Introduction
Lung cancer is the most common cause of
cancer-associated mortality, and according to the data from western
countries, >50% of newly diagnosed patients with lung cancer are
>70 years of age (1). With the
advances in diagnostics and cancer treatment, an increasing number
of elderly patients are being diagnosed with lung cancer and
actively receiving treatment.
Second-line treatments are comparably efficacious in
elderly as well as younger patients. For patients not harboring an
epidermal growth factor receptor (EGFR) mutation, tyrosine
kinase inhibitor (TKI) and cytotoxic chemotherapeutic agents are
currently second-line treatment options. In a retrospective
analysis performed on elderly patients (≥70 years old) enrolled in
the BR.21 trials, elderly patients revealed a benefit from
second-line erlotinib treatment compared with the best supportive
care with similar overall survival (OS) and response rates to those
of younger patients (2). Regarding
cytotoxic chemotherapy, pemetrexed or docetaxel as second-line
treatment demonstrated comparable efficacy and toxicity in elderly,
and younger patients in a retrospective analysis of large
randomized clinical trials (3).
In addition, predicting which elderly patients may
or may not benefit from chemotherapy and investigating suitable
chemotherapeutics is important. Treatment without these
considerations cause unnecessary treatment-associated toxicities,
longer hospital stays, poor quality of life, economic burden and
ultimately shorter survival times. As the majority of clinical
trials have been performed in young and otherwise healthy people,
there is insufficient knowledge regarding the efficacy and safety
of drugs in elderly patients who are particularly prone to
exclusion from clinical trials owing to comorbidities, coexisting
multi-pharmacies or poor performance status (PS).
Comorbidities are assessed using the Charlson
comorbidity index (CCI) and the simplified comorbidity score (SCS)
(4–7).
The CCI is one of the most widely used scoring systems and has been
validated in a number of diseases and the SCS has been designed for
lung cancer (4,7). In addition, there are no data comparing
the efficacy of TKI and chemotherapeutic drugs in elderly patients
without an EGFR mutation. Therefore, the present study
compared the efficacy and toxicity of these two types of drugs in
elderly patients.
Materials and methods
Patients
Elderly patients (≥70 years of age) with wild-type
EGFR NSCLC who previously experienced chemotherapy failure
and received second-line chemotherapy at the Korea University Guro
Hospital (Seoul, Korea) between January 2005 and December 2013 were
included in this retrospective study. For squamous cell carcinoma,
EGFR mutation testing was not a routine practice due to a
lack of possibility for EGFR mutation positivity (8). Therefore, the present study included
patients with squamous cell carcinoma with unknown EGFR
mutation status and confirmed wild-type EGFR. For other
types of histology, only patients confirmed to have wild-type
EGFR were included. EGFR mutation status was
confirmed using direct sequencing or a peptide nucleic acid (PNA)
clamping method using the PNA Clamp™ EGFR Mutation
Detection kit (Panagene, Inc., Daejeon, Korea). Data were collected
from electronic medical records. The present study was approved by
the Institutional Review Board of the Korea University Guro
Hospital (KUGH15250-001).
Between January 2005 and December 2013, 943 patients
with lung cancer received palliative chemotherapy at Korea
University Guro Hospital. Among them, 365 patients were >70
years of age and 203 of these patients received second-line
chemotherapy. Following exclusion of patients harboring an
EGFR mutation, 126 patients were included in the analysis.
The patient characteristics are presented in Table I. The median age was 75 (range, 70–85)
years. There were 101 males (80.2%) and 25 females (19.8%). Of the
126 patients, 58 (46.0%) were diagnosed with adenocarcinoma, 63
(50.0%) were diagnosed with squamous cell carcinoma and 5 (4.0%)
were diagnosed with another histology (3 with undifferentiated
carcinoma, 1 with sarcomatoid carcinoma and 1 with large-cell
carcinoma).
 | Table I.Patient characteristics. |
Table I.
Patient characteristics.
|
Characteristics | All patients | TKI | Chemotherapy |
P-valuea |
|---|
| No. patients
(%) | 126 (100.0) | 52 (41.3) | 74 (58.7) |
|
| Age, years
(range) | 75 (70–85) | 73 (70–85) | 75 (70–81) |
|
| Age, years (%) |
|
|
| 0.456 |
|
<75 | 80 (63.5) | 35 (67.3) | 45 (60.8) |
|
|
≥75 | 46 (36.5) | 17 (32.7) | 29 (39.2) |
|
| Sex (%) |
|
|
| 0.845 |
|
Male | 101 (80.2) | 42 (80.8) | 59 (79.7) |
|
|
Female | 25 (19.8) | 10 (19.2) | 15 (20.3) |
|
| Histology (%) |
|
|
| <0.001 |
|
Non-squamous | 63 (50.0) | 38 (73.1) | 25 (33.8) |
|
|
Squamous | 63 (50.0) | 14 (26.9) | 49 (66.2) |
|
| Smoking history
(%) |
|
|
| 0.413 |
| Never
smoker | 27 (21.4) | 13 (25.0) | 14 (18.9) |
|
| Ever
smoker | 99 (78.6) | 39 (75.0) | 60 (81.1) |
|
| Stage at diagnosis
(%) |
|
|
| 0.360 |
|
I–IIIA | 22 (17.5) | 11 (21.2) | 11 (14.9) |
|
|
IIIB-IV | 104 (82.5) | 41 (78.8) | 63 (85.1) |
|
| Stage at
second-line chemotherapy (%) |
|
|
| 0.549 |
|
M0/M1a | 79 (62.7) | 31 (59.6) | 48 (64.9) |
|
|
M1b | 47 (37.3) | 21 (40.4) | 26 (35.1) |
|
| PFS of first-line
line chemotherapy (%) |
|
|
| 0.383 |
| <4
months | 64 (50.8) | 24 (46.2) | 40 (54.1) |
|
| ≥4
months | 62 (49.2) | 28 (53.8) | 34 (45.9) |
|
| ECOG PS at
second-line chemotherapy (%) |
|
|
| 0.412 |
|
0–1 | 87 (69.0) | 38 (73.1) | 49 (66.2) |
|
|
2–3 | 39 (31.0) | 14 (26.9) | 25 (33.8) |
|
| Subsequent
treatment following failure of second-line chemotherapy (%) |
|
|
| 0.036 |
|
Second-line | 51 (40.5) | 15 (28.8) | 36 (48.6) |
|
|
Third-line | 39 (31.0) | 22 (42.3) | 17 (23.0) |
|
|
Fourth-line or more | 36 (28.6) | 15 (28.8) | 21 (28.4) |
|
| BSA change between
first− and second-line treatment (%) |
|
|
| 0.963 |
| Not
decreased | 73 (57.9) | 30 (57.7) | 43 (58.1) |
|
|
Decreased | 53 (42.1) | 22 (42.3) | 31 (41.9) |
|
| Plasma Hb at the
second-line chemotherapy, g/dl (%) |
|
|
| 0.153 |
|
<10 | 43 (34.1) | 14 (26.9) | 29 (39.2) |
|
|
≥10 | 83 (65.9) | 38 (45.8) | 45 (54.2) |
|
| Serum sodium at the
second-line chemotherapy, mmol/l (%) |
|
|
| 0.482 |
|
<135 | 68 (54.0) | 30 (57.7) | 38 (51.4) |
|
|
≥135 | 58 (46.0) | 22 (42.3) | 36 (48.6) |
|
| Serum albumin at
the second-line chemotherapy, g/dl (%) |
|
|
| 0.224 |
|
<3.5 | 25 (19.8) | 13 (25.0) | 23 (16.2) |
|
|
≥3.5 | 101 (80.2) | 39 (75.0) | 62 (83.8) |
|
Data collection
The following data were collected and analyzed for
each patient: Age, sex, tumor histology, stage at diagnosis,
site(s) of metastasis, comorbidities, previous chemotherapy prior
to second-line treatment, Eastern Cooperative Oncology Group (ECOG)
PS, plasma hemoglobin levels, serum sodium and albumin levels at
the beginning of the second-line chemotherapy, difference in body
surface area difference (BSA) between day 1 of first-line
chemotherapy and day 1 of second-line chemotherapy and the
subsequent treatment following failure of second-line
chemotherapy.
