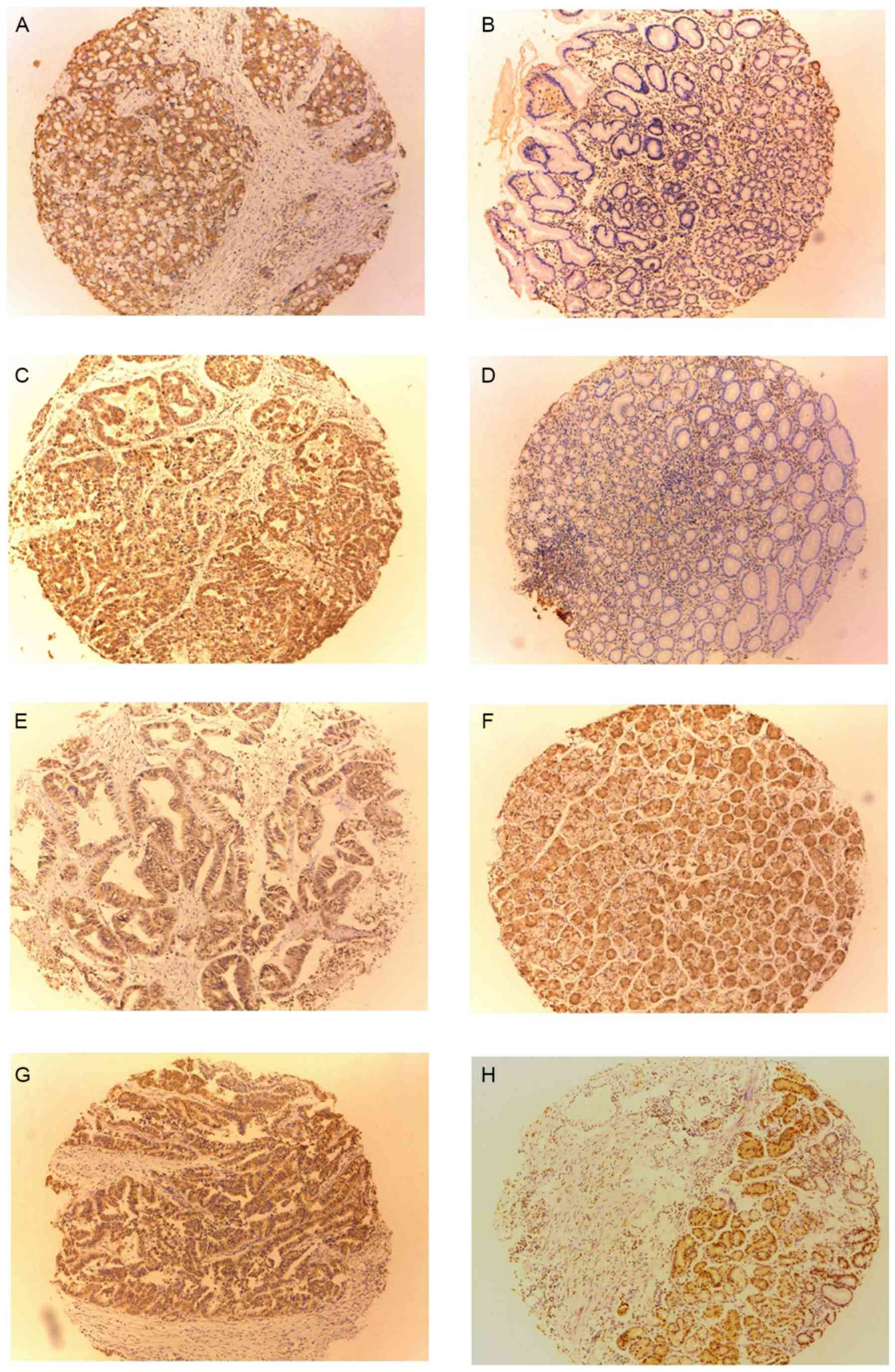
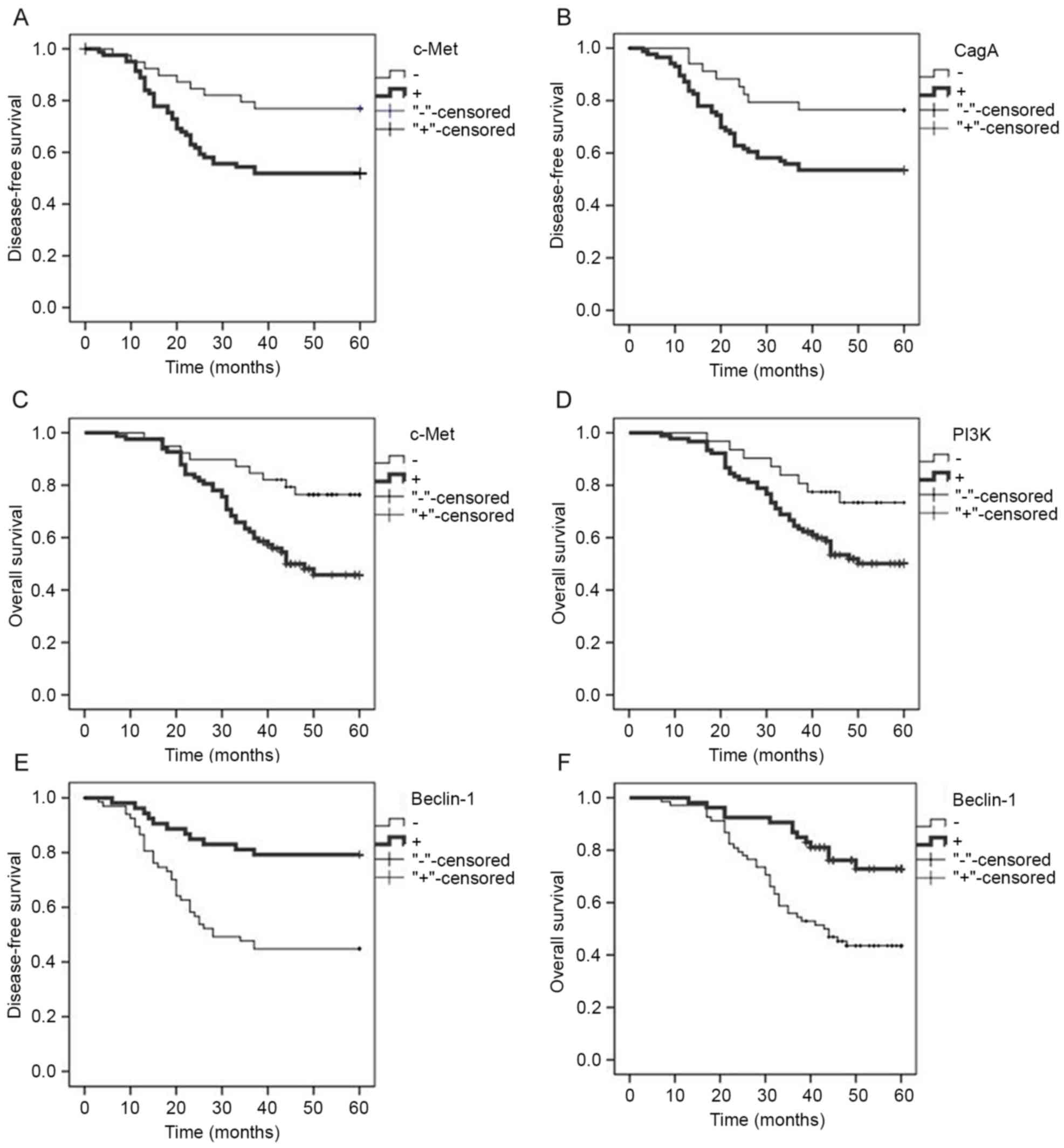
|
1
|
Stewart BW and Wild CP: World Cancer
Report 2014. Geneva: World Health Organization, International
Agency for Research on Cancer, WHO Press; 2015
|
|
2
|
Zhang S, Chen P, Huang Z, Hu X, Chen M, Hu
S, Hu Y and Cai T: Sirt7 promotes gastric cancer growth and
inhibits apoptosis by epigenetically inhibiting miR-34a. Sci Rep.
10:97872015. View Article : Google Scholar
|
|
3
|
WHO, . A Survey of Cancer 2014[DB/OL].
http://www.who.int/cancer/country-profiles/en/2014
|
|
4
|
Siegel R, Naishadham D and Jemal A: Cancer
statistics for Hispanics/Latinos. CA Cancer J Clin. 62:283–298.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Li ZX and Kaminishi MA: Comparison of
gastric cancer between Japan and China. Gastric Cancer. 12:52–53.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
International Agency for Research on
Cancer, . Helicobatcer pylori Eradication as a Strategy for
Preventing Gastric Cancer, IARC Working Group Reports. 8. Lyon:
IARC; 2013, http://www.iarc.fr/en/publications/pdfs-online/wrk/wrk8/Helicobacter_pylori_Eradication.pdfJune
14–2016
|
|
7
|
Odenbreit S, Püls J, Sedlmaier B, Gerland
E, Fischer W and Haas R: Translocation of Helicobacter pylori CagA
into gastric epithelial cells by type IV secretion. Science.
287:1497–1500. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Higashi H, Tsutsumi R, Muto S, Sugiyama T,
Azuma T, Asaka M and Hatakeyama M: SHP-2 tyrosine phosphatase as an
intracellular target of Helicobacter pylori CagA protein. Science.
295:683–686. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Higashi H, Yokoyama K, Fujii Y, Ren S,
Yuasa H, Saadat I, Murata-Kamiya N, Azuma T and Hatakeyama M: EPIYA
motif is a membrane-targeting signal of Helicobacter pylori
virulence factor CagA in mammalian cells. J Biol Chem.
280:23130–23137. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Peek RM Jr and Blaser MJ: Helicobacter
pylori and gastrointestinal tract adenocarcinomas. Nat Rev Cancer.
