Introduction
Antigen-presenting cells (APCs), in particular
dendritic cells (DCs), regulate immune responses and serve key
functions in the induction of antitumor activity (1). DCs exhibit immunomodulatory functions
using major histocompatibility complex (MHC) class I and II
molecules, co-stimulatory molecules [cluster of differentiation
(CD)80, CD86 and CD40] and molecules required for migration and for
the capture and processing of antigens (2). Primed CD4+ and CD8+ T-lymphocytes
secrete cytokines, including interferon gamma (IFN-γ) and tumor
necrosis factor α (TNF-α), which promote the proliferation of
cytotoxic lymphocytes, the destruction of tumor tissue and the
control and elimination of tumor cells (3). DCs present antigens, in complex with MHC
class I and II molecules, to T and B cells, and demonstrate a high
capacity to present tumor-associated antigens in vitro and
in vivo (4). However, the
functional activity of DCs in patients with cancer is significantly
decreased (5). Antigen-specific
activation of dendritic cells with the formation of an antitumor
cytotoxic immune response is considered a promising potential
method of combating cancer (6). The
purpose of the present study was to assess the phenotypic and
functional characteristics of peripheral blood DC subsets in
patients with non-small cell lung cancer (NSCLC) and the
development of an antitumor cytotoxic response by mononuclear cells
(MNCs) derived from patients using in vitro-generated
antigen-primed DCs. It was hypothesized that cancer is associated
with a decreased proportion of immature and mature DCs in the
peripheral blood, and with an impaired migratory capacity, as
compared with healthy donors. Tumor-derived molecules may affect
the maturation of DCs, preventing them from becoming functionally
active (7). DCs are crucial for
generating antitumor immunity; thus, a large number of studies have
aimed to generate methods for loading DCs with tumor antigens for
use in vivo (6,8). These approaches aim to mobilize the
patient's immune system and circumvent any tumor-derived inhibition
of DC maturation.
Materials and methods
Patients and specimens
In the present study, heparinized peripheral venous
blood samples were obtained from 10 healthy donors, 20 patients
with histologically verified non-small cell lung cancer (NSCLC) at
stages IIA, IIB and IIIA, and tumor biopsy material obtained during
surgery (17 males, 3 females; mean age, 60.7±1.5 years). Clinical
material was obtained perioperativelyin the Regional Clinical
Oncology Center (Novosibirsk, Russia) and City Clinical Hospital
No. 1 (Novosibirsk, Russia) from August 2013 to September 2014. The
inclusion criterion was the lack of chemotherapy and/or
radiotherapy prior to surgery. The exclusion criteria were as
follows: Pregnancy; receiving immunocorrective drugs for
concomitant pathology; rapid progression of the underlying disease,
such that the use of immunotherapy is deontologically unjustified;
individual intolerance to the components of the vaccine and/or the
development of severe side effects in response to any of the
components; refusal of the patient to participate in the study
either orally or in writing; and patient involvement in any other
clinical study. All patients provided written informed consent for
the study and all protocols were approved by the Local Ethics
Committee at the Research Institute of Fundamental and Clinical
Immunology (Novosibirsk, Russia).
Phenotypic and functional
characterization of peripheral blood DC subtypes of patients with
NSCLC
The relative proportion of myeloid and plasmacytoid
DCs, and their phenotype, were evaluated in the whole peripheral
blood of patients with NSCLC and healthy donors. Flow cytometry of
labeled monoclonal antibodies was performed on a BD FACSVerse
device (BD Biosciences, Franklin Lakes, NJ, USA). Data analysis was
performed using the BD FACSDiva Software v.6.1.3 (BD Biosciences,
Franklin Lakes, NJ, USA). Peripheral blood dendritic cell subtypes
were characterized by the following phenotype: Myeloid dendritic
cells (mDC) [cluster of differentiation (CD)3−,
CD14−, CD19−, CD45+, Human
Leukocyte Antigen-Antigen D-Related (HLA-DR)+,
BDCA1+ and BDCA2−] and plasmacytoid dendritic
cells (pDC; CD3−CD14−
CD19−CD45+ HLA-DR+
BDCA1− BDCA2+). For cytometric analysis, the
following antibodies were used: CD3-fluorescein isothiocyanate
(FITC; Sorbent, Moscow, Russia), CD19-FITC (Sorbent),
HLA-DR-phycoerythrin (PE) (Sorbent); CD14-FITC (BD Biosciences),
CD45-V450 (BD Biosciences); BDCA1-APC (BioVision, Inc., Milpitas,
CA, USA), BDCA2-PerCP-C5.5 (BioVision, Inc.).
To evaluate the maturation ability and migration
potential, DCs of the two subsets (myeloid and plasmacytoid DCs)
were analyzed using marker expression of CD83, CD86, C-C chemokine
receptor 7 (CCR7; BD Biosciences) prior to and following
stimulation of maturation by agonists of specific toll-like
receptors (TLR) R848 (20 ng/ml; Resiquimod; BioVision, Inc.)
lipopolysaccharide (LPS; 25 ng/ml) [E. coli 0114:B4
(Sigma-Aldrich; Merck KGaA, Darmstadt, Germany)].
Preparation of autologous tumor cells
and tumor antigens
Tumor cells were isolated from surgical biopsy
samples using mechanical homogenization (Potter homogenizer) and a
metal filter to separate the large detritus. The cells were washed
three times with serum-free RPMI-1640 medium (Biolot, St.
Petersburg, Russia) containing 160 µg/ml gentamicin (Samson-Med,
St. Petersburg, Russia), 200 µg/ml ampicillin and 5 µg/ml
amphotericin B. The resulting cells were divided into three equal
aliquots containing 300,000–1,000,000 cells, depending on the
volume of the tumor. The first aliquot was cultured in complete
RPMI-1640 medium containing 10% fetal calf serum (FCS; PAA
Laboratories, Pasching, Austria), 2 mM L-glutamine (Vector,
Novosibirsk, Russia), 10 mM HEPES buffer (Sigma-Aldrich; Merck
KGaA), 5×10−4 M 2-mercaptoethanol (Sigma-Aldrich; Merck
KGaA), 80 µg/ml gentamicin (Samson) and 100 µg/ml ampicillin
(Sintez) at 37°C in an atmosphere containing 5% CO2 for
10 days. A total of half the supernatant was replaced following 3
and 7 days at 37°C with 5% CO2. The second aliquot was
used to prepare a mixture of tumor antigens produced by three
freeze-thaw cycles, followed by filtration through a 0.45-µm filter
(EMD Millipore, Billerica, MA, USA). The third aliquot of tumor
cells was used to prepare total RNA by centrifuging at 300xg for 7
min. The supernatant was removed and 1 ml TRIzol reagent
(Invitrogen; Thermo Fisher Scientific, Inc., Waltham, MA, USA) was
added and incubated at room temperature for 5 min to disrupt the
nucleoprotein complex. Subsequently, 200 µl chloroform (Himreactiv,
Nizhny Novgorod, Russia) was added and incubated for 3 min at room
temperature, followed by centrifugation at 10,000 × g at 4°C for 15
min. Subsequently, 500 µl from the upper phase was transferred to a
new tube and 500 µl isopropanol (ZAO Himreactiv) was added and
incubated for 10 min at room temperature. The resulting mixture was
centrifuged at 10,000 × g at 4°C for 10 min. The pellet was
resuspended in 1 ml ethanol (70%) and centrifuged at 10,000 × g for
5 min at room temperature. The supernatant was discarded and the
tube was left with the lid open for between 15 and 20 min to enable
the sample to dry. The precipitate was resuspended in between 20
and 100 µl double distilled water. The total protein lysate end RNA
concentration was determined using a NanoDrop device (Thermo Fisher
Scientific, Inc.) with an absorbance ratio of 260/280 nm.
Isolation of mononuclear cells from
the peripheral blood of patients with NSCLC and preparation of an
adherent fraction of mononuclear cells (MNCs)
MNCs were isolated by centrifugation of the whole
blood in the Ficoll-Urografin density gradient (ρ=1.077 g/l). For
this purpose, 6 ml whole blood was layered onto 3 ml
Ficoll-Urografin and centrifuged at 300 × g for 30 min at room
temperature. Cells forming an interphase ring were selected, washed
twice in RPMI-1640 medium (Vector) and resuspended in 5 ml complete
RPMI-1640 medium containing 10% FCS (PAA), 2 mM L-glutamine
(Vector), 10 mM HEPES buffer (Sigma-Aldrich; Merck KGaA),
5×10−4 M 2mercaptoethanol (Sigma-Aldrich; Merck KGaA),
80 µg/ml gentamicin (Samson) and 100 µg/ml ampicillin (Sintez),
transferred to a Petri dish (90 mm; Nalge Nunc International,
Penfield, NY, USA), and incubated at 37°C in an atmosphere
containing 5% CO2 for 2 h. The medium with non-adherent
MNCs was removed and cells were pelleted by centrifugation at 300 ×
g for 10 min at room temperature and stored until use at 37°C in an
atmosphere containing 5% CO2. The surfaces of Petri
dishes were rinsed twice with water and cells were selected using a
cell scraper (BD Biosciences).
