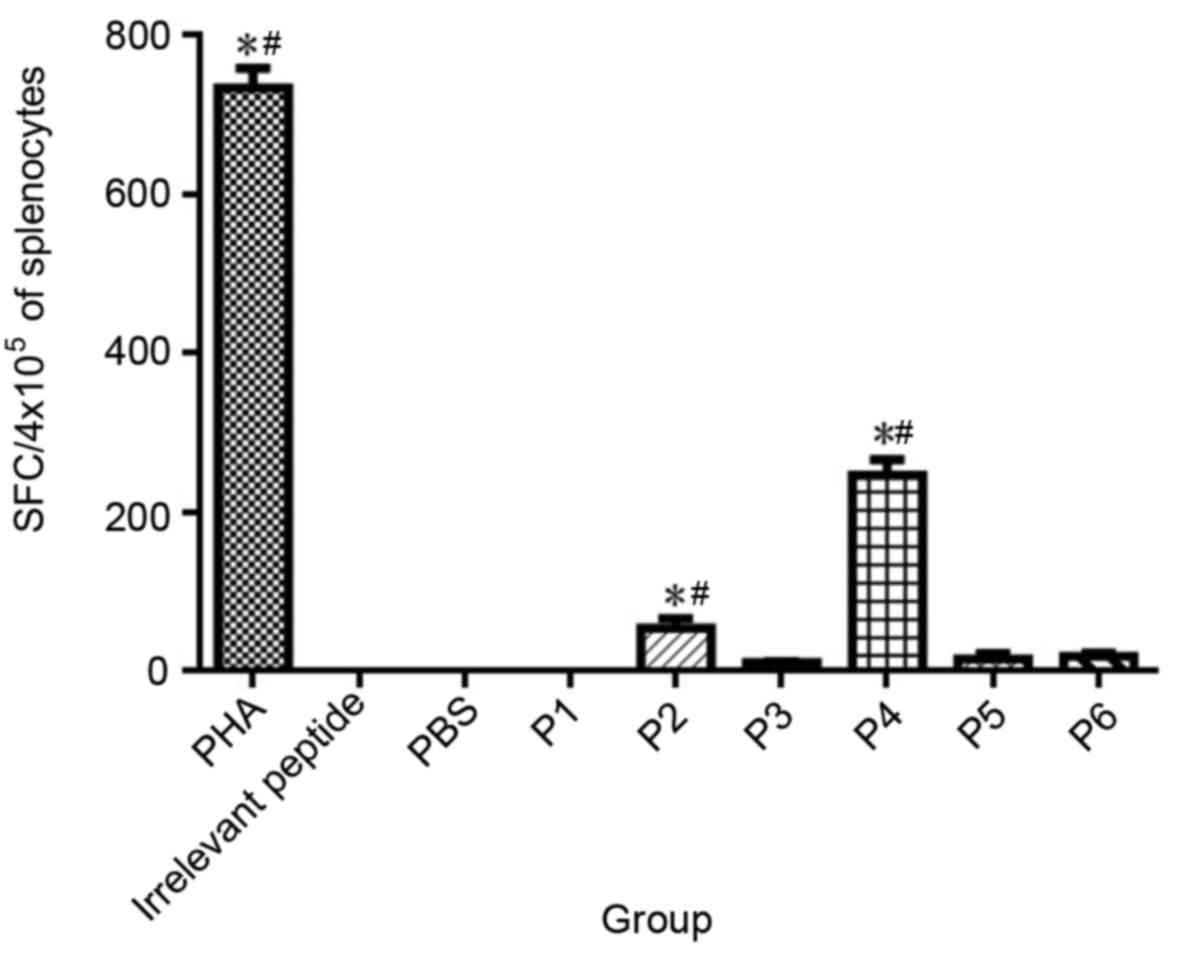
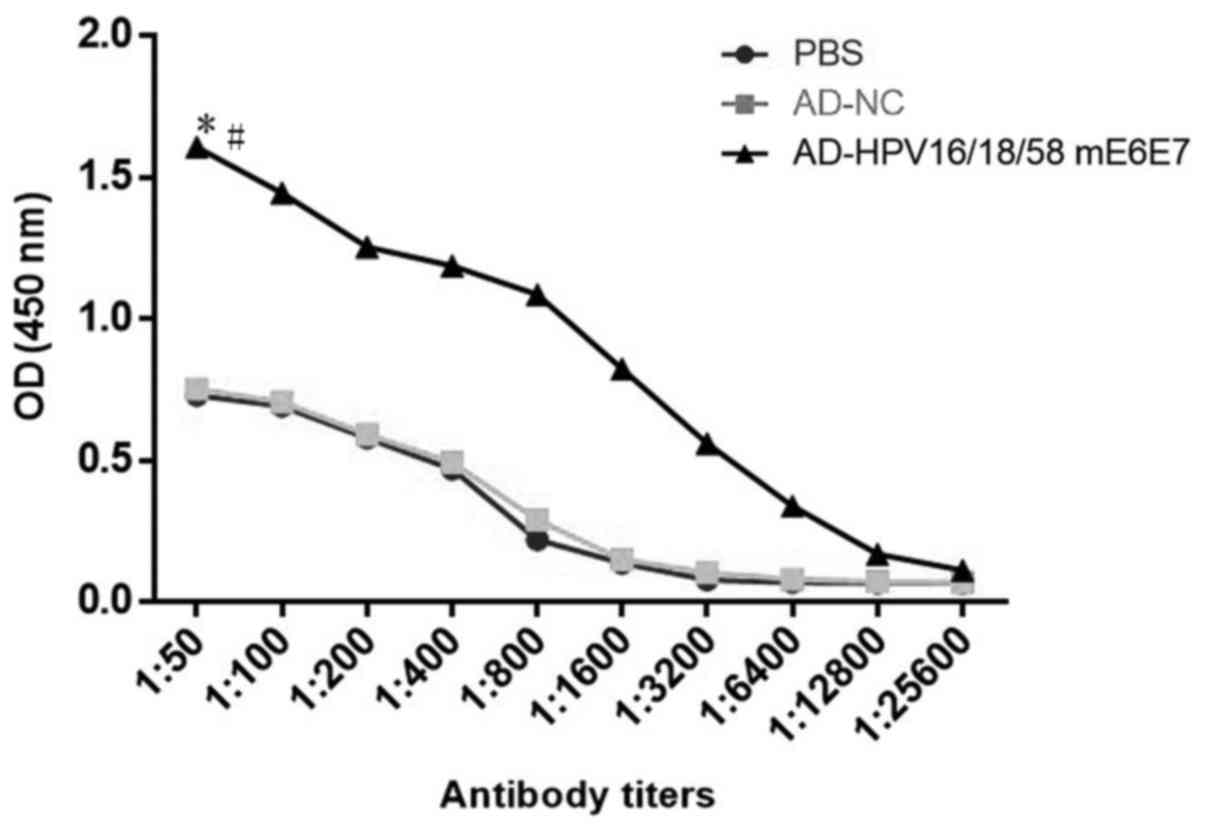
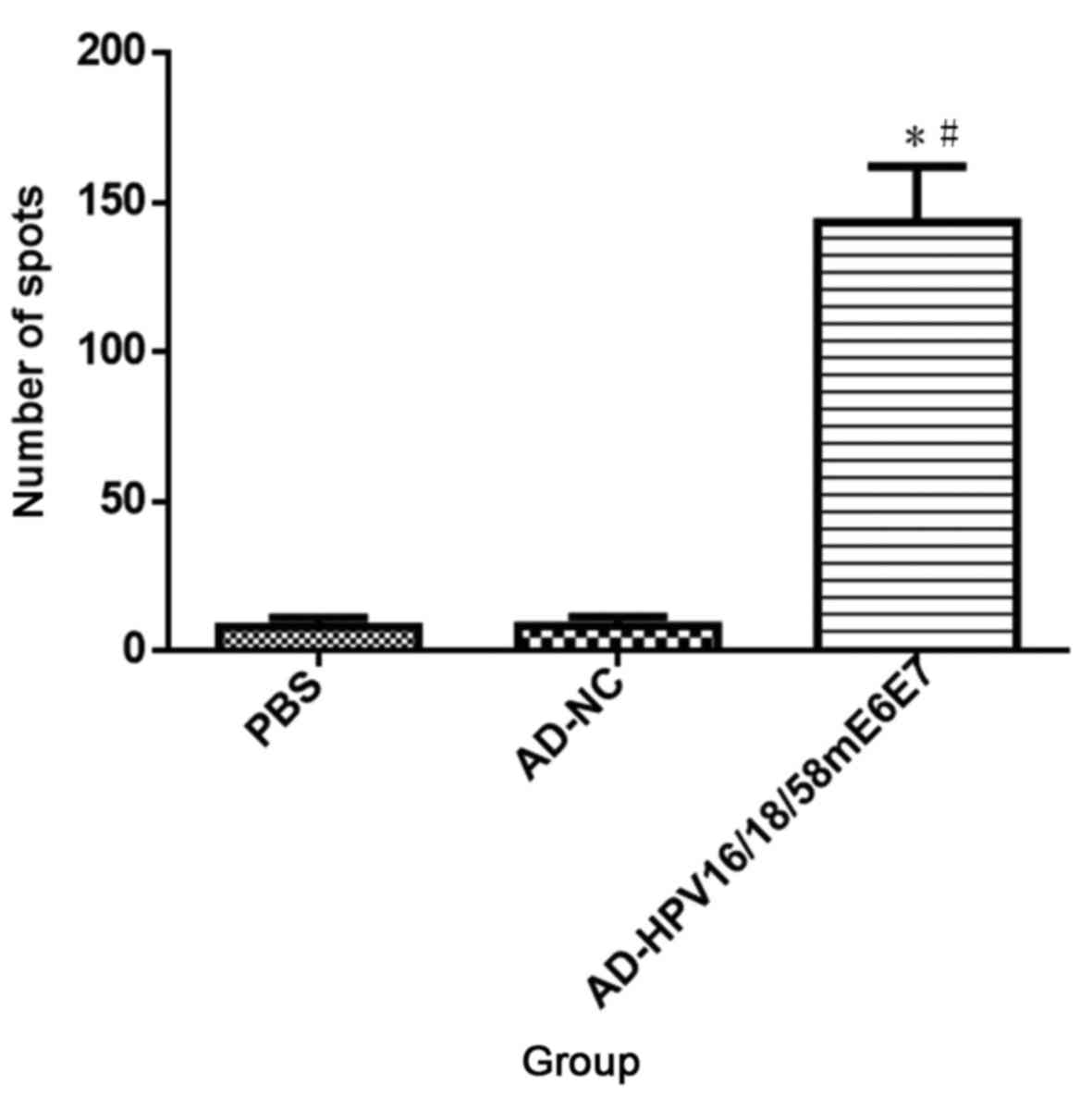
|
1
|
Walboomers JM, Jacobs MV, Manos MM, Bosch
FX, Kummer JA, Shah KV, Snijders PJ, Peto J, Meijer CJ and Muñoz N:
Human papillomavirus is a necessary cause of invasive cervical
cancer worldwide. J Pathol. 189:12–19. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bosch FX, Manos MM, Muñoz N, Sherman M,
Jansen AM, Peto J, Schiffman MH, Moreno V, Kurman R and Shah KV:
Prevalence of human papillomavirus in cervical cancer: a worldwide
perspective. International biological study on cervical cancer
(IBSCC) study group. J Natl Cancer Inst. 87:796–802. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Maleki Z: Human papilloma virus
vaccination: Review article and an update. World J Obstet Gynaecol.
5:16–27. 2016. View Article : Google Scholar
|
|
4
|
Varughese J and Richman S: Cancer care
inequity for women in resource-poor countries. Rev Obstet Gynecol.
