Introduction
Rotor Syndrome (RS) is a rare autosomal recessive
syndrome that is characterized by conjugated hyperbilirubinemia and
coproporphyria (1–4). The main clinical manifestation is
non-pruritic jaundice. When compared with Dubin Johnson Syndrome,
the pigmentation in the liver is not observed in RS, although the
two conditions exhibit similar clinical findings. Histopathological
liver biopsies of these patients have a normal appearance. While
the etiology of RS is not well known, it has been suggested that
the condition occurs as a result of a complete lack or partial
defect of organic anion transporter polypeptides (OATPs) on the
basolateral surface of hepatocytes. The bile ducts are not viewable
in hepatobiliary scintigraphy due to an impaired liver uptake of
radioactive material (5,6). No information is available in the
literature with regard to the association between colon cancer and
RS. The present study aimed to discuss the applied systematic
chemotherapy and its effect on a case of RS with developed colon
cancer.
Case report
A 47-year-old male patient with a diagnosis of
operative-rectal adenocarcinoma was admitted to the Department of
Medical Oncology (Akdeniz University Faculty of Medicine, Antalya,
Turkey) in order to undergo adjuvant chemotherapy. Upon physical
examination, the whole body and sclerae were icteric. A 5–6-cm long
scar track and colostomy was present on the anterior abdominal wall
due to prior surgery. Other systemic examinations were normal. The
patient was evaluated as T2N1M1 (stage four) according to the TNM
staging system. Upon pathological determination of colon serosal
invasion, three regional lymph node metastases were determined
using positron emission tomography (PET). Hypermetabolic nodules
(1.2 cm) on the posterior segment of the lower lobe accounted for
two of the node metastases, while the remaining node was situated
on the anterior portion of the upper lobe (1 cm). When
investigating the blood and biochemical parameters of the patient,
a total bilirubin level of 6.7 mg/dl and a direct bilirubin level
of 5 mg/dl were observed. The transaminase, γ-glutamyl transferase,
alkaline phosphatase, blood urea nitrogen (BUN) and creatine levels
were determined to be within the normal ranges. Due to the