Generation of mature antigen-primed
DCs
To produce immature DCs, isolated cells of the
adherent MNC fraction were cultured (1×105 cells/ml) in
complete medium supplemented with recombinant human (rh)
granulocyte-macrophage colony-stimulating factor (GM-CSF; 50 ng/ml)
and rh interleukin (IL-) 4 (100 ng/ml; PeproTech, Inc., Rocky Hill,
NJ, USA) in 48-well plates (Linbro, San Rafael, CA, USA) for 72 h.
DC populations were identified by evaluating the expression of
CD14, HLA-DR, and CD86 markers at the stage of immature and mature
DCs and the ability of dendritic cells for phagocytosis of
FITC-dextran. After 72 h of culturing, the immature DC culture was
divided into three parts. Antigens in the form of a tumor cell
lysate (with protein concentration of 100 µg/ml, for 18 h) were
added to the first part. In the second part, immature DCs were
transfected with tumor cell RNA using Promokine reagents. DCs from
the third part were not exposed to any antigens and were used as
control group [DCs(0) without any antigens]. After 24 h, immature
DCs were added with 25 ng/ml of rh tumor-necrosis factor (TNF)-α
and 10 ng/ml of rhIL1-β for 24 h to produce mature DCs. To generate
DCs transfected with tumor cell RNA, we performed magnetic
transfection using Promokine reagents (PromoCell GmbH, Heidelberg,
Germany), according to the manufacturer's protocol. RNA was
dissolved in Dulbecco's Modified Eagle's Medium (DMEM; Vector),
added with a MATra-A reagent at the ratio of 0.3 µg RNA/0.3 µl
reagent, and incubated at room temperature for 20 min. During
incubation, the medium in wells with dendritic cells was changed:
RPMI-1640 medium was removed, and 250 µl DMEM was added.
Subsequently, the RNA-MATra-A complex was added to cells in wells
in the amount of 25 µl/well, and a plate was placed on a magnetic
board for 15 min. The medium in wells was replaced following
transfection: DMEM was removed and 300 µl RPMI-1640 medium
supplemented with 10% FCS (PAA), 2 mM L-glutamine (Vector), 10 mM
HEPES (Sigma, USA), 5×10−4 M 2-mercaptoethanol
(Sigma-Aldrich; Merck KGaA), 80 µg/ml gentamicin and 100 µg/ml
ampicillin was added. Transfected cells were incubated at 37°C in
an atmosphere containing 5% CO2 for 24 h, prior to the
addition of maturation factors (25 ng/mlrhTNF-α and 10 ng/ml
rhIL1-β).
Analysis of phenotypic and functional
indicators of generated DCs
The phenotypic characteristics of DCs were studied
using fluorochrome labeled monoclonal antibodies analyzed on a
FACSAria flow cytometer (BD Biosciences). Cells were incubated with
antibodies at 37°C for 1 h, washed in PBS, and fixed in a 1%
formalin solution. The following primary antibodies were used to
evaluate the phenotypic and functional characteristics: CD3-FITC
(Sorbent), CD19-FITC (Sorbent), HLA-DR-phycoerythrin (PE; Sorbent);
CD14-FITC (cat. no. 555937; BD Biosciences), CD45-V450 (cat. no.
560367; BD Biosciences); CD83-APC (cat. no. 551073; BD
Biosciences), CD86-PerCp-Cy5.5 (cat. no. 561129; BD Biosciences),
C-C chemokine receptor 7-PE (CCR7; cat. no. 552176; BD
Biosciences). All antibodies were used according to the
manufacturer's protocol.
To evaluate the capability of produced DCs for
antigen capturing, the receptor-mediated endocytosis efficiency was
determined using FITC-dextran (Sigma-Aldrich; Merck KGaA). For this
purpose, cells were incubated with FITC-dextran (1 µg/ml) in
complete medium containing 10% FCS (PAA), 2 mM L-glutamine
(Vector), 10 mM HEPES buffer (Sigma-Aldrich; Merck KGaA),
5×10−4 M 2-mercaptoethanol (Sigma-Aldrich; Merck KGaA),
80 µg/ml gentamicin (Samson) and 100 µg/ml ampicillin (Sintez) at 4
and 37°C for 30 min each. At 4°C, dextran binds to surface
receptors, and at 37°C the bound dextran enters the cell (direct
endocytosis). The analysis was performed on a FACSAria flow
cytometer (BD Biosciences), on the basis of the fluorescence
intensity in a FITC channel. The results are presented as the
difference in the fluorescence intensity of cells capturing dextran
at 4 and 37°C, described in the following equation: ΔMean
fluorescence intensity (MFI)=[MFI (37°C)-MFI (4°C)].
Co-culturing of antigen-primed DCs and
MNCs of the non-adherent fraction from peripheral blood of patients
with NSCLC
Produced antigen-primed DCs were co-cultured with
MNCs of the non-adherent fraction in complete RPMI 1640 medium
(ratio of DC: MNC, 1:10) for 5 days. Non-adherent MNCs cultured in
the presence of DCs (0), not exposed to tumor antigens, [MNC+DC (0)
group] were used as a control.
Analysis of the impact of
antigen-primed DCs on the cytotoxic activity of MNCs against
autologous NSCLC cells
Following the co-culture of MNCs of the non-adherent
fraction and DCs primed with tumor antigens for 5 days, the cell
suspension was centrifugedat 300 × g for 10 min at room
temperature. Subsequently, the resulting cells and autologous tumor
cells were co-cultured at a ratio of 10:1 ratio, respectively, in
96-well round bottom plates (TPP Techno Plastic Products AG,
Trasadingen, Switzerland), 100 µl culture/well, with the cell
concentration of 1×106 cells/ml for 18 h. The analysis
was conducted using a CytoTox 96 Non-Radioactive Cytotoxicity Assay
(Promega Corporation, Madison, WI, USA), according to the
manufacturer's protocol. The cytotoxic effect was calculated using
the formula proposed by the assay manufacturer and presented as the
cytotoxicity percentage.
Investigation of the effector
potential of MNCs induced by antigen-primed DCs
Following the co-culture of MNCs of the non-adherent
fraction and antigen-primed DCs, and culture of control cells, for
5 days, the cell suspension was washed by centrifugation with PBS
at300 × g for 10 min at room temperature. Subsequently, the level
of MNCs expressing surface markers, including CD178, CD107a,
CD253and the intracellular protein perforin or granzyme B, was
determined in the total lymphocyte population and in
CD8+ lymphocytes using flow cytometry with a BD
FACSVerse device (BD Biosciences). Data analysis was performed
using BD FACSDiva Software v.6.1.3 (BD Biosciences). The following
antibodies were used according to the manufacturer's protocol:
CD178-PE (cat. no. 564261; BD Biosciences), CD107a-FITC (cat. no.
555800; BD Biosciences), CD253-PE (cat. no. 550516; BD
Biosciences), perforin-FITC (cat. no. 556577; BD Biosciences) and
granzyme В-PE (cat. no. 561142; BD Biosciences).
Statistical analysis
Statistical analysis of the data was performed using
Statistica 6.0 software (StatSoft Ltd., Bedford, UK) and GraphPad
Prism v.6.01 (GraphPad Software, Inc., La Jolla, CA, USA). Data
were presented as a median and a quartile range of values (25 and
75%). A non-parametric Wilcoxon test was used to identify
significant differences when comparing two groups. In the analysis
of more than two groups, the statistical significance of
differences was determined using Kruskal-Wallis ANOVA and multiple
comparisons test. To perform the post hoc analysis, Tukey's test
was used in GraphPad Prism software. P<0.05 was considered to
indicate a statistically significant difference. In figure
captions, the number of individuals in a group is denoted as n.