In addition, the present study analyzed the
following data: Type of chemotherapy regimen, start and end dates
of chemotherapy and response and best response to the second-line
chemotherapy according to the Response Evaluation Criteria in Solid
Tumors version 1.1 guidelines (9).
The responses of all patients were assessed at 2- to 3-month
intervals by enhanced computed tomography. Magnetic resonance
imaging, fluorodeoxyglucose positron emission tomography and bone
scanning were performed at the physician's discretion.
Treatment-related toxicity data were collected via medical records
based on the National Cancer Institute Common Terminology Criteria
for Adverse Events version 3 (10).
Comorbidity assessment
For assessment of comorbidities, the present study
investigated the CCI and SCS at the day of second-line chemotherapy
initiation. The CCI has 19 and the SCS has 7 different descriptors,
with a maximum score of 35 for the CCI, and 20 for the SCS
(Tables II and III) (4,7). The
presence of comorbidities was assessed by reviewing electronic
medical charts for individual descriptors of both
comorbidity-scoring systems.
 | Table II.Charlson comorbidity index and
weighting of comorbidities. |
Table II.
Charlson comorbidity index and
weighting of comorbidities.
| Score | Comorbid
condition |
|---|
| 1 | Myocardial
infarction |
|
| Congestive heart
failure |
|
| Cerebral vascular
disease |
|
| Peripheral vascular
disease |
|
| Dementia |
|
| Chronic pulmonary
disease |
|
| Connective tissue
disease |
|
| Peptic ulcer
disease |
|
| Mild liver
disease |
|
| Agea |
| 2 | Diabetes |
|
| Hemiplegia |
|
| Moderate/severe
renal disease |
|
| Diabetes with
end-organ damage |
|
| Any solid
tumor |
|
| Leukemia |
|
| Lymphoma |
| 3 | Moderate/severe
liver disease |
| 6 | Metastatic solid
tumor |
|
| Acquired
immunodeficiency syndrome |
 | Table III.Simplified comorbidity score and
weighting of comorbidities. |
Table III.
Simplified comorbidity score and
weighting of comorbidities.
| Score | Comorbidity |
|---|
| 7 | Tobacco
consumption |
| 5 | Diabetes
mellitus |
| 4 | Renal
insufficiency |
| 1 | Respiratory
comorbidity |
| 1 | Neoplastic
comorbidity |
| 1 | Cardiovascular
comorbidity |
| 1 | Alcoholism |
Chemotherapy regimens
Chemotherapy regimens were classified as TKI or
chemotherapy, with the TKI group including patients who received an
EGFR TKI, and the chemotherapy group including patients who
received any type of cytotoxic chemotherapy regimen. EGFR TKIs
included erlotinib and gefitinib and the cytotoxic chemotherapy
regimen included docetaxel, pemetrexed, a combination of
gemcitabine and vinorelbine, and platinum doublets.
Statistical analyses
All patients who received at least 1 cycle of
second-line chemotherapy were included in the efficacy analysis.
Rates were compared using the χ2 test. The Kaplan-Meier
method was used to estimate the OS and progression-free survival
(PFS). PFS was evaluated from the initiation of the second-line
chemotherapy until the first occurrence of progression, mortality
from any cause or the final follow-up visit if none of the
preceding events had occurred. OS was determined as the interval
between the first day of first-line treatment and mortality or the
final follow-up visit. Differences between the curves were analyzed
using the log-rank test. Following univariate analyses using the
Kaplan-Meier method, variables significantly associated with poor
survival time (P<0.05) were selected, and a Cox proportional
hazards regression was performed for multivariate analyses using
the ‘ENTER’ method in SPSS. SPSS for Windows version 20.0 (IBM
Corp., Armonk, NY, USA) was used for all statistical analyses.
P<0.05 was considered to indicate a statistically significant
difference.
Results
Patient characteristics
Among the 126 patients included in the present
study, 52 (41.3%) were treated with TKIs and 74 (58.7%) were
treated with cytotoxic chemotherapy. In the TKI group, 21 patients
(40.4%) were treated with gefitinib and 31 (59.6%) were treated
with erlotinib. In the chemotherapy group, docetaxel was the most
common agent, which was administered to 34 patients (45.9%).
Pemetrexed was administered to 20 patients (27.0%). A total of 9
patients (12.2%) were treated with a combination of gemcitabine and
vinorelbine, and 7 patients (9.4%) were treated with a platinum
doublet combination. A total of 4 patients (5.4%) were treated with
a singlet agent other than docetaxel or pemetrexed (3 with
gemcitabine and 1 with vinorelbine). A greater percentage of
patients had non-squamous cell carcinoma in the TKI group, and more
patients had squamous cell carcinoma in the chemotherapy group
(P<0.001). The number of patients who received further treatment
was not balanced between the 2 treatment groups. In the TKI group,
71.1% of patients received subsequent chemotherapy following
failure of second-line treatment. Conversely, 51.4% of patients in
the chemotherapy group received further treatment, including
patients who received more than third-line chemotherapy (P=0.036).