3:122–132. 2010.PubMed/NCBI
|
|
5
|
Lo KW, Wong YF, Chan MK, Li JC, Poon JS,
Wang VW, Zhu SN, Zhang TM, He ZG, Wu QL, et al: Prevalence of human
papillomavirus in cervical cancer: A multicenter study in China.
Int J Cancer. 100:327–331. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Callejas-Valera JL, Iglesias-Bartolome R,
Amornphimoltham P, Palacios-Garcia J, Martin D, Califano JA,
Molinolo AA and Gutkind JS: mTOR inhibition prevents rapid-onset of
carcinogen-induced malignancies in a novel inducible HPV-16 E6/E7
mouse model. Carcinogenesis. 37:1014–1025. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
McLachlin CM: Human papillomavirus in
cervical neoplasia. Role, risk factors and implications. Clin Lab
Med. 20:257–270, v. 2000.
|
|
8
|
Ullman CG and Emery VC: Transforming
proteins of human papillomaviruses. Rev Med Virol. 6:39–55. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Münger K, Baldwin A, Edwards KM, Hayakawa
H, Nguyen CL, Owens M, Grace M and Huh K: Mechanisms of human
papillomavirus-induced oncogenesis. J Virol. 78:11451–11460. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wain G: The human papillomavirus (HPV)
vaccine, HPV related diseases and cervical cancer in the
post-reproductive years. Maturitas. 65:205–209. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Bounds CE, Hu J, Cladel NM, Balogh K and
Christensen ND: Vaccine generated immunity targets an HPV16 E7
HLA-A2.1-restricted CD8(+) T cell epitope relocated to an early
gene or a late gene of the cottontail rabbit papillomavirus (CRPV)
genome in HLA-A2.1 transgenic rabbits. Vaccine. 29:1194–1200. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Chen L, Mizuno MT, Singhal MC, Hu SL,
Galloway DA, Hellström I and Hellström KE: Induction of cytotoxic T
lymphocytes specific for a syngeneic tumor expressing the E6
oncoprotein of human papillomavirus type 16. J Immunol.