Results
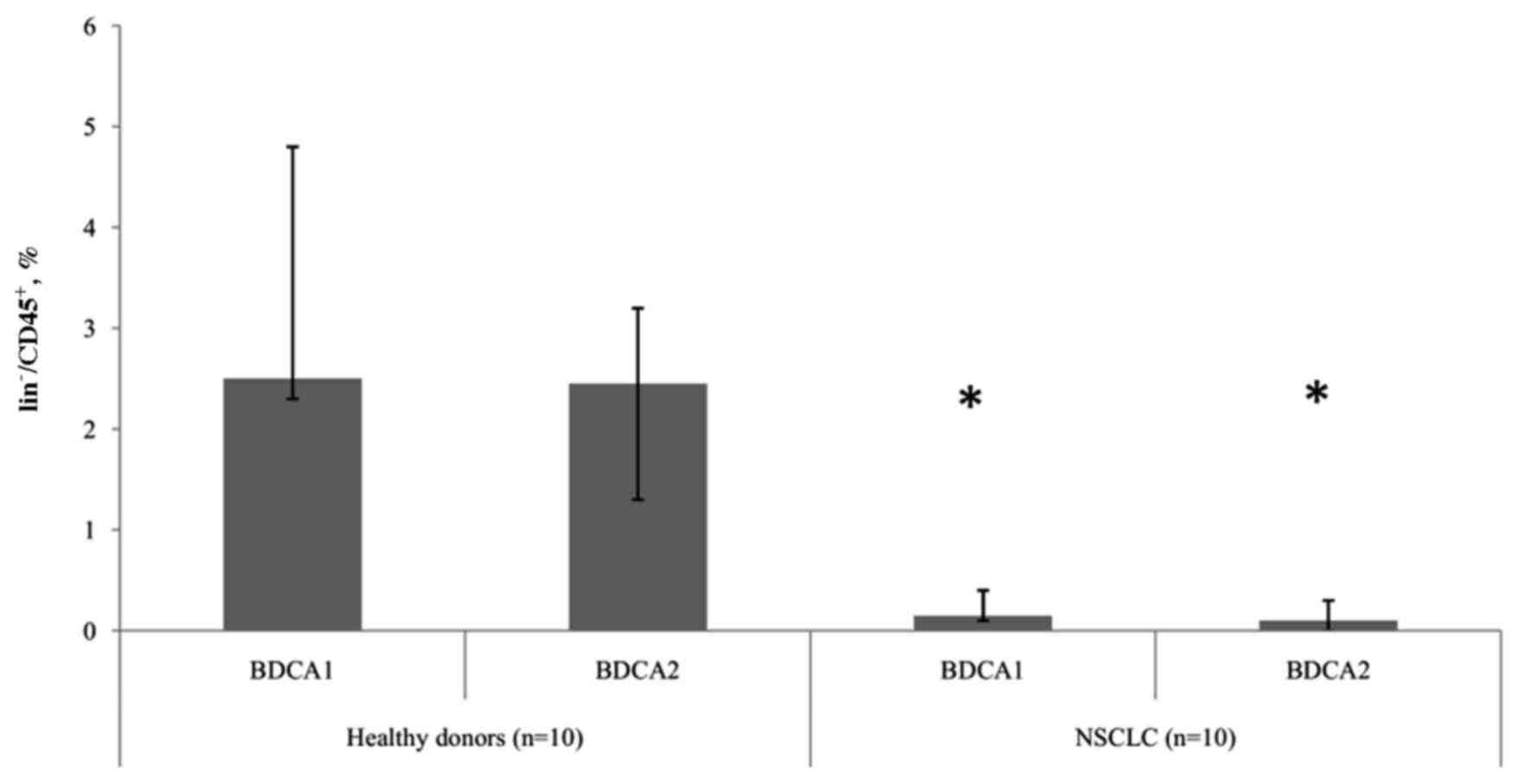
Patients with NSCLC exhibit decreased
levels of mDCs and pDCs
The present study revealed differences in the
relative number of DC subtypes between groups of healthy donors and
patients with NSCLC. In patients with tumors, significantly
(P<0.05) decreased levels of mDCs and pDCs were observed,
compared with healthy donors, indicating a depletion in the
circulating DCs in NSCLC (Fig. 1).
However, there were no statistically significant differences
determined in the ratio of peripheral blood mDC and pDC subtypes
between healthy donors and patients with NSCLC.
CD86 expression is decreased in
patients with NSCLC
Decreased function of DCs in malignant diseases
results from insufficient DC maturation. This is associated with
phenotypic differences in the expression of membrane-bound markers
and differences in their ability to capture an antigen, process it,
and migrate to lymphoid organs for antigen presentation. While DCs
are maturing, the expression of co-stimulatory molecules on their
surface increases (8). Expression of
CD86 begins early during maturation, whereas CD83 expression is
typical of more mature DCs (9). To
analyze the ability of DCs from patients with NSCLC to mature, TLR
4, 7, and 8 agonists, R848 and LPS were used as they are capable of
inducing maturation in mDCs and pDCs. The results of the present
study revealed that, prior to stimulation, the expression of CD86
on mDCs and pDCs in patients with NSCLC was significantly
decreased, compared with that in healthy donors. A significant
increase in the number of mDCs and pDCs expressing CD86 in response
to stimulation with LPS and R848 was observed in healthy controls
and tumor patients. Additionally, the pDC population from patients
with NSCLC expressed significantly decreased CD86 following
stimulation, compared with that in healthy donors (Fig. 2).
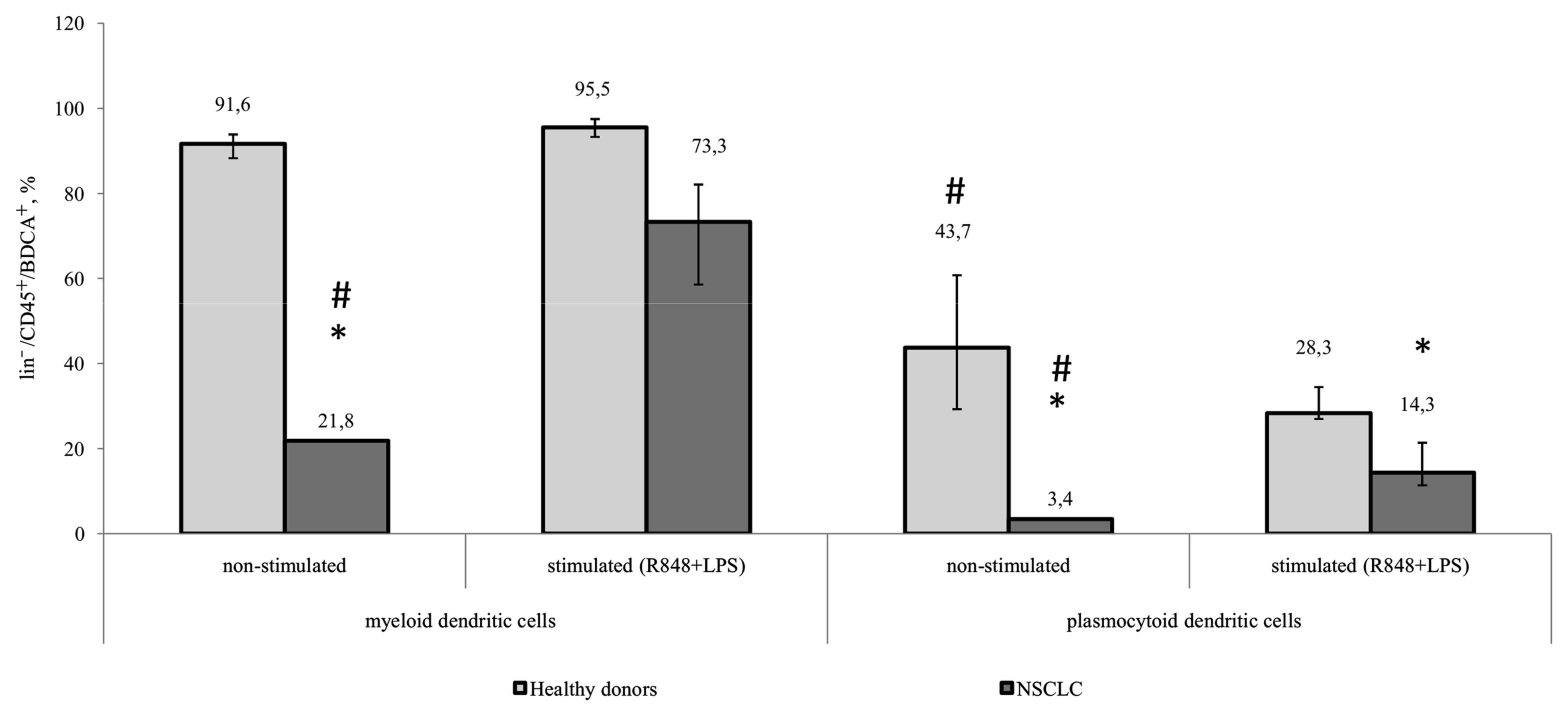
Proportion of CD83+mDCs
from patients with NSCLS is decreased
The expression of CD83 in the myeloid and
plasmocytoid DC subsets in healthy donors and patients with NSCLC
was not determined in unstimulated samples. However, a significant
increase in the number of CD83-expressing cells was observed in all
groups following stimulation. The number of CD83+mDCs
from patients with NSCLC following TLR stimulation was
significantly decreased compared with mDCs from healthy donors
(Fig. 3).
