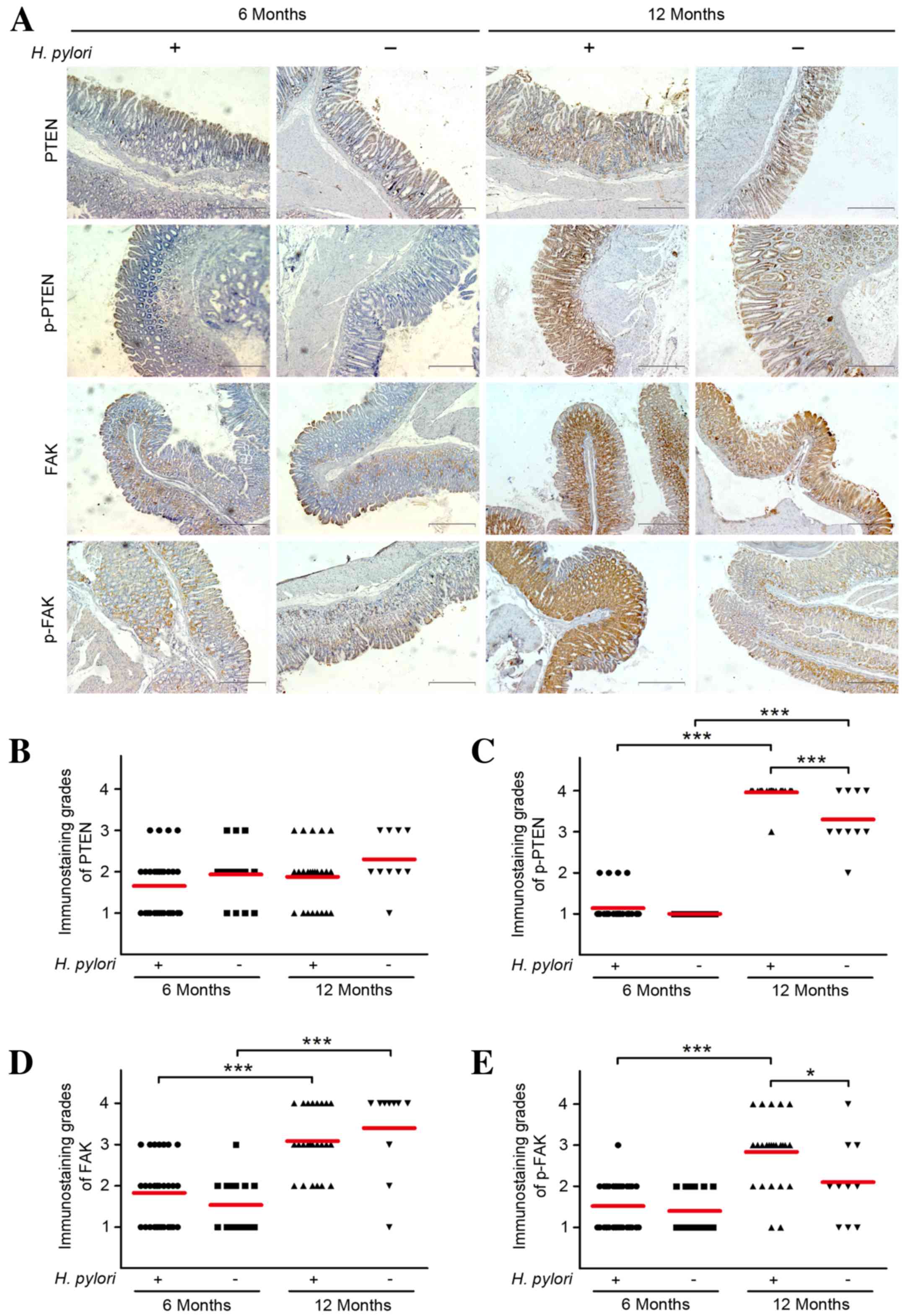
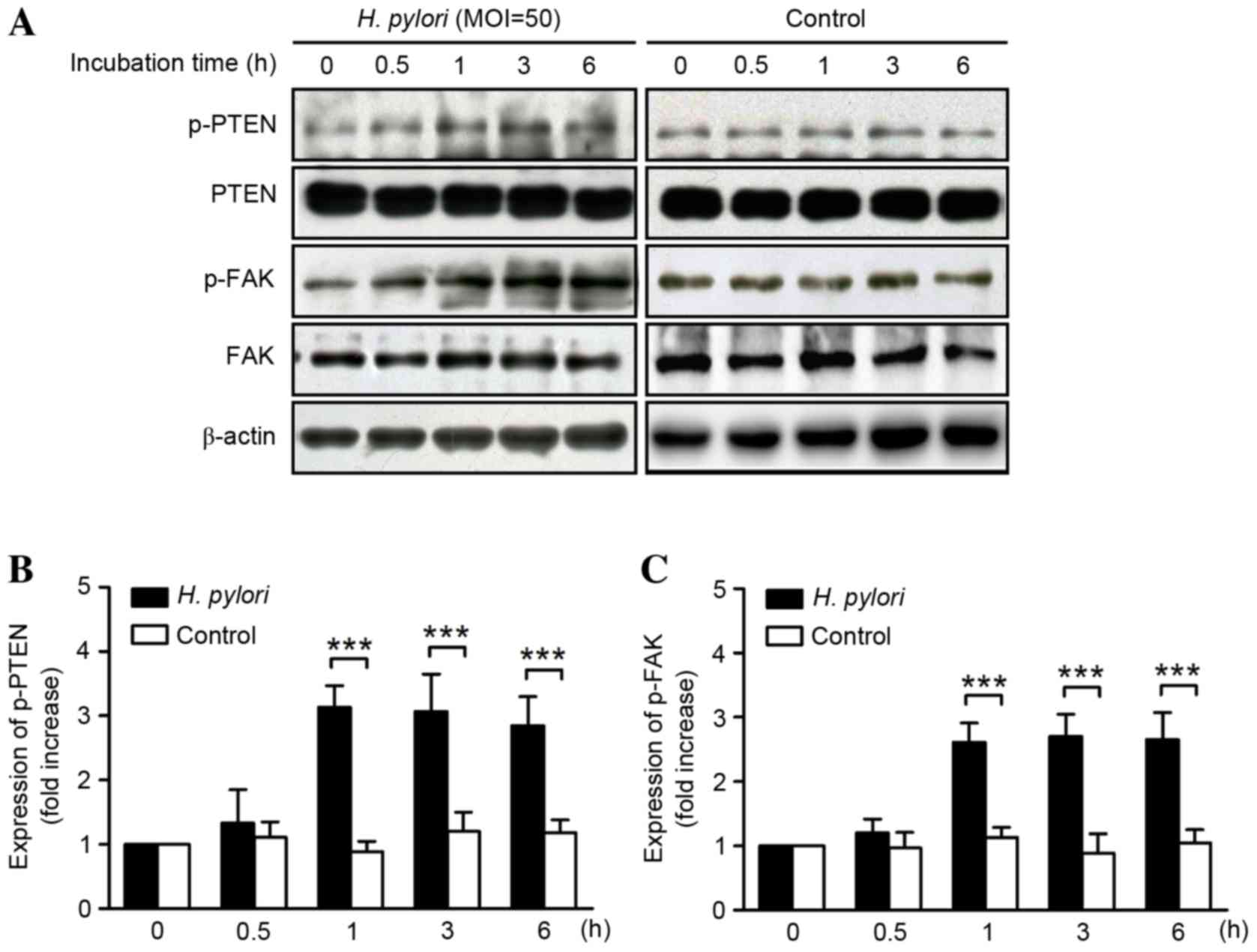
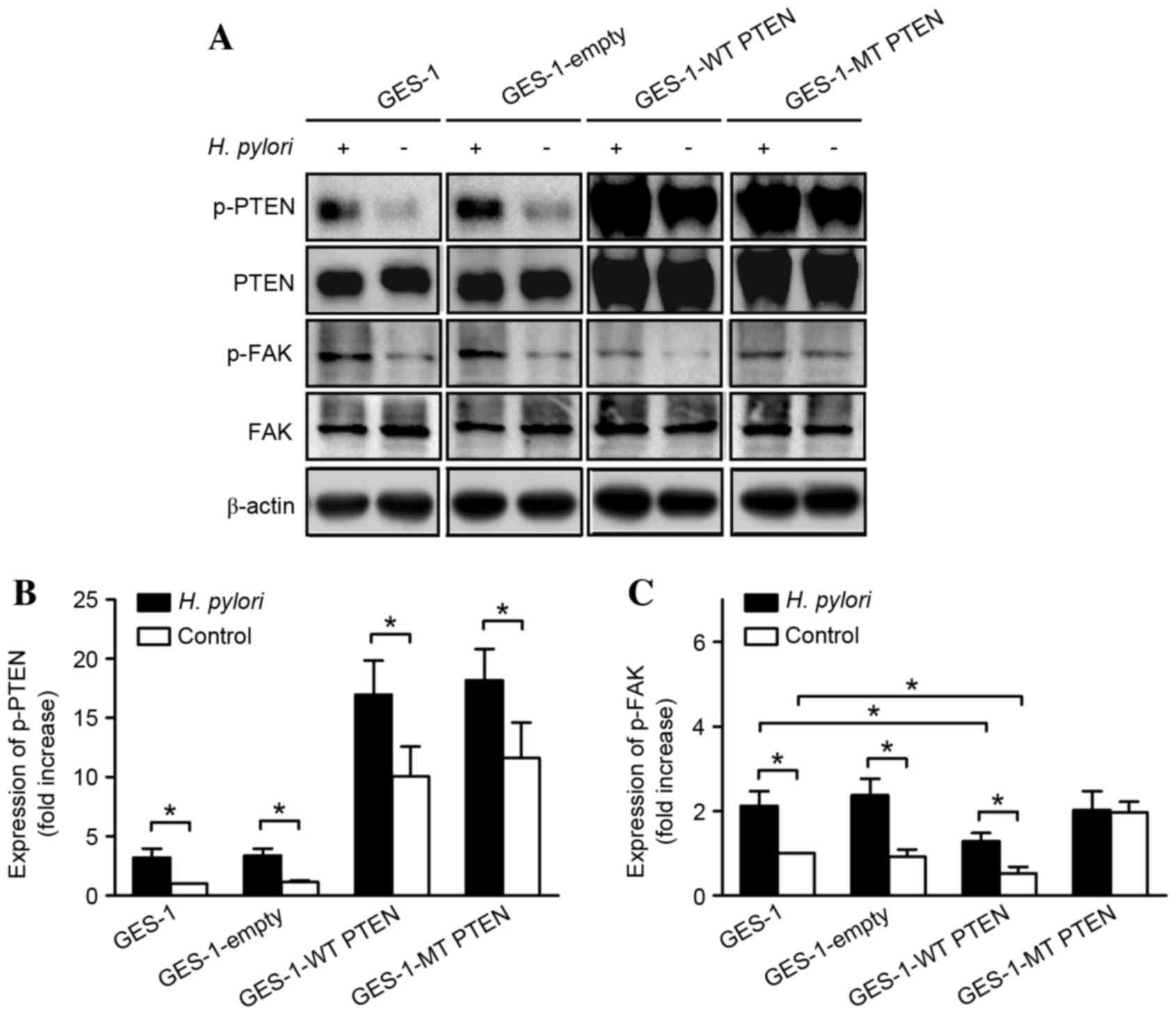
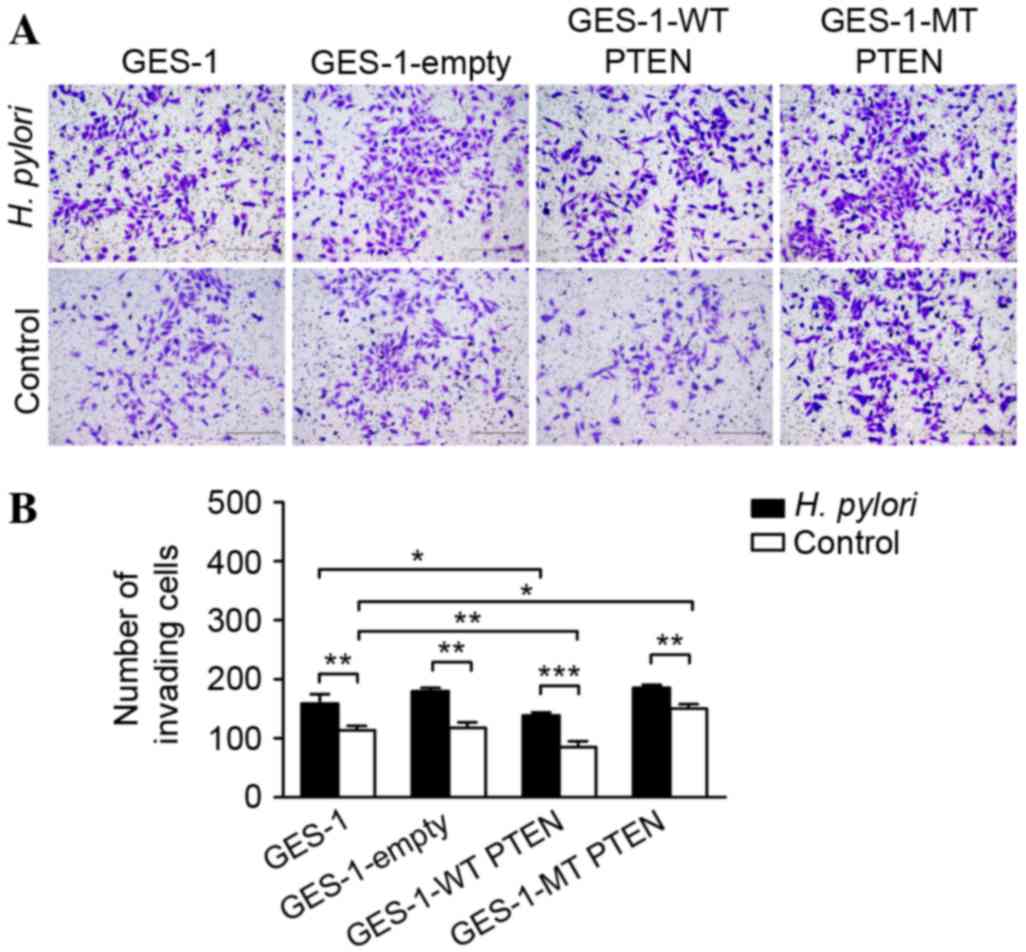
|
1
|
Moss SF and Sood S: Helicobacter pylori.
Curr Opin Infect Dis. 16:445–451. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Peek RM Jr and Crabtree JE: Helicobacter
infection and gastric neoplasia. J Pathol. 208:233–248. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Uemura N, Okamoto S, Yamamoto S, Matsumura
N, Yamaguchi S, Yamakido M, Taniyama K, Sasaki N and Schlemper RJ:
Helicobacter pylori infection and the development of gastric
cancer. N Engl J Med. 345:784–789. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Nomura A, Stemmermann GN, Chyou PH, Kato
I, Perez-Perez GI and Blaser MJ: Helicobacter pylori infection and
