Introduction
Gastric cancer is the fourth most common type of
cancer, and the second leading cause of cancer-associated mortality
worldwide (1). Therefore, novel
therapies for the treatment of gastric cancer are urgently
needed.
The expression of natural killer group 2 member D
(NKG2D) on natural killer (NK) cells, υδ T cells and
CD8+ T cells was first identified in 1991 (2), and its function was subsequently
reported in 1999 (3). A total of 6
UL16 binding proteins (ULBP1-6) and MHC class I chain-related
proteins A and B (MICA/B) are the two major types of NKG2D ligands
(NKG2DLs) for human NKG2D. mRNA export factor, Mult1, and
histocompatibility antigen 60 are the 3 major NKG2DLs for murine
NKG2D (4). NKG2DLs are induced by
cellular or genomic stress. When expressed on CD8+ T
cells, NKG2D functions to receive co-stimulatory signals, thereby
resulting in CD8+ T-cell activation (5). NKG2DL expression is induced upon stress
and malignant transformation (6). In
addition, NKG2D expression on mononuclear cells is significantly
lower in patients with advanced gastric cancer compared with in
patients with early gastric cancer. Additionally, NKG2D expression
is significantly higher following surgery compared with prior to
surgery in patients with gastric cancer (7). The upregulation of NKG2DLs in cancer
cells subjected to stress results in lysis by NK cells (8).
The surface glycoprotein MICA is an NKG2DL broadly
expressed in epithelial and hematopoietic tumors, but not in
healthy tissue. Subsequent to shedding of MICA in tumors, soluble
MICA functions in the immune escape of tumors. Importantly, the
level of soluble MICA has been positively correlated with tumor
node metastasis (TNM) stage in patients with breast cancer
(9). Additionally, soluble MICA has
been demonstrated to reduce the expression of NKG2D, impair
NK-mediated immune surveillance, which leads to immune escape of
breast tumors (9). Interleukin
(IL)-12 may also upregulate the expression of NKG2D and increase
cytotoxicity of NK cells (10).
However, the potential applications of an additional key NKG2DL,
ULBP1, as a biomarker or prognostic factor for patients with
gastric cancer have not been determined, and the role of the
ULBP1/NKG2D interaction in patients with gastric cancer is not yet
clear.
The present study investigated whether ULBP1 and
NKG2D expression levels were associated with the clinical outcomes
of patients with gastric cancer.
Materials and methods
Gastric cancer cell lines and culture
conditions
The human gastric cancer MKN-74 cell line,
established at the National Cancer Center, was maintained in
Dulbecco's modified Eagle's medium/F12 (Sigma-Aldrich; Merck KGaA,
Darmstadt, Germany) supplemented with 10% heat-inactivated fetal
bovine serum (FBS; Thermo Fisher Scientific, Inc., Waltham, MA,
USA) at 37°C in an atmosphere containing 5% CO2.
Patients
A total of 98 samples were obtained from patients
who underwent surgery at the Department of Digestive Surgery and
Surgical Oncology, Yamaguchi University Graduate School of Medicine
(Yamaguchi, Japan) from January 2004 to December 2008. The patients
were diagnosed with gastric cancer with the tumor invaded through
the muscularis propria or deeper. No patients had received
pre-operative therapy. Pathological staging was performed according
to the Japanese Classification of Gastric Carcinoma (14th edition)
based on the Japanese Gastric Cancer Association TNM staging system
(11). The clinicopathological
characteristics of the patients are summarized in Table I. The study protocol was approved by
the Institutional Review Board for Human Use at Yamaguchi Hospital
(Yamaguchi, Japan). Written informed consent for the present study
was obtained from all patients prior to surgery.
 | Table I.Characteristics of the study
population. |
Table I.
Characteristics of the study
population.
| Variables | No. of cases |
|---|
| Number of
patients | 98 |
| Age, years
(range) | 64±14 (23–92) |
| Gender |
|
|
Male/Female | 65/33 |
| Depth of tumor
invasion |
|
|
MP/SS/SE/SI | 21/25/49/3 |
| Histologic grade |
|
| Papillary
adenocarcinoma/tubular adenocarcinoma/poorly differentiated
adenocarcinoma/signet-ring cell carcinoma/mucinous
adenocarcinoma | 3/28/51/9/7 |
| Stroma |
|
| Medullary
type/interstitial connective tissue/scirrhous type | 7/42/43 |
| Pattern of tumor
infiltration |
|
|
a/b/c | 4/34/60 |
| Lymphatic
invasion |
|
|
0/1/2/3 | 7/25/37/29 |
| Venous
invasion |
|
|
0/1/2/3 | 23/50/21/4 |
| Regional lymph
nodes |
|
|
N0/N1/N2/N3 | 27/20/13/38 |
| Stage |
|
|
I/II/III/IV | 11/30/34/23 |
Analysis of ULBP, MICA/MICB expression
in cancer cell lines and NKG2D expression in NK/T cells by flow
cytometric analysis
Flow cytometric analysis was performed to confirm
the expression of NKG2DLs on the MKN-74 gastric cancer cell and
NKG2D on the surface of the NK and T cells. BD FACSFlow (BD
Biosciences, San Jose, CA, USA) was used for buffer of flow
cytometric analysis. ULBP1 against MKN-74 was stained using
phycoerythrin (PE)-conjugated mouse anti-ULBP1 antibody (dilution,
1:10; cat no. FAB1380P; R&D Systems, Inc., Minneapolis, MN,
USA), and ULBP2/5/6 against MKN-74 was stained using
allophycocyanin (APC) -conjugated mouse anti-ULBP2/5/6 (dilution,
1:10; cat no. FAB1298A; R&D Systems, Inc.). CD3 against NK and
T cells was stained using v450-conjugated mouse anti-CD3 (dilution,
1:20; cat no. 560351; BD Biosciences), CD8 against NK and T cells
was stained using APC-Cy7-conjugated mouse anti-CD8 (dilution,
1:20; cat no. 557834; BD Biosciences), CD16 against NK and T cells
was stained using v500-conjugated mouse anti-CD16 (dilution, 1:20;
cat no. 561394; BD Biosciences), and CD56 against NK and T cells
was stained using PE-CF594-conjugated mouse anti-CD56 (dilution,
1:20; cat no. 562289; BD Biosciences). All samples were maintained
at 4°C for 30 min. MKN-74, mononuclear cells, activated T cells and
activated NK cells were stained to identify each cell surface
markers. Following 3 washes with BD FACSFlow buffer, the cells were
resuspended in phosphate-buffered saline (PBS) with 2% FBS at a
density of 2×105 cells/100 µl. Appropriate isotype
control antibodies were stained in the aforementioned manner.