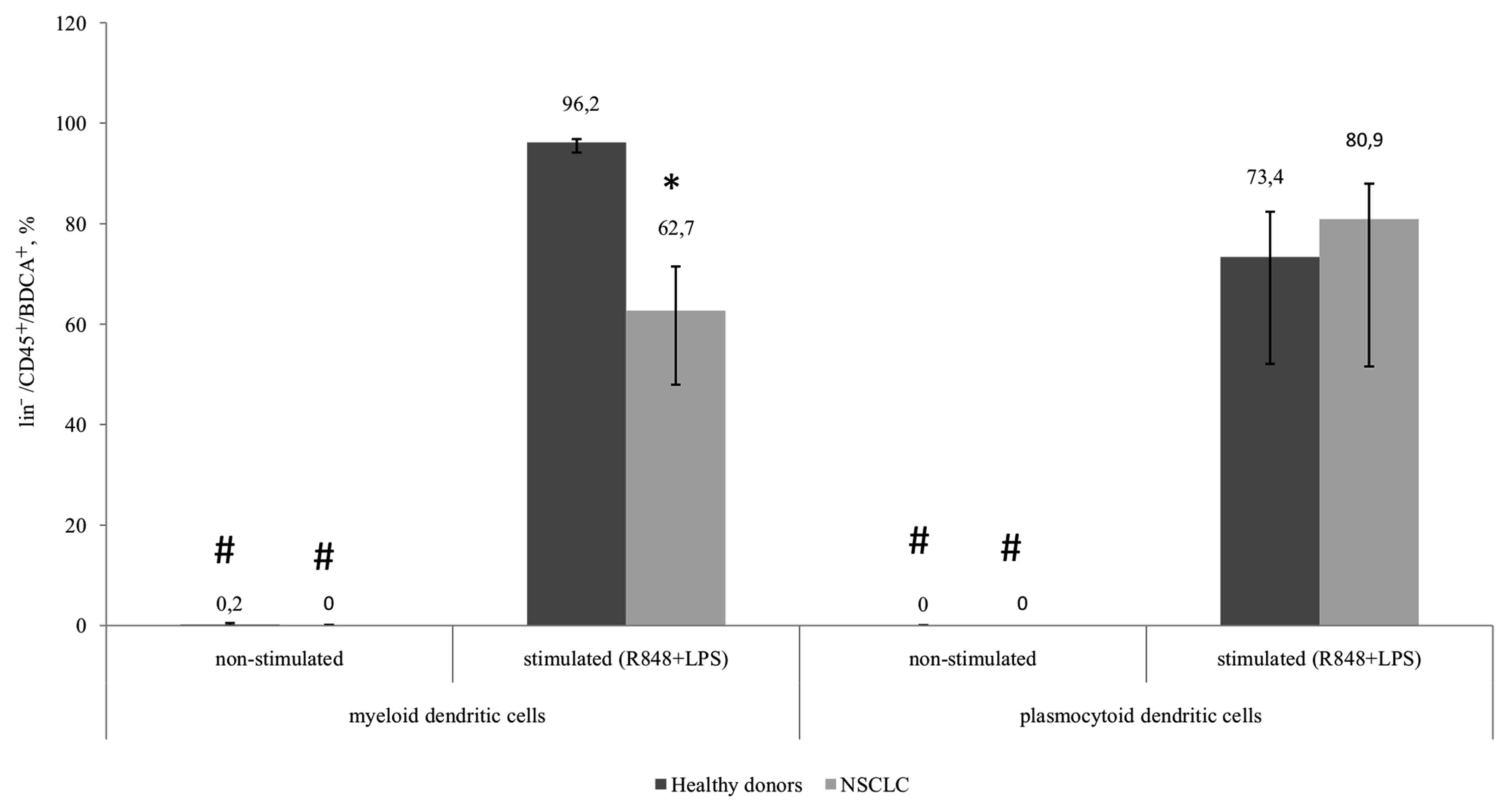 | Figure 3.Relative amount of myeloid and
plasmocytoid dendritic cells expressing the CD83 marker among
lin−/CD45+/BDCA+ cells. Healthy
donors, n=10; patients with NSCLC, n=10. The data are presented as
the median and interquartile range. *P<0.05 vs. corresponding
groups of healthy donors; #P<0.05 vs. corresponding
group of stimulated cultures. CD, cluster of differentiation;
NSCLC, non-small cell lung cancer; BDCA, blood dendritic cell
antigen; Lin, lineage antigen, including СD3, CD14, CD19, CD20,
CD16, CD56; CD, cluster of differentiation; R848, Resiquimod; LPS,
lipopolysaccharide serotype 0114:B4. |
The functional characteristics of tumor-derived DCs
were additionally evaluated. The chemokine receptor, CCR7, was used
as an indicator of migration ability. The number of CCR7-expressing
cells from patients with NSCLC prior to and following stimulation
was lower than the flow cytometer detection limit; whereas, a
significant increase in the number of CCR7-expressing cells in
response to stimulation with R848 and LPS occurred in healthy
donors (data not presented).
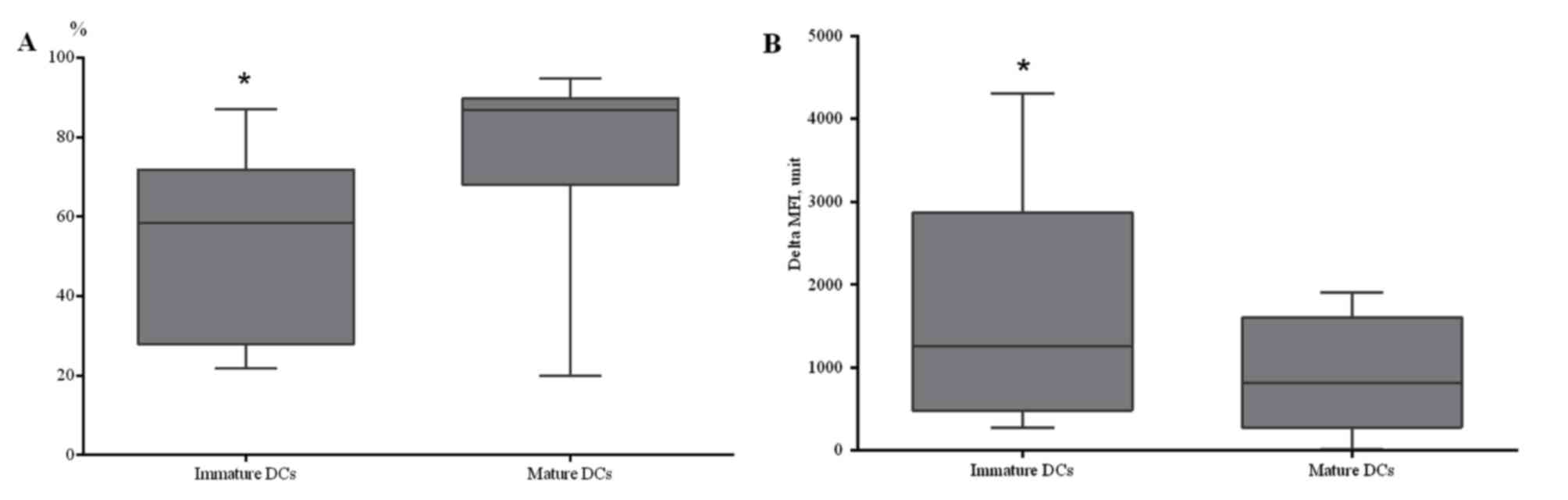
DCs generated from patients with NSCLC
are capable of endocytosis during early maturation stages
A potential approach to the immunotherapy of
patients with NSCLC is the development of vaccines with in
vitro induction of efficient antigen-presenting DCs. In the
present study, the possibility of generating mature, functionally
active DCs, from peripheral blood MNCs isolated from patients with
NSCLC, was investigated. To prepare immature DCs, cells from the
adherent MNC fraction were cultured in complete media supplemented
with rhGM-CSF and rhIL-4 for 4 days. To generate mature DCs, the
immature DCs were added with rhTNF-α and rhIL-1-β and cultured for
24 h. A statistically significant increase in CD86+ DCs
was identified in DCs generated from patients with NSCLC and
stimulated using TNF-α (Fig. 4A).
To determine the capability of in
vitro-derived DCs for antigen capturing, receptor-mediated
endocytosis of dextran was evaluated at 4 and 37°C. A decreased
endocytic ability was identified in mature DCs, compared with
immature DCs, which indicated acquisition of the functional
characteristics typical of the mature state of generated DCs
(Fig. 4B). Therefore, it was
demonstrated that DCs generated from the peripheral blood of
patients with NSCLC are capable of endocytosis during early
maturation stages, while during later maturation stages, DCs lose
the ability to capture antigen.
