, Liu H, Foyi SR, Godar RJ, Weinheimer
CJ, Hill JA and Diwan A: Impaired autophagosome clearance
contributes to cardiomyocyte death in ischemia/reperfusion injury.
Circulation. 125:3170–3181. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Reggiori F, Komatsu M, Finley K and
Simonsen A: Autophagy: More than a nonselective pathway. Int J Cell
Biol. 2012:2196252012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Kabeya Y, Mizushima N, Ueno T, Yamamoto A,
Kirisako T, Noda T, Kominami E, Ohsumi Y and Yoshimori T: LC3, a
mammalian homologue of yeast Apg8p, is localized in autophagosome
membranes after processing. EMBO J. 19:5720–5728. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Lindqvist LM, Simon AK and Baehrecke EH:
Current questions and possible controversies in autophagy. Cell
Death Discov. 1:pii: 150362015. View Article : Google Scholar
|
|
31
|
Zhang Z, Shao Z, Xiong L and Yang S:
Inhibition of autophagy enhances cisplatin-induced apoptosis in the
MG63 human osteosarcoma cell line. Oncol Lett. 10:2941–2946.
2015.PubMed/NCBI
|
|
32
|
Amaravadi RK and Thompson CB: The roles of
therapy-induced autophagy and necrosis in cancer treatment. Clin
Cancer Res. 13:7271–7279. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Bao L, Jaramillo MC, Zhang Z, Zheng Y, Yao
M, Zhang DD and Yi X: Induction of autophagy contributes to
cisplatin resistance in human ovarian cancer cells. Mol Med Rep.
11:91–98. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Le Grazie M, Biagini MR, Tarocchi M,
Polvani S and Galli A: Chemotherapy for hepatocellular carcinoma:
The present and the future. World J Hepatol. 9:907–920. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Cavallo F, Feldman DR and Barchi M:
Revisiting DNA damage repair, p53-mediated apoptosis and cisplatin
sensitivity in germ cell tumors. Int J Dev Biol. 57:273–280. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Zhang HQ, He B, Fang N, Lu S, Liao YQ and
Wan YY: Autophagy inhibition sensitizes cisplatin cytotoxicity in
human gastric cancer cell line SGC7901. Asian Pac J Cancer Prev.
14:4685–4688. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Kimura T, Takabatake Y, Takahashi A and
Isaka Y: Chloroquine in cancer therapy: A double-edged sword of
autophagy. Cancer Res. 73:3–7. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Li YY, Liu J, Yang XM, Dong Y, Liu YL and
Chen M: Ginkgol C17:1 inhibits tumor growth by blunting the
EGF-PI3K/Akt signaling pathway. J Biomed Res. 31:232–239.
2017.PubMed/NCBI
|
|
39
|
Tang JY, Dai T, Zhang H, Xiong WJ, Xu MZ,
Wang XJ, Tang QH, Chen B and Xu M: GDC-0980-induced apoptosis is
enhanced by autophagy inhibition in human pancreatic cancer cells.
Biochem Biophys Res Commun. 453:533–538. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Wu HM, Jiang ZF, Ding PS, Shao LJ and Liu
RY: Hypoxia-induced autophagy mediates cisplatin resistance in lung
cancer cells. Sci Rep. 5:122912015. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Yan MM, Ni JD, Song D, Ding M and Huang J:
Interplay between unfolded protein response and autophagy promotes
tumor drug resistance. Oncol Lett. 10:1959–1969. 2015.PubMed/NCBI
|
|
42
|
Luo J, Manning BD and Cantley LC:
Targeting the PI3K-Akt pathway in human cancer: Rationale and
promise. Cancer Cell. 4:257–262. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Li F, Zeng J, Gao Y, Guan Z, Ma Z, Shi Q,
Du C, Jia J, Xu S, Wang X, et al: G9a inhibition induces autophagic
cell death via AMPK/mTOR pathway in bladder transitional cell
carcinoma. PLoS One. 10:e01383902015. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Nikoletopoulou V, Markaki M, Palikaras K
and Tavernarakis N: Crosstalk between apoptosis, necrosis and
autophagy. Biochim Biophys Acta. 1833:3448–3459. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Okoshi R, Ozaki T, Yamamoto H, Ando K,
Koida N, Ono S, Koda T, Kamijo T, Nakagawara A and Kizaki H:
Activation of AMP-activated protein kinase induces p53-dependent
apoptotic cell death in response to energetic stress. J Biol Chem.
283:3979–3987. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Kefas BA, Cai Y, Ling Z, Heimberg H, Hue
L, Pipeleers D and Van de Casteele M: AMP-activated protein kinase
can induce apoptosis of insulin-producing MIN6 cells through
stimulation of c-Jun-N-terminal kinase. J Mol Endocrinol.
30:151–161. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Nieminen AI, Eskelinen VM, Haikala HM,
Tervonen TA, Yan Y, Partanen JI and Klefström J: Myc-induced
AMPK-phospho p53 pathway activates Bak to sensitize mitochondrial
apoptosis. Proc Natl AcadSci USA. 110:pp. E1839–E1848. 2013;
View Article : Google Scholar
|
|
48
|
Kim J, Kundu M, Viollet B and Guan KL:
AMPK and mTOR regulate autophagy through direct phosphorylation of
Ulk1. Nat Cell Biol. 13:132–141. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Jeon SM: Regulation and function of AMPK
in physiology and diseases. Exp Mol Med. 48:e2452016. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Löffler AS, Alers S, Dieterle AM, Keppeler
H, Franz-Wachtel M, Kundu M, Campbell DG, Wesselborg S, Alessi DR
and Stork B: Ulk1-mediated phosphorylation of AMPK constitutes a
negative regulatory feedback loop. Autophagy. 7:696–706. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Sanli T, Steinberg GR, Singh G and
Tsakiridis T: AMP-activated protein kinase (AMPK) beyond
metabolism: A novel genomic stress sensor participating in the DNA
damage response pathway. Cancer Biol Ther. 15:156–169. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Kondo Y, Kanzawa T, Sawaya R and Kondo S:
The role of autophagy in cancer development and response to
therapy. Nat Rev Cancer. 5:726–734. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Xie BS, Zhao HC, Yao SK, Zhuo DX, Jin B,
Lv DC, Wu CL, Ma DL, Gao C, Shu XM and Ai ZL: Autophagy inhibition
enhances etoposide-induced cell death in human hepatoma G2 cells.
Int J Mol Med. 27:599–606. 2011.PubMed/NCBI
|
|
54
|
Wu T, Wang MC, Jing L, Liu ZY, Guo H, Liu
Y, Bai YY, Cheng YZ, Nan KJ and Liang X: Autophagy facilitates lung
adenocarcinoma resistance to cisplatin treatment by activation of
AMPK/mTOR signaling pathway. Drug Des Devel Ther. 9:6421–6431.
2015. View Article : Google Scholar : PubMed/NCBI
|