Samples were analyzed with a BD LSRFortessa X-20
instrument (BD Biosciences), and data were analyzed with FlowJo
software version ×10.0.7r2 (Tree Star, Inc., Ashland OR, USA). The
relative fluorescence intensity (RFI) was calculated as follows:
RFI = [(MFI) of molecule) - (MFI of corresponding isotype
control)]/MFI of corresponding isotype control, where MFI is the
mean fluorescence intensity.
NK and T cell activation
Mononuclear cells (LP_241; Cellular Technology,
Ltd., Cleveland, OH, USA) were activated to generate NK cells by
addition of 5 µl/ml/well of mixed CD2 and CD335 beads with 500
U/ml/well IL-2 (all from Miltenyi Biotec GmbH, Bergisch Gladbach,
Germany) for 2 h. For T-cell activation, the mononuclear cells were
stimulated with 2 µg/ml/well anti-CD3 agonistic antibodies
(eBioscience; Thermo Fisher Scientific, Inc.) and 1 µg/ml/well
anti-CD28 agonistic antibodies (BioLegend, San Diego, CA, USA) with
100 U/ml/well IL-2 (Miltenyi Biotec GmbH) for 2 h.
Immunohistochemistry (IHC)
Paraffin-embedded tissue blocks were cut into 5 µm
sections, mounted on saline-coated slides, and subsequently dewaxed
and rehydrated using xylene and graded alcohol washes. Antigen
retrieval was performed by microwaving in Target Retrieval solution
(pH 9; Dako; Agilent Technologies, Inc., Santa Clara, CA, USA).
Endogenous peroxidase was blocked at room temperature (RT) for 15
min with 0.3% hydrogen peroxide (Wako Pure Chemical Industries,
Ltd., Osaka, Japan) in PBS containing 0.1% sodium azide. Subsequent
to washing twice in PBS, the sections were blocked with Protein
Block Serum-Free (Dako; Agilent Technologies, Inc.) at RT for 30
min. The primary antibodies were then added. Anti-ULBP1 antibodies
(cat no. HPA007547; 1:100; Sigma-Aldrich; Merck KGaA), anti-NKG2D
antibodies (cat no. ab36136; 1:100; Abcam, Cambridge, UK),
anti-CD56 antibodies (cat no. B159; 1:50), and anti-CD8 antibodies
(cat no. SK1; 1:50) (both from BD Biosciences) were used for IHC
according to the manufacturers' protocol. Briefly, following
incubation with primary antibodies at 4°C for 15 h, the slides were
washed twice in PBS, incubated with horseradish
peroxidase-conjugated rabbit/mouse antibodies with original
concentration at RT for 30 min (K406311-2; EnVision Plus mouse/HRP
system; Dako; Agilent Technologies, Inc.), incubated with
Dako-Chromogen solution, and washed in de-ionized water. Following
background staining with Mayer's hematoxylin, the sections were
dehydrated through ascending alcohols to xylene and mounted. To
confirm the specificity of antibodies, negative control slides were
incubated with mouse IgG1 monoclonal antibodies (ab81032; 1:100;
Abcam). IHC results were evaluated by imaging under low-power
magnification (×100) in eight fields of view to identify regions
containing positive immunoreactivity. Immunostaining was
additionally evaluated at a high-power (×400) magnification
(ECLIPSE E200 and Nikon 1 V2 Micro imaging system; Nikon, Tokyo,
Japan). Microscopic analysis of ULBP1 and NKG2D expression was
performed by three blinded independent observers.
Scoring of ULBP1 expression in cancer
cells
Evaluation of ULBP1 staining in the cancer cells was
performed semi-quantitatively, as described previously by Loos
et al (12) Evaluation of
ULBP1 staining was based on the area and intensity of the staining.