148:2617–2621. 1992.PubMed/NCBI
|
|
13
|
Nurkkala M, Wassén L, Nordström I,
Gustavsson I, Slavica L, Josefsson A and Eriksson K: Conjugation of
HPV16 E7 to cholera toxin enhances the HPV-specific T-cell recall
responses to pulsed dendritic cells in vitro in women with cervical
dysplasia. Vaccine. 28:5828–5836. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zheng Y, Zhang Y, Ma Y, Wan J, Shi C and
Huang L: Enhancement of immunotherapeutic effects of HPV16E7 on
cervical cancer by fusion with CTLA4 extracellular region. J
Microbiol. 46:728–736. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Gan L, Jia R, Zhou L, Guo J and Fan M:
Fusion of CTLA-4 with HPV16 E7 and E6 enhanced the potency of
therapeutic HPV DNA vaccine. PLoS One. 9:e1088922014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ren F, Xu Y, Mao L, Ou R, Ding Z, Zhang X,
Tang J, Li B, Jia Z, Tian Z, et al: Heat shock protin 110 improves
the antitumor effects of the cytotoxic T lymphocyte epitope
E7(49–57) in mice. Cancer Biol Ther. 9:134–141. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Rötzschke O, Falk K, Stevanović S, Jung G
and Rammensee HG: Peptides motif of closely related HLA class I
molecules encompass substantial differences. Eur J Immunol.
22:2453–2456. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Speidel K, Osen W, Faath S, Hilgert I,
Obst R, Braspenning J, Momburg F, Hämmerling GJ and Rammensee HG:
Priming of cytotoxic T lymphocytes by five heat-aggregated antigens
in vivo: Conditions, efficiency, and relation to antibody
responses. Eur J Immunol. 27:2391–2399. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Schäfer K, Müller M, Faath S, Henn A, Osen
W, Zentgraf H, Benner A, Gissmann L and Jochmus I: Immune response
to human papilomavirns 16 L1E7 chimeric virus-1ike particles:
Induction of cytotoxic T cells and specific tumor protection. Int J
Cancer. 81:881–888. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Xu YS, Hao F, Song ZQ, Zhong B, Hao J and
Ye Q: Screening and Identification of Predicted Epitopes of
HLA-A2-restricted Cytotoxic T Lymphocytes Derived from the HPV16 E7
Antigen. Chin J Dermatol. 37:283–284. 2004.(In Chinese).
|
|
21
|
Riemer AB, Keskin DB, Zhang G, Handley M,
Anderson KS, Brusic V, Reinhold B and Reinherz EL: A conserved
E7-derived cytotoxic T lymphocyte epitope expressed on human
papillomavirus 16-transformed HLA-A2+ epithelial cancers. J Biol
Chem. 285:29608–29622. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Harro CD, Pang YY, Roden RB, Hildesheim A,
Wang Z, Reynolds MJ, Mast TC, Robinson R, Murphy BR, Karron RA, et
al: Safety and immunogenicity trial in adult volunteers of a human
papillomavirus 16 L1 virus-like particle vaccine. J Natl Cancer
Inst. 93:284–292. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Chen S, Ou R, Tang J, Deng X, Wu Y, van
Velkinburgh JC, Ni B and Xu Y: Enhanced anti-tumor effects of
HPV16E7(49–57)-based vaccine by combined immunization with
poly(I:C) and oxygen-regulated protein 150. Cancer Epidemiol.
37:172–178. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Yao Y, Huang W, Yang X, Sun W, Liu X, Cun
W and Ma Y: HPV-16 E6 and E7 protein T cell epitopes prediction
analysis based on distributions of HLA-A loci across populations:
An in silico approach. Vaccine. 31:2289–2294. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Chan PK, Liu SJ, Cheung TH, Yeo W, Ngai
SM, Cheung JL, Chong P and Man S: T-cell response to human
papillomavirus type 58 L1, E6, And E7 peptides in women with
cleared infection, cervical intraepithelial neoplasia, or invasive
cancer. Clin Vaccine Immunol. 17:1315–1321. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Sabah SN, Gazi MA, Sthity RA, Husain AB,
Quyyum SA, Rahman M and Islam MR: Designing of epitope-focused
vaccine by targeting E6 and E7 conserved protein sequences: An
immuno-informatics approach in human papillomavirus 58 isolates.
Interdiscip Sci. 17 Sep. 2016, (Epub ahead of print). PubMed/NCBI
|
|
27
|
Tu SH, Huang HI, Lin SI, Liu HY, Sher YP,
Chiang SK, Chong P, Roffler S, Tseng GC, Chen HW and Liu SJ: A
novel HLA-A2-restricted CTL epitope of tumor-associated antigen L6
can inhibit tumor growth in vivo. J Immunother. 35:235–244. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Rudolf MP, Man S, Melief CJ, Sette A and
Kast WM: Human T-cell responses to HLA-A-restricted high binding
affinity peptides of human papillomavirus type 18 proteins E6 and
E7. Clin Cancer Res. 7(3 Suppl): 788s–795s. 2001.PubMed/NCBI
|