Except for histology and subsequent chemotherapy, there were no
significant differences in the clinical characteristics of the
patients between the two treatment groups (Table I).
Comorbidities of patients
The distribution of comorbidities in the included
patients is presented in Table IV. A
total of 69 patients (54.8%) had hypertension, 33 (26.2%) had
diabetes and 46 (36.5%) had chronic pulmonary disease, including
chronic obstructive pulmonary disease and interstitial lung
disease. A history of solid tumor prior to second-line chemotherapy
for lung cancer was observed in 15 patients; all cases were
localized tumors. There were no patients with hemiplegia, dementia
or acquired immune deficiency syndrome. A total of 20 patients
(15.9%) had no underlying disease except metastatic lung cancer.
The CCI scores of all patients ranged between 9 and 12 (median, 10)
points. The SCS of all patients ranged between 1 and 15 (median, 8)
points. There were no significant differences in the distribution
of CCI scores and SCSs between the TKI, and chemotherapy
groups.
 | Table IV.Comorbidities and comorbidity scores
of the patients included in the present study. |
Table IV.
Comorbidities and comorbidity scores
of the patients included in the present study.
| Cormorbity | Number | % |
|---|
| Comorbidities |
|
Diabetes mellitus | 33 | 26.2 |
| Chronic
pulmonary disease | 46 | 36.5 |
|
Congestive heart failure |
5 |
4.0 |
|
Cerebrovascular disease |
9 |
7.1 |
|
Peripheral vascular
disease | 10 |
7.9 |
| Ulcer
disease |
6 |
4.8 |
|
Myocardial infarction |
5 |
4.0 |
| Liver
disease |
3 |
2.4 |
| Chronic
kidney disease |
2 |
1.6 |
| None
except lung cancer | 20 | 15.9 |
| Charlson
comorbidity index |
| 9 | 38 | 30.2 |
| 10 | 56 | 44.4 |
| 11 | 21 | 16.7 |
| 12 | 11 |
8.7 |
|
Total | 126 | 100 |
| Simplified
comorbidity score |
| 1 | 12 |
9.5 |
| 2 |
3 |
2.4 |
| 3 |
6 |
4.8 |
| 6 |
5 |
4.0 |
| 7 |
1 |
0.8 |
| 8 | 53 | 42.1 |
| 9 | 18 | 14.3 |
| 13 | 13 | 10.3 |
| 14 | 10 |
7.9 |
| 15 |
5 |
4.0 |
|
Total | 126 | 100 |
Clinical parameters associated with
treatment outcomes
The median PFS and OS for all patients who received
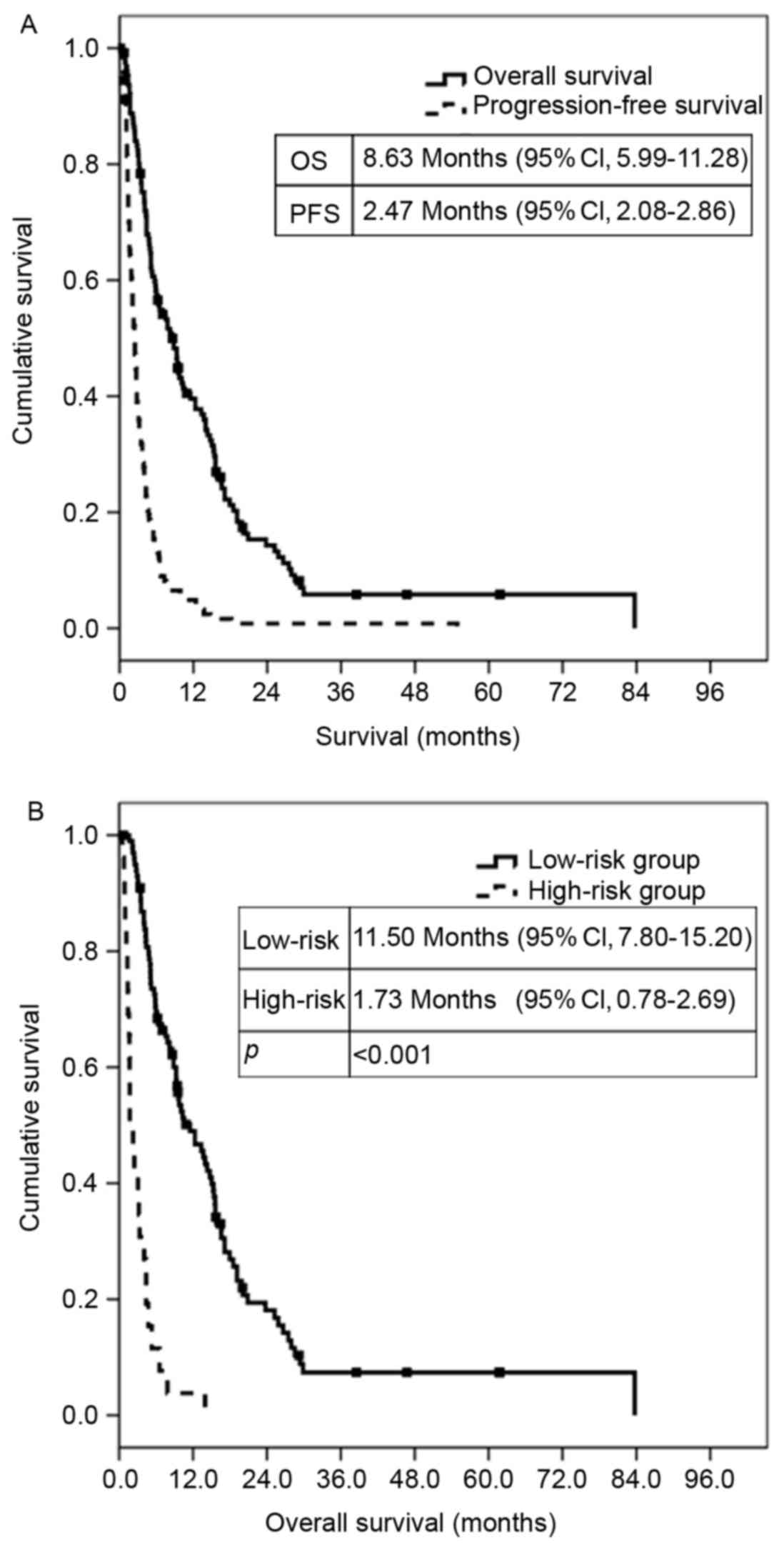
second-line treatment was 2.47 months [95% confidence interval
(CI), 2.08–2.86] and 8.63 months (95% CI, 5.99–11.28), respectively
(Fig. 1A). Univariate analysis
demonstrated that the OS for all patients was significantly
associated with the following factors: Histology (squamous vs.