Scoring system for area of staining is as follows: Score 1, <33%
of cancer cells; score 2, 33–66% of cancer cells; score 3, >66%
of cancer cells. Scoring for staining intensity is as follows:
Score 1, absent/weak staining; score 2, moderately intense staining
and score 3, strong staining. Each section was given a final grade
derived from the combination of the area and intensity scores. The
final score was calculated by adding scores of intensity and area.
Sections with a final score of 3 were classified as exhibiting low
ULBP1 expression (ULBP1 low), whereas sections with a final score
of >3 were classified as exhibiting high ULBP1 expression (ULBP1
high).
Scoring of NKG2D expression on
mononuclear cells around gastric tumors
Absolute numbers of NKG2D-positive cells in 1 µm
invasive tumor core biopsies were counted manually using an
eyepiece reticule by three blinded independent individuals. The
number of NKG2D-positive cells around the tumors was counted using
a computerized image analysis system composed of an Olympus DP70
CCD camera on an Olympus AX70 light microscope (Olympus, Tokyo,
Japan). Under magnification, ×400, there were ≥8 independent and
intact computerized microscopic fields for the duplicates of each
patient sample. A total of four independent microscopic fields
(magnification, ×400), representing the densest lymphocytic
infiltrates, were selected for each patient sample to ensure
representativeness and homogeneity. The scores for four fields were
averaged to calculate the final number for one computerized
microscopic field at magnification, ×400 (0.0768
mm2/field). The evaluation of NKG2D-positive cells was
performed by three independent blinded observers. Discrepancies in
enumeration, within a range of 5%, were re-evaluated and a
consensus decision was made. The ratio of NKG2D-positive
cells/tumor-infiltrating lymphocytes (TILs) was calculated for each
specimen. The median value was selected as the cut-off for defining
TIL subgroups (median, 0.093). High and low ratios of NKG2D were
termed NKG2D high and NKG2D low, respectively.
Statistical analysis
Differences were analyzed using Fisher's exact
tests. Actuarial OS rates and recurrence-free survival rates were
calculated by the Kaplan-Meier method and analyzed by the log-rank
test. Univariate analyses were performed using Fisher's exact
tests. Multivariate analyses were performed to determine factors
affecting overall survival (OS) based on the Cox proportional
hazards regression model. A secondary analysis was performed to
