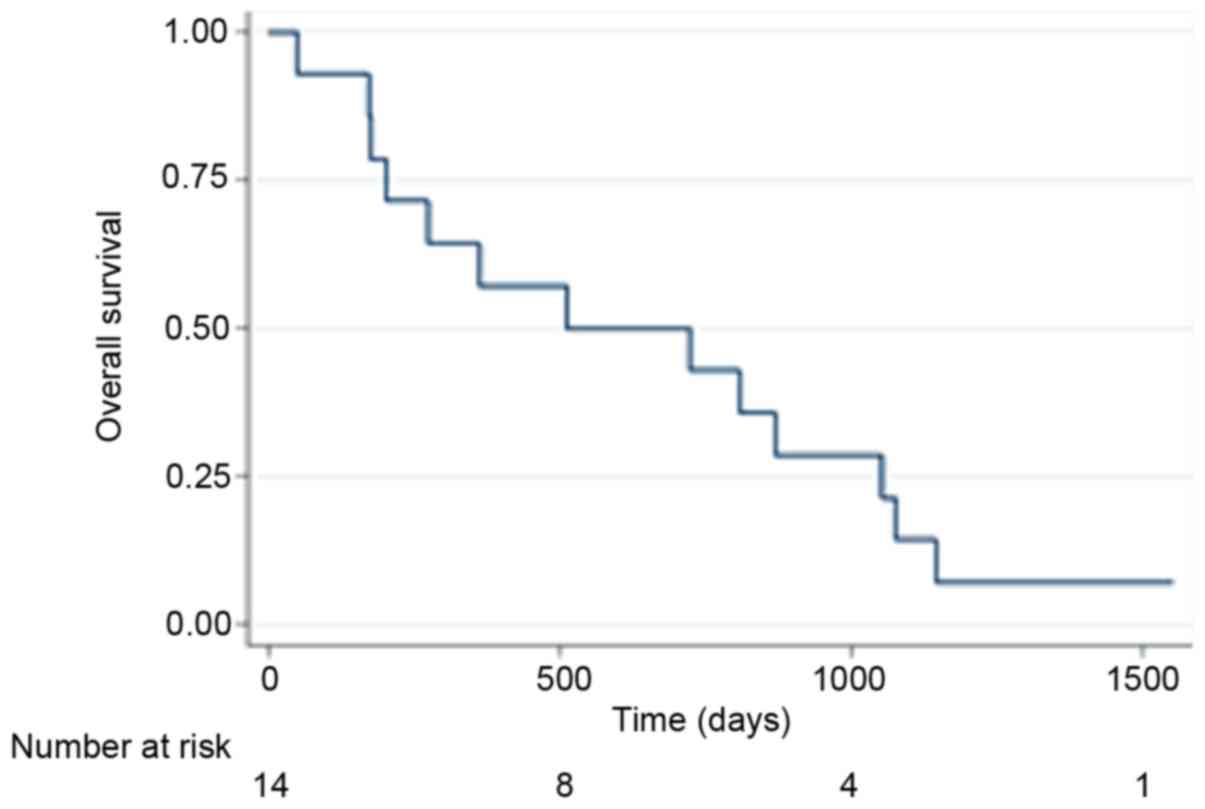
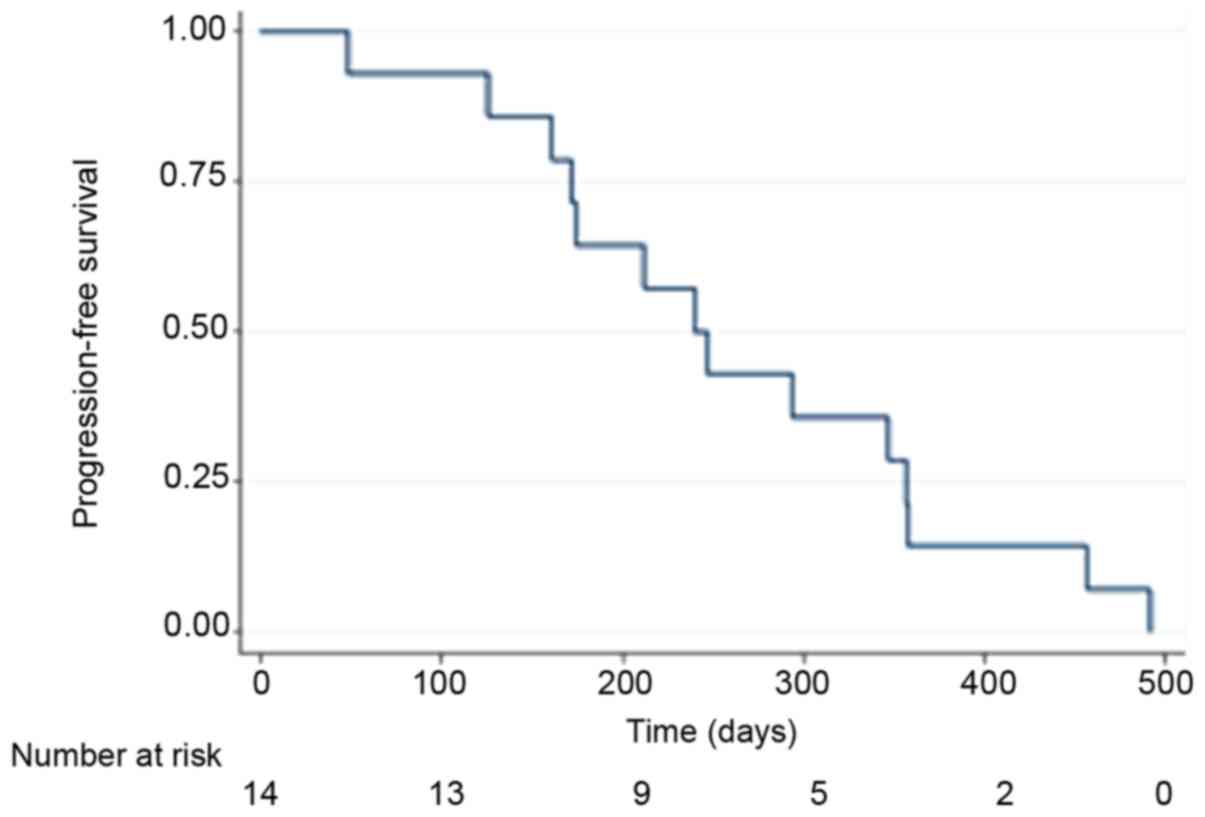
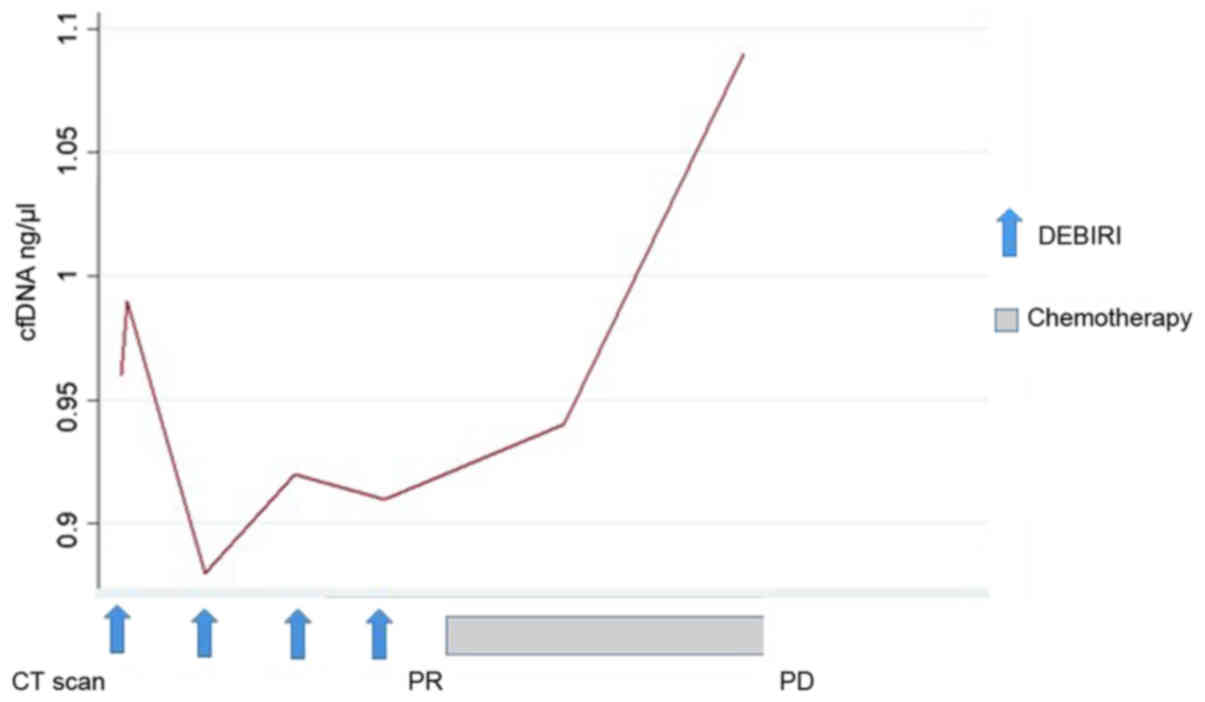
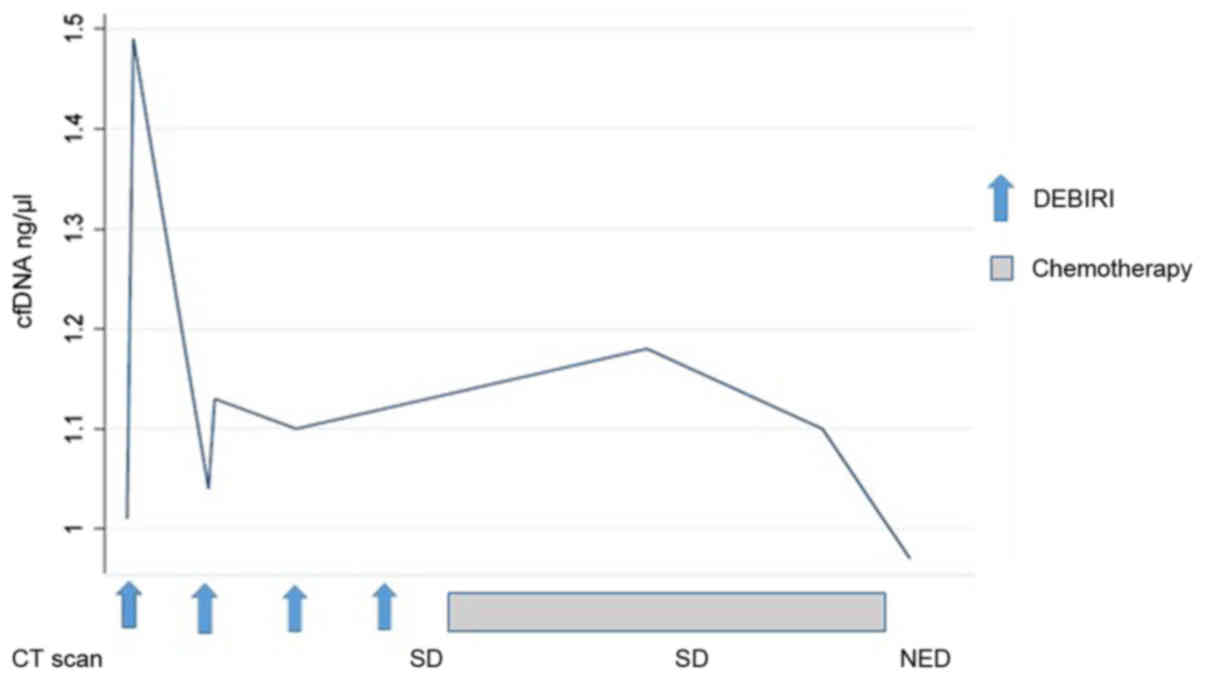
|
1
|
Ferlay J, Soerjomataram I, Dikshit R, Eser
S, Mathers C, Rebelo M, Parkin DM, Forman D and Bray F: CCancer
incidence and mortality worldwide: Sources, methods and major
patterns in GLOBOCAN 2012. Int J Cancer. 136:E359–E389. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Weiss L, Grundmann E, Torhorst I, Hartveit
F, Moberg I, Ider M, Fenoglio-Preiser CM, Napier J, Horne CH, Lopez
MJ, et al: Haematogenous metastatic patterns in colonic carcinoma:
an analysis of 1541 necropsies. J Pathol. 150:195–203. 1986.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kopetz S, Chang GJ, Overman MJ, Eng C,