gastric carcinoma among Japanese Americans in Hawaii. N Engl J Med.
325:1132–1136. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Parsonnet J, Friedman GD, Vandersteen DP,
Chang Y, Vogelman JH, Orentreich N and Sibley RK: Helicobacter
pylori infection and the risk of gastric carcinoma. N Engl J Med.
325:1127–1131. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Correa P: Helicobacter pylori and gastric
cancer: State of the art. Cancer Epidemiol Biomarkers Prev.
5:477–481. 1996.PubMed/NCBI
|
|
7
|
No authors listed: Schistosomes, liver
flukes and Helicobacter pylori. IARC Working Group on the
Evaluation of Carcinogenic Risks to Humans. Lyon, 7–14 June 1994.
IARC Monogr Eval Carcinog Risks Hum. 61:1–241. 1994.PubMed/NCBI
|
|
8
|
Ferlay J, Shin HR, Bray F, Forman D,
Mathers C and Parkin DM: Estimates of worldwide burden of cancer in
2008: GLOBOCAN 2008. Int J Cancer. 127:2893–2917. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Marshall BJ and Warren JR: Unidentified
curved bacilli in the stomach of patients with gastritis and peptic
ulceration. Lancet. 1:1311–1315. 1984. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Zhu Y, Shu X, Chen J, Xie Y, Xu P, Huang
DQ and Lu NH: Effect of Helicobacter pylori eradication on
oncogenes and cell proliferation. Eur J Clin Invest. 38:628–633.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Yang Z, Shu X, Chen L, Chen J, Xie Y and
Lu NH: Expression of p53-MDM2 feedback loop related proteins in
various gastric pathologies in relation to Helicobacter pylori
infection: Implications in gastric carcinogenesis. Clin Res Hepatol
Gastroenterol. 36:235–243. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yang Z, Yuan XG, Chen J and Lu NH: Is
NEDD4-1 a negative regulator of phosphatase and tensin homolog in
gastric carcinogenesis? World J Gastroenterol. 18:6345–6348. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Yang Z, Yuan XG, Chen J, Luo SW, Luo ZJ
and Lu NH: Reduced expression of PTEN and increased PTEN
phosphorylation at residue Ser380 in gastric cancer tissues: A
novel mechanism of PTEN inactivation. Clin Res Hepatol
Gastroenterol. 37:72–79. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Li W, Xie C, Yang Z, Chen J and Lu NH:
Abnormal DNA-PKcs and Ku 70/80 expression may promote malignant
pathological processes in gastric carcinoma. World J Gastroenterol.