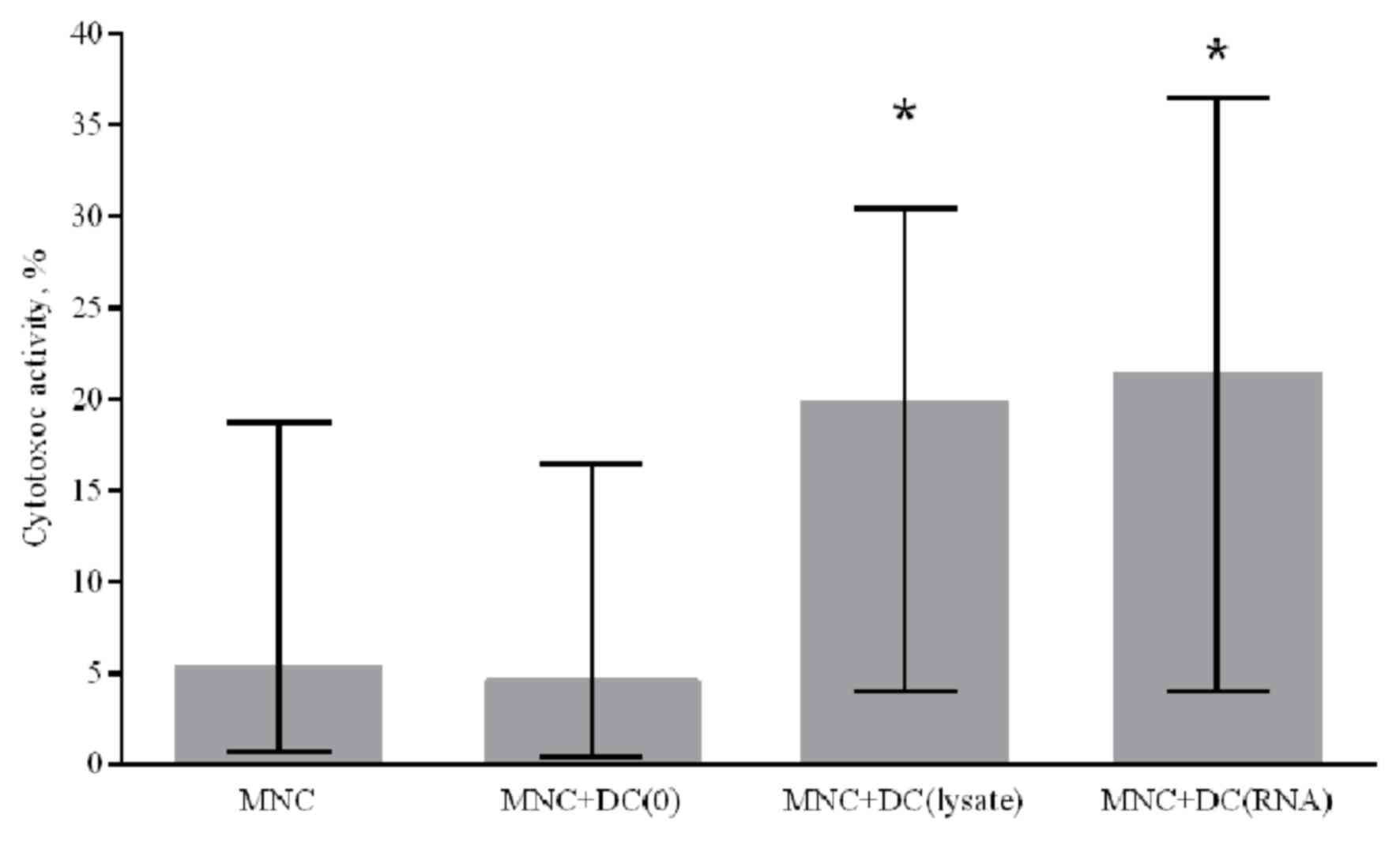
Generated DCs increase the cytotoxic
activity of MNCs against autologous NSCLC cells
To assess the ability of in vitro-derived DCs
to induce a specific antitumor immune response, a cytotoxic
antitumor response was stimulated in the MNC culture in
vitro. Immature DCs generated from patients with NSCLC were
primed with a lysate of tumor cells or transfected with RNA from
tumor cells. To determine the cytotoxic activity of MNCs against
autologous tumor cells, antigen-primed mature DCs were co-cultured
with peripheral blood MNCs of the non-adherent fraction from NSCLC
patients for 5 days. Generated DCs primed with a tumor cell lysate
and DCs transfected with tumor cell RNA were demonstrated to
increase the cytotoxic activity of MNCs against autologous NSCLC
cells by 4.3 and 4.7 times, respectively, compared with the control
group where DCs were not primed with tumor antigens (Fig. 5).
Lysate-primed DCs and RNA-transfected
DCs increase the relative level of perforin-bearing cells
To validate the mechanism of the cytotoxicity
induced during the co-culture of MNCs and DCs, the number of
CD178+, CD107a+, CD253+,
perforin+, and granzyme B+ cells in the total
lymphocyte population and CD8+ cells were determined on
the fifth day of incubation. There were no statistically
significant changes in the expression of CD178, CD107a, and CD253,
which are markers of the Fas ligand and TNF-related apoptosis
inducing ligand (TRAIL)-dependent cytotoxic pathways and direct
cytolysis of target cells (data not presented). Effector cells are
capable of producing lytic granules containing perforin and
granzymes. The accumulation of perforin in the total lymphocyte
population and CD8+ cells following co-culture
demonstrated that lysate-primed DCs and RNA-transfected DCs
increase the relative level of perforin-bearing cells in the
lymphocyte population by 1.6 and 2 times, respectively, and in CD8
cells by 2 times compared with the control group (Fig. 6).
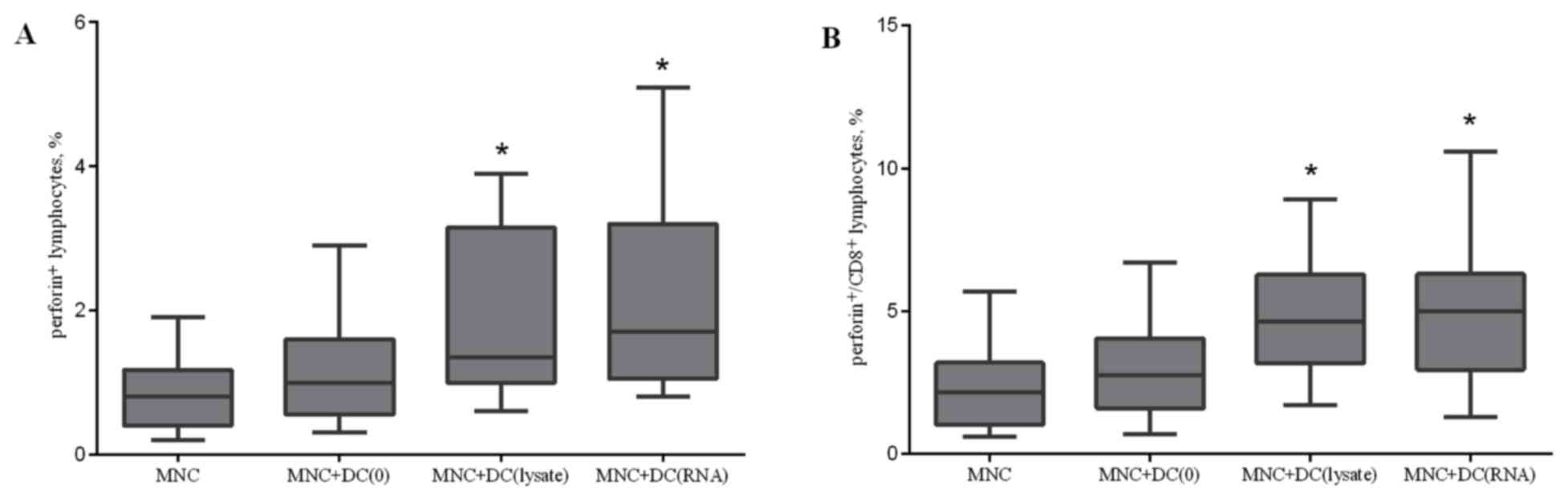 | Figure 6.Effect of antigen-primed DCs on the
relative amount of (A) perforin+ lymphocytes; and (B)
perforin+/CD8+ lymphocytes in the co-culture
of MNCs from patients with NSCLC, n=16. The error bars show a
minimum and maximum of values, boxing is a quartile, and the inner
line is a median. *P<0.05 vs. the group MNC determined using
Kruskal-Wallis ANOVA and multiple comparisons test. NSCLC,
non-small cell lung cancer; DC, dendritic cells; CD, cluster of
differentiation; MNC, mononuclear cells of the non-adherent
fraction; MNC+DC(0), MNCs cultured with non-primed DCs;
MNC+DC(lysate), MNCs cultured with lysate-treated DCs; MNC+DC(RNA),
MNCs cultured with tumor RNA-treated DCs. |
Co-culturing MNCs with antigen-primed
DCs increases the relative level of granzyme B positive cells
The accumulation of granzyme B in lytic granules was
investigated. Co-culturing MNCs with antigen-primed DCs led to an
increase in the relative level of granzyme B positive cells in the
total lymphocyte population and in CD8+ cytotoxic T
lymphocytes (Fig. 7). Similar results
were observed when analysis of the relative level of double
perforin and granzyme B positive cells after co-culturing of MNCs
and DCs was performed (data not presented).