Sargent DJ, Larsson DW, Grothey A, Vauthey JN, Nagorney DM and
McWilliams RR: Improved survival in metastatic colorectal cancer is
associated with adoption of hepatic resection and improved
chemotherapy. J Clin Oncol. 27:3677–3683. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Gillams AR and Lees WR: Five-year survival
in 309 patients with colorectal liver metastases treated with
radiofrequency ablation. Eur Radiol. 19:1206–1213. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Fode MM and Høyer M: Survival and
prognostic factors in 321 patients treated with stereotactic body
radiotherapy for oligo-metastases. Radiother Oncol. 114:155–160.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Yamada R, Sato M, Kawabata K, Nakatsuka H,
Nakamura K and Takashima S: Hepatic artery embolization in 120
patients with unresectable hepatoma. Radiology. 148:397–401. 1983.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Aliberti C, Tilli M, Benea G and
Fiorentini G: Trans-arterial chemoembolization (TACE) of liver
metastases from colorectal cancer using irinotecan-eluting beads:
Preliminary results. Anticancer Res. 26:3793–3795. 2006.PubMed/NCBI
|
|
8
|
Fiorentini G, Aliberti C, Tilli M,
Mulazzani L, Graziano F, Giodani P, Mambrini A, Montagnani F,
Alessandroni P, Catalano V and Coschiera P: IIntra-arterial
infusion of irinotecan-loaded drug-eluting beads (DEBIRI) versus
intravenous therapy (FOLFIRI) for hepatic metastases from
colorectal cancer: Final results of a phase III study. Anticancer
Res. 32:1387–1395. 2012.PubMed/NCBI
|
|
9
|
Martin RC II, Scoggins CR, Schreeder M,
Rilling WS, Laing CJ, Tatum CM, Kelly LR, Garcia-Monaco RD, Sharma
VR, Crocenzi TS and Strasberg SM: Randomized controlled trial of
irinotecan drug-eluting beads with simultaneous FOLFOX and
bevacizumab for patients with unresectable colorectal liver-limited
metastasis. Cancer. 121:3649–3658. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
van Cutsem E, Cervantes A, Adam R, Sobrero
A, van Krieken JH, Aderka D, Aguilar Aranda E, Bardelli A, Benson
A, Bodoky G, et al: ESMO consensus guidelines for the management of
patients with metastatic colorectal cancer. Ann Oncol.