assess the associations among the expression of NKG2DLs/NKG2D and
clinicopathological characteristics by multivariate analysis based
on the proportional hazards regression model. The activation of T
cells and NK cells was analyzed by Mann-Whitney tests. For
comparisons of individual variables, paired-sample t-tests were
performed. Two-tailed P<0.05 was considered to indicate a
statistically significant difference. All statistical analyses were
performed with EZR version 1.35 (Saitama Medical Center, Jichi
Medical University), a graphical user interface for R (The R
Foundation for Statistical Computing, v.2.13.0), and more
specifically a modified version of R commander (v.1.6–3) designed
to add statistical functions frequently used in biostatistics.
Results
Expression of NKG2DLs in cancer cell
lines
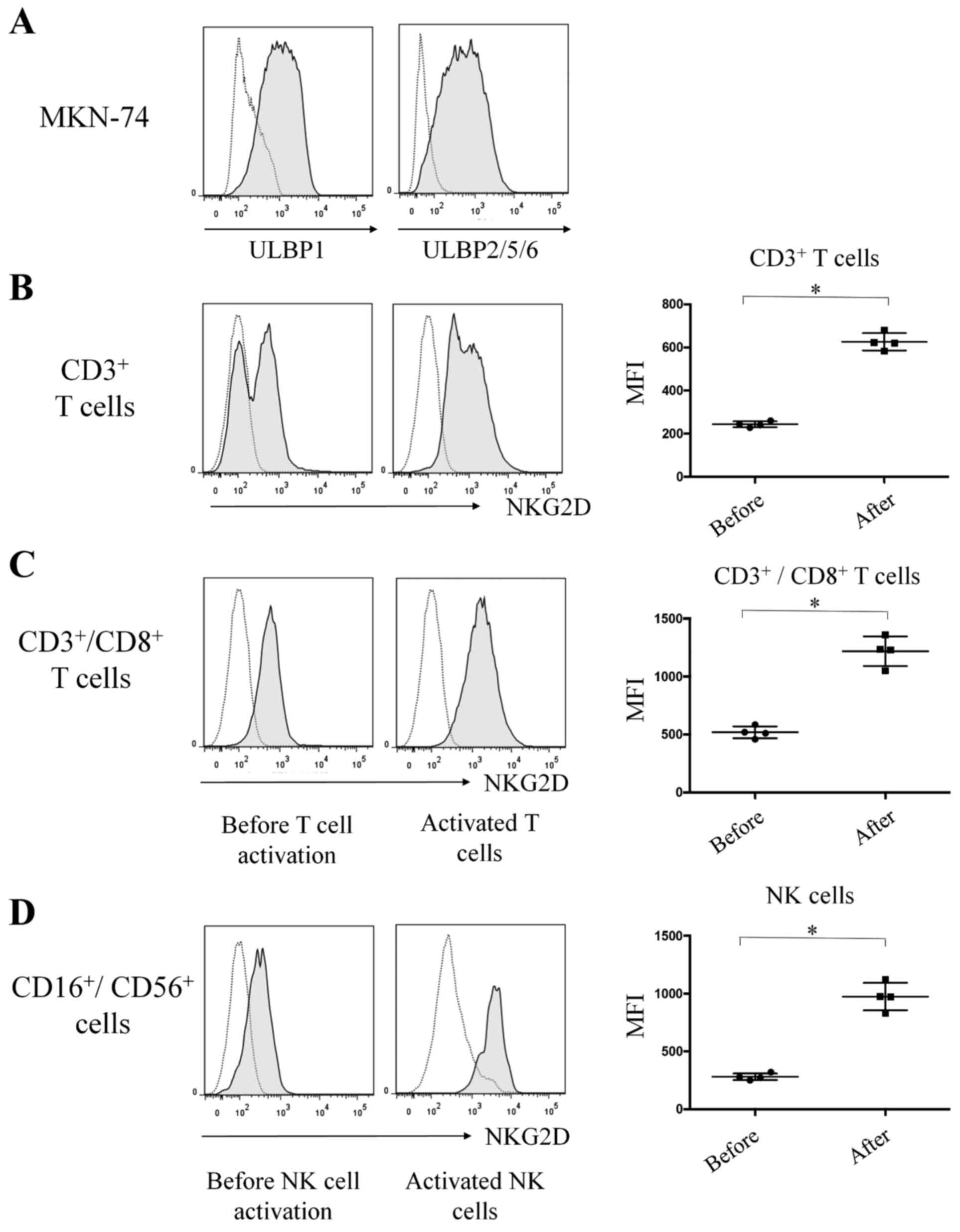
In the flow cytometric analysis, cell surface
expression of ULBP1 and ULBP2/5/6 in MKN-74 cells was
constitutively high (Fig. 1A). By
contrast, cell surface expression of MICA and MICB was not detected
in MKN-74 cells (data not shown).
Expression of NKG2D in mononuclear
cells
The cell surface expression of NKG2D on
CD3+ T cells was significantly increased following
T-cell activation (MFI, 621) compared with the expression (MFI,
243) prior to T-cell activation [P=0.0286, n=4, standard deviation
(SD) = 378; Fig. 1B]. Similarly, cell
surface expression of NKG2D on CD3+CD8+ T
cells was significantly higher following T-cell activation (MFI,
1234) compared with expression (MFI, 515) prior to T-cell
activation (P=0.0286, n=4, SD=719; Fig.
1C). Cell surface expression of NKG2D on
CD16+CD56+ NK cells was significantly higher
following NK cell activation (MFI, 974) compared with expression
(MFI, 276) prior to NK cell activation (P=0.0283, n=4, SD=698;
Fig. 1D).
IHC staining of ULBP1
ULBP1 expression was observed in the cell membrane
and cytoplasm. Fig. 2A and B show
representative images of ULBP1-high staining in gastric cancer
(magnification, ×100 and ×400, respectively). Fig. 2C and D show isotype control staining
in gastric cancer (magnification, ×100 and ×400, respectively). Of
the 98 samples examined in the analysis, 70 were positive for ULBP1
expression.
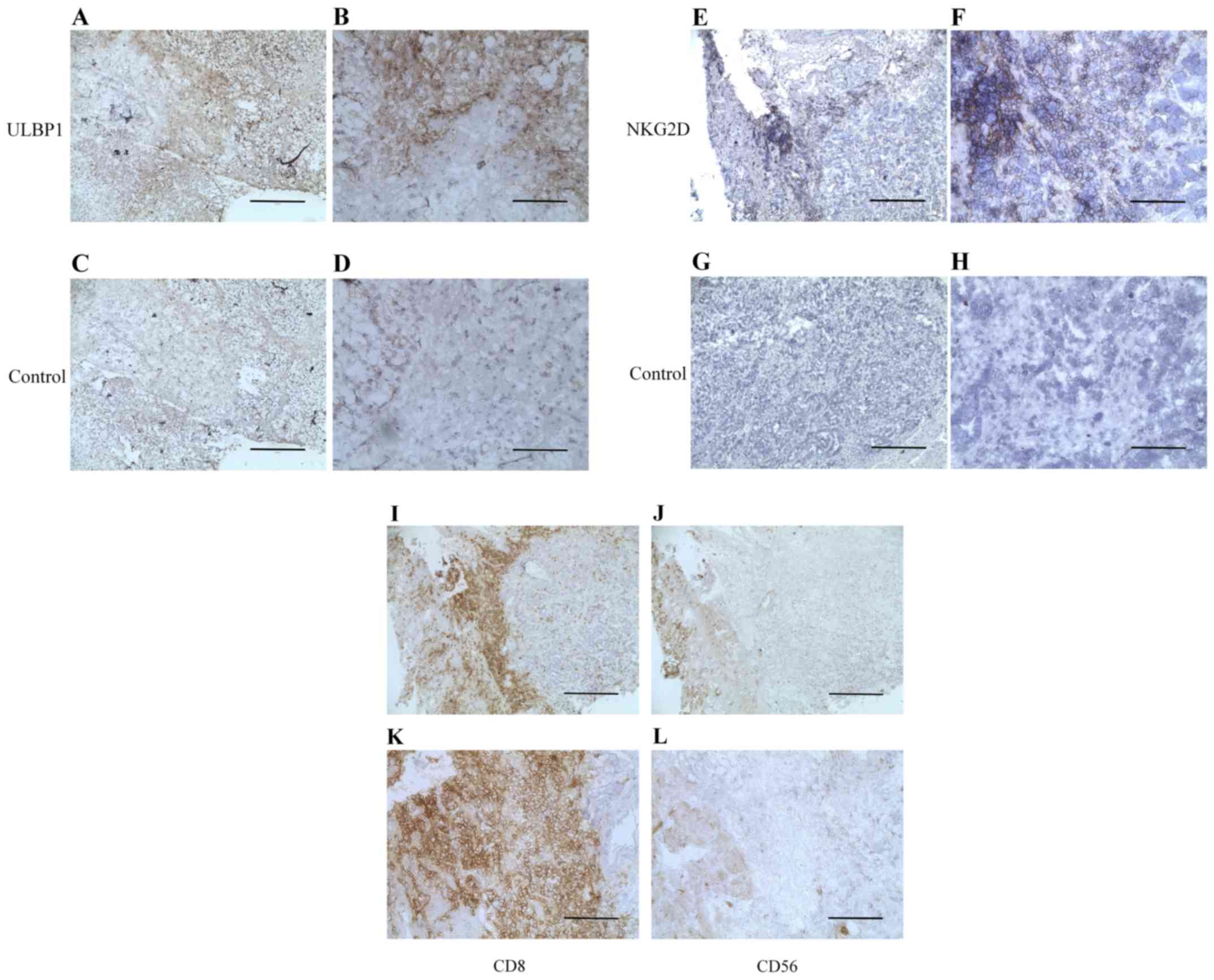 | Figure 2.Representative histopathological
images showing immunohistochemical staining of ULBP1, NKG2D, CD8
and CD56 in gastric cancer tissues. (A) High ULBP1 expression in
gastric cancer tissues. ULBP1 expression is revealed in the cell