non-squamous), stage at diagnosis (stage I–IIIA vs. stage IIIB-IV),
PS (0–1 vs. 2–3), SCS (<13 vs. ≥13), PFS of first-line treatment
(<4 months vs. ≥4 months), change of BSA between first-line and
second-line treatment (not decreased vs. decreased), subsequent
treatment after failure of second-line chemotherapy (at least
third-line chemotherapy vs. no chemotherapy), brain metastasis (no
vs. yes), distant lymph node metastasis (no vs. yes), number of
metastatic organs (<3 vs. ≥3), plasma hemoglobin at the start of
second-line chemotherapy (<10 vs. ≥10 g/dl), serum albumin at
the start of second-line chemotherapy (<3.5 vs. ≥3.5 g/dl) and
serum sodium at the start of second-line chemotherapy (<135 vs.
≥135 mmol/l). CCI, response to second-line treatment or adrenal
gland, liver or bone metastasis were not associated with OS. The
present study then performed multivariate analysis using a Cox
proportional hazards regression model. Histology (squamous vs.
non-squamous), good PS (ECOG 0–1), lower SCS (<13), no brain
metastasis, longer PFS of previous chemotherapy (≥4 months), higher
serum sodium level (>135 mmol/l) and higher serum albumin level
(>3.5 g/dl) were associated with prolonged OS (Table V).
 | Table V.Multivariate analysis of prognostic
factors. |
Table V.
Multivariate analysis of prognostic
factors.
|
| All patients |
|---|
|
|
|
|---|
| Factor | HR | 95% CI |
P-valuea |
|---|
| ECOG PS (0–1 vs.
2–3) | 2.139 | 1.348–3.395 | 0.001 |
| Brain metastasis
(negative vs. positive) | 3.177 | 1.288–7.839 | 0.012 |
| PFS of first-line
treatment, months (≥4 vs. <4) | 2.860 | 1.837–4.452 | <0.001 |
| Serum sodium,
mmol/l (≥135 vs. <135) | 3.228 | 1.685–6.186 | <0.001 |
| Serum albumin, g/dl
(≥3.5 vs. <3.5) | 1.802 | 1.029–3.156 | 0.039 |
| SCS (<13 vs.
≥13) | 1.776 | 1.502–2.996 | 0.031 |
| Histology
(non-squamous vs. squamous) | 1.908 | 1.206–3.017 | 0.006 |
| Distant lymph node
metastasis (negative vs. positive) | 2.229 | 0.968–5.135 | 0.060 |
| Metastatic organ
(M1a vs. M1b) | 1.561 | 0.923–2.641 | 0.097 |
| Number of
metastasis organs (<3 vs. ≥3) | 0.475 | 0.184–1.226 | 0.124 |
| Stage at diagnosis
(I–IIIA vs. IIIB-IV) | 0.865 | 0.490–1.527 | 0.617 |
| Subsequent
treatment (third-line or more chemotherapy vs. no
chemotherapy) | 1.278 | 0.819–1.996 | 0.280 |
| BSA change between
first- and second-line treatment (not decreased vs. decreased) | 1.265 | 0.809–1.978 | 0.303 |
| Plasma Hb, g/dl
(≥10 vs. <10) | 0.983 | 0.613–1.575 | 0.942 |
The present study subsequently performed survival
analysis of patients who exhibited poor prognostic factors to
identify patients who did not benefit from second-line
chemotherapy. As the present study surmised that histology was not
yet validated as prognostic markers to decide second-line
treatment, a subgroup was constructed that did not include
histology, but prognostic factors showing statistical significance
in the Cox proportional hazards regression model were weighted 1
score for each factor. The present study then divided the patients
into 2 groups: Low-risk and high-risk. The low-risk group included
patients who had ≤2 factors and the high-risk group included those
who had ≥3 factors. There were 26 patients (20.6%) in the high-risk
group. The median OS of patients in the low-risk group who received
second-line treatment was 11.50 months (95% CI, 7.80–15.20),
whereas the median OS of the high-risk group was 1.73 months (95%
CI, 0.78–2.69). The difference in survival was statistically
significant (P<0.001; Fig.
1B).
Efficacy of TKI and chemotherapy
The results of efficacy analysis are presented in
Table VI. Of the 126 patients, there
was no case of complete remission. The best responses were partial
remission (PR) in 11 patients (15.5%) and stable disease (SD) in 51
patients (40.5%). In the TKI group, no patient exhibited PR. A
total of 25 patients (50.0%) revealed SD as the best response.
However, 11 patients (15.5%) demonstrated PR and 26 patients
(36.6%) demonstrated SD in the chemotherapy group. Therefore, the
overall response rate, which is defined as the proportion of PR and
CR, was statistically different between the two groups
(P=0.029).
 | Table VI.Efficacy analysis of TKI and
chemotherapy groups. |
Table VI.