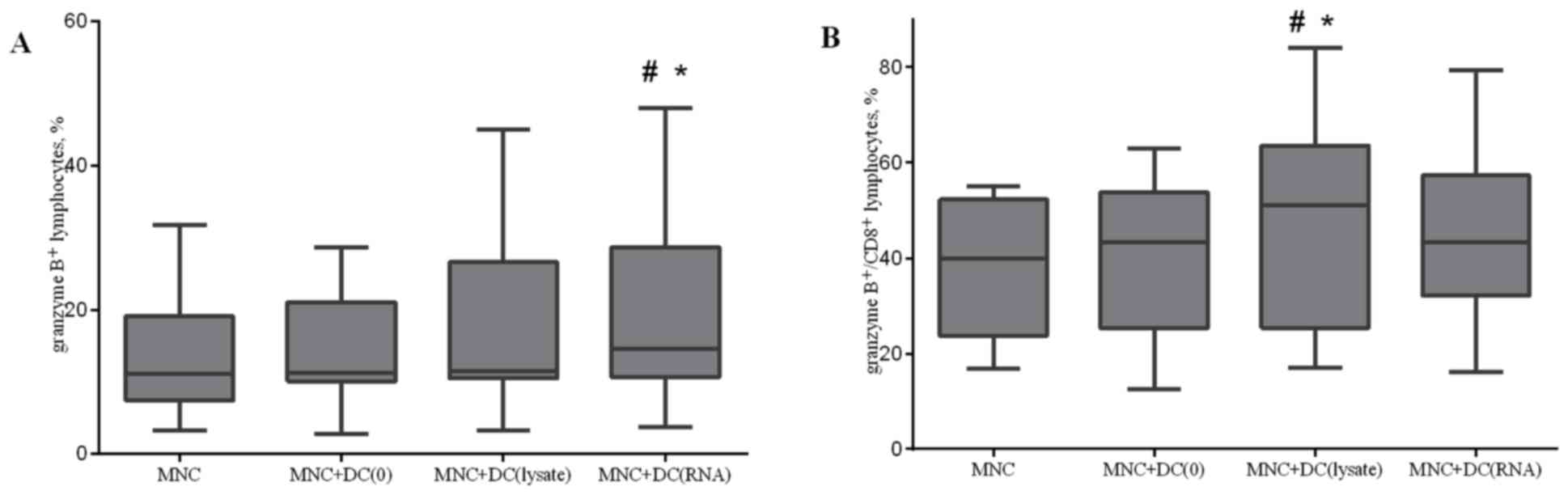 | Figure 7.Effect of antigen-primed DCs on the
relative amount of (A) granzyme B+ lymphocytes; and (B)
granzyme B+/CD8+ lymphocytes in the
co-culture of MNCs from NSCLC patients, n=16. The error bars show a
minimum and maximum of values, boxing is a quartile, and the inner
line is a median *P<0.05 vs. group MNC and #P<0.05
vs. group MNC+DC(0), determined using Kruskal-Wallis ANOVA and
multiple comparisons test. NSCLC, non-small cell lung cancer; DC,
dendritic cells; CD, cluster of differentiation; MNC, mononuclear
cells of the non-adherent fraction; MNC+DC(0), MNCs cultured with
non-primed DCs; MNC+DC(lysate), MNCs cultured with lysate-treated
DCs; MNC+DC(RNA), MNCs cultured with tumor RNA-treated DCs. |
The results of the present study demonstrated that
patients with NSCLC exhibited a decrease in mDCs and pDCs, compared
with healthy donors. Additionally, DCs from patients with NSCLC
exhibited impaired function, resulting in poor induction of an
antitumor immune response. However, stimulation with agonists of
TLR 4, 7, and 8 (R848 and LPS) resulted in partial recovery of the
ability of DCs to reach a mature state. The use of antigen-primed
DCs, which were generated from peripheral blood MNCs of patients
with NSCLC for 5 days, to produce an antitumor cytotoxic response
in vitro led to activation of effector cells, which resulted
in an increased cytotoxic activity of MNCs and increased
perforin+ and granzyme B+ lymphocytes and
CD8+ cells. Despite patients with NSCLC exhibiting
decreased mDCs and pDCs, compared with healthy donors, and impaired
functional capabilities of DCs, it was possible to generate mature,
functionally and phenotypically normal DCs, in vitro,
capable of inducing a cytotoxic immune response upon culture with
autologous MNCs.
Discussion
A number of oncological diseases, including NSCLC,
are associated with a decrease in the absolute and relative number
of immature and mature peripheral blood circulating DCs, compared
with healthy donors (10–12). Additionally, there are various
impairments to the ability of DCs to migrate and interact with
other cells (13). There is
insufficient data regarding peripheral blood DC subsets in NSCLC at
present. The present study revealed differences in the relative
number of DC subtypes between groups of healthy donors and patients
with NSCLC. A significant decrease in the level of mDCs and pDCs
was observed in the patients, compared with healthy donors,
indicating a depletion of the total pool of circulating DCs in
patients with NSCLC, which was observed by Domingues et al
(14). In the present study, there
were no significant differences in the ratio of peripheral blood
mDCs and pDCs between healthy donors and patients with NSCLC. DCs
are required to express appropriate co-stimulatory markers on the
membrane to efficiently present an antigen to cytotoxic T
lymphocytes (15,16). To assess the potential of circulating
DCs for maturation, agonists of TLR 4, 7, and 8 (R-848 and LPS)
were used which are capable of inducing maturation of mDCs and
pDCs, with subsequent evaluation of the expression level of
co-stimulatory molecules. In blood samples prior to LPS and R848
stimulation for the two DC subsets, the number of CD86-expressing
cells in patients with NSCLC was significantly decreased, compared
with that in healthy donors. A significant increase in the
expression of CD86 in mDCs and pDCs, in response to stimulation
with LPS and R848, was observed in all groups. In the present
study, the number of CD86-bearing cells in the pDC population from
patients with NSCLC, following stimulation, was decreased, compared
with that in healthy donors. Expression of CD83 in DC subsets, in
healthy donors and in tumor patients, was not detected. However, a
significant increase in the expression level of CD8, following
after stimulation with R848 and LPS, was observed in all groups.
The relative amount of CD83+ cells among mDCs in
patients with NSCLC following stimulation was significantly
decreased, compared with that in the healthy donors, indicating the
different stages of DC differentiation in NSCLC.
Impaired function of peripheral blood DCs was
observed in patients with NSCLC compared with healthy donors in the
present study. In the present study, the number of cells expressing
CCR7 was analyzed, which is associated with the ability of cells to
migrate to the areas of antigen presentation and co-stimulation of
T cells because its ligand CCL19 is expressed in T cell areas of
secondary lymphoid organs (17). The
number of CCR7-expressing cells in the patients, prior to and
subsequent to stimulation, was below the flow cytometer detection
limit, whereas a significant increase in the number of
CCR7-expressing cells, in response to R848 and LPS stimulators,
occurred in healthy donors. These results indicated an alteration
in the migration ability of antigen-presenting cells into naive T
cell areas within the lymph nodes, and may be the cause of failure
or impairment of the antitumor immune response in NSCLC. Thus, the
study of peripheral circulating DCs in patients with NSCLC revealed
a number of features that may explain a high immunosuppression
level accompanying the development of malignant lung tumors. A
decrease in the number of mDCs and pDCs, compared with healthy
donors, and a disturbance in the differentiation and functional
activity of the DCs were identified in the present study. These
results may prompt the generation of functionally active DCs from
the peripheral blood of patients with NSCLC for use in
immunotherapy regimes. The stimulation-induced recovery of DC
maturation enables DCs to be a target for therapeutic intervention
and indicates that DCs themselves may be a treatment option. To
date, there are a number of approaches for generating DCs in
vitro, including preparation of DCs from peripheral blood
monocytes and bone marrow progenitors (CD34+ cells)
(18). Furthermore, a wide range of
factors may be used for differentiation and maturation of DCs
(GM-CSF, IL-4, interferon-α, IL-2, IL-6, IL-15, TGF-α, stem cell
factor, FLT-3 ligand and prostaglandin E2) that may facilitate, in
a combination or individually, the generation of DCs from monocytes
or CD34+ cells (19–23). The
present study suggested that there is some danger in using numerous
factors for differentiation and maturation of DCs that can lead to
the production of DC subsets with a variety of phenotypes and
functions. In the present study, DCs were generated from peripheral
blood MNCs from patients with NSCLC. To produce immature DCs, the
adherent fraction of MNCs was cultured with rhGM-CSF and rhIL-4. To
generate mature DCs, rhTNF-α and rhIL1-β were added to immature
DCs. This approach resulted in a DC culture with phenotypic and
functional characteristics typical of mature DCs. The main
advantage of using DCs in developing anticancer vaccines is the
possibility to generate, in vitro, antigen presenting cells
with certain characteristics required for a normal antitumor immune
response (24). Additionally, an
important feature of DCs that distinguishes them from other
antigen-presenting cells is their ability to cross-present antigens
(25). It was previously demonstrated
that treatment of a patient with cancer with a DC-based vaccine led
to a considerable reduction in the functional activity of DCs
(26). Therefore, loading DCs with
tumor antigens and induce maturation was required, as this would
promote activation of an antigen-specific immune response and
prevent the immunosuppressive influence of the tumor.
The source of tumor antigens serves an important
function in their immunogenicity (27–29). Tumor
cell lysates, tumor cell RNA, recombinant proteins of
tumor-associated antigens, and DNA constructs encoding
tumor-associated antigens have been used as antigens (21,30,31).
Loading DCs with a tumor lysate enables presentation of a range of
tumor antigens from a patient (32).
The use of a tumor lysate as a tumor associated antigen source may
have the advantage in stimulating a polyclonal immune response, as
they may stimulate CD4+ T helpers and CD8+
cytotoxic T lymphocytes, thereby reducing the likelihood for tumor
escape (33). This method decreases
the time and effort required for identification and synthesis of
certain immunodominant peptide epitopes, allowing DCs to process
naturally occurring tumor antigens and stimulate a natural
cytotoxic response (34,35). The use of a tumor lysate as a source
of antigens for loading DCs is more efficient than the use of
separate peptides (33). Autologous
tumor cells from the patient contain a range of antigens (36), which may lead to the generation of a
wider range of cytotoxic T lymphocytes (37). However, the use of a tumor cell lysate
as a source of antigens is associated with immunosuppression
(38). The use of tumor cell RNA to
transfect DCs may prevent this adverse effect (39,40).