19:6894–6901. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Xie C, Xu LY, Yang Z, Cao XM, Li W and Lu
NH: Expression of gammaH2AX in various gastric pathologies and its
association with Helicobacter pylori infection. Oncol Lett.
7:159–163. 2014.PubMed/NCBI
|
|
17
|
Li J, Yen C, Liaw D, Podsypanina K, Bose
S, Wang SI, Puc J, Miliaresis C, Rodgers L, McCombie R, et al:
PTEN, a putative protein tyrosine phosphatase gene mutated in human
brain, breast, and prostate cancer. Science. 275:1943–1947. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Steck PA, Pershouse MA, Jasser SA, Yung
WK, Lin H, Ligon AH, Langford LA, Baumgard ML, Hattier T, Davis T,
et al: Identification of a candidate tumour suppressor gene, MMAC1,
at chromosome 10q23.3 that is mutated in multiple advanced cancers.
Nat Genet. 15:356–362. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Li DM and Sun H: TEP1, encoded by a
candidate tumor suppressor locus, is a novel protein tyrosine
phosphatase regulated by transforming growth factor beta. Cancer
Res. 57:2124–2129. 1997.PubMed/NCBI
|
|
20
|
Sato K, Tamura G, Tsuchiya T, Endoh Y,
Sakata K, Motoyama T, Usuba O, Kimura W, Terashima M, Nishizuka S,
et al: Analysis of genetic and epigenetic alterations of the PTEN
gene in gastric cancer. Virchows Arch. 440:160–165. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Byun DS, Cho K, Ryu BK, Lee MG, Park JI,
Chae KS, Kim HJ and Chi SG: Frequent monoallelic deletion of PTEN
and its reciprocal associatioin with PIK3CA amplification in
gastric carcinoma. Int J Cancer. 104:318–327. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kang YH, Lee HS and Kim WH: Promoter
methylation and silencing of PTEN in gastric carcinoma. Lab Invest.
82:285–291. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Chun-Zhi Z, Lei H, An-Ling Z, Yan-Chao F,
Xiao Y, Guang-Xiu W, Zhi-Fan J, Pei-Yu P, Qing-Yu Z and Chun-Sheng
K: MicroRNA-221 and microRNA-222 regulate gastric carcinoma cell
proliferation and radioresistance by targeting PTEN. BMC Cancer.
10:3672010. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Xu WT, Yang Z and Lu NH: Roles of PTEN
(Phosphatase and Tensin Homolog) in gastric cancer development and
progression. Asian Pac J Cancer Prev. 15:17–24. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Yang Z, Xie C, Liu G, Cao X, Li W, Xu W
and Lu N: Helicobacter pylori phosphorylates PTEN tumor suppressor
to activate PI3K/Akt signaling pathway. J Gastroenterol Hepatol.
28:1012013.
|
|
26
|
Vazquez F, Ramaswamy S, Nakamura N and
Sellers WR: Phosphorylation of the PTEN tail regulates protein
stability and function. Mol Cell Biol. 20:5010–5018. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Torres J and Pulido R: The tumor
suppressor PTEN is phosphorylated by the protein kinase CK2 at its
C terminus. Implications for PTEN stability to proteasome-mediated
degradation. J Biol Chem. 276:993–998. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Yang Z, Xie C, Xu W, Liu G, Cao X, Li W,
Chen J, Zhu Y, Luo S, Luo Z, et al: Phosphorylation and
inactivation of PTEN at residues Ser380/Thr382/383 induced by
Helicobacter pylori promotes gastric epithelial cell survival
through PI3K/Akt pathway. Oncotarget. 6:31916–31926. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Tamura M, Gu J, Matsumoto K, Aota S,
Parsons R and Yamada KM: Inhibition of cell migration, spreading,
and focal adhesions by tumor suppressor PTEN. Science.
