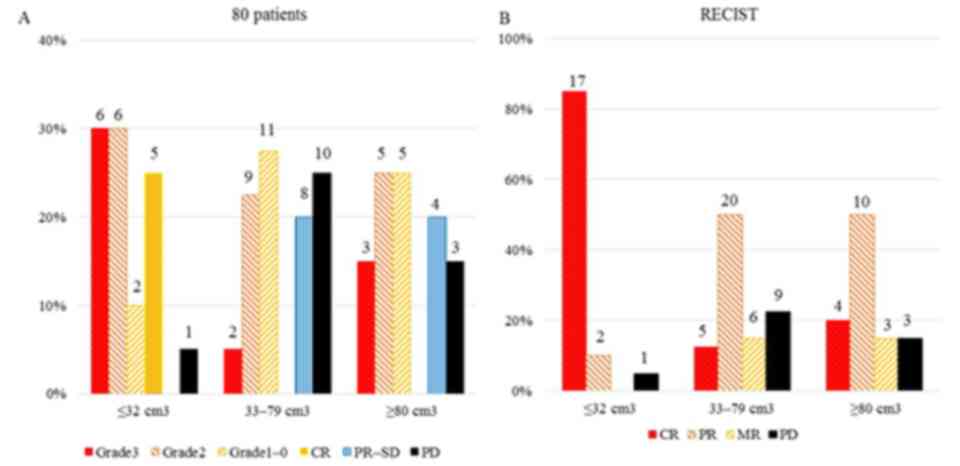
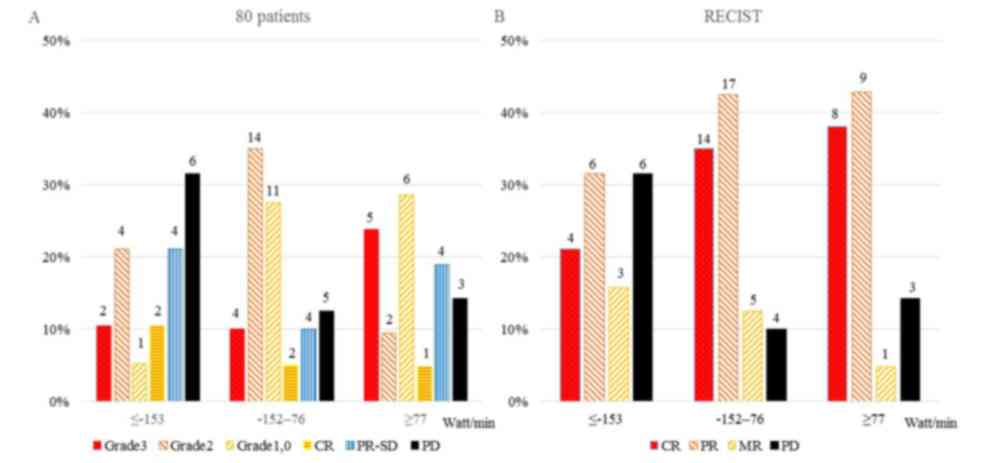
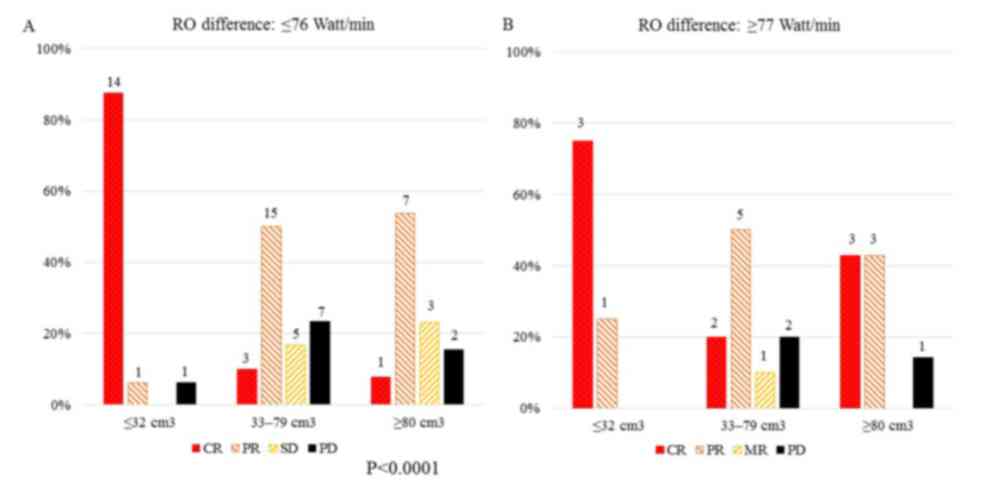
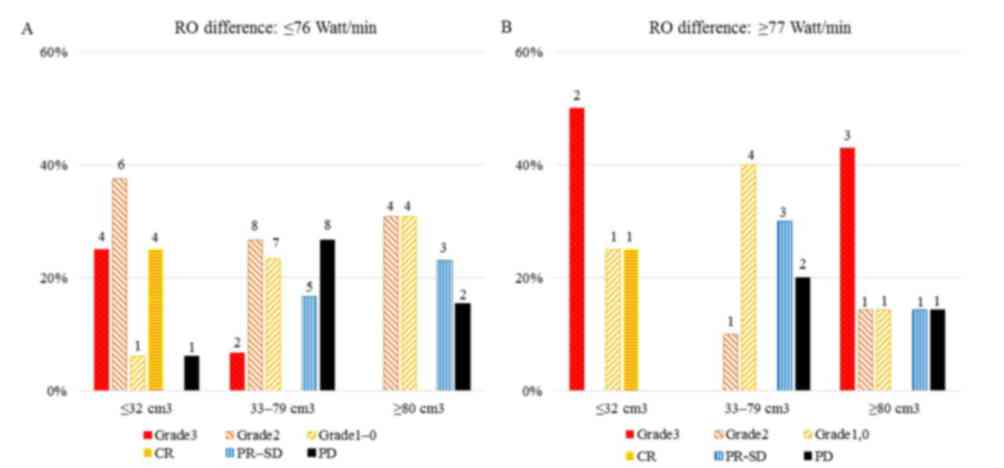
|
1
|
Powathil GG, Adamson DJ and Chaplain MA:
Towards predicting the response of a solid tumour to chemotherapy
and radiotherapy treatments: clinical insights from a computational
model. PLoS Comput Biol. 9:e10031202013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Holck S, Nielsen HJ, Pedersen N and
Larsson LI: Phospho-ERK1/2 levels in cancer cell nuclei predict
responsiveness to radiochemotherapy of rectal adenocarcinoma.
Oncotarget. 6:34321–34328. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Guo Z, Shu Y, Zhou H, Zhang W and Wang H:
Radiogenomics helps to achieve personalized therapy by evaluating
patient responses to radiation treatment. Carcinogenesis.
36:307–317. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bosset JF, Calais G, Mineur L, Maingon P,
Stojanovic-Rundic S, Bensadoun RJ, Bardet E, Beny A, Ollier JC,
Bolla M, et al: Fluorouracil-based adjuvant chemotherapy after
preoperative chemoradiotherapy in rectal cancer: Long-term results
of the EORTC 22921 randomised study. Lancet Oncol. 15:184–190.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Sauer R, Liersch T, Merkel S, Fietkau R,
Hohenberger W, Hess C, Becker H, Raab HR, Villanueva MT, Witzigmann
H, et al: Preoperative versus postoperative chemoradiotherapy for
locally advanced rectal cancer: Results of the German
CAO/ARO/AIO-94 randomized phase III trial after a median follow-up
of 11 years. J Clin Oncol. 30:1926–1933. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Park JH, Yoon SM, Yu CS, Kim JH, Kim TW
and Kim JC: Randomized phase 3 trial comparing preoperative and
postoperative chemoradiotherapy with capecitabine for locally
advanced rectal cancer. Cancer. 117:3703–3712. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Peeters KC, Marijnen CA, Nagtegaal ID,
Kranenbarg EK, Putter H, Wiggers T, Rutten H, Pahlman L, Glimelius
B, Leer JW, et al: The TME trial after a median follow-up of 6
years: Increased local control but no survival benefit in
irradiated patients with resectable rectal carcinoma. Ann Surg.