membrane and cytoplasm (brown staining). (B) High ULBP1 expression
in gastric cancer tissues. Magnification, ×400; scale bar, 100 µm.
(C) Isotype control for staining of ULBP1. Magnification, ×100;
scale bar, 200 µm. (D) Isotype control for staining of ULBP1.
Magnification, ×400; scale bar, 100 µm. (E) High NKG2D expression
in tumor-infiltrating lymphocytes. Magnification, ×100; scale bar,
200 µm. (F) High NKG2D expression in tumor-infiltrating
lymphocytes. Magnification, ×400; scale bar, 100 µm. (G) Isotype
control for staining of tumor-infiltrating lymphocytes.
Magnification, ×100; scale bar, 200 µm. (H) Isotype control for
staining of tumor-infiltrating lymphocytes. Magnification, ×400;
scale bar, 100 µm. (I) Staining of CD8-positive cells in gastric
cancer. Magnification, ×100; scale bar, 200 µm. (J) Staining of
CD8-positive cells in gastric cancer tissues. Magnification, ×400;
scale bar, 100 µm. (K) Staining of CD56-positive cells.
Magnification, ×100; scale bar, 200 µm. (L) Staining of
CD56-positive cells. Magnification, ×400; scale bar, 100 µm.
Immunohistochemical detection of CD8-positive or CD56-positive
mononuclear cells around gastric tumors in I-L used the same
sections as those in E-H. ULBP, UL16 binding protein; CD, cluster
of differentiation; NKG2D, natural killer group 2 member D. |
IHC staining of NKG2D
NKG2D was expressed on the membrane of mononuclear
cells around the gastric tumors. Fig. 2E
and F reveal high NKG2D expression in TILs with (magnification,
×100 and ×400, respectively). Fig. 2G and
H demonstrate isotype control staining in TILs (magnification,
×100 and ×400, respectively) in the same sections as those used in
Fig. 2E and F. Of the 98 samples, 19
were positive for NKG2D.
IHC staining of NKG2D+,
CD8+, and CD56+
Among the NKG2D-expressing tissue sections, a total
of 6 samples were stained for CD8 and CD56 and were used as
representative sections. Fig. 2I-L
displays CD8 or CD56 expression in the same samples as those
analyzed in Fig. 2E-H. In all
tissues, the majority of NKG2D-expressing cells were
CD8+ T cells.
Expression of ULBP1 and NKG2D and
clinicopathological findings
Univariate analysis indicated that NKG2D expression
in mononuclear cells was associated with lymphatic invasion
(P=0.0028) and postoperative recurrence (P=0.0048; Table II). No significant associations were
observed between ULBP1 expression and pathological findings or
postoperative recurrence (Table
II).
 | Table II.Univariate analysis of overall
survival of patients with gastric cancer following gastrectomy. |
Table II.