Efficacy analysis of TKI and
chemotherapy groups.
| Factor | TKI (n=50) | Chemotherapy
(n=73) |
|---|
| Response to
second-line chemotherapy, n=123a (%) |
|
|
| Complete
remission | 0 (0.0) | 0 (0.0) |
| Partial
remission | 0 (0.0) | 11 (15.5) |
| Stable disease | 25 (50.0) | 26 (36.6) |
| Progressive
disease | 25 (50.0) | 34 (47.9) |
| Response rate |
0.0% | 15.5% |
| Disease control
rate | 50.0% | 52.1% |
| Survival of
second-line chemotherapy |
|
|
| Progression-free
survival (months), median (95% CI) | 2.57
(2.18–3.00) | 2.33
(1.75–2.92) |
| Overall survival
(months), median (95% CI) | 8.83
(1.26–16.4) | 7.83
(4.80–10.87) |
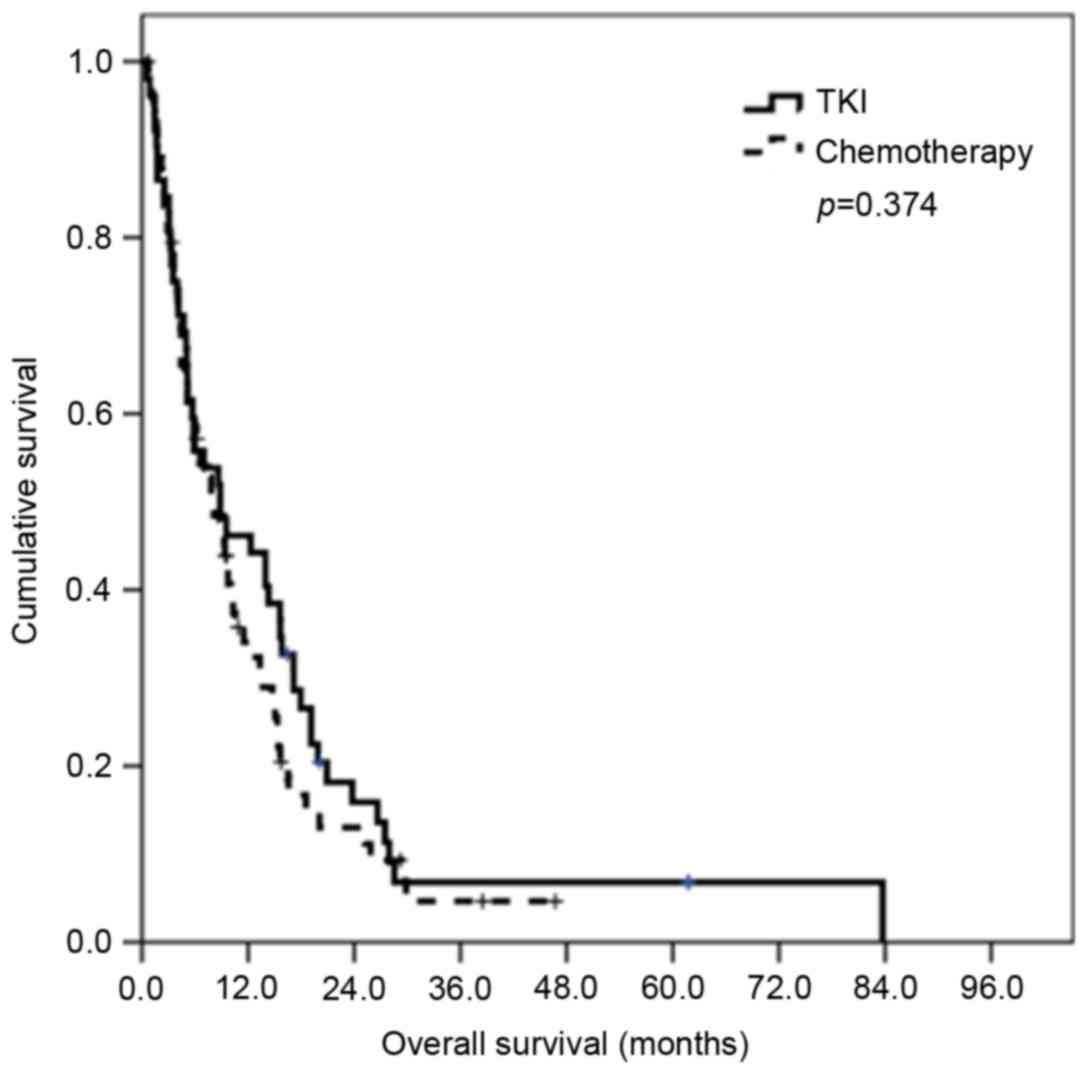
No difference was observed in the median PFS and OS
of second-line chemotherapy between the TKI, and chemotherapy
groups (P=0.287 for PFS and P=0.374 for OS). The survival curves
for PFS and OS are presented in Fig.
2.
Drug delivery and toxicities
The median treatment duration was 3.8 months. Of the
126 patients, 32 patients (25.4%) underwent dose reduction during
second-line chemotherapy, 6 in the TKI group and 26 in the
chemotherapy group. A total of 13 patients (10.3%) underwent
initial dose reduction. The causes of dose reduction were
chemotherapy-associated toxicity (n=18) and decreased PS (n=11). A
total of 7 patients (5.5%) stopped chemotherapy owing to toxicity
and 7 patients (5.5%) stopped treatment due to decreased PS. A
total of 3 patients (2.4%) stopped chemotherapy because they
refused further treatment. A total of 6 patients (11.5%) in the TKI
group and 7 patients (9.4%) in the chemotherapy group stopped
chemotherapy because of toxicity and poorer PS.
The frequencies of hematologic and non-hematologic
adverse events are presented in Table
VII. In the TKI group, non-hematological toxicities, including
skin rash (54.9%), emesis (27.5%) and fatigue (27.5%), were common.
In terms of hematological toxicities, anemia (47.1%) was the most
common. Grade 3/4 toxicities included anemia, neutropenia,
thrombocytopenia, diarrhea, emesis and fatigue, each occurring in
<3.9% of patients. In the chemotherapy group, hematological
toxicities were more common. Anemia (85.1%), neutropenia (36.5%),
thrombocytopenia (16.2%) and febrile neutropenia (10.8%) were
observed. Regarding non-hematological toxicities, emesis and
fatigue were common, occurring in 44.6, and 41.9% of patients,
respectively. Grade 3/4 toxicities included neutropenia (20.3%),
febrile neutropenia (10.8%), anemia (8.1%), thrombocytopenia,
(8.1%), emesis (6.8%) and fatigue (6.8%). There were no
treatment-associated mortalities in either group.
 | Table VII.Treatment-associated adverse events
of TKI and chemotherapy groups. |
Table VII.