One of the potential and promising areas in cancer
immunotherapy is the use of T cells co-cultured, and thereby
‘trained’, in the presence of antigen-primed DCs. One mature
differentiated DC is capable of efficiently activating ~100 T
cells, whereupon they may implement an immune response (41). Therefore, activation of the cellular
immune response may be achieved during in vitro co-culturing
of DCs and T cells, thereby, avoiding the negative influence of
tumor growth products depressing the functional activity of mature
antigen-primed DCs administered to the patient (42,43). After
co-culturing MNCs and DCs, a lactate dehydrogenase (LDH)
cytotoxicity test, which is on the basis of the release of LDH from
lysed tumor cells in vitro, to study the stimulating effect
of antigen-primed DCs on the cytotoxic activity of tumor MNCs
against NSCLC cells. Generated DCs primed with a tumor cell lysate
and DCs transfected with tumor cell RNA were able to enhance the
cytotoxic activity of MNCs against autologous NSCLC cells by 4.3
and 4.7 times, respectively, compared with the control group of DCs
not primed with tumor antigens. In the present study, cytotoxic T
lymphocytes induced apoptosis of target cells via the accumulation
of perforin and granzyme granules. Perforin is a pore-forming
protein that has homology with the C9 complement component and is
synthesized as an inactive precursor cleaved at the C-terminus to
yield the active form (44). Perforin
incorporates into the membrane of the target cell and may promote
cell entry of granzyme B and enhance the cytotoxic effect of T
lymphocytes (45). Additionally, Fas-
and TRAIL-ligands act as apoptotic inducers (46). The Fas-ligand-mediated mechanism of
apoptosis is involved in the elimination of unintended cells,
thereby reducing the risk of developing tumor cells (47). Cytotoxic T lymphocytes use Fas-ligand
to activate caspases that directly induce apoptosis (48). TRAIL is a death receptor that belongs
to the family of TNF receptors (49).
Fas and TRAIL apoptosis responds to certain stimuli (e.g., an
increase in the expression of the corresponding ligands) and causes
DNA damage and protein p53 activation, which leads to the release
of mitochondrial pro-apoptotic factors triggering caspases. The
TRAIL mechanism may be induced by factors including nuclear factor
k-light-chain-enhancer of activated B cells, mitogen-activated
protein kinase and protein kinase B (50). To study the mechanisms of cytotoxic
activity observed in the co-culture of MNCs and DCs, the relative
level of CD178+, CD107a+, CD253+,
perforin+, and granzyme+ cells in the total
lymphocyte population and CD8+ cells was determined on
the fifth day of incubation. There were no significant changes
identified in the expression of CD178, CD107a, and CD253, which are
markers of FasL- and TRAIL-dependent pathways of cytotoxicity. The
accumulation of perforin in the total lymphocyte population and
CD8+ cells, following co-culture, demonstrates a
stimulating effect of lysate-primed DCs and RNA-transfected DCs.
DCs treated with tumor cell RNA as a source of antigens have a
stimulating effect, similar to that of lysate-primed DCs, on the
cytotoxic activity as observed with an increase in
perforin+ and granzyme B+ lymphocytes and
CD8+ cells. These results suggested that tumor cell RNA
may be used with tumor cell lysates, particularly in situations
where antigenic material may be unavailable, undesirable because of
a possible immunosuppressive effect, or insufficient in quantity
for preparation of a tumor cell lysate.
Patients with NSCLC were identified to exhibit a
decreased number of all peripheral blood DC subsets, compared with
healthy donors. Additionally, DCs revealed an impaired ability to
undergo maturation. DCs generated from peripheral blood MNCs of
patients with NSCLC and primed by a lysate and tumor cell RNA were
able to stimulate the cytotoxic activity of MNCs against autologous
tumor cells in vitro. The observed ability of DCs loaded
with tumor antigens (lysate or RNA) to increase the cytotoxic
activity of MNCs against NSCLC cells in vitro indicates
effective activation of T lymphocytes by the generated DCs. The
effector reactions induced by antigen-primed DCs occurred primarily
through a perforin-granzyme B-dependent cytotoxic pathway, which
was demonstrated by an increase in perforin+ and
granzyme B+ CD8+cells, following co-culture
of MNCs and antigen-primed DCs and by the lack of statistically
significant changes in markers of Fas- and TRAIL-dependent
cytotoxic pathways.
To conclude, the disturbances to the phenotype and
functional capabilities of peripheral blood DC subsets in patients
with NSCLC could be overcome by TLR stimulation. Thus, autologous
DCs may be considered for use in patients with NSCLC as a target
for therapeutic strategies, and as the basis for cellular
immunotherapy and induction of an antitumor cytotoxic immune
response.
Acknowledgements
The present study was supported by the Federal
Target Program ‘Research and development in priority areas of the
Russian scientific and technological complex in 2014–2020’,
agreement no. 14.607.21.0043, unique identifier:
RFMEFI60714X0043.
References
|
1
|
Banchereau J and Steinman RM: Dendritic
cells and the control of immunity. Nature. 392:245–252. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Randolph GJ, Ochando J and Patrida-Sánchez
S: Migration of dendritic cell subsets and their precursors. Annu
Rev Immunol. 26:293–316. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Dunn GP, Koebel CM and Schreiber RD:
Interferons, immunity and cancer immunoediting. Nat Rev Immunol.
6:836–848. 2006. View
Article : Google Scholar : PubMed/NCBI
|
|
4
|
Lipscomb MF and Masten BJ: Dendrititc
cells: Immune regulators in health and disease. Physiol Rev.
82:97–130. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Sennikov SV, Obleukhova IA, Kurilin VV,
Kulikova EV and Khristin AA: Features of functional activity of
dendritic cells in tumor growth. Vopr Onkol. 61:556–62.
2015.PubMed/NCBI
|
|
6
|
Bol KF, Schreibelt G, Gerritsen WR, de
Vries IJ and Figdor CG: Dendritic cell-based immunotherapy: State
of the art and beyond. Clin Cancer Res. 22:1897–1906. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kim R, Emi M, Tanabe K and Arihiro K:
Tumor-driven evolution of immunosuppressive networks during
malignant progression. Cancer Res. 66:5527–5536. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
deVries IJ, Eggert AA, Scharenborg NM,
Vissers JL, Lesterhuis WJ, Boerman OC, Punt CJ, Adema GJ and Figdor
CG: Phenotypical and functional characterization of clinical grade
dendritic cells. J Immunother. 25:429–38. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Koski GK, Cohen PA, Roses RE, Xu S and
Czerniecki BJ: Reengineering dendritic cell-based anti-cancer
vaccines. Immunol Rev. 222:256–276. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Huang A, Gilmour JW, Imami N, Amjadi P,
Henderson DC and Allen-Mersh TG: Increased serum transforming
growth factor-beta1 in human colorectal cancer correlates with
reduced circulating dendritic cells and increased colonic
Langerhans cell infiltration. Clin Exp Immunol. 134:270–278. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lissoni P, Malugani F, Bonfanti A, Bucovec
R, Secondino S, Brivio F, Ferrari-Bravo A, Ferrante R, Vigoré L,
Rovelli F, et al: Abnormally enhanced blood concentrations of
vascular endothelial growth factor (VEGF) in metastatic cancer
patients and their relation to circulating dendritic cells, IL-12
and endothelin-1. J Biol Regul Homeost Agents. 15:140–144.
2001.PubMed/NCBI
|
|
12
|
Vetsika EK, Koinis F, Gioulbasani M,
Aggouraki D, Koutoulaki A, Skalidaki E, Mavroudis D, Georgoulias V
and Kotsakis A: Сirculating subpopulation of monocytic
myeloid-derived suppressor cells as an independent
prognostic/predictive factor in untreated non-small lung cancer
patients. J Immunol Res. 2014:6592942014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Liu X, Zhang H, Su L, Yang P, Xin Z, Zou
J, Ren S and Zuo Y: Low expression of dendritic cell-specific
intercellular adhesion molecule-grabbing nonintegrin-related
protein in lung cancer and significant correlations with brain
metastasis and natural killer cells. Mol Cell Biochem. 407:151–160.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Domingues D, Turner A, Silva MD, Marques
DS, Mellidez JC, Wannesson L, Mountzios G and de Mello RA:
Immunotherapy and lung cancer: Current developments and novel
targeted therapies. Immunotherapy. 6:1221–1235. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
deVries IJ, Lesterhuis WJ, Scharenborg NM,
Engelen LP, Ruiter DJ, Gerritsen MJ, Croockewit S, Britten CM,
Torensma R, Adema GJ, et al: Maturation of dendritic cells is a
prerequisite for inducing immune responses in advanced melanoma
patients. Clin Cancer Res. 9:5091–5100. 2003.PubMed/NCBI
|
|
16
|
Stagg AJ, Hart AL, Knight SC and Kamm MA:
The dendritic cell: Its role in intestional inflammation and
relationship with gut bacteria. Gut. 52:1522–1529. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Dieu MC, Vanbervliet B, Vicari A, Bridon
JM, Oldham E, Aït-Yahia S, Brière F, Zlotnik A, Lebecque S and Caux
C: Selective recruitment of immature and mature dendritic cells by
distinct chemokines expressed in different anatomic sites. J Exp
Med. 188:373–386. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Jarnjak-Jankovic S, Hammerstad H,
Sabbøe-Larssen S, Kvalheim G and Gaudernack G: A full scale
comparative study of methods for generation of functional Dendritic
cells for use as cancer vaccines. BMC Cancer. 7:1192007. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Bykovskaia SN, Buffo M, Zhang H, Bunker M,
Levitt ML, Agha M, Marks S, Evans C, Ellis P, Shurin MR and Shogan
J: The generation of human dendritic and NK cells from hemopoietic
progenitors induced by interleukin-15. J Leukoc Biol. 66:659–666.