Univariate analysis of overall
survival of patients with gastric cancer following gastrectomy.
|
|
| ULBP1 | NKG2D |
|---|
|
|
|
|
|
|---|
| Parameters | Cases, n | Positive
(n=70) | Negative
(n=28) | P-value | Positive
(n=19) | Negative
(n=79) | P-value |
|---|
| Depth of tumor
invasion (T) |
|
|
| 0.076 |
|
| 0.132 |
| T2
(muscularis propria or subserosa) | 46 | 37 | 9 |
| 12 | 34 |
|
| T3
(serosal exposure), T4 (invasion) | 52 | 33 | 19 |
| 7 | 45 |
|
| Histological
classification of gastric tumors |
|
|
| 0.814 |
|
| 0.784 |
|
Papillary
adenocarcinoma/tubular adenocarcinoma | 30 | 21 | 9 |
| 5 | 26 |
|
| Poorly
differentiated adenocarcinoma/signet-ring cell carcinoma/mucinous
adenocarcinoma | 68 | 49 | 19 |
| 14 | 53 |
|
| Cancer stromal
volume |
|
|
| 0.647 |
|
| 1.000 |
|
Medullary type/interstitial
connective tissue | 49 | 36 | 13 |
| 9 | 40 |
|
|
Scirrhous type | 48 | 29 | 14 |
| 9 | 39 |
|
| Tumor infiltrative
pattern into the surrounding tissues |
|
|
| 0.819 |
|
| 1.000 |
| a,
b | 38 | 28 | 10 |
| 7 | 31 |
|
| c | 60 | 42 | 18 |
| 12 | 48 |
|
| Lymphatic
invasion |
|
|
| 0.669 |
|
| 0.003 |
| 0 | 7 | 6 | 1 |
| 5 | 2 |
|
| 1, 2,
3 | 91 | 64 | 27 |
| 14 | 77 |
|
| Venous
invasion |
|
|
| 0.798 |
|
| 0.066 |
| 0 | 23 | 16 | 7 |
| 8 | 15 |
|
| 1, 2,
3 | 75 | 54 | 21 |
| 11 | 64 |
|
| Lymph node
metastasis |
|
|
| 0.461 |
|
| 0.776 |
| 0 | 27 | 21 | 6 |
| 6 | 21 |
|
| 1, 2,
3 | 71 | 49 | 22 |
| 13 | 58 |
|
| Stage grouping |
|
|
| 0.262 |
|
| 0.311 |
| I,
II | 41 | 32 | 9 |
| 10 | 31 |
|
| III,
IV | 57 | 38 | 19 |
| 9 | 48 |
|
| Stage grouping |
|
|
| 0.111 |
|
| 0.549 |
| I, II,
III | 75 | 57 | 18 |
| 16 | 59 |
|
| IV | 23 | 13 | 10 |
| 3 | 20 |
|
| Postoperative
recurrence |
|
|
| 0.799 |
|
| 0.005 |
|
Presence | 33 | 25 | 8 |
| 2 | 31 |
|
|
Absence | 45 | 32 | 12 |
| 15 | 30 |
|
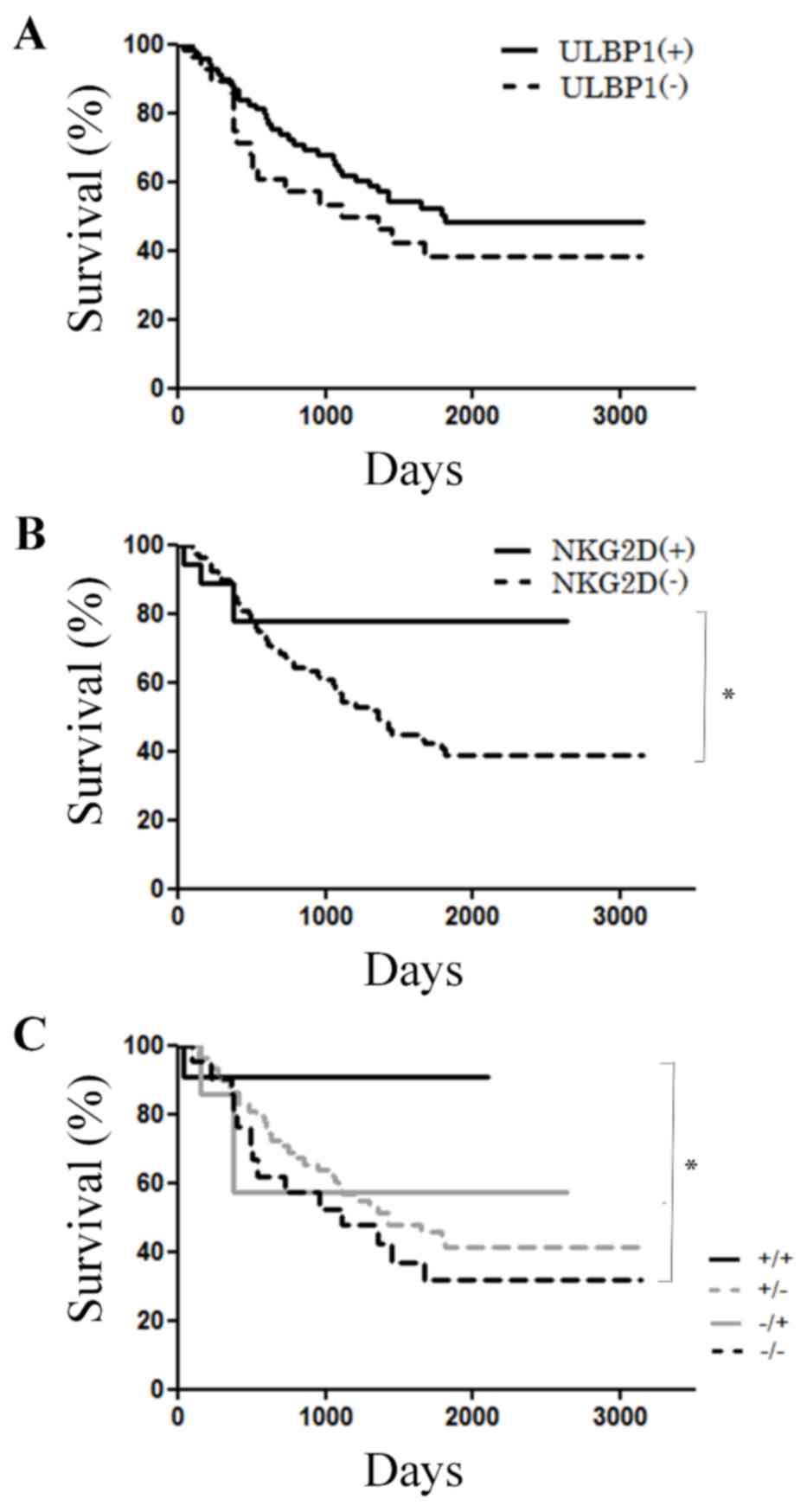
Survival curves following surgery were compared
between high and low ULBP1 and NKG2D expression groups. A total of
70 patients were positive for ULBP1, whereas 28 patients were
negative for ULBP1 (Table II). By
contrast, a total of 19 and 79 patients were positive and negative
for NKG2D, respectively (Table II).