Treatment-associated adverse events
of TKI and chemotherapy groups.
|
| TKI (n=52) | Chemotherapy
(n=74) |
|---|
|
|
|
|
|---|
|
| All | Grade 3–4 | All | Grade 3–4 |
|---|
|
|
|
|
|
|
|---|
| Adverse event | n | (%) | n | (%) | n | (%) | n | (%) |
|---|
| Hematologic |
|
Anemia | 24 | 47.1 | 1 | 2.0 | 63 | 85.1 | 6 | 8.1 |
|
Neutropenia | 1 | 2.0 | 1 | 2.0 | 27 | 36.5 | 15 | 20.3 |
|
Thrombocytopenia | 1 | 2.0 | 1 | 2.0 | 12 | 16.2 | 6 | 8.1 |
| Febrile
neutropenia | 0 | 0.0 | 0 | 0 | 8 | 10.8 | 8 | 10.8 |
|
Non-hematologic |
|
Diarrhea | 10 | 19.6 | 1 | 2.0 | 6 | 8.1 | 0 | 0 |
|
Emesis | 14 | 27.5 | 1 | 2.0 | 33 | 44.6 | 5 | 6.8 |
|
Constipation | 1 | 2.0 | 0 | 0 | 6 | 8.1 | 0 | 0 |
|
Fatigue | 14 | 27.5 | 2 | 3.9 | 31 | 41.9 | 5 | 6.8 |
|
Neuropathy | 0 | 0 | 0 | 0 | 3 | 4.1 | 0 | 0 |
| Skin
rash | 28 | 54.9 | 1 | 2.0 | 1 | 1.4 | 0 | 0 |
|
ILD | 2 | 3.9 | 1 | 2.0 | 0 | 0 | 0 | 0 |
Discussion
In the present study, clinical factors, including
comorbid conditions is the most important factor affecting survival
in elderly patients with lung cancer without EGFR mutations
receiving second-line treatment. Additionally, clinical factors,
including PS, PFS of first-line chemotherapy, presence of brain
metastasis, serum albumin levels and serum sodium levels, were
strong prognostic factors for elderly patients in the present
study. A good PS is a well-known prognostic factor and a long PFS
of previous chemotherapy reflects the less aggressive nature of the
cancer (3,11). Brain metastasis is also a well-known
risk factor with a poor outcome. In particular, brain metastasis in
elderly patients may affect tolerability to chemotherapy, quality
of life and neurotoxicities depending on the surgery or
radiotherapy of the brain (12,13).
Furthermore, numerous studies evaluated low serum albumin and
sodium levels as independent poor prognostic factors in patients
with cancer (14–16). Serum albumin is a commonly used marker
for assessing a patient's nutritional status, inflammation and
hepatic function. Serum albumin is decreased in cases of
malnutrition due to cancer cachexia, treatment-associated poor oral
intake or response to acute inflammation as these situations are
major causes of albumin synthesis suppression (17). Hyponatremia is caused by inappropriate
antidiuretic hormone secretion syndrome, diuretic drugs and
salt-wasting conditions in patients with brain metastases,
meningitis, and salt-losing nephropathy following cisplatin use
(16). Furthermore, poor oral intake
accompanying hypovolemia may induce hyponatremia in numerous
patients with cancer (16).
Therefore, serum albumin and sodium are reliable factors
representing nutritional status, inflammatory conditions or cancer
activities. Clinicians should pay close attention to these
laboratory findings when treating elderly patients with cancer.
In the present study, patients with non-squamous
cell histology without an EGFR mutation demonstrated better
survival, compared with the patients with squamous cell histology.
The OS in lung adenocarcinoma patients has been reported to be
higher compared with that in patients with lung squamous cell
carcinoma owing to the introduction of EGFR TKI therapy (18). However, the reasons for the survival
difference between patients with lung adenocarcinoma not harboring
EGFR mutations and patients with squamous cell carcinoma in
the present study remain unclear. Further investigations of
survival according to histology in patients with lung cancer with
or without target therapy options may aid in explaining this
difference.
The present study divided patients into subgroups to
investigate reliable clinical factors to predict which patients do
not benefit from second-line chemotherapy. Therefore, the present
study identified low-risk and high-risk groups according to the six
factors that revealed statistical significance in the multivariate
analysis. The OS of the high-risk group revealed ≥3 risk factors
were 1.73 months, a very short duration compared with the OS of the
low-risk group patients (11.50 months). Therefore, the present
study concluded that second-line treatment is not beneficial and
may even be harmful to patients with numerous risk factors.
In general, comorbid conditions of patients with
cancer are associated with poorer survival (19,20). The
present study evaluated CCI and SCS to analyze the association of
comorbidities and efficacy of chemotherapy. CCI was not associated
with survival, whereas SCS was associated with OS in univariate and
multivariate analyses. SCS was suggested to be more sensitive to
validate comorbidities. In other studies, the SCS represented an
independent prognostic factor for NSCLC as well as small-cell lung
cancer (7,21) and was more informative compared with
CCI to predict outcomes in patients with NSCLC (7), although this is still debated. Numerous
studies reported that SCS did not provide prognostic information in
patients with lung cancer (6,5). To more precisely predict the outcome of
patients with lung cancer with underlying disease, more validation
is required.
For patients with lung cancer without EGFR
mutations, second-line treatment includes cytotoxic agents, such as
docetaxel, pemetrexed, gemcitabine and EGFR TKIs (gefitinib and
erlotinib). A number of studies reported that EGFR TKI treatment in
second-line treatment had similar efficacy to cytotoxic
chemotherapy in patients with NSCLC (22–25).
Patients with squamous cell carcinoma or NSCLC who do not harbor an
EGFR mutation revealed a benefit from TKI compared with the
best supportive care as second-line treatment (25–28).
However, previous meta-analyses reported that cytotoxic
chemotherapy is better compared with TKI for patients without
EGFR mutations (29–31).
Numerous factors other than survival alone have to
be considered to select therapies for elderly patients who failed
previous chemotherapy as they are regarded as having a poorer
physical status compared with younger patients. Physicians should
take into account a number of factors in addition to age, including
the patient's life expectancy, functional status and comorbidities.
Therefore, TKI is an attractive choice for elderly patients as TKIs
are convenient to take and require less frequent hospital visits.
Furthermore, TKIs have less severe toxicities compared with
cytotoxic chemotherapeutic agents. However, the treatment choice
for more than second-line treatment of elderly patients with
wild-type EGFR remains unclear, as there have been no
reports on this patient group to date.
The present study was performed with elderly
patients with NSCLC not harboring EGFR mutations in order to
determine a solution for the practical treatment of these patients.
