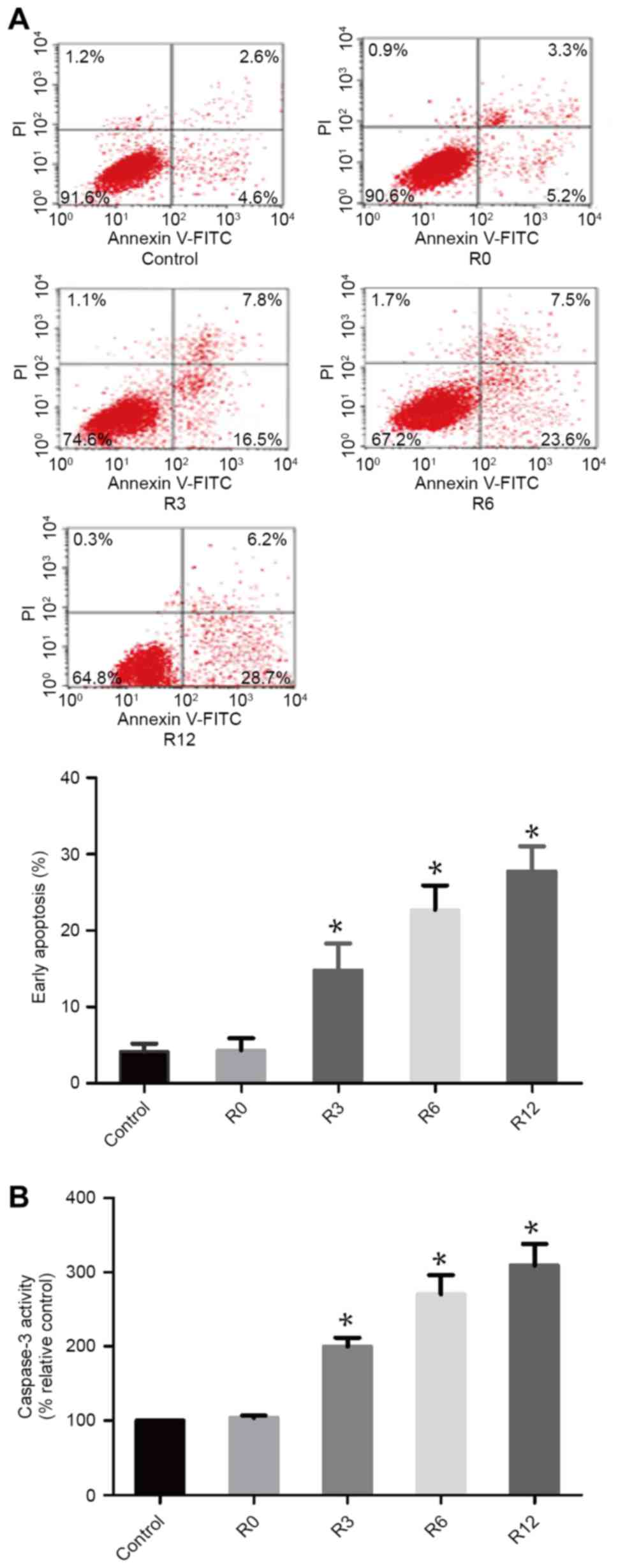
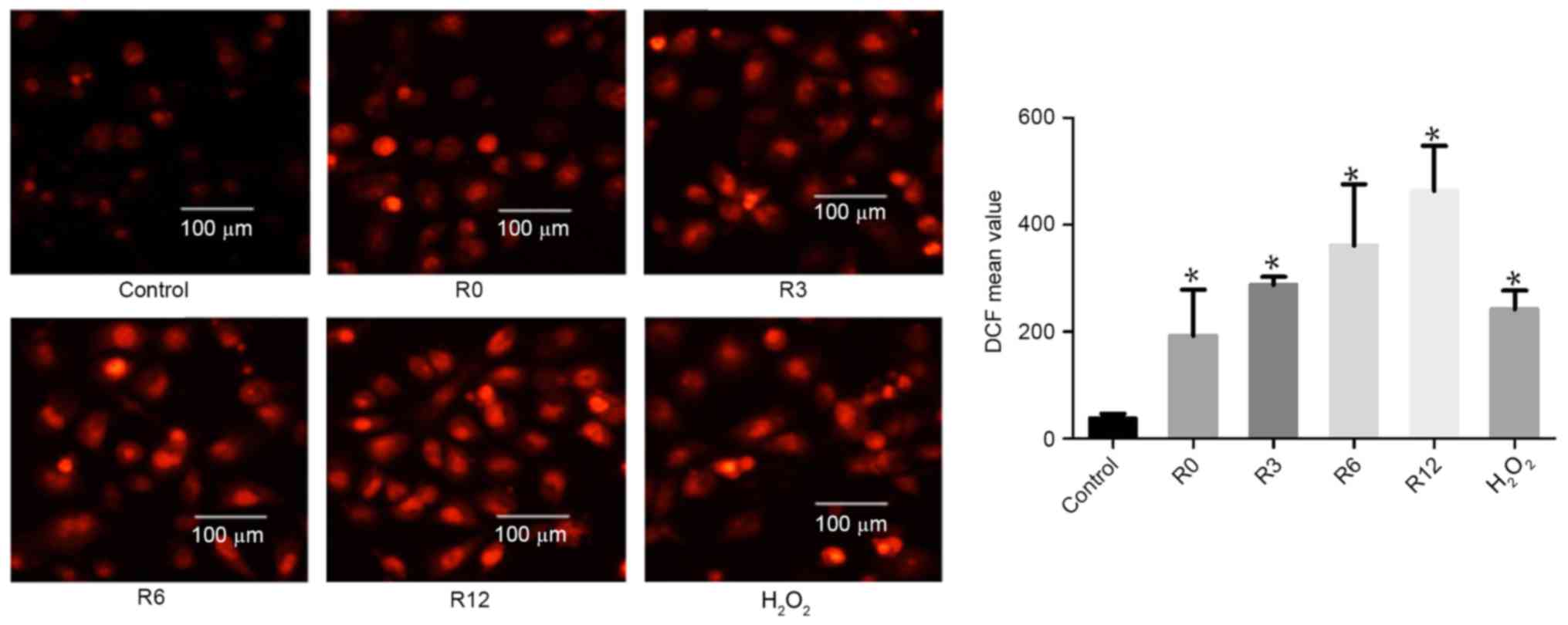
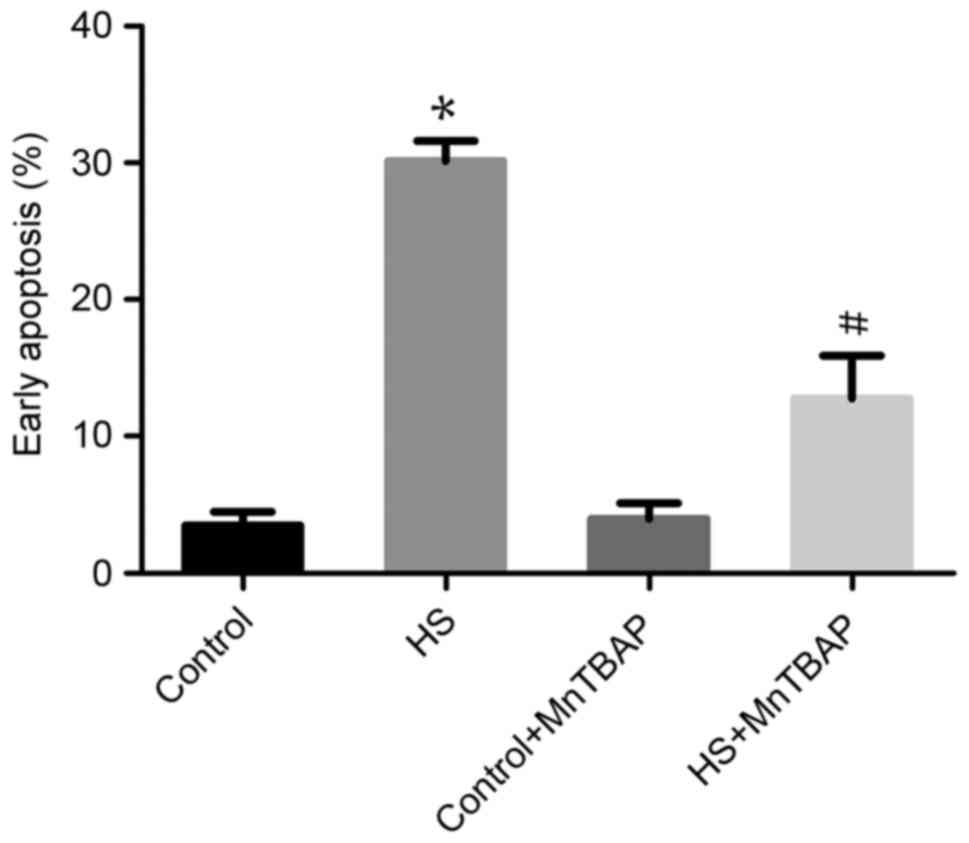
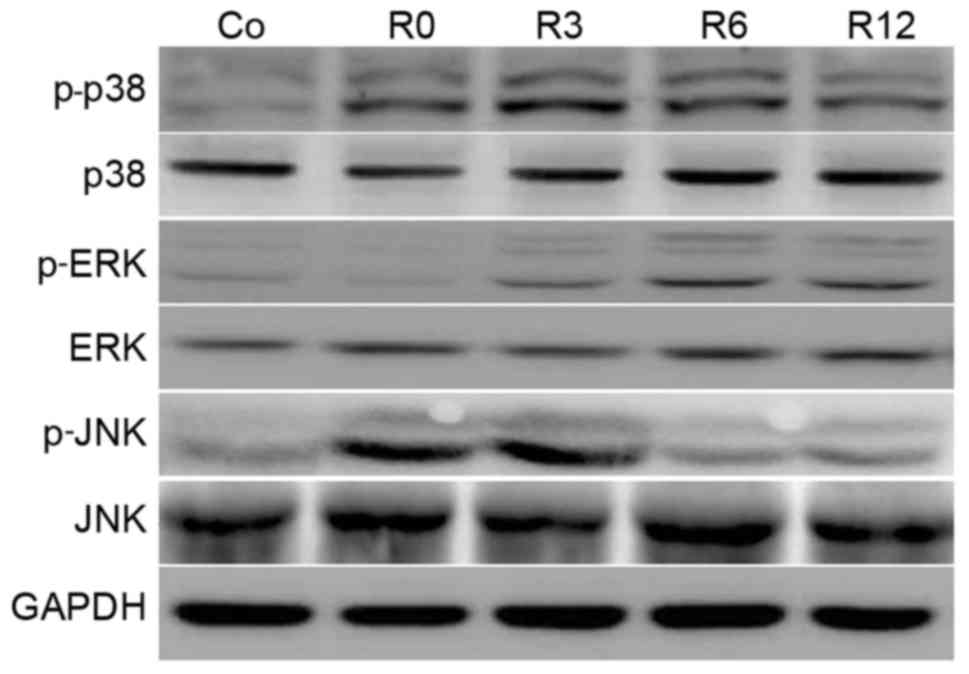
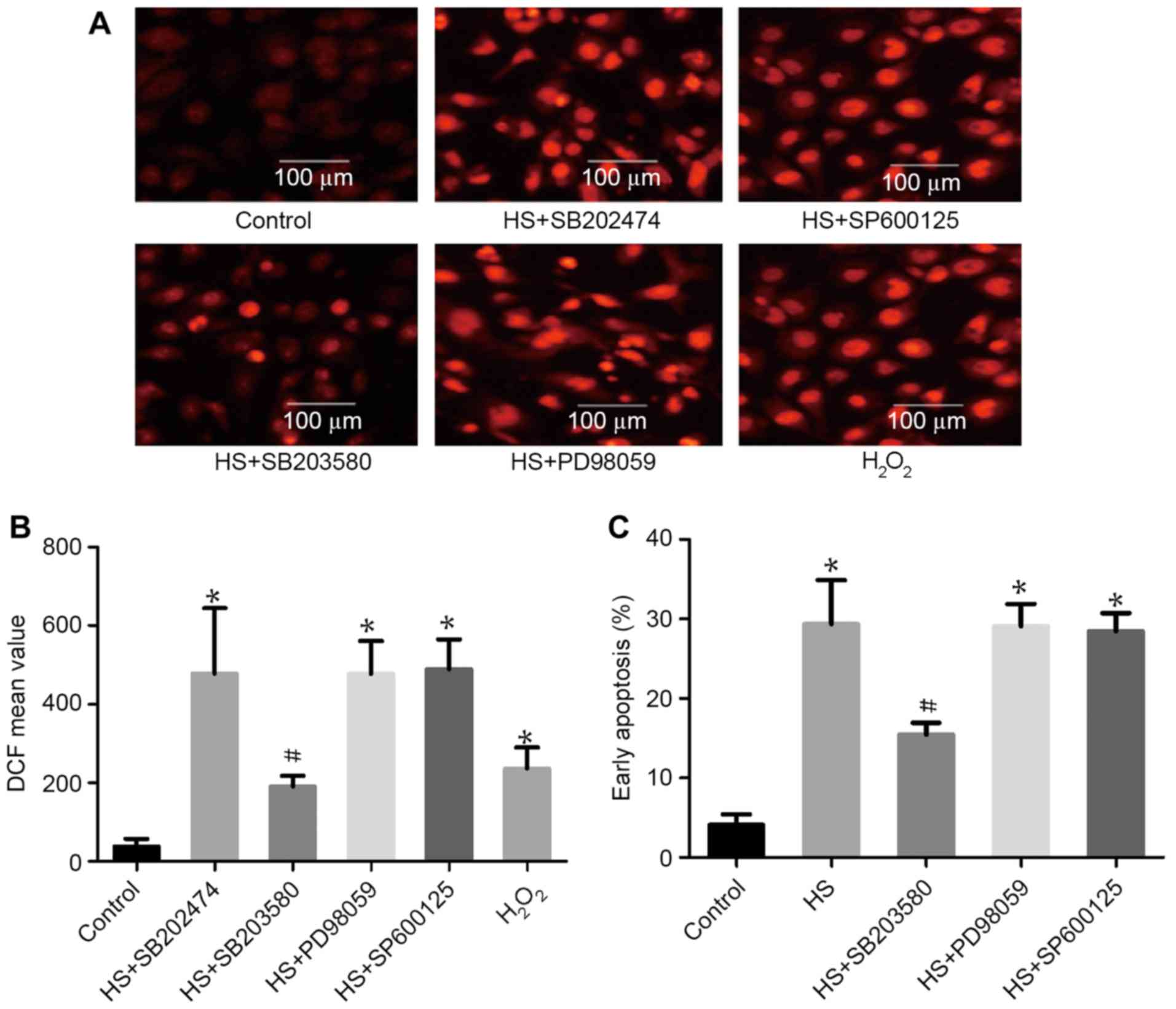
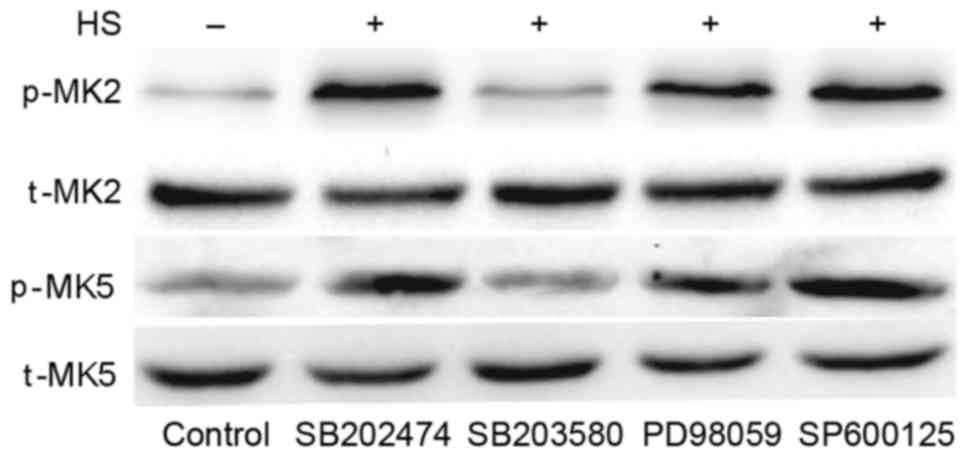
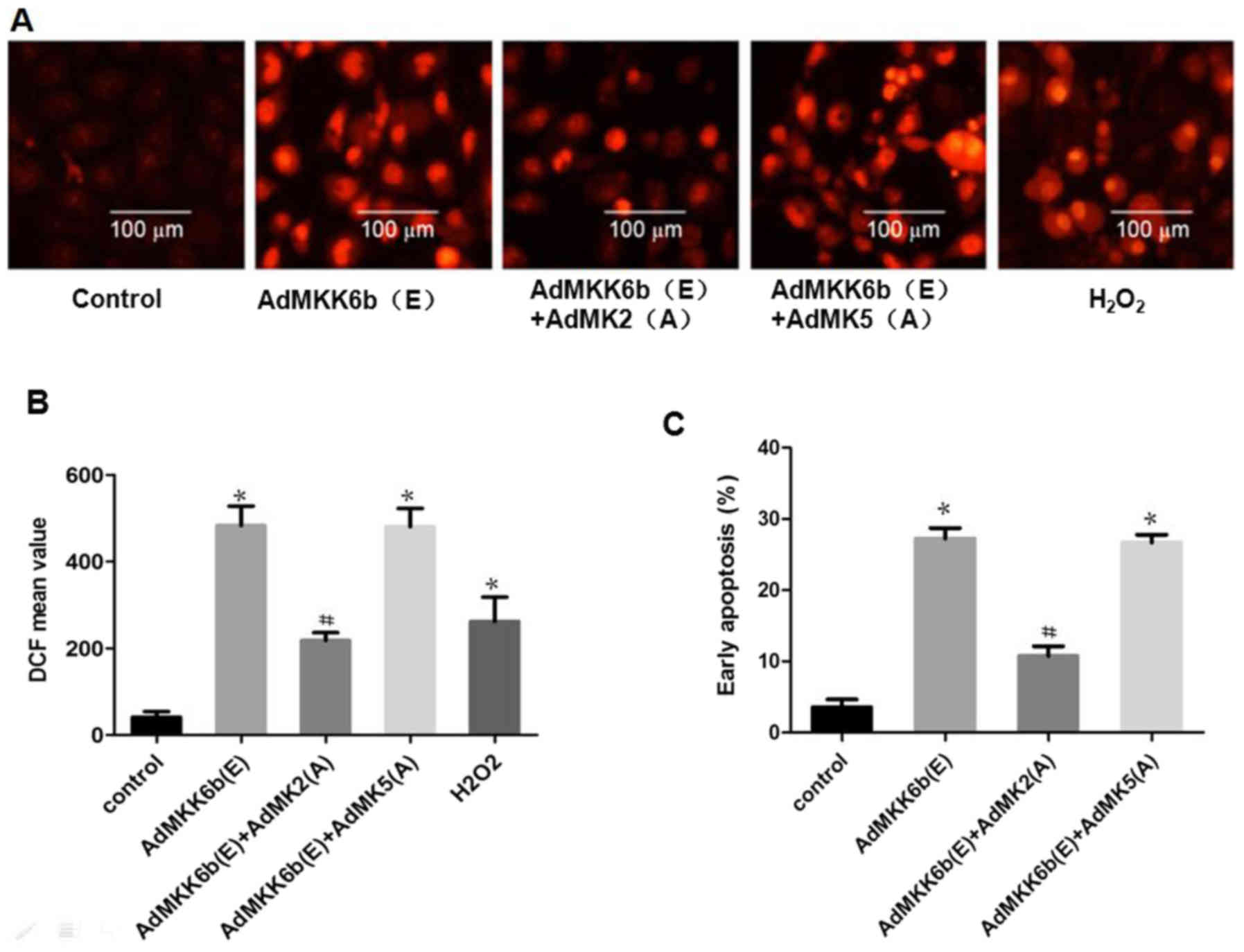