280:1614–1617. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Tamura M, Gu J, Takino T and Yamada KM:
Tumor suppressor PTEN inhibition of cell invasion, migration, and
growth: Differential involvement of focal adhesion kinase and
p130Cas. Cancer Res. 59:442–449. 1999.PubMed/NCBI
|
|
31
|
Tamura M, Gu J, Danen EH, Takino T,
Miyamoto S and Yamada KM: PTEN interactions with focal adhesion
kinase and suppression of the extracellular matrix-dependent
phosphatidylinositol 3-kinase/Akt cell survival pathway. J Biol
Chem. 274:20693–20703. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Gu J, Tamura M, Pankov R, Danen EH, Takino
T, Matsumoto K and Yamada KM: Shc and FAK differentially regulate
cell motility and directionality modulated by PTEN. J Cell Biol.
146:389–403. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Kornberg LJ: Focal adhesion kinase and its
potential involvement in tumor invasion and metastasis. Head Neck.
20:745–752. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
McLean GW, Carragher NO, Avizienyte E,
Evans J, Brunton VG and Frame MC: The role of focal-adhesion kinase
in cancer-a new therapeutic opportunity. Nat Rev Cancer. 5:505–515.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Tabassam FH, Graham DY and Yamaoka Y: OipA
plays a role in Helicobacter pylori-induced focal adhesion kinase
activation and cytoskeletal re-organization. Cell Microbiol.
10:1008–1020. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Tegtmeyer N, Wittelsberger R, Hartig R,
Wessler S, Martinez-Quiles N and Backert S: Serine phosphorylation
of cortactin controls focal adhesion kinase activity and cell
scattering induced by Helicobacter pylori. Cell Host Microbe.
9:520–531. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Wang C, Yang R, Yue D and Zhang Z:
Expression of FAK and PTEN in bronchioloalveolar carcinoma and lung
adenocarcinoma. Lung. 187:104–109. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Wang SY, Hao HL, Deng K, Li Y, Cheng ZY,
Lv C, Liu ZM, Yang J and Pan L: Expression levels of phosphatase
and tensin homolog deleted on chromosome 10 (PTEN) and focal
adhesion kinase in patients with multiple myeloma and their
relationship to clinical stage and extramedullary infiltration.
Leuk Lymphoma. 53:1162–1168. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Zhang LL, Liu J, Lei S, Zhang J, Zhou W
and Yu HG: PTEN inhibits the invasion and metastasis of gastric
cancer via downregulation of FAK expression. Cell Signal.
26:1011–1020. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Kodama M, Murakami K, Nishizono A and
Fujioka T: Animal models for the study of Helicobacter-induced
gastric carcinoma. J Infect Chemother. 10:316–325. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Yan J, Luo YH and Mao YF: Establishment of
Helicobacter pylori infection model in Mongolian gerbils. World J
Gastroenterol. 10:852–855. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Myers MP, Pass I, Batty IH, van der Kaay
J, Stolarov JP, Hemmings BA, Wigler MH, Downes CP and Tonks NK: The
lipid phosphatase activity of PTEN is critical for its tumor
supressor function. Proc Natl Acad Sci USA. 95:pp. 13513–13518.
1998; View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Wroblewski LE, Noble PJ, Pagliocca A,
Pritchard DM, Hart CA, Campbell F, Dodson AR, Dockray GJ and Varro
A: Stimulation of MMP-7 (matrilysin) by Helicobacter pylori in
human gastric epithelial cells: Role in epithelial cell migration.
J Cell Sci. 116:3017–3026. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Al-Ghoul L, Wessler S, Hundertmark T,
Kruger S, Fischer W, Wunder C, Haas R, Roessner A and Naumann M:
Analysis of the type IV secretion system-dependent cell motility of
Helicobacter pylori-infected epithelial cells. Biochem Biophys Res
Commun. 322:860–866. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Schneider S, Weydig C and Wessler S:
Targeting focal adhesions: Helicobacter pylori-host communication
in cell migration. Cell Commun Signal. 6:22008. View Article : Google Scholar : PubMed/NCBI
|