conjugated hyperbilirubinemia the patient was diagnosed with RS
subsequent to being admitted for jaundice. Treatment with
ursodeoxycholic acid was administered for a long period of time. At
the multidisciplinary oncology council, a metastasectomy was
decided upon for the metastatic foci on the left side of the lung.
The surgical margins were determined as positive when the
metastasectomy materials were examined. The patient provided
written informed consent.
Conformal radiotherapy was administered at 3,000 cGy
to the metastasectomy area and at 5,040 cGy to the pelvis. Six
cycles of systemic chemotherapy were administered to the patient
following radiotherapy, every three weeks with 85
mg/m2/day oxaliplatin. The disease was evaluated as
stable as a result of a control PET scan following the sixth cycle.
The treatment of the patient was arranged as a combination
chemotherapy and consisted of 1,000 mg/m2 capecitabine
twice a day for two weeks in combination with 85
mg/m2/day oxaliplatin every three weeks. The patient’s
biluribin level increased after one week. Capecitabine treatment
was interrupted, as the total blood bilirubin level, which was
drawn two weeks following the first cycle of chemotherapy using
oxaliplatin + capecitabine, was determined as 18 mg/dl. In the
liver and bile tracts, ultrasonography was performed due to the
total blood bilirubin levels. The intrahepatic bile tracts were
evaluated as normal in the patient, who was diagnosed with grade I
hepatosteatosis, which was not a focal lesion in the liver
parenchyma. Two more cycles of oxaliplatin monotherapy were
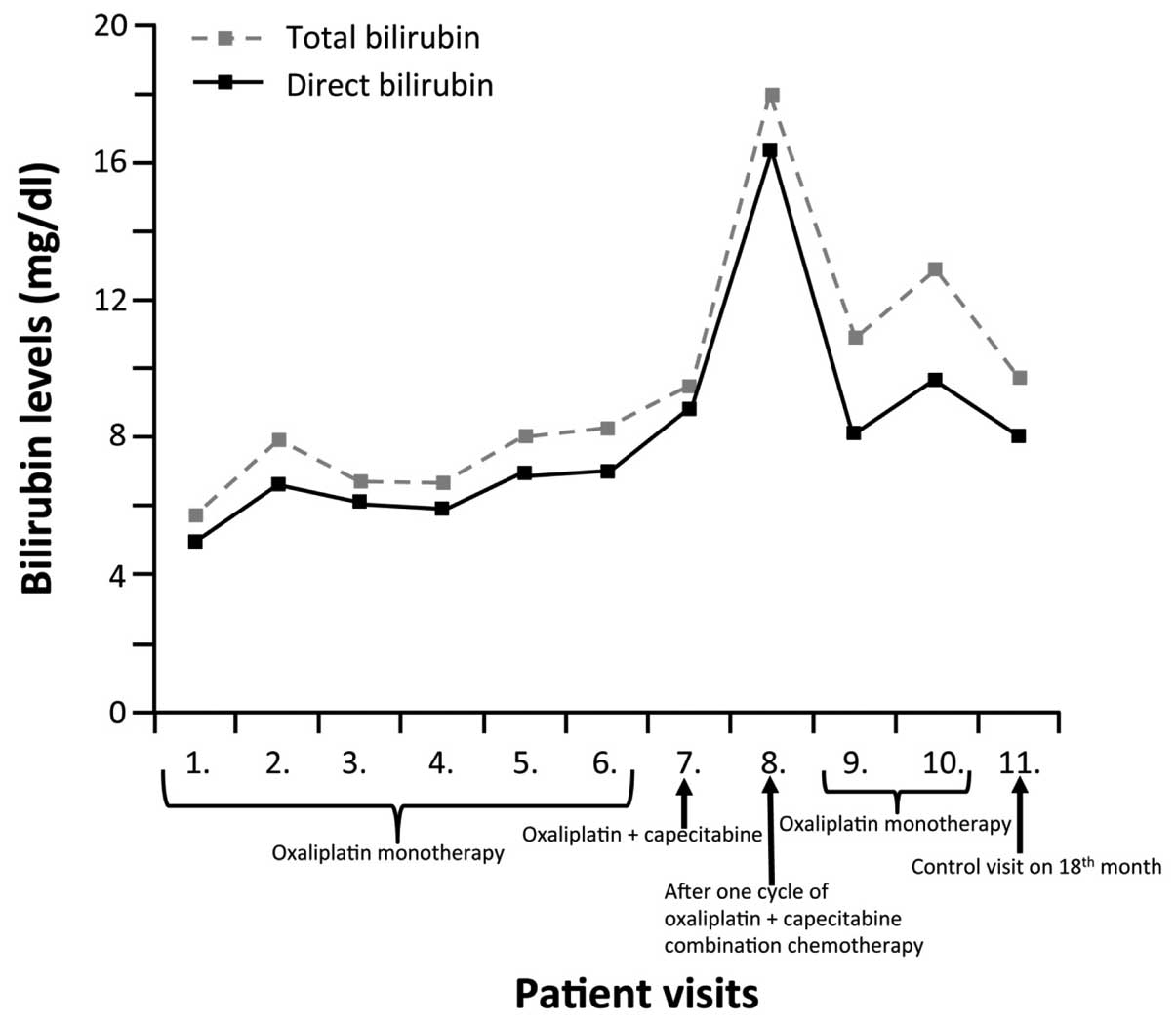
administered to the patient. The changes in the total blood
bilirubin levels of the patient are shown in Fig. 1. PET was administered to the
patient, who underwent a total of nine cycles of oxaliplatin
therapy. Pathological findings were not determined in the
colonoscopy. However, hypermetabolic wall thickening was determined
in the right lower quadrant of the gut wall. It was decided at the
multidisciplinary oncology council that the post-operative and
metastatic situations of the patient, who had tumor markers in the
normal range during the pre-operative period, should be followed up
without treatment. The patient was followed up without treatment or
progression for 18 months.