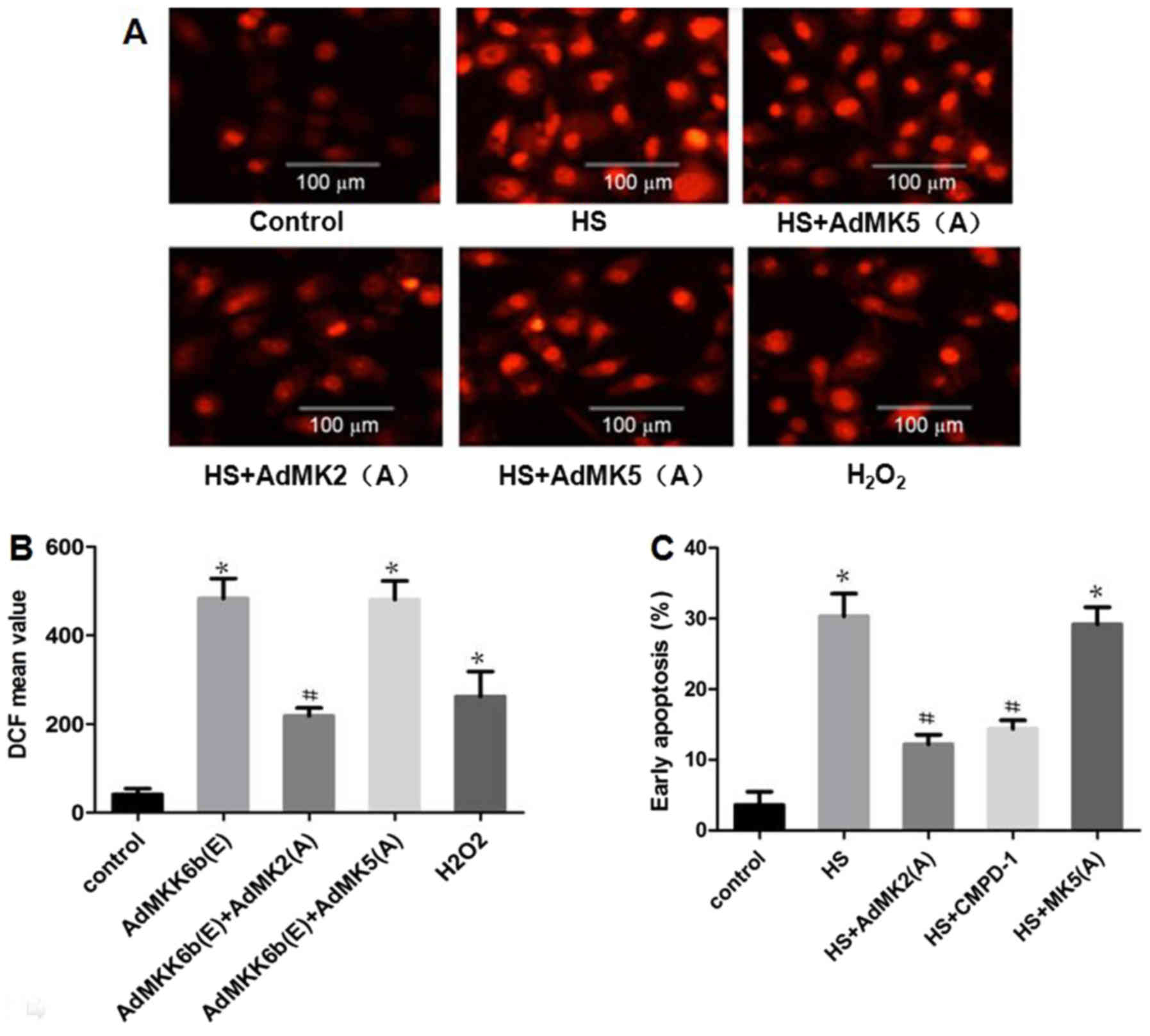
|
1
|
Malamud N, Haymaker W and Custer RP: Heat
stroke: A clinicoorgan pathological study of 125 fatal cases. Mil
Surg. 99:394–449. 1946.
|
|
2
|
Hausfater P, Megarbane B, Dautheville S,
Patzak A, Andronikof M, Santin A, André S, Korchia L, Terbaoui N,
Kierzek G, et al: Prognostic factors in non-exertional heatstroke.
Intensive Care Med. 36:272–280. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Dematte JE, O'Mara K, Buescher J, Whitney
CG, Forsythe S, McNamee T, Adiga RB and Ndukwu IM: Near-fatal heat
stroke during the 1995 heat wave in Chicago. Ann Intern Med.
129:173–181. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Mastrangelo G, Fedeli U, Visentin C, Milan
G, Fadda E and Spolaore P: Pattern and determinants of
hospitalization during heat waves: An ecologic study. BMC Public
Health. 7:2002007. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kovats RS and Kristie LE: Heatwaves and
public health in Europe. Eur J Public Health. 16:592–599. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Edwards MJ, Walsh DA and Li Z:
Hyperthermia, teratogenesis and the heat shock response in
mammalian embryos in culture. Int J Dev Biol. 41:345–358.
1997.PubMed/NCBI
|
|
7
|
Yu J, Liu F, Yin P, Zhao H, Luan W, Hou X,
Zhong Y, Jia D, Zan J, Ma W, et al: Involvement of oxidative stress
and mitogen-activated protein kinase signaling pathways in heat
stress-induced injury in the rat small intestine. Stress.