246:693–701. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Das P, Lin EH, Bhatia S, Skibber JM,
Rodriguez-Bigas MA, Feig BW, Chang GJ, Hoff PM, Eng C, Wolff RA, et
al: Preoperative chemoradiotherapy with capecitabine versus
protracted infusion 5-fluorouracil for rectal cancer: A matched
pair analysis. Int J Radiat Oncol Biol Phys. 66:1378–1383. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Maas M, Nelemans PJ, Valentini V, Das P,
Rödel C, Kuo LJ, Calvo FA, García-Aguilar J, Glynne-Jones R,
Haustermans K, et al: Long-term outcome in patients with a
pathological complete response after chemoradiation for rectal
cancer: A pooled analysis of individual patient data. Lancet Oncol.
11:835–844. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yeo SG, Kim DY, Kim TH, Chang HJ, Oh JH,
Park W, Choi DH, Nam H, Kim JS, Cho MJ, et al: Pathologic complete
response of primary tumor following preoperative chemoradiotherapy
for locally advanced rectal cancer: Long-term outcomes and
prognostic significance of pathologic nodal status (KROG 09–01).
Ann Surg. 252:998–1004. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Park IJ, You YN, Agarwal A, Skibber JM,
Rodriguez-Bigas MA, Eng C, Feig BW, Das P, Krishnan S, Crane CH, et
al: Neoadjuvant treatment response as an early response indicator
for patients with rectal cancer. J Clin Oncol. 30:1770–1776. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Fokas E, Liersch T, Fietkau R, Hohenberger
W, Beissbarth T, Hess C, Becker H, Ghadimi M, Mrak K, Merkel S, et
al: Tumor regression grading after preoperative chemoradiotherapy
for locally advanced rectal carcinoma revisited: Updated results of
the CAO/ARO/AIO-94 trial. J Clin Oncol. 32:1554–1562. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zhu J, Gu W, Lian P, Sheng W, Cai G, Shi
D, Cai S and Zhang Z: A phase II trial of neoadjuvant IMRT-based
chemoradiotherapy followed by one cycle of capecitabine for stage
II/III rectal adenocarcinoma. Radiat Oncol. 8:1302013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Roh MS, Colangelo LH, OConnell MJ, Yothers
G, Deutsch M, Allegra CJ, Kahlenberg MS, Baez-Diaz L, Ursiny CS,
Petrelli NJ and Wolmark N: Preoperative multimodality therapy
improves disease-free survival in patients with carcinoma of the
rectum: NSABP R-03. J Clin Oncol. 27:5124–5130. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Habr-Gama A, Perez RO, Nadalin W, Sabbaga
J, Ribeiro U Jr, Silva e Sousa AH Jr, Campos FG, Kiss DR and
Gama-Rodrigues J: Operative versus nonoperative treatment for stage
0 distal rectal cancer following chemoradiation therapy: Long-term
results. Ann Surg. 240:711–718. 2004.PubMed/NCBI
|
|
16
|
Shoji H, Motegi M, Osawa K, Okonogi N,
Okazaki A, Andou Y, Asao T, Kuwano H, Takahashi T and Ogoshi K:
Does standardization of radiofrequency hyperthermia benefit
patients with malignancies? Ann Cancer Res Ther. 22:28–35. 2014.
View Article : Google Scholar
|
|
17
|
Shoji H, Motegi M, Osawa K, Okonogi N,
Okazaki A, Andou Y, Asao T, Kuwano H, Takahashi T and Ogoshi K:
Output-limiting symptoms induced by radiofrequency hyperthermia.
Are they predictable? Int J Hyperthermia. 32:199–203. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Japanese Society for Cancer of the Colon
and Rectum: Japanese classification of colon carcinoma. 8th
edition. Kanehara, Tokyo: 2013
|
|
19
|
Shoji H, Motegi M, Osawa K, Okonogi N,
Okazaki A, Andou Y, Asao T, Kuwano H, Takahashi T and Ogoshi K: A
novel strategy of radiofrequency hyperthermia (neothermia) in
combination with preoperative chemoradiotherapy for the treatment
of advanced rectal cancer: A pilot study. Cancer Med. 4:834–843.