In the present study, TKI produced a poor response rate compared
with chemotherapy. However, PFS and OS were not different between
the two treatment groups. As expected, severe toxicities exceeding
grade 3 were more common in the chemotherapy group and dose
reduction was also more common in the chemotherapy group, compared
with the TKI group. Therefore, the results of the present study
suggested that TKI may be an appropriate second-line treatment
option for elderly patients. Furthermore, the present study
suggested that careful dose adjustment is necessary during
cytotoxic chemotherapy treatment in this age group. Hematologic
toxicities are generally more common with chemotherapy compared
with TKI therapy, and these toxicities affect the quality of life
as well as the dose intensity during chemotherapy. In addition,
elderly patients frequently have numerous comorbidities and these
comorbidities also affect the dose of drugs. A low dose intensity
may result in shorter survival times and hematologic toxicities may
sometimes cause treatment-associated mortality. Therefore,
physicians should administer well-managed chemotherapy and pay
careful attention to elderly patients.
As studies of the parameters associated with
treatment outcomes of second-line therapy in this clinical setting
are scarce, the prognostic factors identified in the present study
should be useful for designing prospective, randomized clinical
trials on the efficacy of second-line chemotherapy in these
patients. In addition, the present study investigated the
usefulness of comorbidity scores, including the CCI and SCS, as
prognostic factors. Comorbidity analysis is important in studies of
elderly patients, but few such studies have been reported to date
(32,33). Based on the results of the present
study, it is suggested that clinical factors as well as comorbidity
factors have a strong predictive value with respect to survival. To
the best of our knowledge, this is the first study comparing
second-line TKI and chemotherapy treatment in elderly patients with
NSCLC and wild-type EGFR. Although the findings were limited
by the small number of patients evaluated and the retrospective
study design, they provide a practical guide for selecting
second-line chemotherapy in elderly patients without EGFR
mutations.
Previously, novel immunotherapeutics, including
programmed death ligand-1 immune check point blockade revealed high
efficacy in patients with lung cancer. Nivolumab, pembrolizumab and
atezolizumab demonstrated benefits in terms of survival compared
with docetaxel as second-line treatment. Furthermore, these agents
revealed good safety profiles (34)
and therefore would be more beneficial to elderly patients. Further
investigation of immunotherapeutics is required.
In conclusion, significant prognostic factors
affecting survival identified in the current study were squamous
histology, poor PS, higher SCS, short PFS of first-line treatment,
presence of brain metastasis, low serum albumin level and
hyponatremia. Careful consideration should be given in deciding
second-line treatment for patients having ≥3 of these factors,
except histology. TKI and cytotoxic chemotherapy as second-line
treatment revealed similar survival results and different toxicity
profiles. Both are good options for elderly patients with NSCLC not
harboring EGFR mutations if careful management is provided.
Therefore, physicians should consider clinical conditions of each
patient as the most important factors affecting survival in the
second-line treatment of elderly patients.
References
|
1
|
Vora N and Reckamp KL: Non-small cell lung
cancer in the elderly: Defining treatment options. Semin Oncol.
35:590–596. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Wheatley-Price P, Ding K, Seymour L, Clark
GM and Shepherd FA: Erlotinib for advanced non-small-cell lung
cancer in the elderly: An analysis of the National Cancer Institute
of Canada clinical trials group study BR.21. J Clin Oncol.
26:2350–2357. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Weiss GJ, Langer C, Rosell R, Hanna N,
Shepherd F, Einhorn LH, Nguyen B, Paul S, McAndrews P, Bunn PA Jr
and Kelly K: Elderly patients benefit from second-line cytotoxic
chemotherapy: A subset analysis of a randomized phase III trial of
pemetrexed compared with docetaxel in patients with previously
treated advanced non-small-cell lung cancer. J Clin Oncol.
24:4405–4411. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Charlson M, Szatrowski TP, Peterson J and
Gold J: Validation of a combined comorbidity index. J Clin
Epidemiol. 47:1245–1251. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Alexander M, Evans SM, Stirling RG, Wolfe
R, Officer A, MacManus M, Solomon B, Burbury K and Ball D: The
influence of comorbidity and the simplified comorbidity score on
overall survival in non-small cell lung cancer-a prospective cohort
study. J Thorac Oncol. 11:748–757. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ball D, Thursfield V, Irving L, Mitchell
P, Richardson G, Torn-Broers Y, Wright G and Giles G: Evaluation of
the simplified comorbidity score (Colinet) as a prognostic
indicator for patients with lung cancer: A cancer registry study.
Lung Cancer. 82:358–361. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Colinet B, Jacot W, Bertrand D, Lacombe S,
Bozonnat MC, Daurès JP and Pujol JL: oncoLR health network: A new
simplified comorbidity score as a prognostic factor in
non-small-cell lung cancer patients: Description and comparison
with the Charlson's index. Br J Cancer. 93:1098–1105. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Lindeman NI, Cagle PT, Beasley MB, Chitale
DA, Dacic S, Giaccone G, Jenkins RB, Kwiatkowski DJ, Saldivar JS,
Squire J, et al: Molecular testing guideline for selection of lung
cancer patients for EGFR and ALK tyrosine kinase inhibitors:
Guideline from the college of American pathologists, international
association for the study of lung cancer and association for
molecular pathology. J Mol Diagn. 15:415–453. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Eisenhauer EA, Therasse P, Bogaerts J,
Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S,
Mooney M, et al: New response evaluation criteria in solid tumours:
Revised RECIST guideline (version 1.1). Eur J Cancer. 45:228–247.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
National Cancer Institute, . Common
Terminology Criteria for Adverse Events v3.0 (CTCAE). 2006,
https://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/ctcaev3.pdfAugust
9–2006
|
|
11
|
Imai H, Takahashi T, Mori K, Ono A,
Akamatsu H, Shukuya T, Taira T, Kenmotsu H, Naito T, Murakami H, et
al: Individual-level data on the relationships of progression-free
survival, post-progression survival, and tumor response with
overall survival in patients with advanced non-squamous non-small
cell lung cancer. Neoplasma. 61:233–240. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
DeAngelis LM, Delattre JY and Posner JB:
Radiation-induced dementia in patients cured of brain metastases.
Neurology. 39:789–796. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Aoyama H, Tago M, Kato N, Toyoda T, Kenjyo
M, Hirota S, Shioura H, Inomata T, Kunieda E, Hayakawa K, et al:
Neurocognitive function of patients with brain metastasis who
received either whole brain radiotherapy plus stereotactic
radiosurgery or radiosurgery alone. Int J Radiat Oncol Biol Phys.