16:99–113. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Liu Y, Zhou G, Wang Z, Guo X, Xu Q, Huang
Q and Su L: NF-κB signaling is essential for resistance to heat
stress-induced early stage apoptosis in human umbilical vein
endothelial cells. Sci Rep. 5:135472015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Chang L and Karin M: Mammalian MAP kinase
signalling cascades. Nature. 410:37–40. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Seger R and Krebs EG: The MAPK signaling
cascade. FASEB J. 9:726–735. 1995.PubMed/NCBI
|
|
11
|
Roux PP and Blenis J: ERK and p38
MAPK-activated protein kinases: A family of protein kinases with
diverse biological functions. Microbiol Mol Biol Rev. 68:320–344.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kim EK and Choi EJ: Pathological roles of
MAPK signaling pathways in human diseases. Biochim Biophys Acta.
1802:396–405. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Cargnello M and Roux PP: Activation and
function of the MAPKs and their substrates, the MAPK-activated
protein kinases. Microbiol Mol Biol Rev. 75:50–83. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Gaestel M: MAPKAP kinases-MKs-two's
company, three's a crowd. Nat Rev Mol Cell Biol. 7:120–30. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
New L, Jiang Y, Zhao M, Liu K, Zhu W,
Flood LJ, Kato Y, Parry GC and Han J: PRAK, a novel protein kinase
regulated by the p38 MAP kinase. EMBO J. 17:3372–3384. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Liu R, Wu CX, Zhou D, Yang F, Tian S,
Zhang L, Zhang TT and Du GH: Pinocembrin protects against
β-amyloid-induced toxicity in neurons through inhibiting receptor
for advanced glycation end products (RAGE)-independent signaling
pathways and regulating mitochondrion-mediated apoptosis. BMC Med.
10:1052012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Gu ZT, Wang H, Li L, Liu YS, Deng XB, Huo
SF, Yuan FF, Liu ZF, Tong HS and Su L: Heat stress induces
apoptosis through transcription-independent p53-mediated
mitochondrial pathways in human umbilical vein endothelial cell.
Sci Rep. 4:44692014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Fiers W, Beyaert R, Declercq W and
Vandenabeele P: More than one way to die: Apoptosis, necrosis and
reactive oxygen damage. Oncogene. 18:7719–7730. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Sneed PK, Matsumato K, Stauffer PR, Fike
JR, Smith V and Gutin PH: Interstitial microwave hyperthermia in a
canine brain model. Int J Radiat Oncol Biol Phys. 12:1887–1897.
1986. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Wu W, Huan Q, Miao J, Xiao M, Liu H, Zhao
K and Zhao M: MK2 plays an important role for the increased
vascular permeability that follows thermal injury. Burns.
39:923–934. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Dorozynski A: Chirac announces
investigation into heat wave's death toll. BMJ. 327:4652003.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Harris AB, Erickson L, Kendig JH, Mingrino
S and Goldring S: Observations on selective brain heating in dogs.
J Neurosurg. 19:514–521. 1962. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Burdon RH, Gill VM and Rice-Evans C:
Oxidative stress and heat shock protein induction in human cells.
Free Radic Res Commun. 3:129–139. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Skibba JL, Powers RH, Stadnicka A,
Cullinane DW, Almagro UA and Kalbfleisch JH: Oxidative stress as a
precursor to the irreversible hepatocellular injury caused by
hyperthermia. Int J yperthermia. 7:749–761. 1991. View Article : Google Scholar
|
|
25
|
McAnulty SR, McAnulty L, Pascoe DD,
Gropper SS, Keith RE, Morrow JD and Gladden LB: Hyperthermia
increases exercise-induced oxidative stress. Int J Sports Med.
26:188–192. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Cuenda A and Rousseau S: p38 MAP-kinases
pathway regulation, function and role in human diseases. Biochim
Biophys Acta. 1773:1358–1375. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Cuadrado A and Nebreda AR: Mechanisms and
functions of p38 MAPK signaling. Biochem J. 429:403–417. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Zhou J, Wan B, Liu XM, Li R, Wang Y and Yu
L: MK5 is degraded in response to doxorubicin and negatively
regulates doxorubicin-induced apoptosis in hepatocellular carcinoma
cells. Biochem Biophys Res Commun. 427:581–586. 2012. View Article : Google Scholar : PubMed/NCBI
|