1999.PubMed/NCBI
|
|
20
|
Dauer M, Obermaier B, Herten J, Haerle C,
Pohl K, Rothenfusser S, Schnurr M, Endres S and Eigler A: Mature
dendritic cells derived from human monocytes within 48 h: A novel
strategy for dendritic cell differentiation from blood precursors.
J Immunol. 170:4069–4076. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kulikova EV, Kurilin VV, Shevchenko JA,
Obleukhova IA, Khrapov EA, Boyarskikh UA, Filipenko ML, Shorokhov
RV, Yakushenko VK, Sokolov AV and Sennikov SV: Dendritic cells
transfected with a DNA construct encoding tumor-associated antigen
epitopes induce a cytotoxic immune response against autologous
tumor cells in a culture of mononuclear cells from colorectal
cancer patients. Scand J Immunol. 82:110–117. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Leplina OY, Stupak VV, Kozlov YP,
Pendyurin IV, Nikonov SD, Tikhonova MA, Sycheva NV, Ostanin AA and
Chernykh ER: Use of interferon-alpha-induced dendritic cells in the
therapy of patients with malignant brain gliomas. Bull Exp Biol
Med. 143:528–534. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Obleukhova IA, Kurilin VV, Goncharov MA,
Tarkhov AV, Krasil'nikov SE and Sennikov SV: Effect of mature
dendritic cells primed with autologous tumor antigens from patients
with epithelial ovarian cancer on stimulation of the cytotoxic
immune response in culture of mononuclear cells. Bull Exp Biol Med.
156:161–164. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Almand B, Resser JR, Lindman B, Nadaf S,
Clark JI, Kwon ED, Carbone DP and Gabrilovich DI: Clinical
significance of defective dendritic cell differentiation in cancer.
Clin Cancer Res. 6:1755–1766. 2000.PubMed/NCBI
|
|
25
|
Ruben JM, Bontkes HJ, Westers TM,
Hooijberg E, Ossenkoppele GJ, de Gruijl TD and van de Loosdrecht
AA: Differential capacity of human interleukin-4 and interferon-α
monocyte-derived dendritic cells for cross-presentation of free
versus cell-associated antigen. Cancer Immunol Immunother.
64:1419–1427. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Almand В, Clark JI, Nikitina E, van Beynen
J, English NR, Knight SC, Carbone DP and Gabrilovich DI: Increased
production of immature myeloid cells in cancer patients: A
mechanism of immunosuppression in cancer. J Immunol. 166:678–689.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Palucka K and Banchereau J: Cancer
immunotherapy via dendritic cells. Nat Rev Cancer. 12:265–277.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Türeci Ö, Vormehr M, Diken M, Kreiter S,
Huber C and Sahin U: Targeting the heterogeneity of cancer with
individualized neoepitope vaccines. Clin Cancer Res. 22:1885–1896.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Markiewicz MA and Kast WM: Progress in the
development of immunotherapy of cancer using ex vivo-generated
dendritic cells expressing multiple tumor antigen epitopes. Cancer
Invest. 22:417–434. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Sennikov SV, Kulikova E, Obleukhova IA and
Shevchenko JA: Technologies of cellular antitumor immune response
induction in vitro. Genes Cells. 10:16–22. 2015.
|
|
31
|
Sennikov SV, Shevchenko JA, Kurilin VV,
Khantakova JN, Lopatnikova JA, Gavrilova EV, Maksyutov RA, Bakulina
AY, Sidorov SV, Khristin AA and Maksyutov AZ: Induction of an
antitumor response using dendritic cells transfected with DNA
constructs encoding the HLA-A*02:01-restricted epitopes of
tumor-associated antigens in culture of mononuclear cells of breast
cancer patients. Immunol Res. 64:171–180. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Robson NC, Hoves S, Maraskovsky E and
Schnurr M: Presentation of tumour antigens by dendritic cells and
challenges faced. Curr Opin Immunol. 22:137–144. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Delirezh N, Moazzeni SM, Shokri F,
Shokrgozar MA, Atri M and Kokhaei P: Autologous dendritic cells
loaded with apoptotic tumor cells induce T cell-mediated immune
responses against breast cancer in vitro. Cellular Immunol.
257:23–31. 2009. View Article : Google Scholar
|
|
34
|
Galluzzi L, Senovilla L, Vacchelli E,
Eggermont A, Fridman WH, Galon J, Sautès-Fridman C, Tartour E,
Zitvogel L and Kroemer G: Trial watch. Dendritic cell-based
interventions for cancer therapy. OncoImmunology. 1:1111–1134.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Hargadon KM: Tumor-altered dendritic cell
function: Implications for antitumor immunity. Front Immunol.
4:1922013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Chang AE, Redman BG, Whitfield JR,
Nickoloff BJ, Braun TM, Lee PP, Geiger JD and Mulé JJ: A phase i
trial of tumor lysate-pulsed dendritic cells in the treatment of
advanced cancer. Clin Cancer Res. 8:1021–1032. 2002.PubMed/NCBI
|
|
37
|
Win SJ, McMillan DG, Errington-Mais F,
Ward VK, Young SL, Baird MA and Melcher AA: Enhancing the
immunogenicity of tumour lysate-loaded dendritic cell vaccines by
conjugation to virus-like particles. British J Cancer. 106:92–98.
2012. View Article : Google Scholar
|
|
38
|
Dong B, Dai G, Xu L, Zhang Y, Ling L, Sun
L and Lv J: Tumor cell lysate induces the immunosuppression and
apoptosis of mouse Immunocytes. Mol Med Rep. 10:2827–2834. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Gilboa E and Vieweg J: Cancer
immunotherapy with mRNA-transfected dendritic cells. Immunol Rev.
199:251–63. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Kreiter S, Diken M, Selmi A, Türeci Ö and
Sahin U: Tumor vaccination using messenger RNA: Prospects of a
future therapy. Curr Opin Immunol. 23:399–406. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Degli-Esposti MA and Smyth MJ: Close
encounters of different kinds: Dendritic cells and NK cells take
centre stage. Nat Rev Immunol. 5:112–124. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Bernhard H, Neudorfer J, Gebhard K, Conrad
H, Hermann C, Nährig J, Fend F, Weber W, Busch DH and Peschel C:
Adoptive transfer of autologous, HER2-specific, cytotoxic T
lymphocytes for the treatment of HER2-overexpressing breast cancer.
Cancer Immunol Immunother. 57:271–280. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Gelao L, Criscitiello C, Esposito A, De
Laurentiis M, Fumagalli L, Locatelli MA, Minchella I, Santangelo M,
De Placido S, Goldhirsch A and Curigliano G: Dendritic cell-based
vaccines: Clinical applications in breast cancer. Immunotherapy.
6:349–360. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Cullen SP, Brunet M and Martin SJ:
Granzymes in cancer and immunity. Cell Death Differ. 17:616–623.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Smyth MJ, Kelly JM, Sutton VR, Davis JE,
Browne KA, Sayers TJ and Trapani JA: Unlocking the secrets of
cytotoxic granule proteins. J Leukoc Biol. 70:18–29.
2001.PubMed/NCBI
|
|
46
|
Walczak H and Krammer PH: The CD95
(APO-1/Fas) and the TRAIL (APO-2L) apoptosis systems. Exp Cell Res.
256:58–66. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Poehlein CH, Hu HM, Yamada J, Assmann I,
Alvord WG, Urba WJ and Fox BA: TNF plays an essential role in tumor
regression after adoptive transfer of perforin/IFN-gamma double
knockout effector T cells. J Immunol. 170:2004–2013. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Henkler F, Behrle E, Dennehy KM, Wicovsky
A, Peters N, Warnke C, Pfizenmaier K and Wajant H: The
extracellular domains of FasL and Fas are sufficient for the
formation of supramolecular FasL-Fas clusters of high stability. J
Cell Biol. 168:1087–1098. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Rahman M, Davis SR, Pumphrey JG, Bao J,
Nau MM, Meltzer PS and Lipkowitz S: TRAIL induces apoptosis in
triple-negative breast cancer cells with a mesenchymal phenotype.
Breast Cancer Res Treat. 113:217–230. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Stuckey DW and Shah K: TRAIL on trial:
Preclinical advances for cancer therapy. Trends Mol Med.
19:685–694. 2013. View Article : Google Scholar : PubMed/NCBI
|