2015. View
Article : Google Scholar : PubMed/NCBI
|
|
20
|
Shoji H, Motegi M, Osawa K, Okonogi N,
Okazaki A, Andou Y, Asao T, Kuwano H, Takahashi T and Ogoshi K:
Radiofrequency thermal treatment with chemoradiotherapy for
advanced rectal cancer. Oncol Rep. 35:2569–2575. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Therasse P, Arbuck SG, Eisenhauer EA,
Wanders J, Kaplan RS, Rubinstein L, Verweij J, Van Glabbeke M, van
Oosterom AT, Christian MC and Gwyther SG: New guidelines to
evaluate the response to treatment in solid tumors. European
Organization for Research and Treatment of Cancer, National Cancer
Institute of the United States, National Cancer Institute of
Canada. J Natl Cancer Inst. 92:205–216. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
US and National Cancer Institute Cancer
Therapy Evaluation Program: Common Terminology Criteria for Adverse
Events (CTCAE). http://ctep.cancer.gov/protocolDevelopment/electronic_applications/ctc.htm#ctc_40Feb
14–2017
|
|
23
|
Krishnan S, Janjan NA, Skibber JM,
Rodriguez-Bigas MA, Wolff RA, Das P, Delclos ME, Chang GJ, Hoff PM,
Eng C, et al: Phase II study of capecitabine (Xeloda) and
concomitant boost radiotherapy in patients with locally advanced
rectal cancer. Int J Radiat Oncol Biol Phys. 66:762–771. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Yerushalmi R, Idelevich E, Dror Y, Stemmer
SM, Figer A, Sulkes A, Brenner B, Loven D, Dreznik Z, Nudelman I,
et al: Preoperative chemoradiation in rectal cancer: Retrospective
comparison between capecitabine and continuous infusion of
5-fluorouracil. J Surg Oncol. 93:529–533. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
De Paoli A, Chiara S, Luppi G, Friso ML,
Beretta GD, Del Prete S, Pasetto L, Santantonio M, Sarti E,
Mantello G, et al: Capecitabine in combination with preoperative
radiation therapy in locally advanced, resectable, rectal cancer: A
multicentric phase II study. Ann Oncol. 17:246–251. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Craven I, Crellin A, Cooper R, Melcher A,
Byrne P and Sebag-Montefiore D: Preoperative radiotherapy combined
with 5 days per week capecitabine chemotherapy in locally advanced
rectal cancer. Br J Cancer. 97:1333–1337. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Elshazly WG, Farouk M and Samy M:
Preoperative concomitant radiotherapy with oral capecitabine in
advanced rectal cancer within 6 cm from anal verge. Int J
Colorectal Dis. 24:401–407. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Valentini V, Gambacorta MA, Barbaro B,
Chiloiro G, Coco C, Das P, Fanfani F, Joye I, Kachnic L, Maingon P,
et al: International consensus guidelines on clinical target volume
delineation in rectal cancer. Radiother Oncol. 120:195–201. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
De Ridder M, Tournel K, Van Nieuwenhove Y,
Engels B, Hoorens A, Everaert H, de Beeck Op B, Vinh-Hung V, De
Grève J, Delvaux G, et al: Phase II study of preoperative helical
tomotherapy for rectal cancer. Int J Radiat Oncol Biol Phys.
70:728–734. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Huang MY, Chen CF, Huang CM, Tsai HL, Yeh
YS, Ma CJ, Wu CH, Lu CY, Chai CY, Huang CJ and Wang JY: Helical
tomotherapy combined with capecitabine in the preoperative
treatment of locally advanced rectal cancer. Biomed Res Int.
2014:3520832014.PubMed/NCBI
|
|
31
|
Maluta S, Romano M, Dalloglio S, Genna M,
Oliani C, Pioli F, Gabbani M, Marciai N and Palazzi M: Regional
hyperthermia added to intensified preoperative chemo-radiation in
locally advanced adenocarcinoma of middle and lower rectum. Int J
Hyperthermia. 26:108–117. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Schroeder C, Gani C, Lamprecht U, von
Weyhern CH, Weinmann M, Bamberg M and Berger B: Pathological
complete response and sphincter-sparing surgery after neoadjuvant
radiochemotherapy with regional hyperthermia for locally advanced
rectal cancer compared with radiochemotherapy alone. Int J
Hyperthermia. 28:707–714. 2012. View Article : Google Scholar : PubMed/NCBI
|