Discussion
RS is a rare syndrome that is characterized by
familial conjugated hyperbilirubinemia. The etiology of RS is not
well known, however, it has been suggested that RS occurs due to
the partial defect or lack of OATPs, including OATP1B1 and OATP1B3,
which are located on the basolateral surface of hepatocytes and
function to excrete organic anions, bile acids and bilirubin bile
sinusoids (5,6). There is no information in the
literature with regard to patients with RS and colon carcinoma. In
one previous study, it was determined that the de novo
expression of the SLC21 gene, which encodes OATP proteins, was
elevated in breast, colon, pancreas, stomach and prostate cancers
(7). It is indicated in the
literature that various single gene polymorphisms on the OATP1 gene
cause an elevation in the drug levels in the tissues and blood by
reducing the elimination of certain drugs, including statins,
methotrexate and irinotecan (8).
There is no information with regard to the use of
5-flourourasil (5-FU) and capecitabine for the treatment of
patients with RS and colon carcinoma. 5-FU and capecitabine are
antimetabolite chemotherapeutic drugs that are analogs of
pyrimidine and are metabolized and excreted in the liver. However,
oxaliplatin is primarily eliminated from the kidneys (9). The present study determined the
presence of a significant increase in the total blood bilirubin
levels in the patient following the administration of oxaliplatin
chemotherapy. The patient tolerated the oxaliplatin chemotherapy
well. However, following the capecitabine chemotherapy, a
three-fold increase in the basal total blood bilirubin levels
(basal total blood bilirubin of the patient was 6 mg/dl) was
observed and the Eastern Cooperative Oncology Group performance
increased from 0 to 3. Therefore, capecitabine chemotherapy is not
the right agent to administer to patients with RS and colon
carcinoma.
In conclusion, there is no information in the
literature with regard to RS with colon carcinoma. Oxaliplatin was
an applicable agent for use in the patient with RS and colon
carcinoma as there were no side-effects in determining the total
blood bilirubin levels and was well tolerated. The patient has been
followed up for 18 months without treatment or progression.
References
|
1
|
Rotor AB, Manahan L and Florentin A:
Familial nonhemolytic jaundice with direct van den Bergh reaction.
Acta Med Phil. 5:37–49. 1948.
|
|
2
|
Wolpert E, Pascasio FM, Wolkoff AW and
Arias IM: Abnormal sulfobromophthalein metabolism in Rotor’s
syndrome and obligate heterozygotes. N Engl J Med. 296:1099–1101.
1977.
|
|
3
|
Bar-Meir S, Baron J, Seligson U,
Gottesfeld F, Levy R and Gilat T: 99mTc-HIDA cholescintigraphy in
Dubin-Johnson and Rotor syndromes. Radiology. 142:743–746. 1982.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
LeBouthillier G, Morais J, Picard M,
Picard D, Chartrand R and Pommier G: Scintigraphic aspect of
Rotor’s disease with technetium-99m-mebrofenin. J Nucl Med.
33:1550–1551. 1992.
|
|
5
|
Kartenbeck J, Leuschner U, Mayer R and
Keppler D: Absence of the canalicular isoform of the MRP
gene-encoded conjugate export pump from the hepatocytes in
Dubin-Johnson syndrome. Hepatology. 23:1061–1066. 1996.PubMed/NCBI
|
|
6
|
Paulusma CC, Kool M, Bosma PJ, Scheffer
GL, ter Borg F, Scheper RJ, Tytgat GN, Borst P, Baas F and Oude
Elferink RP: A mutation in the human canalicular multispecific
organic anion transporter gene causes the Dubin-Johnson syndrome.
Hepatology. 25:1539–1542. 1997. View Article : Google Scholar
|
|
7
|
Obaidat A, Roth M and Hagenbuch B: The
expression and function of organic anion transporting polypeptides
in normal tissues and in cancer. Annu Rev Pharmacol Toxicol.
52:135–151. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kalliokoski A and Niemi M: Impact of OATP
transporters on pharmacokinetics. Br J Pharmacol. 158:693–705.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Graham MA, Lockwood GF, Greenslade D, et
al: Clinical pharmacokinetics of oxaliplatin: a critical review.
Clin Cancer Res. 6:1205–1218. 2000.PubMed/NCBI
|