27:1386–1422. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Spindler KL, Appelt A, Pallisgaard N,
Andersen RF, Brandslund I and Jakobsen A: Cell-free DNA in healthy
individuals, noncancerous disease and strong prognostic value in
colorectal cancer. Int J Cancer. 135:2984–2991. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Mandel P and Metasis P: Les acides
nucléiques du plasma sanguin chez l'homme. C R Seances Soc Biol
Fil. 142:241–243. 1948.(In Undetermined Language). PubMed/NCBI
|
|
13
|
Leon SA, Shapiro B, Sklaroff DM and Yaros
MJ: Free DNA in the serum of cancer patients and the effect of
therapy. Cancer Res. 37:646–650. 1977.PubMed/NCBI
|
|
14
|
Boysen AK, Wettergren Y, Sorensen BS,
Taflin H, Gustavson B and Spindler KG: Cell-free DNA levels and
correlation to stage and outcome following treatment of locally
advanced rectal cancer. Tumour Biol. 39:10104283177309762017.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Spindler KG, Boysen AK, Pallisgård N,
Johansen JS, Tabernero J, Sørensen MM, Jensen BV, Hansen TF,
Sefrioui D, Andersen RF, et al: Cell-free DNA in metastatic
colorectal cancer: A systematic review and meta-analysis.
Oncologist. 22:1049–1055. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Eisenhauer EA, Therasse P, Bogaerts J,
Schwartz LH, Sargant D, Ford R, Dancey J, Arbuck S, Gwyther S,
Mooney M, et al: New response evaluation criteria in solid tumours:
Revised RECIST guideline (version 1.1). Eur J Cancer. 45:228–247.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Cancer Therapy Evaluation Program, Common
Terminology Criteria for Adverse Events, . Version 3.0. DCTD, NCI,
NIH, DHHS March 31. 2003.http://ctep.cancer.govAugust 9–2006
|
|
18
|
Price DD, McGrath PA, Rafii A and
Buckingham B: The validation of visual analogue scales as ratio
scale measures for chronic and experimental pain. Pain. 17:45–56.
1983. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Goldshtein H, Hausmann MJ and Douvdevani
A: A rapid direct fluorescent assay for cell-free DNA
quantification in biological fluids. Ann Clin Biochem. 46:488–494.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Schou JV, Larsen FO, Sørensen BS, Abrates
R, Boysen AK, Johansen JS, Jensen BV, Nielsen DL and Spindler KL:
Circulating cell-free DNA as predictor of treatment failure after
neoadjuvant chemo-radiotherapy before surgery in patients with
locally advanced rectal cancer. Ann Oncol. 29:610–615. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Eichler K, Zangus S, Mack MG, Hammerstring
R, Gruber-Rouh T, Galluc C and Vogl TJ: First human study in
treatment of unresectable liver metastases from colorectal cancer
with irinotecan-loaded beads (DEBIRI). Int J Oncol. 41:1213–1220.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Fiorentini G, Aliberti C, Turrisi G, del
Conte A, Rossi S, Benea G and Giovanis P: IIntraarterial hepatic
chemoembolization of liver metastases from colorectal cancer
adopting irinotecan-eluting beads: Results of a phase II clinical
study. In Vivo. 21:1085–1091. 2007.PubMed/NCBI
|
|
23
|
Loupakis F, Cremolini C, Masi G, Lonardi
S, Zagonel V, Salvatore L, Cortesi E, Tomasello G, Ronzoni M, Spadi
R, et al: Initial therapy with FOLFOXIRI and bevacizumab for
metastatic colorectal cancer. N Engl J Med. 371:1609–1618. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Tie J, Kinde I, Wang Y, Wong HL, Roebort
J, Christie M, Tacey M, Wong R, Singh M, Karapetis CS, et al:
Circulating tumor DNA as an early marker of therapeutic response in
patients with metastatic colorectal cancer. Ann Oncol.
26:1715–1722. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Reinert T, Schøler L, Thomsen R, Tobiasen
H, Vang S, Nordentoft I, Lamy P, Kannerup AS, Mortensen FV,
Stribolt K, et al: Analysis of circulating tumour DNA to monitor
disease burden following colorectal cancer surgery. Gut.
65:625–634. 2016. View Article : Google Scholar : PubMed/NCBI
|