There were no significant differences in OS observed according to
ULBP1 expression (P=0.334; Fig. 3A).
However, patients with NKG2D expression were indicated to have
significantly longer OS compared with patients without NKG2D
expression (P=0.0217; Fig. 3B).
Combined expression of NKG2D and ULBP1 was also analyzed and are as
follows: ULBP1-high/NKG2D-high (n=12), ULBP1-high/NKG2D-low (n=58),
ULBP1-low/NKG2D-high (n=7), and ULBP1-low/NKG2D-low (n=21). With
regards to OS, the findings indicated that the
ULBP1-high/NKG2D-high group had the best prognosis, and the
ULBP1-low/NKG2D-low group had the poorest prognosis (P=0.0096;
Fig. 3C).
Multivariate analysis
Multivariate analysis was used to determine the
hazard ratios for OS conferred by ULBP1, NKG2D, sex, lymph node
metastasis, lymphatic invasion and venous invasion. NKG2D
expression and lymph node metastasis were identified to be
independent prognostic factors for OS (P=0.0431 and P=0.0030,
respectively; Table III).
 | Table III.Multivariate proportional hazards
analysis of overall survival patients with gastric cancer following
gastrectomy. |
Table III.
Multivariate proportional hazards
analysis of overall survival patients with gastric cancer following
gastrectomy.
| Variable | Hazard ratio | 95% CI | P-value |
|---|
| Lymph node
metastasis | 1.5670 | 1.1640–2.1100 | 0.0030 |
| NKG2D
expression | 0.3369 | 0.1173–0.9672 | 0.0431 |
Discussion
The present study examined the association between
prognosis and expression of NKG2D and ULBP1. The results indicated
that detecting a combination of ULBP1 and NKG2D expression might be
an effective strategy for prediction of OS in patients with gastric
cancer, and therefore this may provide important insights into the
roles of these two proteins in gastric cancer. A number of solid
tumors constitutively express NKG2DLs and are receptive to
NKG2D-dependent immunosurveillance (6). However, soluble NKG2DLs are released
from tumors and block NKG2D activation, resulting in tumor immune
escape (6). It has been demonstrated
that the levels of soluble NKG2DLs in serum affect tumor
progression (6). In addition, MICA
shed from the cell surface may affect NKG2D/NKG2DL interactions
(6). No prior studies had examined
the role of the soluble form of ULBP1. In the present study, ULBP1
did not exhibit any antagonistic effects on tumor immunity through
NKG2D expression.
NKG2DLs are often expressed in breast cancer tissues
(13). In particular, MIC-A/B and
ULBP2 expression is associated with an improved prognosis in
patients with cancer (13) and
immunological function of NKG2DLs have not been demonstrated
(13). However, other studies have
suggested that soluble MICA is involved in tumor immune escape
(9). In the present study, ULBP1
expression in gastric cancer may have acted as a positive ligand by
binding to NKG2D, thereby prognoses are improved when these
proteins were co-expressed. Additionally, it was suggested in Osaki
et al (14) that decreased
NKG2D expression may be responsible for immune evasion by tumors in
gastric cancer, which is consistent with the results of the present
study in which patients with low NKG2D expression exhibited poorer
survival.
The present study revealed that the interaction of
NKG2DLs and NKG2D was involved in cancer and T-cell activation in
tumors. This interaction is also thought to be important for NK
activation in cancer (3). Therefore,
the NKG2D/ULBP1 interaction may be involved in the association
between cancer and T cells. ULBP1 and NKG2D, and the ULBP1/NKG2D
interaction, may be molecular targets for immunotherapy. In
patients with advanced non-small-cell lung cancer, melanoma and
renal-cell cancer, the inhibition of programmed death ligand 1
(PD-L1) exhibited a robust response and blocked disease progression
(15). Anti-PD-1 antibodies produced
objective responses in ~20–25% of patients with non-small-cell lung
cancer, melanoma or renal-cell cancer (16). Therefore, one approach for blocking
the ULBP1/NKG2D interaction is to use specific antibodies against
ULBP1 and NKG2D, which may target T cells at sites of solid tumors,
and therefore accelerates the activation of T cells. Additional
studies are required to determine the effects of the ULBP1/NKG2D
interaction on the adaptive immune system.