2:28–37. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
11
|
Blaser MJ, Perez-Perez GI, Kleanthous H,
Cover TL, Peek RM, Chyou PH, Stemmermann GN and Nomura A: Infection
with Helicobacter pylori strains possessing cagA is associated with
an increased risk of developing adenocarcinoma of the stomach.
Cancer Res. 55:2111–2115. 1995.PubMed/NCBI
|
|
12
|
Yang JJ, Yang JH, Kim J, Ma SH, Cho LY, Ko
KP, Shin A, Choi BY, Kim HJ, Han DS, et al: Soluble c-Met protein
as a susceptible biomarker for gastric cancer risk: A nested
case-control study within the Korean Multicenter Cancer Cohort. Int
J Cancer. 132:2148–2156. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Taniguchi K, Yonemura Y, Nojima N, Hirono
Y, Fushida S, Fujimura T, Miwa K, Endo Y, Yamamoto H and Watanabe
H: The relation between the growth patterns of gastric carcinoma
and the expression of hepatocyte growth factor receptor (c-met),
autocrine motility factor receptor, and urokinase-type plasminogen
activator receptor. Cancer. 82:2112–2122. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Toiyama Y, Yasuda H, Saigusa S, Matushita
K, Fujikawa H, Tanaka K, Mohri Y, Inoue Y, Goel A and Kusunoki M:
Co-expression of hepatocyte growth factor and c-Met predicts
peritoneal dissemination established by autocrine hepatocyte growth
factor/c-Met signaling in gastric cancer. Int J Cancer.
130:2912–2921. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Li Y, Chen CQ, He YL, Cai SR, Yang DJ, He
WL, Xu JB and Zan WH: Abnormal expression of E-cadherin in tumor
cells is associated with poor prognosis of gastric carcinoma. J
Surg Oncol. 106:304–310. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Yıldız Y, Sokmensuer C and Yalcin S:
Evaluation of c-Met, HGF, and HER-2 expressions in gastric
carcinoma and their association with other clinicopathological
factors. Onco Targets Ther. 9:5809–5817. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Churin Y, Al-Ghoul L, Kepp O, Meyer TF,
Birchmeier W and Naumann M: Helicobacter pylori CagA protein
targets the c-Met receptor and enhances the motogenic response. J
Cell Biol. 161:249–255. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wang RC, Wei Y, An Z, Zou Z, Xiao G,
Bhagat G, White M, Reichelt J and Levine B: Akt-mediated regulation
of autophagy and tumorigenesis through Beclin 1 phosphorylation.
Science. 338:956–959. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Vasconcelos AC, Wagner VP, Meurer L,
Vargas PA, de Souza LB, Fonseca FP, Squarize CH, Castilho RM and
Martins MD: Immunoprofile of c-MET/PI3K signaling in human salivary
gland tumors. Oral Surg Oral Med Oral Pathol Oral Radiol.
120:238–247. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zhuo B, Li Y, Li Z, Qin H, Sun Q, Zhang F,
Shen Y, Shi Y and Wang R: PI3K/Akt signaling mediated Hexokinase-2
expression inhibits cell apoptosis and promotes tumor growth in
pediatric osteosarcoma. Biochem Biophys Res Commun. 464:401–406.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Jung KA, Choi BH and Kwak MK: The
c-MET/PI3K signaling is associated with cancer resistance to
doxorubicin and photodynamic therapy by elevating BCRP/ABCG2
expression. Mol Pharmacol. 87:465–476. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wu J, Chen C and Zhao KN:
Phosphatidylinositol 3-kinase signaling as a therapeutic target for
cervical cancer. Curr Cancer Drug Targets. 13:143–156. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Edge SB, Byrd DR, Compton CC, Fritz AG,
Greene FL and Trotti A: AJCC Cancer Staging Manual. 7th. Springer;
New York: pp. 191–200. 2010
|
|
24
|
Okada F: Inflammation-related
carcinogenesis: Current findings in epidemiological trends, causes
and mechanisms. Yonago Acta Med. 57:65–72. 2014.PubMed/NCBI
|
|
25
|
Wajih N, Walter J and Sane DC: Vascular
origin of a soluble truncated form of the hepatocyte growth factor
receptor (c-met). Circ Res. 90:46–52. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Porter J: Small molecule c-Met kinase
inhibitors: A review of recent patents. Expert Opin Ther Pat.
20:159–177. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Oliveira MJ, Costa AC, Costa AM, Henriques
L, Suriano G, Atherton JC, Machado JC, Carneiro F, Seruca R, Mareel
M, et al: Helicobacter pylori induces gastric epithelial cell
invasion in a c-Met and type IV secretion system-dependent manner.
J Biol Chem. 281:34888–34896. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Snider JL and Cardelli JA: Helicobacter
pylori induces cancer cell motility independent of the c-Met
receptor. J Carcinog. 8:72009. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Hu Y, Li L, Yin W, Shen L, You B and Gao
H: Protective effect of proanthocyanidins on anoxia-reoxygenation
injury of myocardial cells mediated by the PI3K/Akt/GSK-3β pathway
and mitochondrial ATP-sensitive potassium channel. Mol Med Rep.
10:2051–2058. 2014. View Article : Google Scholar : PubMed/NCBI
|