68:1388–1395. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Tanriverdi O, Avci N, Oktay E, Kalemci S,
Pilanci KN, Cokmert S, Menekse S, Kocar M, Sen CA, Akman T, et al:
Pretreatment serum albumin level is an independent prognostic
factor in patients with stage IIIB non-small cell lung cancer: A
study of the turkish descriptive oncological researches group.
Asian Pac J Cancer Prev. 16:5971–5976. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kobayashi N, Usui S, Yamaoka M, Suzuki H,
Kikuchi S, Goto Y, Sakai M and Sato Y: The influence of serum
sodium concentration on prognosis in resected non-small cell lung
cancer. Thorac Cardiovasc Surg. 62:338–343. 2014.PubMed/NCBI
|
|
16
|
Castillo JJ, Vincent M and Justice E:
Diagnosis and management of hyponatremia in cancer patients.
Oncologist. 17:756–765. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Simons JP, Schols AM, Buurman WA and
Wouters EF: Weight loss and low body cell mass in males with lung
cancer: Relationship with systemic inflammation, acute-phase
response, resting energy expenditure, and catabolic and anabolic
hormones. Clin Sci (Lond). 97:215–223. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Chang JS, Chen LT, Shan YS, Lin SF, Hsiao
SY, Tsai CR, Yu SJ and Tsai HJ: Comprehensive analysis of the
incidence and survival patterns of lung cancer by histologies,
including rare subtypes, in the era of molecular medicine and
targeted therapy: A nation-wide cancer registry-based study from
Taiwan. Medicine (Baltimore). 94:e9692015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Islam KM, Jiang X, Anggondowati T, Lin G
and Ganti AK: Comorbidity and survival in lung cancer patients.
Cancer Epidemiol Biomarkers Prev. 24:1079–1085. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Jacot W, Colinet B, Bertrand D, Lacombe S,
Bozonnat MC, Daurès JP and Pujol JL; Onco LR health network, :
Quality of life and comorbidity score as prognostic determinants in
non-small-cell lung cancer patients. Ann Oncol. 19:1458–1464. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kuo YW, Jerng JS, Shih JY, Chen KY, Yu CJ
and Yang PC: The prognostic value of the simplified comorbidity
score in the treatment of small cell lung carcinoma. J Thorac
Oncol. 6:378–383. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kim ES, Hirsh V, Mok T, Socinski MA,
Gervais R, Wu YL, Li LY, Watkins CL, Sellers MV, Lowe ES, et al:
Gefitinib versus docetaxel in previously treated non-small-cell
lung cancer (INTEREST): A randomised phase III trial. Lancet.
372:1809–1818. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Sun JM, Lee KH, Kim SW, Lee DH, Min YJ,
Yun HJ, Kim HK, Song HS, Kim YH, Kim BS, et al: Gefitinib versus
pemetrexed as second-line treatment in patients with nonsmall cell
lung cancer previously treated with platinum-based chemotherapy
(KCSG-LU08-01): An open-label, phase 3 trial. Cancer.
118:6234–6242. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Garassino MC, Martelli O, Broggini M,
Farina G, Veronese S, Rulli E, Bianchi F, Bettini A, Longo F,
Moscetti L, et al: Erlotinib versus docetaxel as second-line
treatment of patients with advanced non-small-cell lung cancer and
wild-type EGFR tumours (TAILOR): A randomised controlled trial.
Lancet Oncol. 14:981–988. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kawaguchi T, Ando M, Asami K, Okano Y,
Fukuda M, Nakagawa H, Ibata H, Kozuki T, Endo T, Tamura A, et al:
Randomized phase III trial of erlotinib versus docetaxel as second-
or third-line therapy in patients with advanced non-small-cell lung
cancer: Docetaxel and Erlotinib Lung Cancer Trial (DELTA). J Clin
Oncol. 32:1902–1908. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zhao HY, Zhang Y, Huang H, Chen LK, Xu GC
and Zhang L: Efficacy of erlotinib on advanced non-small cell lung
cancer. Ai Zheng. 27:393–399. 2008.(In Chinese). PubMed/NCBI
|
|
27
|
Li N, Ou W, Yang H, Liu QW, Zhang SL, Wang
BX and Wang SY: A randomized phase 2 trial of erlotinib versus
pemetrexed as second-line therapy in the treatment of patients with
advanced EGFR wild-type and EGFR FISH-positive lung adenocarcinoma.
Cancer. 120:1379–1386. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ma K, Cohen V, Kasymjanova G, Small D,
Novac K, Peterson J, Levit A and Agulnik J: An exploratory
comparative analysis of tyrosine kinase inhibitors or docetaxel in
second-line treatment of EGFR wild-type non-small-cell lung cancer:
A retrospective real-world practice review at a single tertiary
care centre. Curr Oncol. 22:e157–e163. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Lee JK, Hahn S, Kim DW, Suh KJ, Keam B,
Kim TM, Lee SH and Heo DS: Epidermal growth factor receptor
tyrosine kinase inhibitors vs conventional chemotherapy in
non-small cell lung cancer harboring wild-type epidermal growth
factor receptor: A meta-analysis. JAMA. 311:1430–1437. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zhao N, Zhang XC, Yan HH, Yang JJ and Wu
YL: Efficacy of epidermal growth factor receptor inhibitors versus
chemotherapy as second-line treatment in advanced non-small-cell
lung cancer with wild-type EGFR: A meta-analysis of randomized
controlled clinical trials. Lung Cancer. 85:66–73. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Vale CL, Burdett S, Fisher DJ, Navani N,
Parmar MK, Copas AJ and Tierney JF: Should tyrosine kinase
inhibitors be considered for advanced non-small-cell lung cancer
patients with wild type EGFR? Two systematic reviews and
meta-analyses of randomized trials. Clin Lung Cancer.
16:173–182.e4. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Gironés R, Torregrosa D, Gómez-Codina J,
Maestu I, Tenias JM and Rosell R: Prognostic impact of comorbidity
in elderly lung cancer patients: Use and comparison of two scores.
Lung Cancer. 72:108–113. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Haruki T, Yurugi Y, Wakahara M, Matsuoka
Y, Miwa K, Araki K, Taniguchi Y and Nakamura H: Simplified
comorbidity score for elderly patients undergoing thoracoscopic
surgery for lung cancer. Surg Today. 47:718–725. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Meng X, Liu Y, Zhang J, Teng F, Xing L and
Yu J: PD-1/PD-L1 checkpoint blockades in non-small cell lung
cancer: New development and challenges. Cancer Lett. 405:29–37.
2017. View Article : Google Scholar : PubMed/NCBI
|