Therefore, the interaction between ULBP1 and NKG2D
in gastric cancer may be associated with OS. In addition, the
results of the present study suggested that ULBP1 and NKG2D may be
applied in immunotherapy to induce acquired immunity in patients
with cancer and for the prediction of prognosis following surgical
resection in patients with gastric cancer. The detailed
interactions between NKG2DL and NKG2D require additional
investigation. The development of NKG2DL-targeted immunotherapy may
have important future clinical applications.
Acknowledgements
The present study was supported in part by the
National Cancer Center Research and Development Fund (grant no.
27-A-2). The authors would like to thank Ms Akiko Sano (Yamaguchi
University, Ube, Japan) for technical support. The authors would
also like to thank Editage (Tokyo, Japan) and Ms Manami Kobayashi
(National Cancer Center, Tokyo, Japan) for assistance with English
language editing.
Glossary
Abbreviations
Abbreviations:
|
ULBP1
|
UL16 binding protein 1
|
|
MICA
|
MHC class I chain-related proteins
A
|
|
NKG2D
|
natural killer group 2 member D
|
References
|
1
|
Edge S, Byrd DR, Compton CC, Fritz AG,
Greene FL and Trotti A: AJCC Cancer Staging Manual. Springer;
2010
|
|
2
|
Houchins JP, Yabe T, McSherry C and Bach
FH: DNA sequence analysis of NKG2, a family of related cDNA clones
encoding type II integral membrane proteins on human natural killer
cells. J Exp Med. 173:1017–1020. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Bauer S, Groh V, Wu J, Steinle A, Phillips
JH, Lanier LL and Spies T: Activation of NK cells and T cells by
NKG2D, a receptor for stress-inducible MICA. Science. 285:727–729.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Raulet DH: Roles of the NKG2D
immunoreceptor and its ligands. Nat Rev Immunol. 3:781–790. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
González S, López-Soto A, Suarez-Alvarez
B, López-Vázquez A and López-Larrea C: NKG2D ligands: Key targets
of the immune response. Trends Immunol. 29:397–403. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chitadze G, Bhat J, Lettau M, Janssen O
and Kabelitz D: Generation of soluble NKG2D ligands: Proteolytic
cleavage, exosome secretion and functional implications. Scand J
Immunol. 78:120–129. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Saito H, Osaki T and Ikeguchi M: Decreased
NKG2D expression on NK cells correlates with impaired NK cell
function in patients with gastric cancer. Gastric Cancer. 15:27–33.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zafirova B, Wensveen FM, Gulin M and Polić
B: Regulation of immune cell function and differentiation by the
NKG2D receptor. Cell Mol Life Sci. 68:3519–3529. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Holdenrieder S, Stieber P, Peterfi A,
Nagel D, Steinle A and Salih HR: Soluble MICA in malignant
diseases. Int J Cancer. 118:684–687. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zhang C, Zhang J, Niu J, Zhou Z, Zhang J
and Tian Z: Interleukin-12 improves cytotoxicity of natural killer
cells via upregulated expression of NKG2D. Hum Immunol. 69:490–500.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Japanese Gastric Cancer Association, .
Japanese classification of gastric carcinoma-2nd english edition.
Gastric Cancer. 1:10–24. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Loos M, Hedderich DM, Ottenhausen M, Giese
NA, Laschinger M, Esposito I, Kleeff J and Friess H: Expression of
the costimulatory molecule B7-H3 is associated with prolonged
survival in human pancreatic cancer. BMC Cancer. 9:4632009.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
de Kruijf EM, Sajet A, van Nes JG, Putter
H, Smit VT, Eagle RA, Jafferji I, Trowsdale J, Liefers GJ, van de
Velde CJ and Kuppen PJ: NKG2D ligand tumor expression and
association with clinical outcome in early breast cancer patients:
An observational study. BMC Cancer. 12:242012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Osaki T, Saito H, Yoshikawa T, Matsumoto
S, Tatebe S, Tsujitani S and Ikeguchi M: Decreased NKG2D expression
on CD8+ T cell is involved in immune evasion in patients with
gastric cancer. Clin Cancer Res. 13:382–387. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Brahmer JR, Tykodi SS, Chow LQ, Hwu WJ,
Topalian SL, Hwu P, Drake CG, Camacho LH, Kauh J, Odunsi K, et al:
Safety and activity of anti-PD-L1 antibody in patients with
advanced cancer. N Engl J Med. 366:2455–2465. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Topalian SL, Hodi FS, Brahmer JR,
Gettinger SN, Smith DC, McDermott DF, Powderly JD, Carvajal RD,
Sosman JA, Atkins MB, et al: Safety, activity, and immune
correlates of anti-PD-1 antibody in cancer. N Engl J Med.
366:2443–2454. 2012. View Article : Google Scholar : PubMed/NCBI
|